Abstract
Imaging is used in human medicine to diagnose disease and monitor treatment efficacy. Computed tomography (CT) positron emission tomography (PET) and magnetic resonance (MR) are applied to animal models of infectious diseases to increase data quality, enhance their relevance to the clinical situation, and to address ethical issues through reduction of numbers and refinement of study designs. The time required for collection of MR and PET-CT scans means that normal breathing produces motion artefacts that can render images unacceptable. We report, for the first time, the use of high frequency jet ventilation (HFJV) for respiratory management during imaging of macaques. HFJV enables continuous gaseous exchange, resulting in cessation of spontaneous breathing motion thus providing a motionless field without the potential stresses induced by repeated breath-hold strategies.
To maximise information obtained and to refine clinical end points we apply serial imaging in our macaque model of tuberculosis (TB). Whilst acceptable images can be captured from sedated, normally breathing animals using computed tomography (CT), the extended time required for image collection using positron emission tomography (PET)-CT or magnetic resonance (MR) means that respiratory movement can impact adversely on the image quality of mobile areas such as the lung. Other research centres testing TB interventions in macaque models have reported the importance of PET-CT in assessing levels of disease and minimise respiratory motion effects during image capture by respiratory gating to exclude motion from reconstructed images and by employing a succession of imposed breath-holds during scan collection, using positive pressure respiratory equipment. 1 Respiratory control through breath-hold at positive end-expiratory pressure risks the accumulation of carbon dioxide (CO2) to potentially dangerous levels unless the breath-hold is released in a timely manner, bringing the potential for considerable physiological stress and even death. To address this welfare issue, we introduced high frequency jet ventilation (HFJV) to minimise respiratory movement. HFJV is established in the clinic and characterised by the delivery of small tidal volumes (1–3 ml/kg) from a high-pressure jet at supra-physiological frequencies (1–10 Hz) followed by passive expiration. The effective elimination of CO2 from the airways, and maintenance of normocapnoea, results in the cessation of spontaneous breathing motion, providing a relatively motionless field during procedures such as CT-guided radio frequency ablation of tumours.2,3
This report describes the establishment of HFJV for macaques and demonstrates its effectiveness for respiratory management during collection of PET-CT images. Macaques obtained from UK breeding colonies were housed in compatible social groups, in accordance with Home Office (UK) and NC3Rs guidelines,4,5 and provided with environmental stimulation and dietary enrichment. Study designs and procedures were approved by the Establishment Animal Welfare and Ethical Review Committee and authorized by the UK Home Office.
Subjects were sedated by intramuscular injection (IM) of 0.15 ml/kg of a mixture of 5 ml ketamine (Anesketin, 100 mg/ml Dechra Veterinary Products Ltd, Shrewsbury, UK) to 2 ml acepromazine (2 mg/ml) Elanco Animal Health, Basingstoke, UK) deepened by an IM injection of 0.02 ml/kg of medetomidine hydrochloride (Sedator, 1 mg/ml, Dechra Veterinary Products, UK). Anaesthetised animals were placed in ventral recumbency and a laryngoscope was used to locate access to the larynx. The area was sprayed with lidocaine (Intubeaze, Dechra Veterinary Products, UK) and a 10 FG flexible catheter (Vygon (UK) Ltd, Swindon, UK) introduced into the trachea and positioned above the carinal bifurcation. Respiration was controlled using an electrically powered, solenoid cycled, automatic, high frequency jet ventilator (Mistral Universal jet ventilator, Acutronic Medical Systems). Using compressed air and oxygen passed through a pressure regulator the ventilator delivers pulses of small tidal volumes (1–3 ml/kg) from a high-pressure jet at supra-physiological frequencies (1–10 Hz) followed by passive expiration. The volume of each pulse was determined by setting the respiratory frequency and driving pressure (DP). End tidal CO2 levels were measured by sampling exhaled gas from a side port attached to the jet ventilation tube after pausing the automated HFJV and delivering several tidal-volume breaths manually at a normal rate. The gas sampling tube was attached to a Datex Capnomac Ultima oxicapnograph (Datex-Engstrom, Helsinki, Finland), providing a real-time display of the capnogram.
HFJV was established with a mycobacterium-free, 10-year-old female cynomolgus macaque (weight 5.71 kg). Following transition from free breathing to controlled HFJV, the frequency was increased by stages from 120 to 300 cycles per minute (CPM). Once HFJV was established, cycle time was demonstrated to be inversely related to the ventilation frequency and the amplitude of chest movement and tidal excursion (Table 1). With small adjustments to DP, HFJV was compatible with normocarbia at frequencies of 120, 150, 200 or 300, and did not adversely affect animal welfare. A plethysmography chamber (Buxco, Wilmington, NC, USA) 4 was used to measure the volume of air in the lungs, to visualise the amplitude of chest movement and evaluate the impact of jetting frequency (Figure 1a). CT scans were collected using a 16-slice Lightspeed CT scanner (General Electric Healthcare, Milwaukee, WI, USA) 1 when free-breathing (Figure 1b) and with respiration managed using HFJV (Figure 1c). A venting frequency of 150–200 CPM, with a DP of 0.3 mBar was sufficient to provide refined images. Whereas images collected during free breathing provided an acceptable level of detail, the impact of diaphragmatic motion at the lung bases caused image blur (Figure 1b), which was not detected on images collected during HFJV (Figure 1c), demonstrating that HFJV improved definition and diagnostic quality. Following cessation of ventilation, spontaneous breathing returned within 2 min.
Example of respiration volume changes over time and CO2 levels in a cynomolgus macaque during jet ventilation.
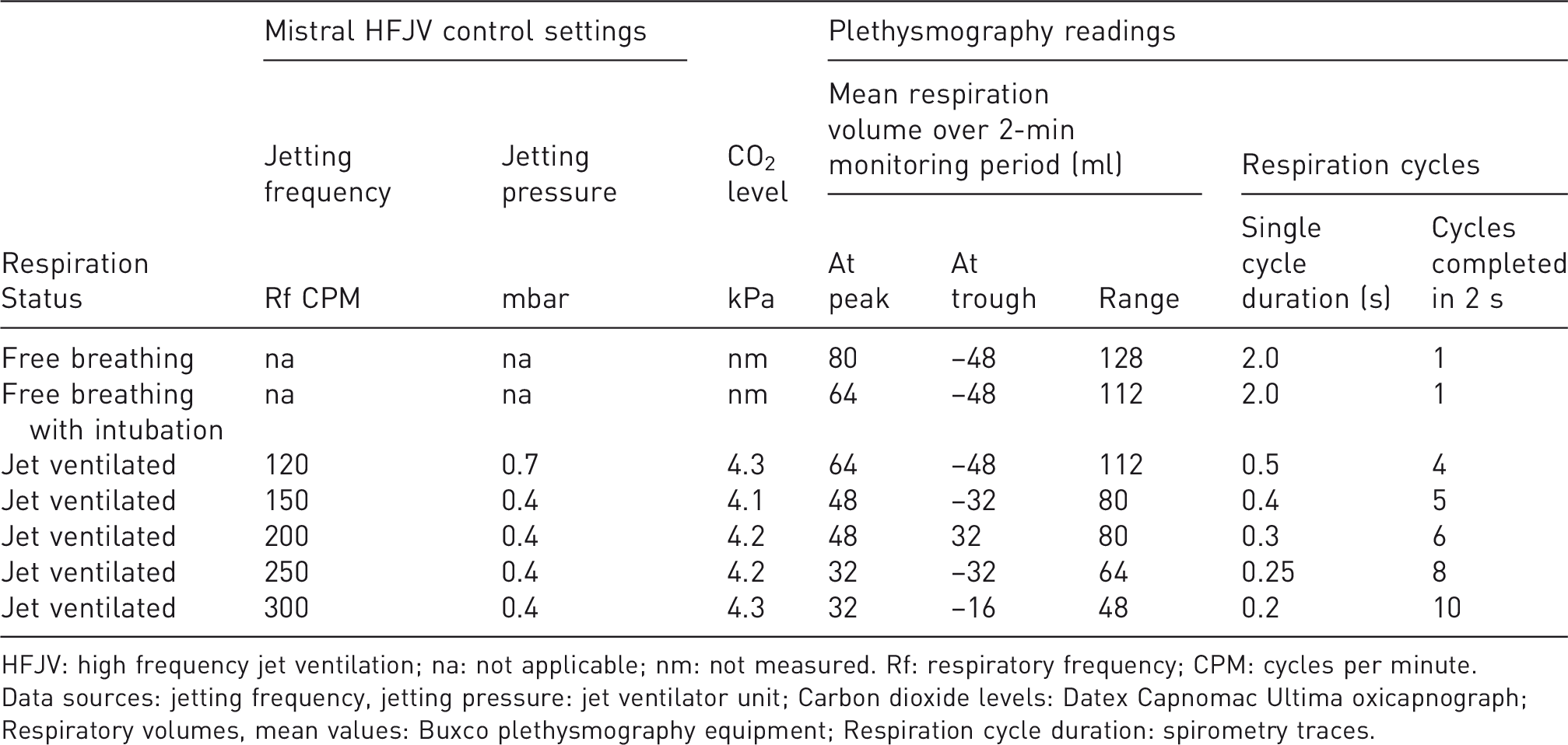
HFJV: high frequency jet ventilation; na: not applicable; nm: not measured. Rf: respiratory frequency; CPM: cycles per minute.
Data sources: jetting frequency, jetting pressure: jet ventilator unit; Carbon dioxide levels: Datex Capnomac Ultima oxicapnograph; Respiratory volumes, mean values: Buxco plethysmography equipment; Respiration cycle duration: spirometry traces.
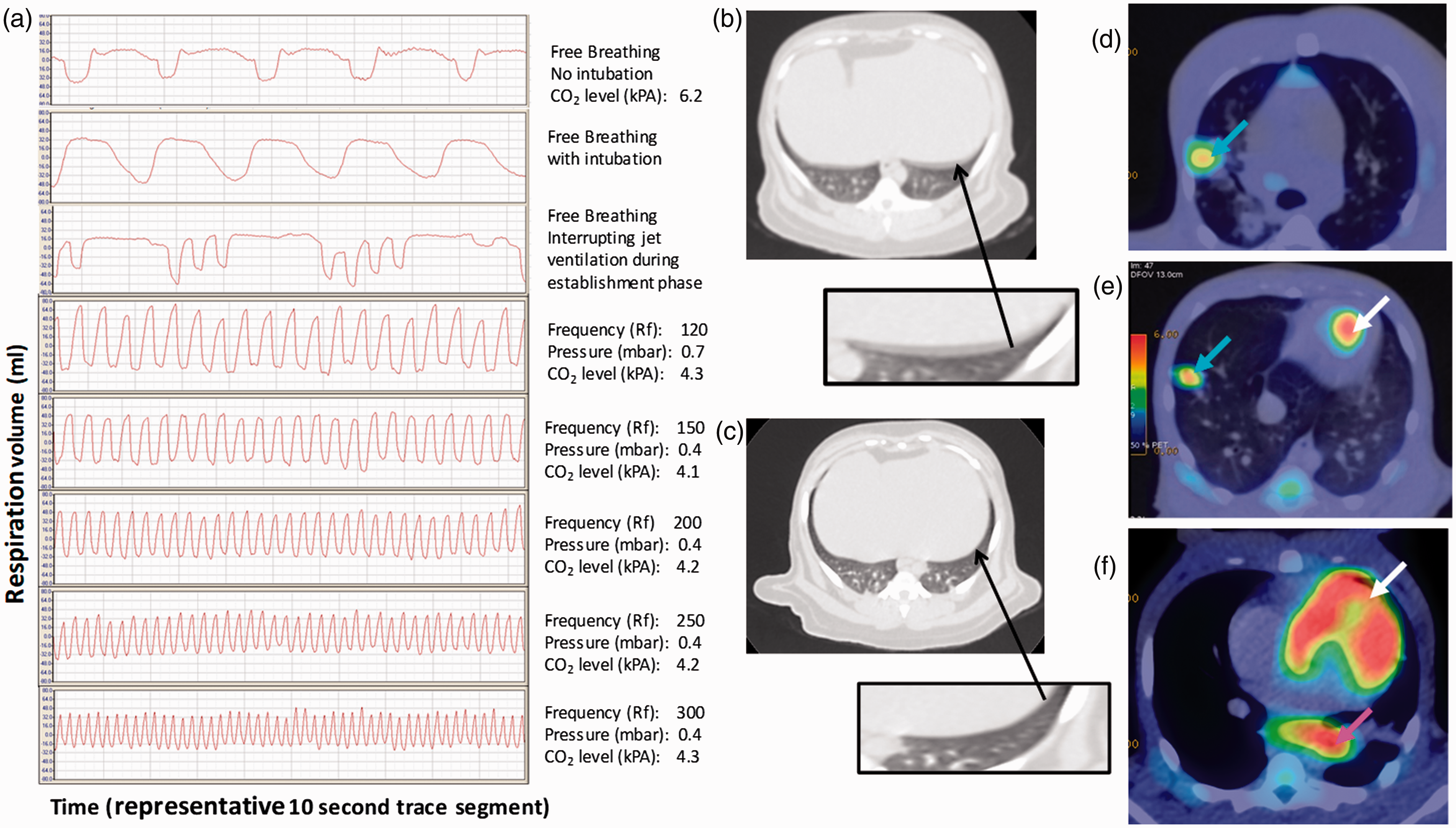
Illustrative spirometry traces (a); CT images from a cynomolgus macaque during free breathing (b) or with HFJV at 150-200 CPM (c); PET-CT images from HFJ-ventilated TB-infected rhesus macaques (56W: d, 65W: e, f) showing FDG avid areas in the lung parenchyma (blue arrow), lung-associated lymph node (pink arrow) and heart (white arrow).
HFJV was applied to minimise respiratory motion to enable collection of PET-CT scans from two, 4-year-old female rhesus macaques (56W: weight 5.40 kg, blood glucose 3.8 mmol/l; 65W: weight 5.36 kg, blood glucose 4.6 mmol/l) 6 weeks after aerosol exposure to M. tuberculosis, using a GE discovery 710 PETCT scanner. Following sedation, fluorodeoxyglucose (18F) (FDG) was administered via a catheter placed into a saphenous vein at a dose of 6 MBq/kg 60 min before PET scan collection. The subject was intubated, placed into an isolator and transferred to the scanner. HFJV was established at a frequency of 150–180 CPM, with a DP of approximately 0.3 mBar (range: 0.2–0.5 mBar) and maintained for up to 90 min for scan collection. Capnography and pulse oximetry monitoring demonstrated that oxygen saturation levels, CO2 levels and heart rate remained within normal ranges. HFJV was well tolerated and minimised spontaneous respiratory motion, enabling the collection of high quality images from both subjects. Images revealed FDG avid areas in lung-associated lymph nodes (Figure 1f) and lung parenchyma (Figure 1d,e) caused by TB-induced disease and FDG avidity in the heart due to metabolic activity associated with cardiac function (Figure 1e,f). Following ventilation, spontaneous respiration resumed within 2 min.
We have demonstrated, for the first time, the use of HFJV as a technique to enhance the clarity of lung images obtained from macaques. Respiratory motion control, a key challenge in thoracic imaging, was achieved whilst maintaining normal gas exchange in a steady state throughout the process, without the use of muscle relaxant or apnoea. Whilst adverse effects of HFJV in the frequency range of 120–300 CPM were not observed in uninfected subjects, scanning protocols were standardized to a frequency of 150–180 CPM to avoid compromising efficiency of gaseous exchange in animals in more advanced stages of tuberculosis. Mechanical ventilation strategies are accompanied by risks associated with intubation, HFJV provides welfare advantages over strategies requiring protracted serial breath-holding, by reducing dangers due to adjuvant drugs by reduction of anaesthetic dosing requirements and avoidance of muscle relaxants; stress-related sequelae by minimisation of physiological distress due to cardiorespiratory challenge, or intensive ventilatory control. Subjects were maintained using HFJV for periods up to 90 min without adverse effects. However, for longer periods of controlled ventilation, experience from human medical use shows that the risk of drying of mucosal surfaces can be mitigated through the use of a moisture exchanger. This suggests that HFJV could offer a potential refinement in other areas of research that require prolonged respiratory management and in the use of other imaging modalities such as MRI.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Department of Health, UK and The Bill and Melinda Gates Foundation. The views expressed in this publication are those of the authors and not necessarily those of the Department of Health.
