Abstract
For investigating the effects of mechanical ventilation on the respiratory system, experiments in small mammal models are used. However, conventional ventilators for small animals are usually limited to a specific ventilation mode, and in particular to passive expiration. Here, we present a computer-controlled research ventilator for small animals which provides conventional mechanical ventilation as well as new type ventilation profiles. Typical profiles of conventional mechanical ventilation, as well as flow-controlled expiration and sinusoidal ventilation profiles can be generated with our new ventilator. Flow control during expiration reduced the expiratory peak flow rate by 73% and increased the mean airway pressure by up to 1 mbar compared with conventional ventilation without increasing peak pressure and end-expiratory pressure. Our new ventilator for small animals allows for the application of various ventilation profiles. We could analyse the effects of applying conventional ventilation profiles, pressure-controlled ventilation and volume-controlled ventilation, as well as the novel flow-controlled ventilation profile. This new approach enables studying the mechanical properties of the respiratory system with an increased freedom for choosing independent ventilation parameters.
Introduction
Mechanical ventilation is a key life-saving therapy in modern medicine, being applied during general anaesthesia and in intensive care therapy. Small animals such as rats or guinea pigs are generally used in experiments to study the behaviour of the respiratory system under mechanical ventilation. Specifically, for investigating ventilation-induced lung injury (VILI), models in rats are internationally recognised and well established.1,2 Aiming at studying the effects of ventilation modes, and in particular the effects of our new concept of lung-protective mechanical ventilation, i.e. ‘flow-controlled expiration’ (FLEX) in rats, 3 we have developed a mechanical ventilator for small animals allowing the application of freely definable ventilation profiles for inspiration and expiration. In a proof of principle investigation, rats were ventilated with this machine with various different combinations of inspiratory and expiratory ventilation profiles, and the respiratory variables were recorded.
In the present paper, we provide a detailed description of the design and operation options of our new type mechanical ventilator for small animals.
Animals, materials and methods
Configuration of the ventilator
Our small animal ventilator is based on linear motor-driven (PS-XY, LinMot, Spreitenbach, Switzerland) piston pumps for generating inspiratory and expiratory volume shifts. The linear motor is controlled by a position feedback controller (E1100, LinMot, Spreitenbach, Switzerland).
Four piston pumps (two for inspiration and two for expiration) are operated synchronously. For generating tidal volumes of up to 4.5 ml, two pistons are uncoupled. For generating tidal volumes from 4.5 ml to 9 ml these two pumps are coupled and run in parallel to the respective other inspiration or expiration pump. The position of the pistons is continuously measured using a linear potentiometer (Y-Fader, 10 kΩ, Conrad, Hirschau, Germany) as a surrogate for the current volume status during inspiration and expiration.
In conventional ventilators, passive expiration can be realised by simply opening the expiration valve against atmosphere or a positive end-expiratory pressure (PEEP) generating device. By contrast, the active modulation of the expiration requires enhanced control and safety mechanisms; first, in order to generate the required expiration profile, and second, in order to prevent successive accumulation of retained volume (dynamic hyperinflation) and progressive induction of negative airway pressure by continuous over-expiration. Therefore, three airway switches, controlled via solenoid valves (Isliker Magnete, Andelfingen, Switzerland), were synchronised to separate inspiratory airways from expiratory airways and to maintain the required level of PEEP in the airways. The inspiration switch opens and closes the inspiratory airways, the expiration switch opens and closes the expiratory airways, and the PEEP switch connects the expiratory airways to the PEEP source. During inspiration, the expiratory switch and the PEEP switch disconnect the airways from the expiratory pathway. The inspiration switch connects the inspiratory pumps to the inspiratory airways and disconnects the inspiratory airways from the gas refill source. Then gas is insufflated to the ventilated lungs by forward piston movement. At the beginning of the expiration, the inspiration switch disconnects the inspiratory pumps from the inspiratory airways and connects them to the gas refill and the PEEP source. At the same time, the expiratory switch connects the airways to the expiratory pumps (Figure 1). Then gas is withdrawn from the lungs in a controlled fashion by backward piston movement.
Schematic representation of the ventilator for small animals with the respective elements and connections.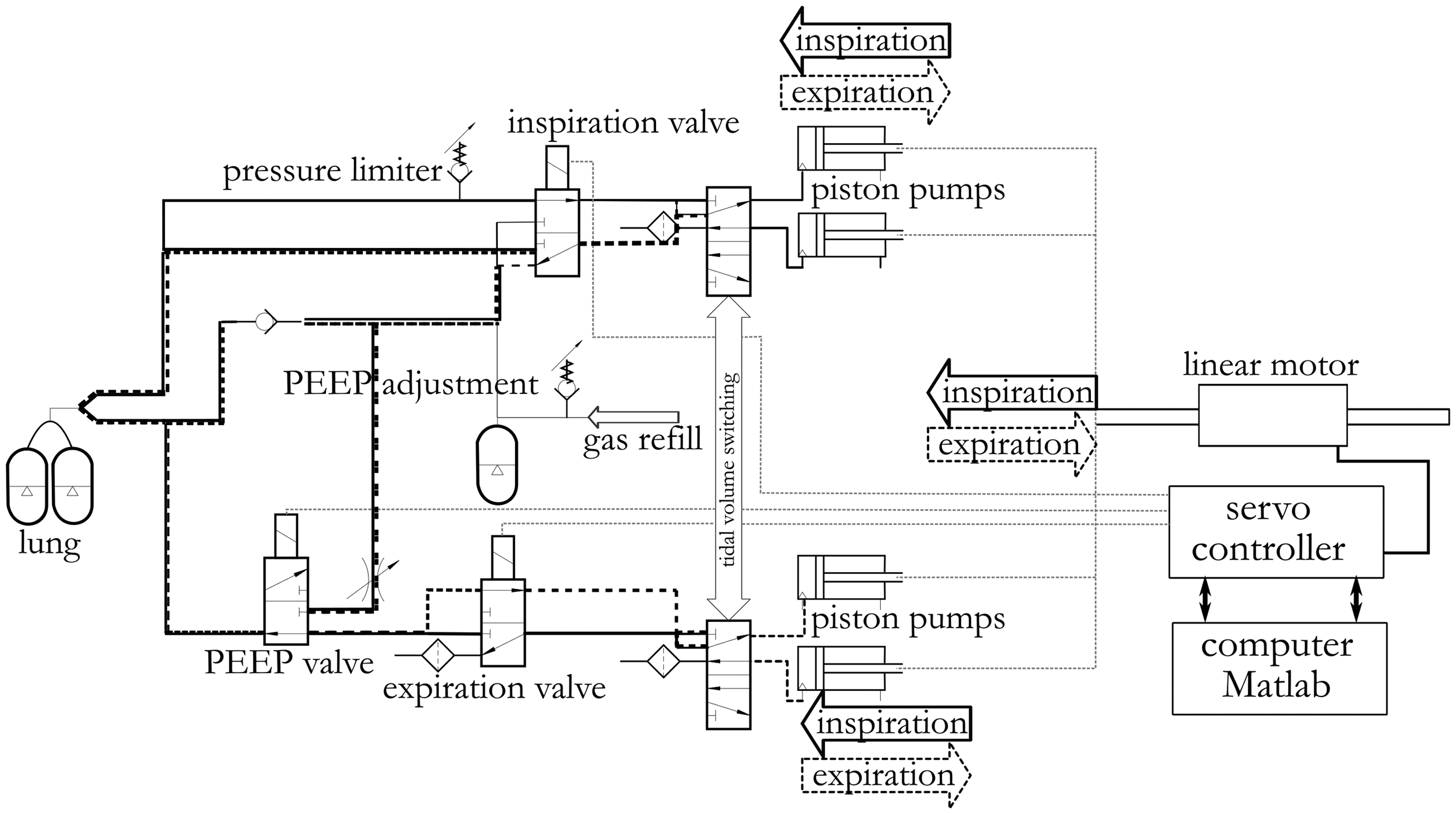
The PEEP level is generated by a pressurised volume reservoir and a PEEP valve that is always connected to the airways to guarantee PEEP as the minimal pressure. At the final phase of expiration, the expiration switch and the PEEP switch connect the expiratory airways to a throttle, dimensioned to allow for deflation of potentially accumulated end-expiratory volume and thus to prevent hyperinflation.
The maximal inspiration pressure can be adjusted by a pressure limiter. Valves and pneumatic lines were designed with the smallest possible internal volume. The external port for fresh gas is accessible, so that the animal may be ventilated with any dedicated gas mixture. All components of the ventilator were mounted onto a rigid 280 × 480 mm polyvinyl chloride table.
Software
To control the motor movement, the motor’s controller was connected to a laptop computer via an RS232 interface (LinRS protocol, LinMot). The controlling and timing were integrated into a self-programmed MatLab (v2017, The MathWorks, Natick, MA) software package. This software package allows any user-defined ventilation profile, stored as volume–time curves in the controller, to be applied. So far, linear (analogous to volume-controlled inspiration and FLEX expiration), sinusoidal and exponential (analogous to pressure-controlled inspiration and conventional expiration) ventilation profiles have been realised. Characteristic ventilation settings such as respiratory rate, tidal volume and inspiration and expiration times are also freely adjustable.
The ventilator-controlling software allows the combination of any inspiratory curve with any expiratory curve (currently three inspiration and three expiration curve profiles are available). Examples of the combinations used for this study are represented in Figure 2. The ventilator can provide respiratory rates up to 120 breaths per minute.
Flow and volume time courses of the presented ventilation profiles.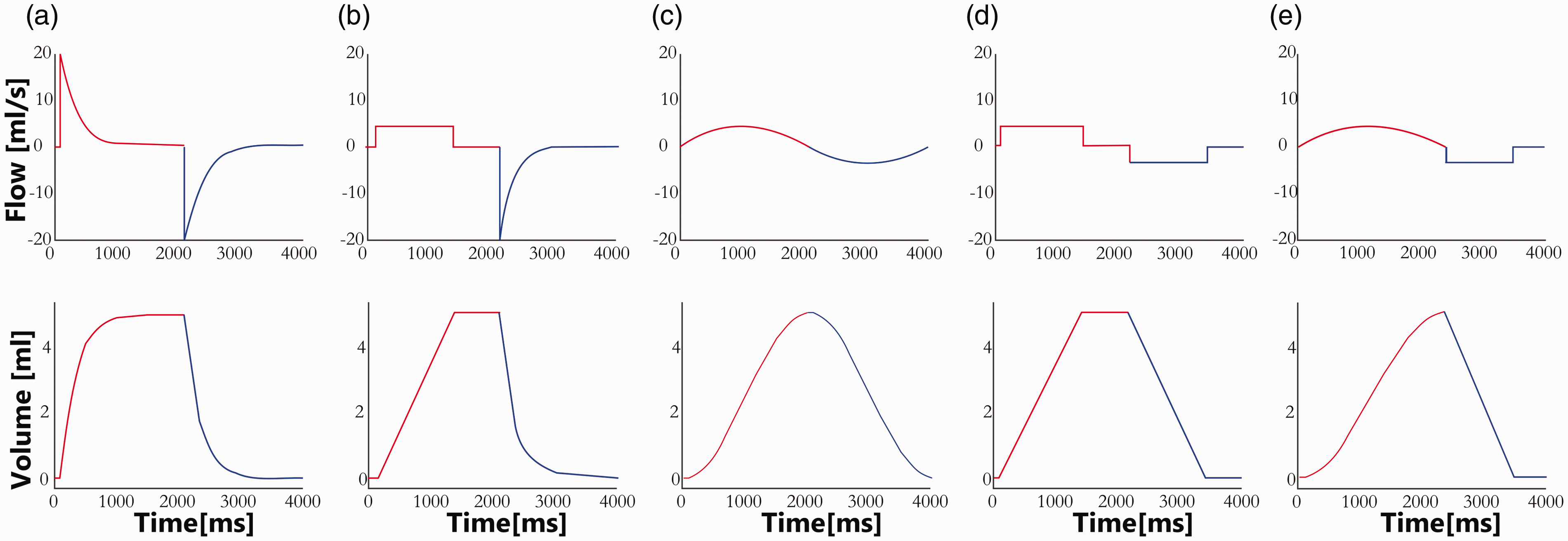
Animal experiments
The primary purpose of this paper is to present our new design of a mechanical ventilator for small animals. To prevent unnecessary animal experiments solely for this purpose, the presented animal data were taken from as yet unpublished experiments focusing on the lung-protective effects of controlled expiration. The rats were randomly assigned to one of five groups with different ventilation profiles: (i) pressure-controlled ventilation (PCV) profile, (ii) volume-controlled ventilation (VCV) profile, (iii) sinus profile, (iv) control of expiration with linear inspiration profile and (v) control of expiration with sinusoidal inspiration profile.
All animal experiments were approved by the review board for the care of animal subjects of the government executive (Regierungspräsidium, Freiburg, Germany) and carried out in accordance with the German law for animal protection and the animal care guidelines of the European Community. In total 48 female Wistar rats 263–337 g body weight (Charles River Laboratories, Sulzburg, Germany) were used for the study. Female rats were used with the intention to eliminate potential sex-dependent variability. 4 The animals were housed with an inverse 12-hour day–night cycle with a room temperature of 22 ± 2℃. Before surgery, the animals were housed five by five in type 3 cages. All rats were allowed free access to water and a maintenance diet.
The rats were anaesthetised with 100 mg/kg ketamine (Medistar Arzneimittelvertrieb GmbH Ascheberg Germany) intramuscular (i.m.) and 1 mg/kg medetomidine (Orion Pharma GmbH Hamburg Germany) i.m. and placed on a heating pad to maintain body temperature at 36 (±0.2)℃. The animals’ tracheae were tracheotomised and catheterised. Before onset of mechanical ventilation, muscular relaxation was induced by intraperitoneal (i.p.) instillation of 1 mg/kg pancuronium (Inresa Arzneimittel GmbH, Freiburg Germany).
Electrocardiogram (ECG,) blood pressure and body temperature were monitored (Haemodyn v.2.0.1, Hugo Sachs Electronics-Harvad Apparatus GmbH, March, Germany) and blood oxygen saturation was continuously measured with a small animal pulse oximeter (MouseOx Plus v.1.5, STARR Life Science Corporation, Oakmont, PA, US). In addition, blood samples were taken and analysed using an automated blood gas analyser (Cobas b221, Rosch, Indianapolis, US) to ensure normal ventilation. Anaesthesia was maintained by continuous i.p. administration of ketamine/midazolam and pancuronium as needed. Saline boli were injected intravenously to prevent dehydration during ventilation. After intubation, the animals were assigned to one of five different ventilation profiles for 4 h. The ratio of inspiration to expiration time was 1:2, the applied tidal volume was 8 ml/kg bodyweight, and the inspiratory fraction of oxygen was 1. Respiratory rate was set to maintain normocapnia (arterial carbon dioxide partial pressure between 35 and 45 mmHg).
Airway pressure was measured using a piezoelectric pressure transducer (SI – special instruments GmbH, Nordlingen, Germany). Inspiratory and expiratory flow rates were measured separately using two pneumotachographs (Fleisch 000, Dr. Fenyves und Gut, Hechingen, Germany) to reduce dead space. Volume was calculated by numerical integration of flow. Data were recorded using self-written software programmed in LabVIEW (ver. 7.1, National Instruments Corp., Austin, TX) and processed and analysed using MatLab (Matlab®, Version R2017a, 2017, The MathWorks Inc., Natick, MA).
After the experiments, the animals were euthanised by exsanguination.
Statistics
Values are given as mean [range] or mean ± SD. The MatLab (Matlab R2017a, The MathWorks, Natick, MA) statistics toolbox was used for all statistical analyses. To compare the effects of the five different ventilation modes on the response variables, respiratory rate, maximal airway pressure and maximal absolute expiratory flow, one-way ANOVA was performed. A p-value of less than 0.05 was considered statistically significant. In cases of rejection of the null hypothesis, multiple post-hoc comparisons with the Tukey’s test were used to determine which ventilation mode was different from the others. The ‘Honest Significant Difference’ criterion was used to represent the difference between the five ventilation profiles comparing every mean with every other mean.
Results
We were able to achieve all required ventilation profiles. In total 48 rats were successfully ventilated under five different ventilation profiles (Figure 3). Of these, 20 received ‘conventional’ ventilation forms (pressure (n = 10) or volume-controlled (n = 10) ventilation), 10 received a sinusoidal profile and 18 received a linear expiration profile (10 with linear inspiration profile and eight with a sinusoidal inspiration profile). The tidal volume in the experiments covered a range from 1.84 ml to 3.25 ml.
Representative airway pressure (top), flow (middle), and volume (bottom) curves measured during conventional pressure-controlled ventilation (PCV), conventional volume-controlled ventilation (VCV), sinusoidal ventilation (SINUS) and flow-controlled ventilation profiles: linear inspiration and expiration (FLEX), and sinusoidal inspiration with linear expiration (SINUS-FLEX).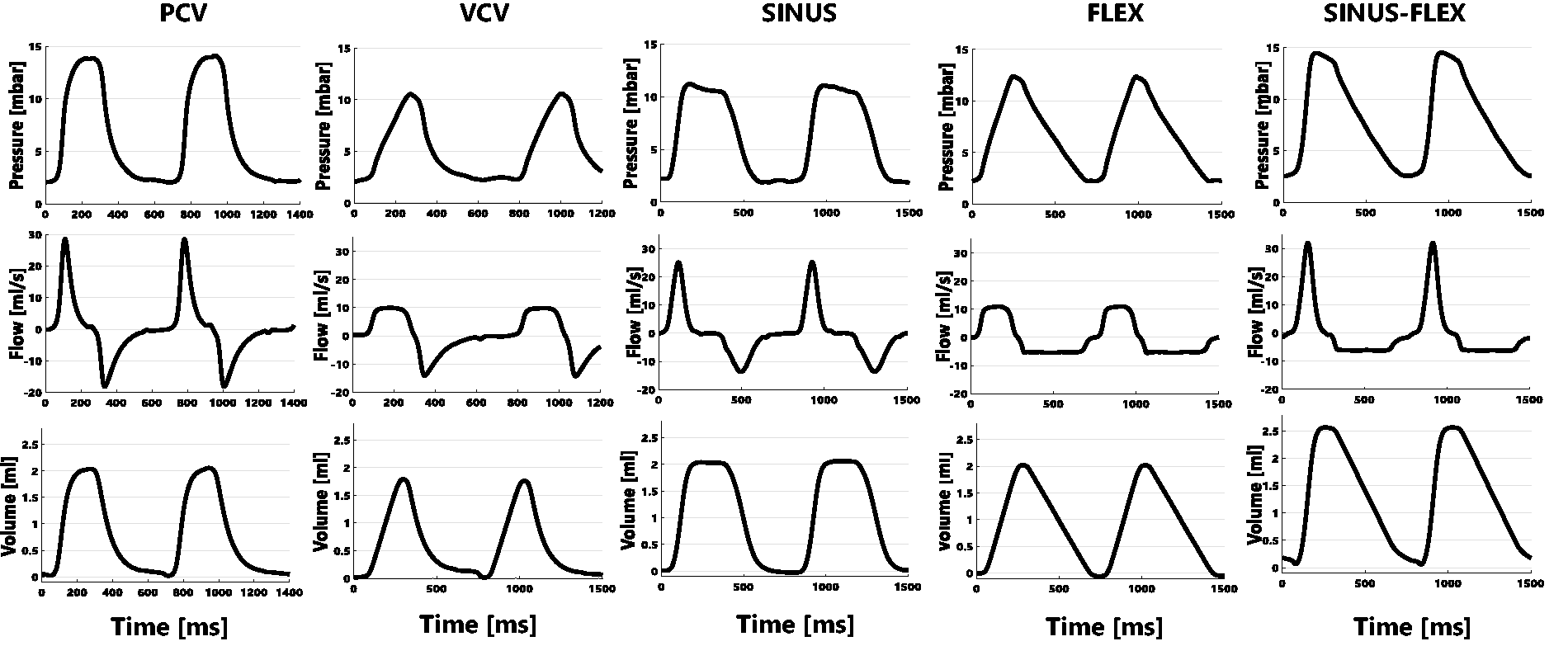
The maximal airway pressure was not significantly different between the ventilation profiles. The mean airway pressure was highest during ventilation with the FLEX and the sinusoidal inspiration combined with FLEX expiration and lowest during VCV. The peak expiratory flow was clearly higher during conventional than during controlled expiration forms.
Physiological variables during breathing with five ventilation profiles.
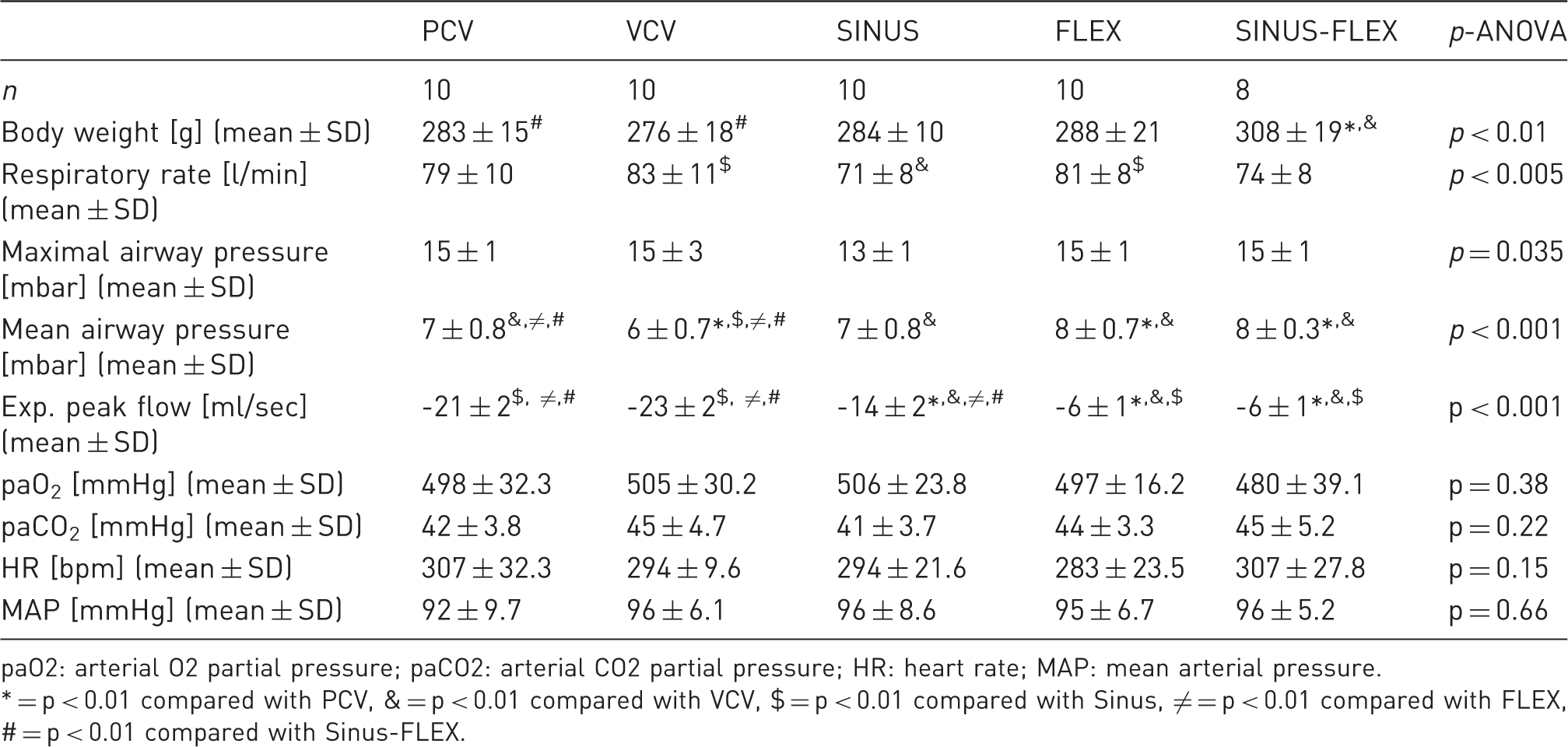
paO2: arterial O2 partial pressure; paCO2: arterial CO2 partial pressure; HR: heart rate; MAP: mean arterial pressure.
* = p < 0.01 compared with PCV, & = p < 0.01 compared with VCV, $ = p < 0.01 compared with Sinus, ≠ = p < 0.01 compared with FLEX, # = p < 0.01 compared with Sinus-FLEX.
Discussion
This paper describes the design of a new mechanical ventilator for small animals which is able to apply various ventilation profiles separately for inspiration and expiration. Small animals such as mice, rats and rabbits are commonly used for investigating the systemic effects of mechanical ventilation. So far, comparative studies on mammalian respiratory mechanics have often been performed during spontaneous breathing, which allows animals to vary tidal volume and respiratory rate. 5 Therefore, investigating respiratory system mechanics during controlled mechanical ventilation may be necessary, as it reduces the variability in measurements. With the exception of some specialised systems, for example the flexiVent device, 6 commercially available ventilators support the setting of the respiratory rate, PEEP, as well as peak pressure or tidal volume; however, they are usually limited to one ventilation mode only. In addition, all systems are limited to passive expiration.
With increasing interest in the effects of energy transfer from the ventilator to the lungs in the case of VILI, 7 more sophisticated ventilation devices for investigating this kind of scientific question become necessary.
Our ventilator for small animals is able to apply a volume range of up to 9 ml at respiratory rates of up to 120 breaths per minute. In this study we demonstrated the ability to perform conventional ventilation profiles such as PCV and VCV, and two new types of ventilation profile including the control of expiration: linear inspiration and expiration, and sinusoidal inspiration and linear expiration.
Currently, research on ventilation profiles is done mostly using available applications in the hospital setting (e.g. VCV vs. PCV), and is therefore restricted to investigating the effects of different modifications of the inspiratory phase. With exception of the effects of PEEP, modifications of the expiratory phase have rarely been investigated. In contrast to conventional ventilation profiles, our FLEX and sinusoidal ventilation modes modify the expiration pattern. This results in reduced expiratory peak flow in the early phase and maintenance of a prolonged active flow towards the final phase of expiration. Therefore, the mean airway pressure was higher compared with conventional ventilation profiles with passive expiration. These characteristics of FLEX are expected to stabilise the lung by maintaining airway pressure during expiration and therefore leaving less time for lung collapse. 8 We were able to demonstrate that this is associated with an improved oxygenation. It is important to note that during this process neither PEEP nor peak airway pressure were increased. 9 Further studies corroborate the lung-protective effects of FLEX by means of electrical impedance tomography, and the efficacy of FLEX for lung-protective ventilation was closely related to setting an appropriate PEEP. 10 In animal experiments, PEEP could be reduced when FLEX was applied. 3 For a recent study in lung-healthy adult patients during intraoperative mechanical ventilation, FLEX improved the homogeneous distribution of ventilation in the lungs by increasing the mean airway pressure and by reducing the expiratory peak flow with FLEX compared with conventional expiration. 8 Eventually, an enhanced analysis of respiratory system mechanics is enabled by the controlled expiration, allowing for separate inspiratory and expiratory intratidal compliance measurement. 11
The complexity of the physiological conditions during mechanical ventilation, particularly with respect to the variability of different lung injuries, cannot be represented in currently available mathematical models. However, new treatment strategies must be investigated for their effects and side effects before they can be applied in patients. The assessment of the effects of different ventilation profiles, directly on the fine structure of the lung, on gas exchange, and also on the circulatory system, can only be achieved within in vivo studies in ventilated mammals. For studying the mechanical properties of the respiratory system small animals such as rats and mice are widely used, because they can be obtained in large numbers and in a statistical sense produce more reliable results than large animal models. 12 Essential scientific findings about respiratory physiology, such as the protective effect of small tidal volumes, were first obtained in rats. 13 The challenge in working with small animals is that it is essential to apply small tidal volumes with precision, and that the equipment’s total dead space needs to be insignificant in relation to the animal’s lung volume. 14 Commercial equipment for ventilation of small animals supports only passive expiration. This has been demonstrated to be associated with suboptimal lung protection during mechanical ventilation. 9 Mechanical ventilation of laboratory animals with the proposed controlled expiration might therefore not only be of interest for research on the respiratory system but also for research within other areas of interest, since the ventilation-induced systematic biological response is expected to be ameliorated with a decelerated expiratory flow profile. It is important to note that further studies should be conducted to assess the generalisability of the small animal ventilator, for example exploring different animal species, sex and age.
Conclusion
We present a mechanical ventilator with freely selectable time-profiles for inspiration and expiration. With the option for novel ventilation modes including controlled expiration, our ventilator increases flexibility in studying the mechanically ventilated respiratory system in small animals.
Footnotes
Acknowledgements
The authors would like to thank Gerd Strohmeier of the ‘Wissenschaftliche Werkstätten’, University Medical Center Freiburg, for his help in building up the electronical controller for the airway switches and Katja Tutor for thorough language editing of the manuscript.
During the course of this work Matthias Schneider died unexpectedly. We have lost a skilled technician and a close friend.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the German Research Foundation Grant # SCHU2499/5-1.
