Abstract
Ventilator-induced lung injury is a severe complication mainly caused from mechanical ventilation (MV), associated with the upregulation of inflammation response. The mechanism still remains unclear. This study aims to explore the effects of pathological damage, neutrophil infiltration, expression of P2X7 receptor, and activation of Caspase-1 in lung tissue using a rat model. Sprague Dawley (SD) rats were randomly divided into sham group, conventional MV group, and high-tidal-volume ventilation group and fed with clean water and rat food. The sham group received tracheotomy without MV; conventional MV group was given 7 mL/kg tidal volume ventilation, and high-tidal-volume MV group was given 28 mL/kg tidal volume ventilation. All the rats were sacrificed after 4 h of ventilation or spontaneous breath. Lung wet/dry ratio was measured, and paraffin sections were prepared for pathological injury assessment and immunohistochemistry of P2X7 and myeloperoxidase levels. Lung homogenate was used for Western blot analysis of P2X7 receptor and Caspase-1 levels and real-time polymerase chain reaction (PCR) analysis of P2X7 gene expression level. Compared to sham group and conventional MV group, high-tidal-volume MV led to an increase in lung wet/dry ratio and histology score. High-tidal-volume ventilation also led to chemotaxis of neutrophils. The expression levels of protein and messenger RNA (mRNA) of P2X7 receptor were significantly upregulated. Cleaved-caspase-1 expression was also upregulated. All data provide the evidence that high-tidal-volume MV can lead to lung injury, neutrophils infiltration, and upregulation of cleaved-Caspase-1 level. This result may be related to the upregulation of P2X7 receptor expression.
Introduction
Mechanical ventilation (MV) is a widely used lifesaving intervention in intensive care unit. It can help to support respiratory function, increase oxygen consumption, and clear CO2. However, MV can aggravate preexisting lung injury and even induce lung injury in previously healthy lungs, which is called ventilator-induced lung injury (VILI). 1 In recent years, scientists have found that innate immune response plays an important role in the onset of VILI. In those lungs injured by MV, pro-inflammatory mediators such as interleukin-1β (IL-1β) and interleukin-18 (IL-18) can be upregulated, 2 but the mechanisms are still incompletely clear. The inflammasome is the key point of inflammatory mediators formation. P2X7 receptor is a type of gate-controlled purinoceptor, which exists on the surface of cell membrane. After binding with adenosine triphosphate (ATP), it can be activated and modulated to ion efflux, which is the main reason of intracellular inflammasome activation.3–5 Studies have shown that P2X7 receptor plays an important role in the pathogenesis of acute lung injury induced by lipopolysaccharide and/or hyperoxia,6–8 but whether P2X7 receptor is involved in lung injury induced by aseptic pure mechanical strain was unknown. The aim of this study was to test the hypothesis that P2X7 receptor activation is related to inflammatory mediator formation in an animal model of VILI.
Materials and methods
Animals
In all, 24 male Sprague Dawley (SD) rats aged 8 weeks (250–320 g in weight) were purchased from Changsheng Biotech Co. Ltd. (China). All the procedures of the experiment were approved by the animal ethics committee of Shengjing Hospital of China Medical University (approval number: 2015PS319K). Rats were kept in a specific pathogen-free environment and fed with free food and water intake until the experiments.
Induction of VILI
Animal model was established using high-tidal-volume MV.9,10 Briefly, rats were anesthetized by intraperitoneal injection of pentobarbital (75 mg/kg; Sigma-Aldrich, USA) and intubated with a 14-gauge needle through tracheotomy at the middle of the neck. Then, rats were randomly divided into three groups: spontaneous group (S group), low-tidal-volume ventilation group (L group), and high-tidal-volume ventilation group (H group), eight rats in each group. Rats in S group were allowed to breath room air spontaneously, while rats in L group and H group were connected to a rodent ventilator (TaiMeng Software Co. Ltd, China) and received MV with a respiratory rate of 80 per minute, fraction of oxygen 21%, I:E ratio 1:2, and 0 cm H2O post end-expiratory pressure. Rats in L group were ventilated with a conventional tidal volume of 7 mL/kg and rats in H group were ventilated with tidal volume of 28 mL/kg instead.
Histological assessments
Anesthesia was maintained with intraperitoneal injection of pentobarbital 20 mg/kg 2 h after tracheotomy. After 4 h of ventilation or spontaneous breath, the rats were sacrificed with exsanguination and the lung was flushed with 100 mL pre-cooled phosphate-buffered saline. The upper right lobe of lung was excised, fixed with 4% paraformaldehyde for 72 h, embedded in paraffin, and sectioned in 2.5-μm-thick slices, which were stained with hematoxylin and eosin. The severity of lung injury was evaluated by a pathologist blinded to the experimental conditions using a modified histology scoring system. In short, four pathologic parameters were scored on a scale of 1–4: (1) congestion, (2) hemorrhage, (3) neutrophil infiltration, and (4) thickness of alveolar wall. A score of 0 represents normal lungs; 1, mild; 2, moderate; and 3, severe. The total histology score was expressed as the average of the four parameters.
Wet-to-dry lung weight ratio
The left lung was used for the measurement of wet-to-dry (W/D) ratio, an index of lung water accumulation and lung injury. Briefly, after flushing with phosphate-buffered saline, the upper right lung was excised, and the surface water was removed with filter paper and then weighed immediately as the wet tissue weight. The lung tissue was then dried in an oven at 60°C for 72 h and re-weighed as dry weight. The W/D weight ratio was calculated by the wet dividing by the dry weight.
Immunohistochemistry
Paraffin-embedded lung section slices were used for immunohistochemistry with the antibodies of P2X7 (1:600 diluted; Abcam, UK) and MPO (1:100 diluted; Santa Cruz Biotechnology, USA). Sections were counterstained with hematoxylin. The sections were observed under high power fields (400×), and then, the integrated optical density (IOD) was measured by Nikon Eclipse NI software.
Western blotting
The lower right lobe of lung was excised to extract total proteins and RNA. Proteins were extracted with radioimmunoprecipitation assay (RIPA) lysis buffer (Beyotime Biotechnology, China) and then separated by electrophoresis. Proteins separated were transferred to a polyvinylidene fluoride membrane. The primary antibodies used in this study included the following: rabbit polyclonal anti-rat cleaved-caspase-1 antibody (Sigma-Aldrich), rabbit polyclonal anti-rat P2X7 antibody (Abcam), and rabbit polyclonal anti-rat glyceraldehyde 3-phosphate dehydrogenase (GAPDH) antibody (Santa Cruz Biotechnology). Secondary horseradish peroxidase (HRP)-conjugated anti-rabbit IgG (Abcam, UK) was used as secondary antibodies. Signals were detected with enhanced chemiluminescence (Azure C300 system).
Real-time polymerase chain reaction
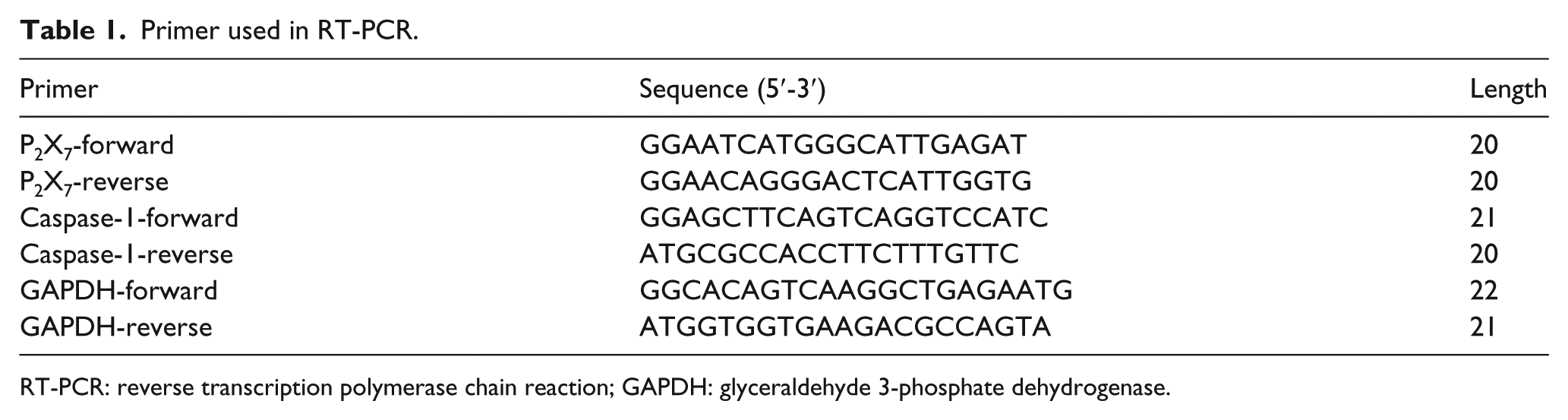
Total RNA was extracted from lower right lung homogenates, using the RNAiso® reagent (TAKARA Bio Inc., Japan) according to the manufacturer’s guide. First-strand complementary DNA (cDNA) was synthesized from total RNA using a two-step reverse transcript polymerase chain reaction (PCR) kit (Promega, USA). PCR primers for P2X7, caspase-1, and glyceraldehyde-3-phosphate dehydrogenase (GAPDH) were purchased from Sangon Biotech Co., Ltd. (China). Messenger RNA (mRNA) levels were analyzed by SYBR green II Kit (TAKARA Bio Inc.) on an ABI 7500 Fast Real-Time PCR system (Applied Biosystems, USA). Relative amount of expression of target genes were calculated as a ratio compared with GAPDH. The primers used in real-time PCR were listed in Table 1.
Primer used in RT-PCR.
RT-PCR: reverse transcription polymerase chain reaction; GAPDH: glyceraldehyde 3-phosphate dehydrogenase.
Statistical analysis
All data were collected as mean ± standard error of the mean (SEM). The data of different tests and groups were first evaluated for homogeneity of variance and then compared using either one-way analysis of variance (ANOVA) with parametric least-significant difference test or the non-parametric Kruskal–Wallis test with Dunnett’s T3 multiple comparison.
11
Statistics were performed using SPSS 21.0 (IBM Corporation, USA). A
Results
High-tidal-volume MV enhances lung injury compared to low-tidal-volume MV
Following the high-tidal-volume ventilation, the lung W/D ratio of rats in H group increased compared to L group and S group (S group: 4.36 ± 0.17, L group: 4.25 ± 0.35, H group: 5.01 ± 0.27,

Histological examination and W/D rate of the lung showed that compared to (b) low-tidal-volume ventilation (L group) and (a) spontaneous breath (Sham group), (c) high-tidal-volume ventilation (H group) significantly increases the W/D rate and histological score (*
High-tidal-volume MV increases the expression of P2X7 protein and cleaved-Caspase-1 protein in lung tissue
Expression of the P2X7 protein in lung tissue was examined through the immunohistochemistry and Western blot analysis. Immunohistochemistry showed enhanced protein expression of P2X7 in the lungs from the H group compared with L and S groups, with the immunohistochemistry integrated optic density (IOD) score increased significantly (S group: 21.29 ± 8.45, L group: 21.76 ± 13.31, H group: 31.13 ± 9.83,

Immunohistochemistry examination of the lung showed that compared to low-tidal-volume ventilation (L group) and spontaneous breath (Sham group), high-tidal-volume ventilation (H group) significantly increases the IOD score of P2X7 stain and MPO stain. (a) P2X7 stain and (b) MPO stain (*

Western blotting analysis of the lung homogenates showed that compared to low-tidal-volume group and sham group, P2X7 expression and cleaved-Caspase-1 expression in high-tidal-volume group were significantly upregulated (*
High-tidal-volume MV increases the infiltration of neutrophils in lung tissue
MPO was expressed specifically in neutrophils, which can be used as a parameter of neutrophils infiltration.
10
Expression of MPO was significantly elevated in the lungs injured with high-tidal-volume MV compared to low-tidal-volume MV and spontaneous breath (IOD score: S group: 26.17 ± 8.13, L group: 27.12 ± 4.53, and H group: 40.06 ± 6.92,
High-tidal-volume MV increases the relative mRNA expression of P2X7 but not Caspase-1 in lung tissue
Relative P2X7 mRNA levels were higher in lung homogenates after 4 h of high-tidal-volume MV than low-tidal-volume MV and spontaneous breath (standard by 2−ΔCT compared to GAPDH: S group: 1.28 ± 0.55, L group: 1.16 ± 0.35, and H group: 2.22 ± 0.76,

Real-time PCR analysis showed compared to sham group and low Vt group, rats in high Vt group had a higher P2X7 mRNA level, while the Caspase-1 mRNA level had no significant differences between different groups (*
Discussion
A number of literatures have revealed the relationship between P2X7 receptor and lung injury induced by ATP injection, higher oxygen concentration, lipopolysaccharide, ischemia–reperfusion, trauma, smoking, drugs, and so on and have came to a conclusion, which has been expanded by this study. The lung injury in this study is induced by aseptic inflammatory method using pure high-tidal-volume ventilation, without other damage factors. Our results showed that the lung wet/dry weight ratio and histological score increased in VILI rats. MPO immunohistochemistry stain showed significant infiltration of neutrophils. At the same time, immunohistochemistry and western blotting of the lung tissue showed that the expression of P2X7 protein on the membranes of the epithelial cell and macrophage cell was upregulated significantly, indicating that there may be certain relationship between the upregulation of P2X7 receptor and mechanical stimulation. Caspase-1 is the key enzyme for the transform of precursors IL-1β and IL-18 for their mature form. Cleaved-Caspase-1 level in high-tidal-volume group was higher than conventional ventilation group, suggesting that pulmonary pro-inflammatory reaction has been activated and that the elevation of P2X7 level and cleaved-Caspase-1 may be related. The Caspase-1 mRNA level did not increase based on our observation. The reason might be that only the expression of cleaved-Caspase-1 was upregulated, which may be initiated by the editing of nucleotide oligomerization domain (NOD)-like receptor (NLR) after its activation. 12
Recently, more studies show that inflammation reaction plays an important role in the pathogenesis of VILI. 13 Among them, toll-like-receptor (TLR), such as TLR-4, can mediate the generation of the precursor of inflammatory factor through the signaling pathway of nuclear factor (NF)-κB. 14 While NLR assembles into inflammasome and cuts the precursor of inflammatory factor into mature form and then secretes into the extracellular fluid, participate in the overexpression of inflammatory response. 15 Between these two key pathways, NLR may play more important roles in the pathogenesis of VILI as it is an aseptic inflammation and TLR is activated by lipopolysaccharide, bacterial infection and other pathogen associated with molecular patterns (PAMP). But NLR is located in the cytoplasm, while as external stimuli, the shear stress of MV acts directly on the surface of the cell membrane. There must be a receptor located on the surface of the membrane, which acts as a sensor of shear stress and transduces into cytoplasm. This receptor is likely to be P2X7 receptor, which is a member of the ion-gated purinergic receptor family, widely distributed on the cell membrane of the macrophages, dendritic cells, lymphocytes, endothelial cells, mast cells, and type 1 alveolar epithelial cells. It can be activated after binding to its nature agonist ATP and then open the ion gate, leading to the potassium outflow.3,16 Studies have confirmed that potassium efflux can cause NLR activation. And studies have also confirmed that the extracellular ATP concentration in VILI is significantly elevated. 17 ATP has been shown as a representative of inflammation in the animal trials of VILI in literatures. Therefore, P2X7 receptor may play an important role in the regulation of VILI via ATP and inflammation. When ATP level increases in the injured lung tissue, it binds to the P2X7 receptor, followed by activation of pro-inflammatory in the lung. Wang et al. have shown that blocking P2X7 receptor attenuated the lung injury induced by lipopolysaccharide injection, despite the presence of ATP in their recent animal trial. The above-mentioned study suggests the importance of P2X7 receptor in the lung injury induced by elevated level of ATP, which is in accordance with the findings in this study. 16
Based on the data presented in this preliminary study, we observe the involvement of P2X7 receptor expression influenced by high-tidal-volume ventilation without lipopolysaccharide. As one of the key factors of NLR activation, P2X7 may play an important role in the assembly of inflammasome, which will lead to the amplification of pro-inflammatory factors. In the future studies, we will use P2X7 gene knockout animals to verify whether knockout the expression of P2X7 may reduce the inflammation of lung in VILI, in order to find an effective method for clinical intervention for alleviating VILI.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
