Abstract
Volatile agents are widely used to anaesthetise laboratory non-human primates as they allow a rapid induction and recovery as well as an easy adjustment of the anaesthesia plan. Desflurane is currently the volatile agent with the lowest solubility in blood, and hence enables the most rapid onset of anaesthesia and most rapid recovery. This study aimed to investigate the suitability of desflurane for maintenance of general anaesthesia in rhesus macaques undergoing elective experimental neurosurgery. Fourteen primates (five males and nine females) were sedated with ketamine (10 mg kg−1) and anaesthesia was induced with propofol (usually 8 mg kg−1 IV). Anaesthesia was maintained with desflurane (5.9 ± 0.8 %) and alfentanil (0.2–0.5 µg kg−1 min−1 IV). Animals were mechanically ventilated. Meloxicam (0.3 mg kg−1) and methylprednisolone infusion (5.4 mg kg−1 h−1) were also administered. All the primates were successfully anaesthetised and no severe complications related to the procedure or the anaesthesia regimen occurred. No major differences in physiological parameters and recovery times between the male and female groups were found. Emergence from anaesthesia was rapid (male 5.2 ± 2.4 min; female 4.1 ± 1.7 min) but its quality was assessed as equivalent to two other volatile anaesthetics, isoflurane and sevoflurane. These had previously been assessed for neuroanaesthesia in rhesus macaques. In conclusion, this study demonstrated that desflurane was suitable for maintenance of general anaesthesia for elective experimental neurosurgical procedures in rhesus macaque. However the vasodilatory action of the desflurane may limit its use in cases of severe intracranial hypertension or systemic hypotension.
Introduction
Non-human primates provide a valuable model in neuroscience, 1 and prolonged anaesthesia to perform either invasive procedures (e.g. craniotomy chambers, headpiece) or non-invasive procedures (e.g. magnetic resonance imaging) may be required. Volatile anaesthetic agents are widely used for this purpose as they allow a rapid induction and recovery, as well as easy adjustment of the anaesthesia plan. 2 In cases of neurosurgery, cardiovascular stability and the quality of recovery are considered as critical factors to decrease the risk of intra- or post-operative complications such as a rise in intra-cranial pressure or subdural haemorrhage.3–5 Desflurane is currently the volatile anaesthetic agent with the lowest solubility in blood and hence has the most rapid onset of anaesthesia and the most rapid recovery. 2 In previous studies in dogs and people, desflurane was considered suitable for maintenance of general anaesthesia and rapidity of recovery was considered a potential advantage.6,7 The use of isoflurane and sevoflurane for experimental neurosurgery in macaque has been described previously. 5 This previous study reported use of sevoflurane was associated with a shorter recovery time but emergence quality was similar between the two anaesthetic agents. This study aimed to investigate the suitability of desflurane for maintenance of general anaesthesia in rhesus macaque undergoing elective experimental neurosurgery, and to assess potential benefits during anaesthetic emergence when compared to isoflurane and sevoflurane.
Material and methods
Ethical statement
The use of these animals for research was authorised by the Home Office (United Kingdom authority) (PPL60/4560; PPL70/7976; PPL60/4041; PPL60/4095), and the Newcastle University Animal Welfare and Ethical Review Body.
Animals and husbandry
The animals were purpose bred for research in the UK, and were supplied from the Centre for Macaques (Medical Research Council, Porton Down, Salisbury, UK) to Newcastle University. Animals were housed in a Home Office licensed facility and complied with the Animal Scientific Procedure Act 1986 and the European Directive 2010/63/EU. Animals were free of the following infectious agents: SIV, SRV(D), STLV-1, tuberculosis, Campylobacter spp., Salmonella spp., Shigella spp. Animals were pair housed in indoor pens with a solid floor and windows allowing a view of the other pens and corridors. A minimum floor area of 4.40 m2 was provided for each pair. A smaller pen with a squeeze-back system was located between each housing pen. Enrichment devices and substrate for foraging were provided. Animals were maintained on a light:dark cycle of 12 h:12 h, at a temperature of 22 ℃ with 15 air changes per hour, a relative humidity of 24% and with natural lighting via sky-lights. Primates were fed with adapted old world primate diet (Special Diets Service, Witham, UK) and received tap water ad libitum during the study. Forage mix was provided daily to all animals (LBS Biotechnology, UK). The primates had not been sedated or anaesthetised for at least two weeks prior to the start of the study.
General anaesthesia maintenance with desflurane
Nature of neurosurgical procedures.
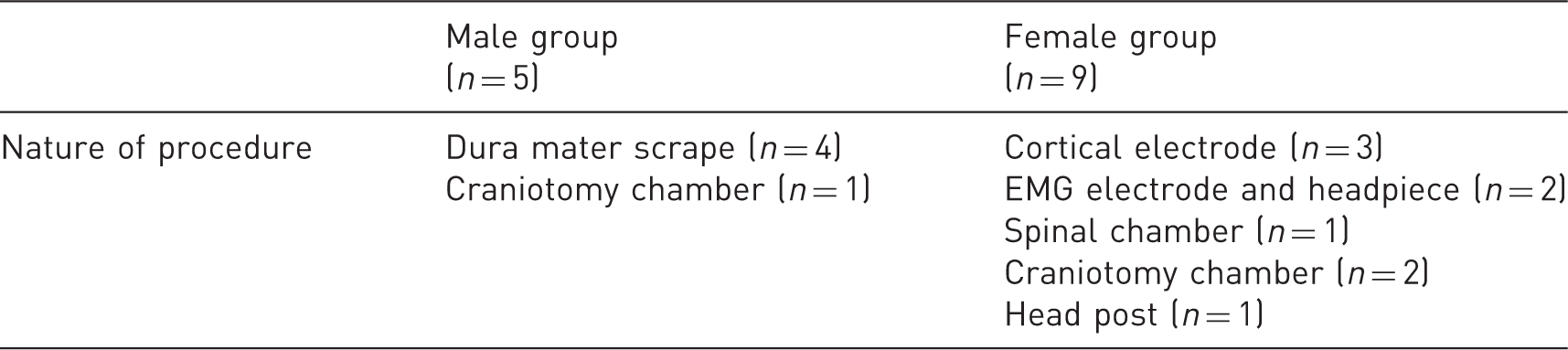
The number between brackets is the number of procedures performed. EMG: electromyography.
American Society of Anesthesiologists (ASA) physical status classification system.

From the American Society of Anesthesiologists website (https://www.asahq.org/resources/clinical-information/asa-physical-status-classification-system; consulted on 2 August 2017).
Anaesthesia procedure
Food was withheld for a minimum period of 12 h prior to surgery but primates still had access to water ad libitum. Animals were sedated in the holding unit with 10 mg kg−1 IM of ketamine hydrochloride (Ketaset 100 mg/ml solution for injection, Zoetis, London, UK). Once a level of sedation allowing safe handling was attained, animals were transported to an anaesthesia preparation room. Animals received 100% oxygen delivered at 4 l min−1 via a facemask for 5 min and a dose of 0.3 mg kg−1 of meloxicam (Metacam 5 mg/ml solution for injection for dogs and cats, Boehringer Ingelheim, Bracknell, UK) was administered subcutaneously. One saphenous vein was catheterised and anaesthesia was induced with slow intravenous administration of propofol (Fresenius Propoven 1% Emulsion for injection or infusion, Fresenius Kabi Ltd, Runcorn, UK) to effect (usually 8 mg kg−1) to allow endotracheal intubation. After intubation, using a cuffed tube, the animal was connected to a circle breathing system (Clear-Flo™ circle breathing systems with soda lime, Intersurgical Ltd, Wokingham, UK) and mechanically ventilated (Merlin Small Animal Ventilator, Vetronic Services Ltd, Abbotskerswell, UK) with the respiratory frequency adjusted to maintain an expired CO2 consistent with the surgical procedure (e.g. craniotomy required an end-tidal CO2 (EtCO2) below 35 mmHg). Anaesthesia was maintained with desflurane (Desflurane 100% w/w inhalation vapour, liquid, Baxter Healthcare Ltd, Thetford, UK) mixed with 100% oxygen as the carrier gas. The vaporiser (Tec 6, Datex-Ohmeda, Hatfield, UK) settings were adjusted to maintain an adequate surgical plane of anaesthesia throughout the surgical procedure. Initially a fresh gas flow of 4 l min−1 was maintained, and the flow decreased after 10 min to between 0.3 and 1 l min−1. Intravenous infusion of alfentanil (Alfentanil 500 µg/ml solution for injection, Hameln Pharmaceuticals, Gloucester, UK) at 0.2–0.5 µg kg−1 min−1 and methylprednisolone (Solu-Medrone 500 mg, Pfizer Ltd, Sandwich, UK) at 5.4 mg kg−1 h−1 were administered during the surgical procedures.
Anaesthesia monitoring
Physiological parameters were constantly monitored during the surgery and recorded every 10 min. A Vitalogik 4500 monitoring system (Charter-Kontron Ltd, Milton Keynes, UK) was used to measure vital signs. Heart rate (HR) and oxygen saturation (SpO2) were assessed using an absorbance pulse oximeter probe placed on a finger. Blood pressure was assessed using an oscillometric method with a blood pressure cuff (Critikon Dura-Cuf, GE Healthcare, Hatfield, UK) appropriate for the size of the animal. The cuff was placed to record the blood pressure from the brachial artery. The respiration rate (RR) and EtCO2 were measured using a side stream gas analyser (sampling rate: 50 ml min−1), integrated into the electronic monitoring system. The animal's core and skin temperature were also monitored and maintained using a homeothermic heating pad set at 38 ℃ (Homeothermic Blanket system, Harvard Apparatus, Cambridge, UK) and a forced-air warming blanket set between 32 and 42 ℃ (Bair hugger model 505, Augustine Medical, Eden Prairie, MN, USA).
Recovery procedure
At the end of the surgical procedure, alfentanil and methylprednisolone infusions were terminated and the primate was placed in lateral recumbency. Buprenorphine (Vetergesic multidose 0.3 mg/ml solution for injection for dogs, cats and horses, Ceva Animal Health Ltd, Amersham, UK) (20 µg kg−1 IV) was administered to reverse the respiratory depression caused by the alfentanil and to provide prolonged post-operative analgesia. The concentration of gas administered was set at 8%, and the assist-mode of the ventilator was used during the return of spontaneous ventilation. Once spontaneous respiration was observed, the endotracheal tube was connected to a Bain system (Mapleson D deluxe Bain coaxial breathing system with 2L bag 1.6 m, Intersurgical Ltd, Wokingham, UK), assisted ventilation withdrawn and the anaesthesia vaporiser turned off. Once the concentration of expired volatile agent was close to zero, the animal was disconnected from the breathing system and a short period allowed to elapse (2–3 min) to ensure oxygen saturation remained above 85% when breathing room air. The endotracheal tube was removed once pharyngeal and laryngeal reflexes began to return and the primate was placed in a recovery cage once it was able to maintain sternal recumbency. The animal was kept under continuous observation and transferred to the animal holding unit when it was judged stable by the attending veterinarian.
Emergence quality assessment
Recovery clinical scoring scheme.
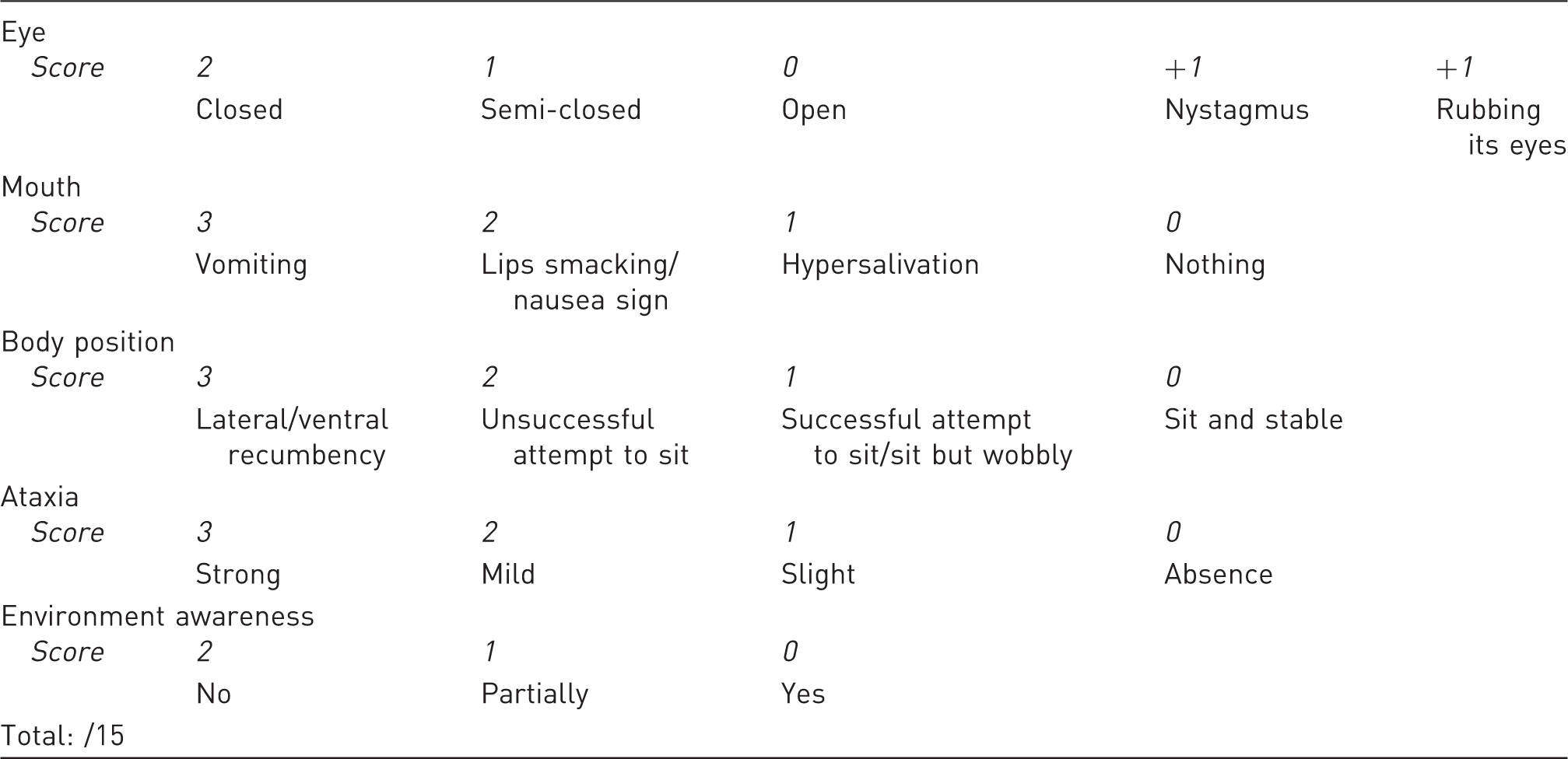
System based on 15 points with the principle that a higher score indicates a poorer recovery.
Statistical analysis
The Shapiro–Wilk test was used to assess the normal distribution of the data. A comparison of cardiovascular parameters between female and male animals was performed using Student's t-test. The Friedman test was used to compare the recovery scores between each of the three time points in the three treatment groups. The Kruskall–Wallis test was used to assess the treatment effect at each time point.
Statistical significance was set at 5%. The statistical software R (vers. 3.3.2, The R Foundation for Statistical Computing) was used to perform the analyses.
Results
Maintenance of general anaesthesia
Anaesthesia data for desflurane anaesthesia.
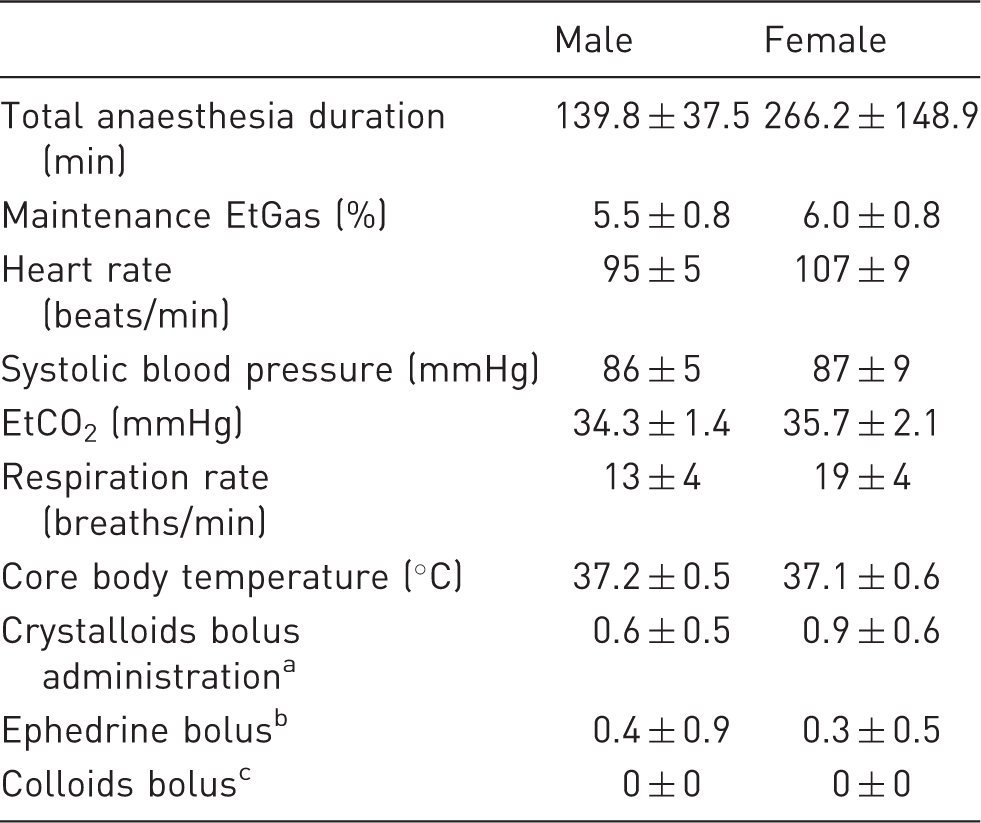
The data were collected over the maintenance period of anaesthesia and are reported as mean ± 1 SD.
Crystalloids bolus dose of 10 ml kg−1 over 10 min.
Ephedrine (ephedrine hydrochloride injection 30 mg in 1 ml for slow intravenous injection, Martindale Pharmaceuticals, Romford, UK) bolus of 0.1 mg kg−1 intravenously.
Colloids bolus (Isoplex 4% w/v solution for infusion, Beacon Pharmaceutical, Tunbridge Wells, UK) dose of 3 ml kg−1.
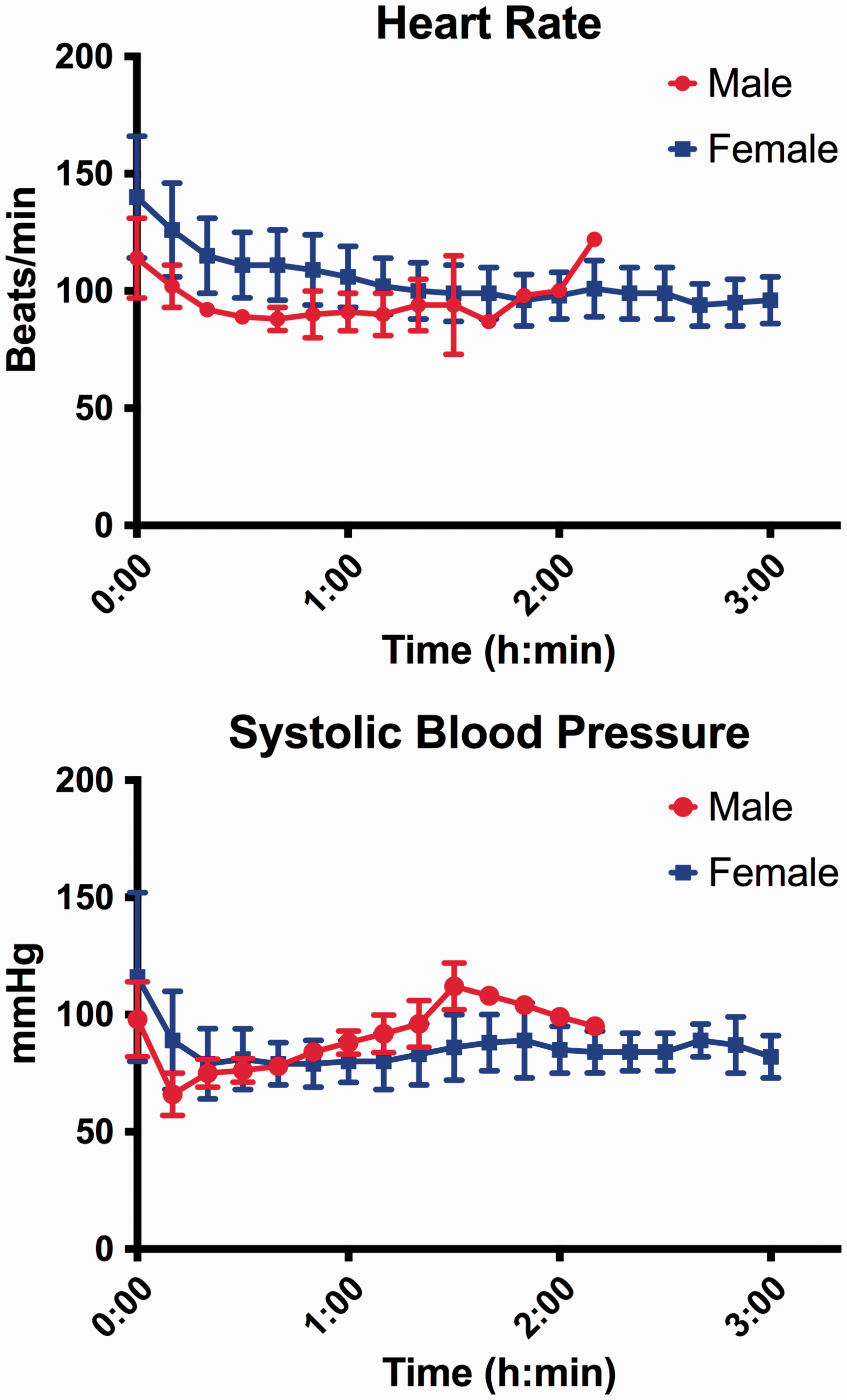
Cardiovascular parameters over the time. The data are presented as the mean ± 1 SD. The plots at the right extremity of the graph represent data from only one animal so no SD is shown.
Emergence time
The time between the withdrawal of desflurane and extubation of the animal was not significantly different between males and females (male 5.2 ± 2.4 min; female 4.1 ± 1.7 min; p = 0.401). The total anaesthesia emergence time (time recorded between the discontinuation of desflurane administration and the animal being able to sit) was slightly longer in the male group (male 13.9 ± 9.6 min; female 5.9 ± 2.7 min; p = 0.135).
Emergence quality
The Friedman test showed a significant improvement in the recovery quality over time in all treatment groups (ISO, p = 0.002; SEVO, p = 0.002; DESF, p = 0.006). However, no difference was found between the three treatment groups at any time point (Figure 2) (p > 0.1). The variability between the observer scores was also compared and no difference was found (p > 0.1).
Recovery quality results over the time. The results are presented as the mean ± 1 SD of all the scores for each treatment group at each time point. Treatment groups were compared at each time point with the Kruskall–Wallis test. The following results are expressed as mean ± 1SD. t0, ISO: 5.6 ± 1.0, SEVO: 6.1 ± 3.0, DESF: 4.5 ± 2.2 p = 0.470; t+5, ISO: 3.5 ± 2.3, SEVO: 4.1 ± 2.1, DESF: 2.8 ± 2.2 p = 0.611; t+12, ISO: 1.8 ± 2.1, SEVO: 1.8 ± 1.7, DESF: 1.3 ± 1.6 p = 0.657). A higher score indicates a poorer quality of recovery.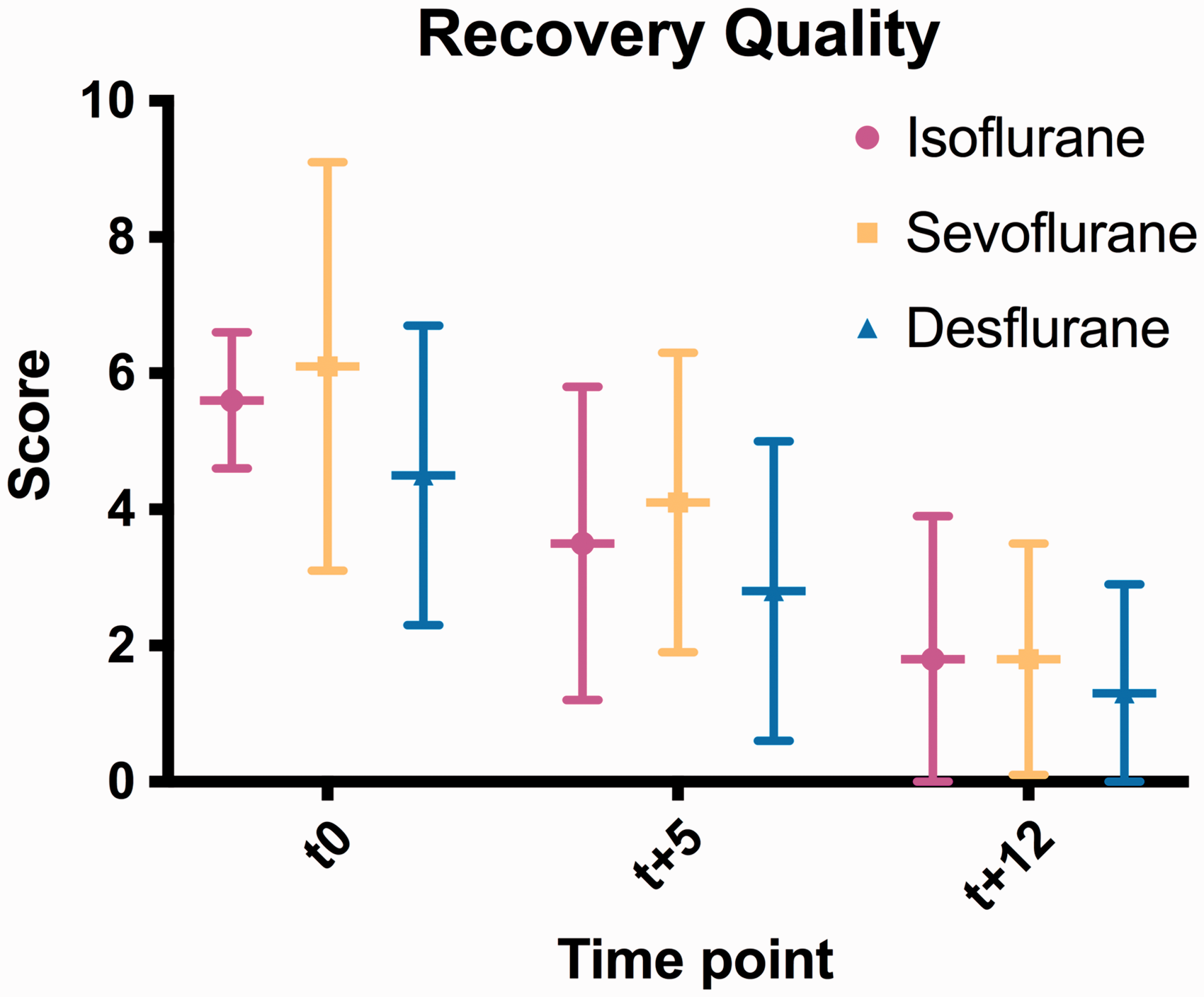
Discussion
In the present study, the administration of desflurane in combination with an opioid was suitable for maintenance of a surgical plane of general anaesthesia for an elective, experimental neurosurgical procedure. Desflurane recovery quality was similar to isoflurane and sevoflurane, two other volatile anaesthetics that have been previously described as suitable for maintenance of general anaesthesia in rhesus macaques. 5 The inspired gas concentration to maintain general anaesthesia in the present study was close to the minimal alveolar concentration (MAC) of desflurane described in adult humans (6%). 2 Cardiovascular stability during neurosurgery is considered essential to maintain the auto-regulation of cerebral blood flow and to decrease the risk of complications (e.g. cerebral ischemia, haemorrhage). 3 In the present study, cardio-vascular parameters (heart rate and systolic blood pressure) decreased at the beginning of desflurane administration and then stabilised over time. This is a similar trend to that observed during administration of isoflurane or sevoflurane to animals undergoing similar procedures in our laboratory. 5 The influence of volatile anaesthetics on systemic blood pressure is a well-recognised phenomenon and is usually proportional to the inspired concentration of anaesthetic. 2 Moderate hypotension was observed in most animals in this study but was successfully managed by the administration of fluids or a vasopressor agent (ephedrine). 9 The concomitant administration of an opioid decreased the concentration of desflurane required to maintain a surgical plane of general anaesthesia and this likely avoided more severe depression of the cardio-vascular system.10–12 Previous publications demonstrated that in pigs and children, administration of 1 MAC desflurane in hypocapnia produced greater vasodilatation than isoflurane and sevoflurane. This could result in a moderate increase in intracranial pressure but also a decrease in the systemic mean arterial blood pressure (MAP). This risk of hypotension requires close monitoring of systemic blood pressure which is important to reduce the risk of a fall in cerebral perfusion pressure.13,14 This suggests that desflurane would not be the anaesthetic of choice in a clinical case associated with severe intracranial hypertension.
Quality and speed of recovery are important considerations in general anaesthesia for neurosurgery. A rapid and smooth recovery allows preliminary neurological assessment to be performed and any supportive treatment to be administered if necessary. 15 Desflurane should result in the fastest onset of anaesthesia and recovery in comparison to isoflurane and sevoflurane, 2 and hence it should enable neurological assessments to be performed at the earliest stage post-operatively and so allow decisions on supportive treatments to be adjusted if necessary. The recovery times in this study were shorter than those with isoflurane, but were similar to those reported for sevoflurane anaesthesia in rhesus macaques undergoing similar procedures. 5 The recovery times in this study tended to be longer in the male group which could potentially be explained by the physiological differences between males and females. 16
Any excitement, delirium or vomiting during anaesthesia recovery after neurosurgery could increase intracranial pressure and result in the development of cerebral oedema or haemorrhage. 17 In the present study, recovery from desflurane anaesthesia was smooth in all animals with no adverse effects observed. Although desflurane tended to have a lower score (indicating a smoother recovery) at each time point, no significant difference in the recovery quality was found in comparison with isoflurane and sevoflurane. The system used to perform this comparison was developed in a previous study and enabled discrimination between poor and good anaesthesia recovery in Rhesus macaques. 8 This finding is similar to reports in other species including dogs and people.6,7,18 A rapid recovery can be associated with safety issues for animal care staff, such as being scratched, bitten or coming into contact with a primate's secretions The use of small doses of sedative agents (e.g. benzodiazepines) during the emergence period has been suggested. 19 However, providing all staff involved are aware that emergence is likely to be rapid, then the management of the animal can be adjusted to avoid these risks. We strongly recommend the use of a mobile recovery cage that allows immediate transfer of the animal from the operating table into a secure enclosure. With any anaesthetic regimen, the use of personal protective equipment (e.g. gloves, face mask) is recommended to avoid exposure to potential biohazards.
In conclusion, this study demonstrated desflurane was suitable for the maintenance of general anaesthesia for elective experimental neurosurgical procedures in Rhesus macaques. However, the greater vasodilatory action of desflurane may limit its use in cases of severe intracranial hypertension or systemic hypotension. Desflurane was associated with rapid and smooth recovery from anaesthesia but with a quality similar to that seen with isoflurane and sevoflurane.
Footnotes
Acknowledgements
The authors would like to thank Mrs Denise Reed, Ms Jennifer Murray, Mr Ashley Waddle, Ms Stevie O'Keefe, Ms Carrie Todd, Mr Joseph Middleton and Ms Adele Kitching for their technical support and help during this work.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
