Abstract
Phenotype-driven screens using chemical mutagens, such as
ENU is a commonly used mutagen for phenotype-driven screens in mice. As an alkylating agent its ethyl group is transferred to nucleophilic sites on each of the four nucleotides of DNA. This results in mispairing at the next round of DNA replication. ENU predominantly mutates A:T base pairs, and in mouse phenotypic screens approximately 60% of the mutations detected are A:T to T:A transitions or A:T to G:C transversions. In descending order of occurrence, ENU produces missense, nonsense and splice-site mutations and these tend to occur in genes with longer coding sequences and higher exon numbers. 2
The most important factors in the design of a mutagenesis experiment are the rate at which germline mutations are induced and the ability to recover healthy, fertile male mice for breeding. Several studies have been published on optimizing ENU dosage in different inbred strains of mouse and these are summarized in recent reviews. 7,8 Although the exact dose that produces the highest mutation frequency may vary from one strain to another when administered in a single dose, increasing the dose of ENU increases the mutation rate. However, this effect does not remain linear at high, single doses. Instead, the maximum efficiency is achieved when high doses are administered in a fractionated form with a short period separating each injection, typically a week.
Published data on the pathology of ENU-treated males in the context of a mutagenesis programme are generally scant. In one of the few examples, a tumour survey in retired ENU-treated (C57BL/6 × CBA) F1 males, lung tumours, thymoma (taken as a synonym for thymic lymphoma) and liver tumours were the most common and skin, kidney, prostate and colon tumours occurred, but were less frequent. 7 In this paper, we have explored the breeding and pathology records from our experience with the mutagenesis programme at the Medical Research Council (MRC) Harwell to optimize the programme by developing breeding management practices and to help address welfare issues. This paper is a retrospective case-control study of the breeding and fate of 760 male ENU-mutagenized mice (BALB/cAnNCrl and latterly C57BL/6J) and a more detailed analysis of tumour pathology in 226 of these mice. We have focused on how ENU treatment affects lifetime fecundity in relation to ENU-induced pathology, especially clinically important tumour entities that necessitated premature culling. This information has been used to refine the breeding programme.
Materials and methods
Study design
The MRC Harwell operates phenotype-driven dominant and recessive screens. During the course of the early MRC mutagenesis projects, the inbred strain of mice used was BALB/cAnNCrl. For the latter mutagenesis projects commencing in 2004, C57BL/6J was the major strain used though smaller numbers of BALB/cAnNCrl, BALB/cByJ and CeH/HeH were also used.
Three ENU dosage regimens were used. The ENU dose was titrated in an attempt to optimize mutagenic rate in offspring as determined by heritable phenotypes. The experimental aim was to be just above the borderline of infertility as higher ENU doses increase the mutation frequency. The numbers of mice of each strain used in each dosage regimen and necropsy sampling statistics are given in Table 1. A general overview of the timeline for the programme is given in Figure 1a.
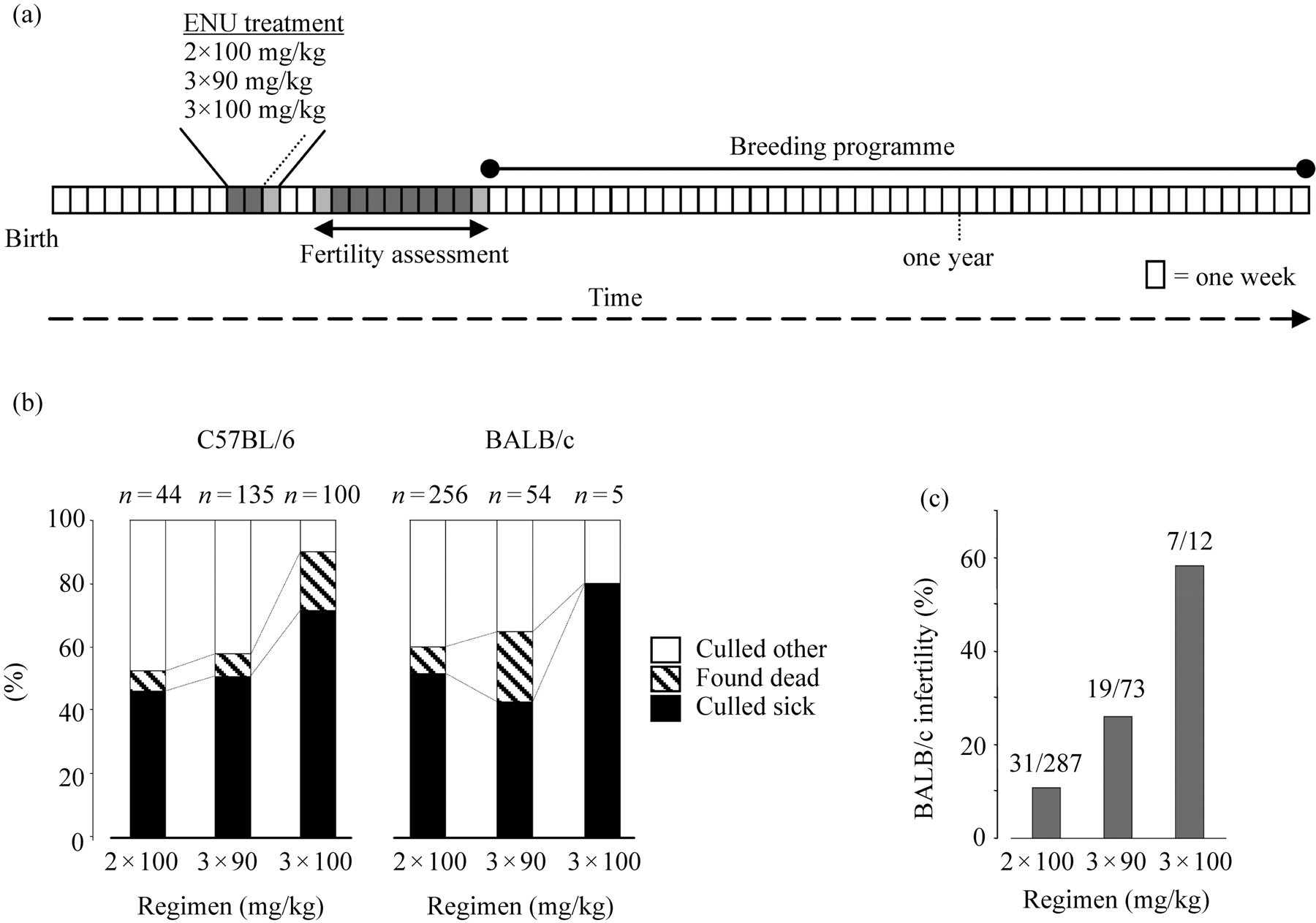
(a) Major timeline events for G0 ENU-treated male mice used for the phenotype-driven screens. Starting at 10 weeks each mouse was injected intraperitoneally with ENU at weekly intervals. The regimens employed were two doses of 100 mg/kg at one-week intervals, three weekly doses of 90 mg/kg or three weekly doses of 100 mg/kg. (b) Categories of males lost to the breeding programme for C57BL/6J and BALB/cAnNCrl strains. The number of mice culled sick or found dead increases with ENU dose. (c) In BALB/cAnNCrl mice the highest dosage regimen was poorly tolerated and resulted in a high percentage of mice failing to regain fertility. These were excluded from analysis in (b) for clarity. A similar effect is seen in C57BL/6J mice which were analysed in more detail (Figure 3)
Mutagenesis programme population sizes in this study
*Because this dosage was not tolerated by BALB/cAnNCrl mice this was discontinued
†Includes mice that were culled after losing 20% body weight acutely post-treatment and excluded for further analysis (36 BALB/cAnNCrl and 1 C57BL/6J)
Mice, husbandry and health screening
The humane care and use of mice in the rederivation programme was conducted under the Home Office regulations. The earlier MRC Harwell mutagenesis projects were performed in a largely conventional mouse house facility; however, the ENU-treated mice were kept in individually ventilated cages (IVCs). This old low health status mouse house has now been replaced by a new high health status, mouse only, specific pathogen free (SPF) barrier facility, the Mary Lyon Centre (MLC). The new centre also has digital record-keeping that has greatly facilitated data mining.
BALB/cAnNCrl males were purchased from Charles River and imported as 4–6-week-old mice. They were protected from exposure to infectious agents (see below) by housing them in IVCs (Charles River Laboratories UK, Margate, UK) in a 12 h light/dark (07:00–19:00 light phase) cycle at 19–23°C and 45–65% relative humidity on grade 6 sawdust bedding (Datesand Ltd, Manchester, UK). All equipment was autoclaved before use. Husbandry and ENU treatments were performed in a class II biological safety cabinet. Mice were fed rat and mouse No. 3 breeding diet (Special Diets Services, Witham, Essex, UK) and provided with chlorinated water
Sentinels were challenged by infectious agents through airborne exposure. Live mice were submitted to an outside laboratory (Harlan UK Ltd Technical Services Department, Loughborough, UK) for microbiological testing based on FELASA recommendations.
9
Health screening in the pre-existing conventional colony (before rederivation of colonies into the MLC) showed the presence of mouse hepatitis virus (judged by histology to be enteropathic strains), mouse adenovirus type 2, Theiler's murine encephalomyelitis virus, the intestinal flagellates
In some of the more recent ENU mutagenesis projects, male C57BL/6J (Jackson Laboratories, Bar Harbor, Maine, USA) mice were used from a SPF colony established in the MLC rederived by embryo transfer as described. 10 Mouse husbandry in the MLC was essentially the same as in the pre-existing mouse facilities except that the mice were housed in Techniplast IVCs (Techniplast UK Ltd, Kettering, UK) and the diet was irradiated.
The microbiological screening programme in the MLC involves sending mice from sentinel cages (one per 56 cages) that are challenged with samples of dirty bedding from separate columns in each rack on an eight-week rotation as previously described. 11 Health screening has shown that the SPF status described above has been maintained since the facility opened more than two years ago.
ENU treatment and mouse breeding
As the BALB/cAnNCrl in the earlier mutagenesis projects were imported at approximately six weeks of age, they were assigned an estimated birth date. Records of exact birth dates were available for the C57BL/6J and BALB/cAnNCrl males bred in the MLC. At approximately 10–12 weeks of age (target 10 weeks), ENU doses were administered intraperitoneally at weekly intervals: 3 × 90 mg/kg or 2 × 100 mg/kg in BALB/cAnNCrl males and 3 × 90 mg/kg, 2 × 100 mg/kg or 3 × 100 mg/kg in C57BL/6J males (Figure 1a). These ‘generation zero’ ENU-treated males are referred to hereafter as G0 males to distinguish them from their first generation offspring hereafter termed as G1 mice.
Successful ENU treatment causes a period of sterility of approximately eight weeks starting 3–4 weeks post-treatment 12 reflecting exposure of pre-spermatid stages and significant germcell death. For the first three weeks, post-injection progeny may arise from spermatozoa that were in the later stages of development when exposed to ENU. Therefore, ENU-treated males were test-bred (one male and one female) with similarly aged females from week 4 to week 11 after the last dose. The females from these matings were culled at 11 weeks and examined at postmortem for the presence of fetuses. The males were judged to have regained fertility very early (see below) if a pregnancy was detected and the males were subsequently withdrawn from the programme.
At 12 weeks after ENU treatment, males were then set up for matings with two C3H/HeH females with an aim to produce 50–60 offspring. A target of 50–60 offspring was chosen to avoid the theoretical possibility of producing multiple identical mutations in offspring due to the oligoclonality of ENU-mutagenized spermatogonial stem cells that repopulate the testis following ENU treatment. ENU treatment can result in poor fertility in G0 males (see below), so the number of G1 offspring produced in each mating was carefully monitored. If an ENU-treated male did not produce offspring in two months, or was a poor breeder (i.e. had one litter but had not produced a litter for 3 months), the decision on whether to continue with the mating was made based on the number of active matings in the breeding colony as a whole and the availability of new replacement ENU-mutagenized G0 males. In a later pilot study to increase male productivity, some ENU-treated males were rotated between two cages each with two C3H/HeH females.
Approximations of the efficacy of ENU treatment were made at regular intervals as the number of heritable phenotypes per G0 male. This is used for guidance in programme management meeting requirements for the various screens. Because of the differences in the screen protocols, like-for-like inter-group comparisons cannot be made for the output of the various screens and are, therefore, refractory to rigorous statistical analysis. Furthermore, the various heritable mutations are usually in various stages of mapping again complicating rigorous analysis.
Case definitions for analysis of breeding and pathology
To analyse the breeding and pathology data for ENU-treated males, mice were categorized as those ‘found dead’ (i.e. sudden unexpected death without premonitory signs), ‘culled sick’, ‘culled regained fertility too early’ or ‘culled did not regain fertility’. The remaining category ‘culled other’ consisted of mice that were culled because they were poor breeders or those that had reached their required offspring target (50 progeny for each G0 male), males that were injured by fighting or were priapistic. The case definition of culled sick included both neoplastic causes and some non-neoplastic causes of morbidity, although this latter category comprised <5% of cases categorized as ‘culled sick’. Cases that were excluded from analysis were males with acute post-treatment toxicity (>20% weight loss was considered as grounds for culling).
Welfare assessments, necropsy and histology
ENU-treated mice were examined every day at 08:00 h and 14:30 h and those which were found ill (clinical signs most often noted were rapid respirations, hunched, piloerect) were culled immediately by an overdose of barbiturate administered intraperitoneally (Pentoject, Animalcare, York, UK). They were weighed, then necropsied and gross pathology findings were recorded. Histological analysis and tissues examined were as previously described.
11
Later, testes and sternebrae were included as additional tissues. Testes were collected and fixed in Bouins solution for 8 h. Bones were decalcified in Formical (Decal Corp, Congers, NY, USA) for 48 h. Tissues were embedded in wax and 4
To compare the incidence of lymphoma in ENU-treated C57BL/6J males with spontaneous rates in untreated males, 28 one-year-old retired breeders from the same stock colonies were euthanized and examined at necropsy for gross tumour burden. The mediastinum including the thymus lobes were fixed and examined histologically.
Tissue array manufacture and immunohistochemistry
Tumours were cored in duplicate (2 × 600
Statistical analysis
Dosage effects on tumour latencies were analysed by the analysis of variance (ANOVA) across multiple groups or by a two-sample
Results
ENU-induced pathology
The higher ENU dosage regimens (3 × 90 mg/kg and 3 × 100 mg/kg) were associated with an increasing percentage of C57BL/6J and BALB/cAnNCrl males that were culled due to illness (‘culled sick’) or were found dead without premonitory signs (Figure 1b). However, in BALB/cAnNCrl males there was an increasing dose-dependent proportion of mice that never regained fertility (Figure 1c). Therefore, this regimen was discontinued prior to initiating the pathology studies. In the earlier mutagenesis projects conducted in the old mouse house, some batches of ENU-mutagenized BALB/cAnNCrl males were culled because of acute 20% weight loss after ENU treatment (Table 1), but this has not recurred under SPF conditions. 10
Morbidity and mortality in the vast majority (>95%) of the G0 mice we studied were due to the development of neoplasia. A few individuals had conditions such as hydronephrosis, bite wounds or priapism (categorized as ‘culled other’ for subsequent analysis). However, even these mice were usually found to have some additional occult tumour burden, usually lung adenomas. By far, the most common tumours in both the BALB/cAnNCrl and C57BL/6J strains were multisystemic lymphomas and lung tumours (see below, Table 2). Small benign lung adenomas were commonly found in ENU-treated C57BL/6J and BALB/cAnNCrl mice of all ages, but by gross and histological assessment these were not sufficiently large or numerous enough to be classified as a primary cause of morbidity or mortality. In contrast, large lung carcinomas were likely to be clinically significant in BALB/cAnNCrl mice, as clinical signs could not be attributed to multisystemic lymphoma (only three of 42 of these mice had multisystemic lymphoma). Some morbidity in C57BL/6J mice was associated with intestinal tumours causing a degree of bowel obstruction, and the incidence of such tumours appeared to increase with ENU dosage. The incidence of intestinal tumours appeared lower in BALB/cAnNCrl males. In contrast, renal neoplasia was a common, clinically non-significant finding in both BALB/cAnNCrl and C57BL/6J males. Fewer than 7% of mice had neoplastic lesions in other tissue origins, for instance there were five myeloid leukaemias and two oligodendrogliomas (data not shown). Other pathological findings included a spectrum of endothelial lesions in the spleen (7–27% in both strains; the most common was angiectasis, then haemangioma, whereas haemangiosarcoma was rare) and liver (biliary) cysts (9–21% incidence in both strains).
Major tumour incidences in G0 mice culled sick
The figure in the body of the table represents percentage incidence and in parentheses are the number of case observed of the total (
*The incidence of lymphoma in 2 × 100 mg/kg dosage regimen is significantly lower in BALB/cAnNCrl males (
†Incidence of lymphoma is not significant at 3 × 90 mg/kg (
A subset of lymphomas from C57BL/6J (
Temporal pattern of tumour development in relation to the breeding programme
The potential impact of tumourigenesis on the breeding programme was evaluated by plotting the date of necropsy or sudden unexpected death after ENU treatment. This latency for the four most common tumours is presented in Figure 2. In cohorts of BALB/cAnNCrl mice, there was an early cluster of lymphomas followed by a later cluster of lung carcinomas. In contrast, in C57BL/6J mice morbidity was almost exclusively due to lymphoma early in the breeding programme. In both strains, the peak of lymphoma incidence was well within the breeding period for these mice (Figure 2) significantly curtailing the number of G0 mice that would produce a target of 50 progeny (see below in this section). The significantly later onset of lung carcinoma in BALB/cAnNCrl mice suggests that these lesions were far less likely to impact on the breeding programme as many more of these mice would have lived long enough to reach their pup limit.
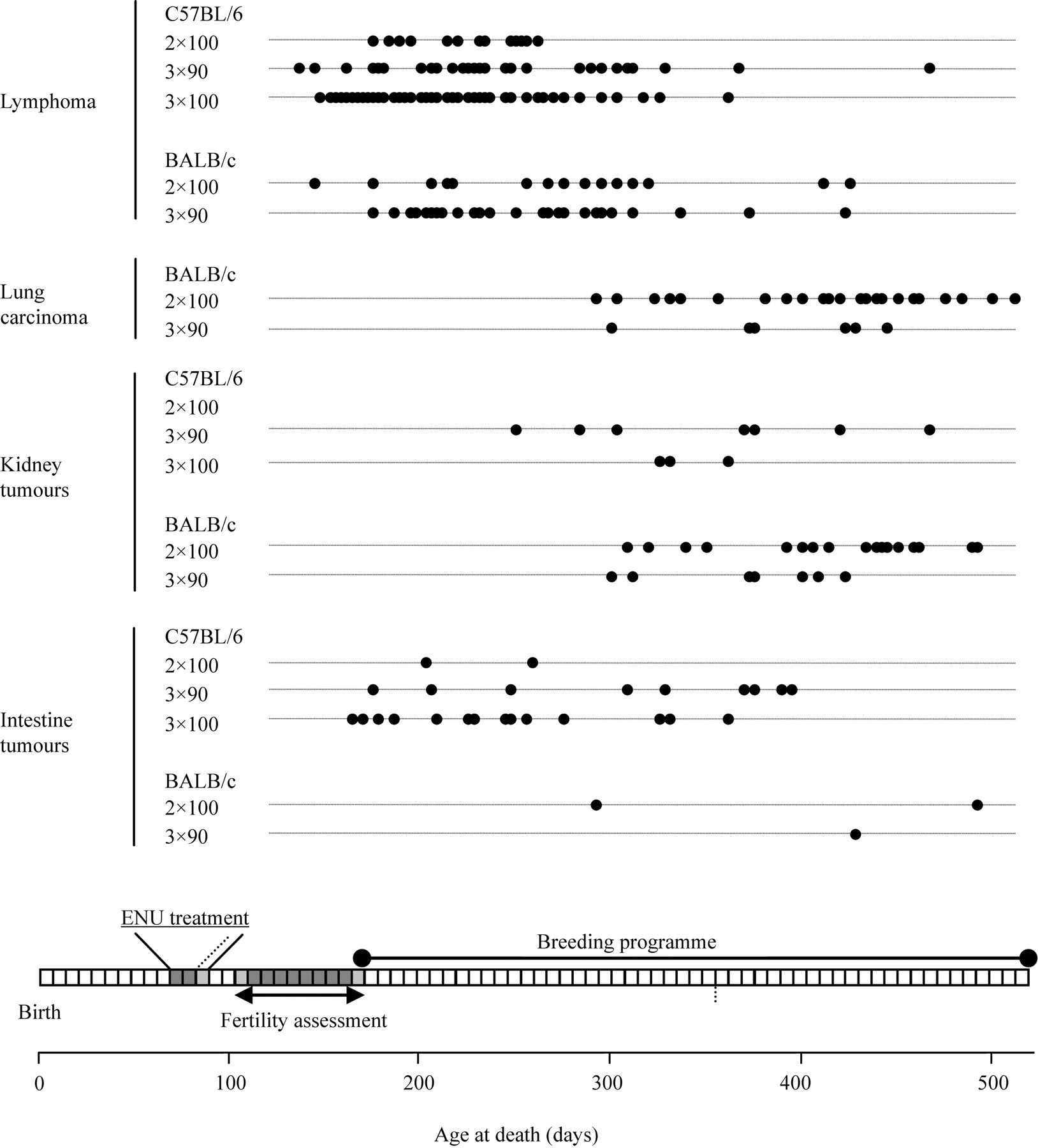
Age-related incidence of the four most common tissue neoplasms found at necropsy. In C57BL/6J mice, morbidity is almost exclusively due to early-onset lymphomas, though some mice develop early-onset intestinal neoplasms and late-onset renal tumours. Early-onset lymphomas are also seen in BALB/cAnNCrl mice, but these mice also develop late-onset lung carcinomas (not seen in C57BL/6J mice) and renal tumours. Note: in this series of C57BL/6J males did not develop lung carcinoma
Although the incidence of lymphoma may be influenced by ENU dosage, the latency was not significantly different between the dosage groups for either strain. For C57BL/6J mice, the mean age for lymphoma development on each dosage regimen were 218 ± 8, 234 ± 11 and 214 ± 7 days (mean ± SEM) for the 2 × 100 (
In contrast to early morbidity associated with high incidence of thymic lymphoma in ENU-treated males, a cohort of 28 retired untreated C57BL/6J breeding males experienced no morbidity or sudden deaths between six months and 12 months and, at necropsy, none had any macroscopic tumour burdens or histological evidence of thymic lymphoma.
The effect of ENU dosage regimen on breeding potential in C57BL/6J males
ENU administration causes a temporary period of infertility in C57BL/6J males and also has the potential to cause permanent infertility. ENU also induces neoplasia that may impact the breeding potential of G0 mice either by premature removal (e.g. infertility, morbidity, sudden death, etc.) or subclinical neoplasia may reduce male productivity in the terminal stages.
Each of these possible contributory factors was assessed (Figure 3) for the C57BL/6J mice. The most dramatic perturbation of fecundity was evident in an apparently dose-dependent increase in the number of G0 males that failed to produce any offspring. This was possibly related to the cumulative dose for each regimen, and in the highest regimen of 3 × 100 mg/kg results in 40% of the males failing to produce any offspring at all (Figure 3a). This was not due to a dose-dependent increase in the time from the end of ENU dosage to first litter, as the time to first litter was comparable for each of the three regimens. These were 121 ± 3.1, 123 ± 2.8 and 126 ± 2.7 days (mean ± SEM) for the dosage regimens 2 × 100 (
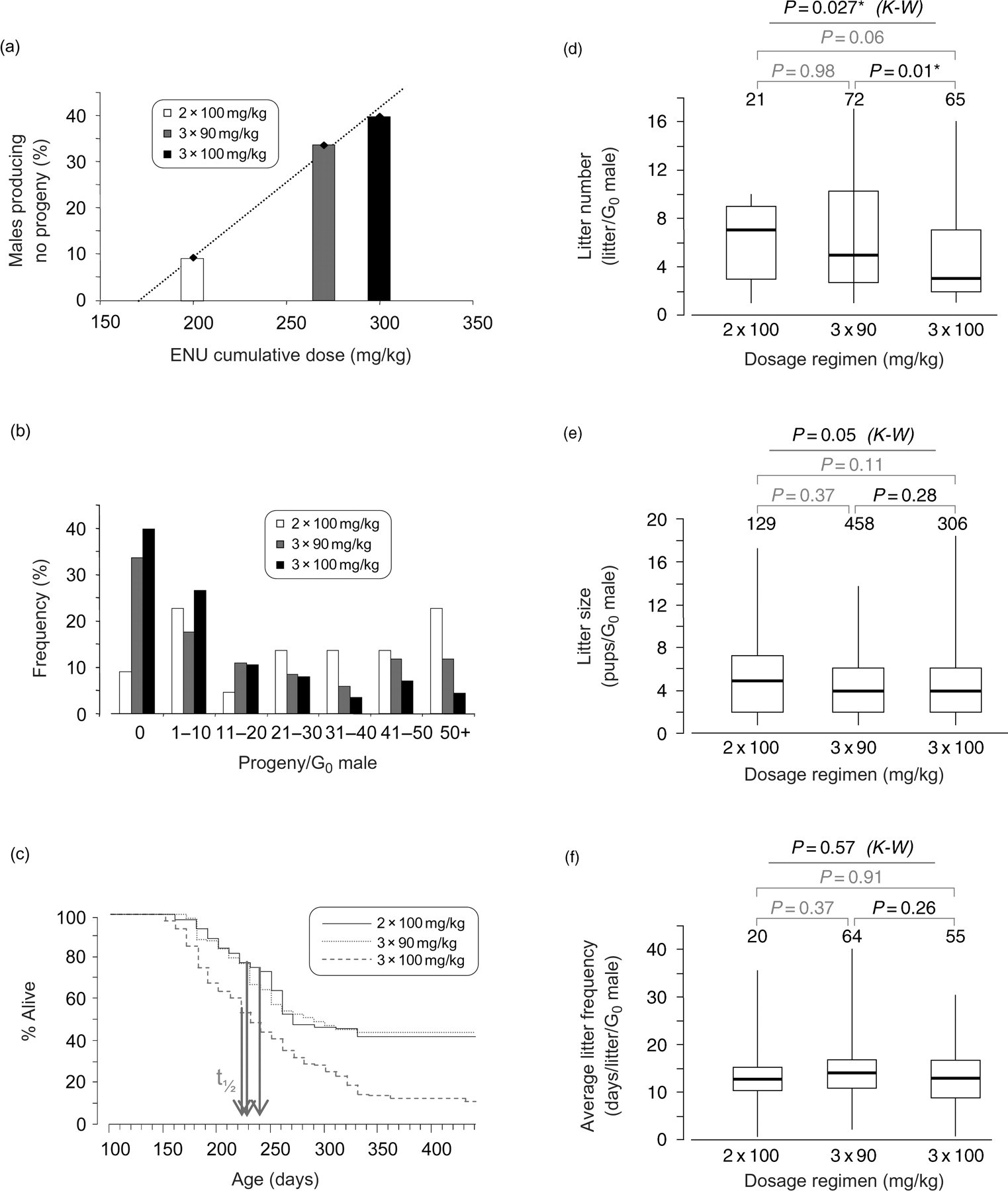
Decreased reproductive potential of ENU-treated C57BL/6J mice. ENU treatment decreases the reproductive potential by three possible mechanisms, impairment of fertility, premature death and a terminal decline in productivity. (a) Increasing dosage regimen, or more precisely increasing cumulative dose, results in more males rendered permanently infertile. (b) Loss of reproductive potential through premature death is manifested in fewer males producing the set target of 50 progeny. (c) Survival analysis demonstrates that in mice treated with 3 × 100 mg/kg loss from the breeding programme is through higher tumour incidence rather than significantly decreased tumour latency;
The most important contributory factor to the reduction in fecundity of males that did breed was a reduction in the number of litters produced (Figure 3d) due to increased incidence of morbidity in the highest ENU dosage cohort. The average litter size (Figure 3e) and frequency of litter production (Figure 3f) were not altered. We assessed the effect of subclinical lymphoma in C57BL/6J on male productivity (assuming that C3H/HeH female fertility was not impaired) and found C57BL/6J males showed the same pattern of siring litters up to the time that they were ‘culled sick’ in all dosage regimens (data not shown). Histological assessment of testes in C57BL/6J males culled sick (
Effect on productivity of providing extra females to fertile ENU-treated C57BL/6J males
In view of the limited breeding lifespan of ENU-treated males in the 3 × 100 mg/kg dosage group, a pilot study was undertaken to determine the effect of providing more female mates. Once males had sired their first litter(s), they were then rotated between this cage and a second cage with two C3H/HeH females. The median number of litters increased from three to seven (
Discussion
We present here the findings from more than five years data on the effects of ENU dosage regimens on the breeding performance and incidence of tumour pathology in BALB/cAnNCrl and C57BL/6J mice mutagenized in the ENU mouse mutagenesis programmes at MRC Harwell. We go on to describe what influence the findings of these studies have had on our management practices.
The management of large-scale mutagenesis programmes, such as those at Harwell, are driven by phenotype demand from the various screens that are undertaken. These are subject to regular review and management practices alter to meet these demands. The value of reviewing breeding and pathology, such as with the current study, is to provide insight into the fate of the G0 males in different dosage regimens and the impact on production of G1 progeny. As it is known that ENU dosing increases the mutation rate linearly in the dose range we used, 13 the optimal dose regimen is the highest possible that does not reduce fertility or welfare of the mice to an unacceptable extent.
Much of the published ENU literature focuses on mechanisms of mutagenesis, mutation rates and specific mouse models generated. 2 There is relatively little information regarding colony management practices and productivity in relation to welfare and pathology. The issue of productivity of males following ENU treatment is of considerable importance in ENU mutagenesis programmes because of the need to generate sufficient numbers of G0 offspring for different phenotypic screens. Others have assessed mortality in the sterile period after exposure to ENU, and regained fertility but primarily with the view to optimize ENU dosage regimen for each strain. 7,8 Initial studies in small cohorts identified 3 × 100 mg/kg as an optimal regimen for both C57BL/6J and BALB/cJ strains with regained fertility rates of 6/10 (60%) and 5/6 (83%) in males for each strain, respectively. 8 Although the BALB/c is a different substrain from the one in our study, the C57BL/6J strain is comparable. In our larger sample size, we can confirm a relationship between cumulative dose and infertility rates suggested in smaller cohorts of these strains. Indeed, a more recent study of comparable population size, but in C3HeB/FeJ mice, 14 shows a similar finding to ours and is also in agreement with previously published rates in smaller cohorts of this inbred strain. 8 We were particularly interested in our study to extend this analysis and to explore the components of lifetime fecundity: regained fertility and lifetime productivity.
One good example of detailed analysis of fecundity in chemically mutagenized males is that by Russell
In a similar way, we have examined litter frequency, size and number of ENU-treated males after they have regained fertility. Using these indices coupled with the results of the pathology analysis, we can also show loss from the breeding programme through morbidity and premature death. The biggest impact of ENU on the fecundity or lifetime breeding potential of G0 males was the adverse effect of cumulative ENU dose, increasing the number of males being rendered permanently infertile. Subsequently, the loss from the breeding programme through morbidity and premature death has a greater effect than a modest decline in productivity during subclinical stages of lymphoma. This is supported by data from those males that did breed indicating that while neither average litter size nor littering frequency were affected, the number of litters per G0 male was significantly decreased in the highest dosage regimen.
The types of ENU-related pathology are as follows. In the earlier mutagenesis experiments, acute post-treatment toxicity with ENU treatment resulting in 20% weight loss was a problem when BALB/cAnNCrl mice were exposed to infection as a result of IVC barrier breakdown in an otherwise low health status mouse. ENU is a stem-cell mutagen and affects haematopoietic stem cells causing immunosuppression. 7 In our low health status conventional mouse colony this appeared to predispose to MHV infection. This complication of acute post-treatment toxicity has been overcome by rederivation of the stocks and performing ENU treatments in SPF conditions in the MLC. 10 The major pathology was tumourigenesis and in our analysis we found that in both BALB/cAnNCrl and C57BL/6J strains, ENU-treated males had a very high incidence of malignant T-lymphoblastic lymphoma. In addition, in BALB/cAnNCrl lung carcinoma was also important. The case for lymphoma being induced by ENU is clear-cut because the very high incidence (>75% of those culled sick) at a relatively young age is considerably greater than background incidence (9–34%) in untreated BALB/cAnNCrl and C57BL/6J mice on two-year ageing studies. 15 Furthermore, our survey of 28 retired untreated C57BL/6J breeding males found no morbidity or sudden death between six months and 12 months and no thymic lymphomas.
The incidence of other less common tumours can be age-related and we are cautious not to over-interpret the differences between strains because early lymphomas abrogate the development of a wider tumour spectrum. Furthermore, it is possible that low health status of the conventional BALB/cAnNCrl mice in the earlier mutagenesis experiments has had some effect on the development of tumours. In both BALB/cAnNCrl and C57BL/6J, the peak of lymphoma incidence within the breeding period produces a significant decrease in productivity. Lymphoma incidence in BALB/cAnNCrl and C57BL/6J mice ‘culled sick’ was similar at the 3 × 90 mg/kg dosage (no data are available for comparison with the 3 × 100 mg/kg dosage), but in the 2 × 100 mg/kg dosage group, the incidence of lymphoma in BALB/cAnNCrl was lower than in C57BL/6J mice (27% vs. 82%,
In recognition of the early onset of lymphoma in ENU-treated males, and as part of the continual monitoring of our practices, we have implemented a major refinement to our current mutagenesis programme. Once G0 males have sired a litter, we now rotate them to a second cage that holds two C3H/HeH females. The rationale is to maximize productivity within a limited breeding lifetime. In C57BL/6J males treated with 3 × 100 mg/kg ENU this is, on average, 88 days between the birth of the first litter(s) (126 ± 3 days) and lymphoma-associated death (214 ± 7 days). This refinement in breeding strategy has increased productivity by more than two-fold.
This retrospective case-control study of breeding and pathology in an ongoing ENU mutagenesis programme does not attempt to address the question of efficiency of mutagenesis for different dosage regimens using standard measures, such as a specific-locus test 12 or the number of molecularly characterized mutations per mutagenized gamete. An assessment can be made after several more years’ work to identify all the mutated genes underlying the phenotypes revealed in the screens. Truly comparative estimates of ENU dosage and mutagenesis efficiency will have to take into account the diverse and changing collection of dominant and recessive phenotypic screens across all Harwell mutagenesis projects. Under these circumstances, the question of cost/benefit analysis cannot be completely resolved because an ENU dosage that might have a high impact on animal welfare (cost) might be very efficient at producing mutant pedigrees (high scientific benefit). The closest we can come to reconcile this problem is to use published data as a guideline for the highest tolerable ENU dose to produce the greatest mutagenesis efficiency. The question then becomes how to maximize the productivity of ENU-injected males because one major outcome of the highest dose will be increased sterility. The measurement and analysis of breeding success and the pathology outcomes then becomes the key to identifying timelines to breed as many offspring in the shortest period.
In conclusion, to our knowledge, this is the first large-scale analysis of deleterious effects of ENU dosage regimens on the breeding performance, pattern and incidence of tumour pathology in an ENU mouse mutagenesis programme. This has provided new insights into lifetime productivity and allowed us to refine our breeding strategy to improve breeding productivity while minimizing the numbers of males that are treated with ENU.
Footnotes
ACKNOWLEDGEMENTS
A P L Smith is the recipient of an MRC Career Development Award.
We thank all the members of the Mutagenesis Team for their contributions to the generation and collation of these data. Specifically, we thank Russell Joynson and Rosie Maunder who care for the mice in the mutagenesis programme. The generation of breeding data for the MLC was facilitated by the work of the Anonymus software team designers and technical support led by Alison Walling. Kate Vowell and Dave Shipston performed necropsies, and Adele Austin, Caroline Barker, Jenny Corrigan and Terry Hacker produced the histology slides for review. Beverley Haynes, Clare Brookes and Diane Walters performed the tissue array manufacture, and Beverley Haynes performed the immunohistochemistry. We thank Martin Fray and the Frozen Embryo and Sperm Archive core facility for rederiving mouse strains.
