Abstract
Fifty-four pigs (
Introduction
An innovative surgical technique for laparoscopic kidney transplantation has been developed in a pig model over the last six years 1 with the ultimate goal of translating a method for laparoscopic orthotopic kidney grafting to human patients. 2 The development of this technique in a pig model has presented a number of challenges to the research team as the surgery involved two procedures: laparoscopic donor nephrectomy followed by laparoscopic kidney auto-transplantation. In addition, the pigs recovered from surgery for up to one month. Multiple anaesthetic refinements have been incorporated during this six-year period.
Application of the principle of refinement is a continuous process incorporating methods that avoid or minimise potential pain and distress, and enhance animal wellbeing. 3 This concept applies to all aspects of animal care from basic husbandry to major surgical procedures. In this project there have been many opportunities to introduce refinements, particularly in the peri-operative period. Herein is a brief description of six such refinements, and their perceived value, applied to this pig model.
Materials and methods
Animals
The surgical project was approved over three consecutive applications by the Animal Ethics Committee of the University of Western Australia in accordance with the guidelines of the
This retrospective review details procedural refinements introduced as 54 pigs (
The procedural stages for each pig included initial instrumentation for intra-operative monitoring, laparoscopic nephrectomy,
5
preparation of the graft, orthotopic auto-transplantation by either a laparoscopic (
Anaesthesia
During the course of the project the drugs for anaesthesia have not changed per se
Multi-modal analgesia was provided from tiletamine, xylazine, local anaesthetic, opioids and tramadol. 4 Bupivacaine (maximum dose 2 mg/kg SC) was injected at each of the surgery sites at the end of surgery. Opioids were administered in the peri-operative period: morphine prior to the start of surgery (0.1–0.2 mg/kg IV) and as intermittent bolus doses or as an IV infusion (0.2 mg/kg/h) during surgery; and morphine (0.2–0.5 mg/kg IM) every 6–8 h after surgery for 1-2 days. Ketamine was administered to some pigs during surgery (0.3 mg/kg/h IV). Tramadol (2–4 mg/kg IM) was also administered every 12 h after surgery for 3 days.
Post-procedural monitoring in the first six hours involved observation and scoring of six parameters: body temperature (normal, 39.8–41℃, >41℃); ventilation (normal, slightly/intermittently laboured, moderately laboured); capillary refill time (normal, slightly increased, moderately increased); mucous membrane colour (normal, slightly pale, moderately pale or cyanotic); demeanour (normal, slightly or intermittently deviated from normal, moderately or consistently deviated from normal given the pig was recovering from anaesthesia and had received analgesic drugs); and other presenting clinical signs. These parameters were scored 0 (normal) to a maximum of 2 and summed to generate a total score. Interventions were triggered by the summed score with prompt euthanasia required if the score was >6.
Post-procedural monitoring from days 1 to 28 after surgery was performed twice daily for the first 14 days and once daily for the subsequent 14 days. Observation and scoring of 10 parameters was performed: ventilation as described above; gait (normal, intermittent or slight lameness or paresis, moderate lameness or paresis); secretions from eyes/nose (normal, mildly increased, moderately increased); demeanour (normal, slight or intermittent malaise, moderate malaise); appetite (normal, reduced feed intake, no feed intake); thirst (normal, no drinking); urination (normal, not observed and dry flooring); faecal output and texture (normal, reduced or soft, no output or watery consistency); wound appearance (normal, slightly inflamed, moderately inflamed with or without discharge); and other presenting clinical signs. These parameters were scored 0 (normal) to a maximum of 2 and summed to generate a total score. Interventions were triggered by the summed score, with veterinary assessment required if the score was >1 and prompt euthanasia required if the score was >8.
Experimental refinements
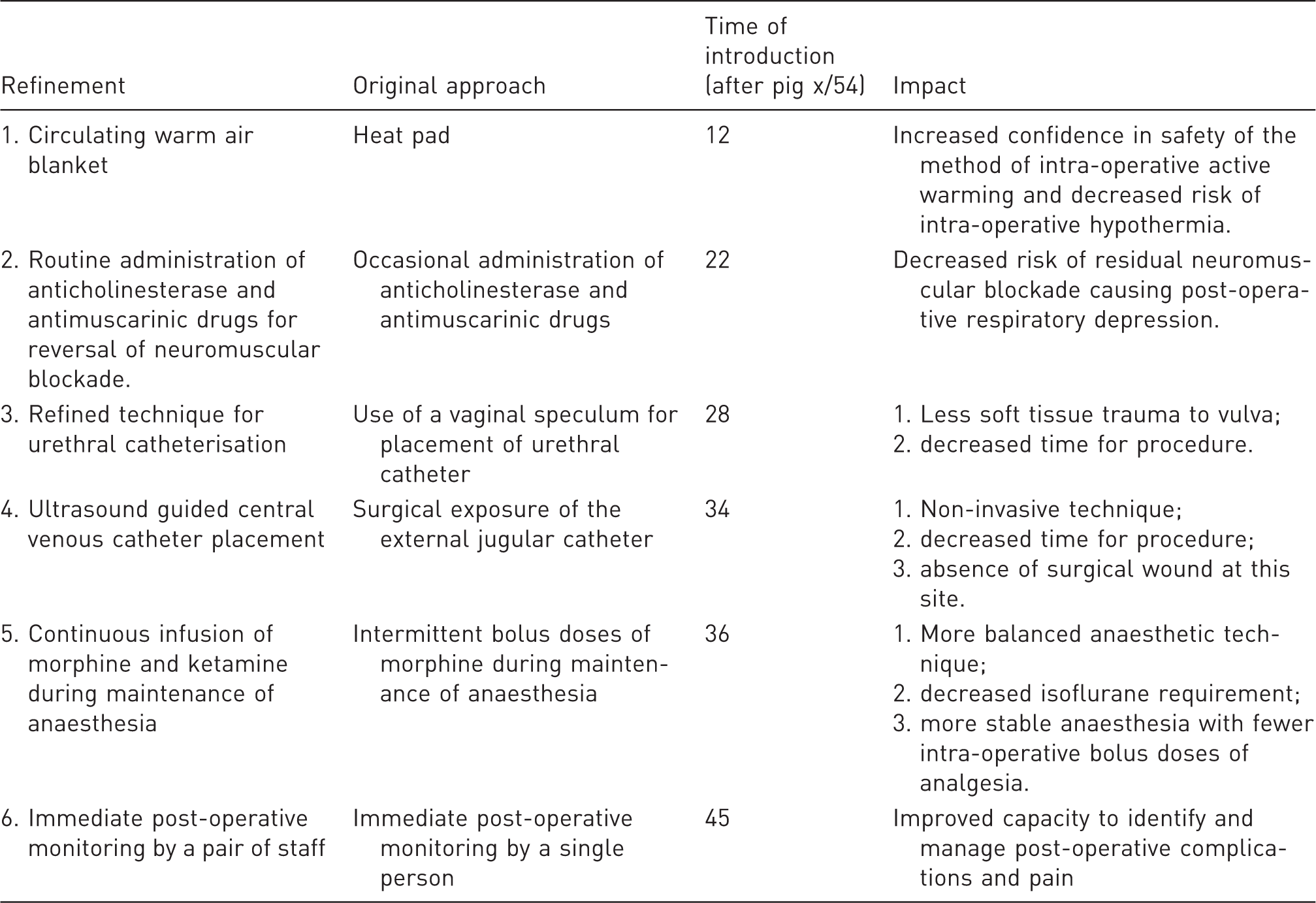
During the evolution and establishment of this pig model multiple refinements were introduced: (1) a heat pad was changed to a circulating warm air blanket to safely minimise the severity of intra-operative hypothermia; (2) routine administration of anticholinesterase (neostigmine 0.04 mg/kg IV) and antimuscarinic (glycopyrrolate 0.01 mg/kg IV) drugs for reversal of neuromuscular blockade was introduced; (3) a technique for urethral catheterisation for measurement of intra-operative urine output was developed and described; 6 (4) ultrasound guidance for placement of a central line for measurement of central venous pressure and administration of intravenous mannitol and other therapies was learned; (5) intravenous infusions of morphine and ketamine were instituted for a more balanced anaesthetic protocol; and (6) post-operative monitoring was performed by two technical staff for at least the first six hours after extubation of the trachea.
Results
Following the administration of zolazepam, tiletamine and xylazine, propofol was administered to 27 pigs to achieve a depth of anaesthesia for oral tracheal intubation. The time between IM injection of zolazepam, tiletamine and xylazine and the IV injection of propofol (
Summary of the refinement and its perceived impact.
Discussion
The six anaesthetic refinements were introduced to the anaesthetic management of pigs undergoing orthotopic kidney auto-transplantation when an opportunity for improvement and efficiency was identified. The aims of these refinements were to decrease total anaesthesia time, morbidity and mortality. Measurement of the impact of the refinements is difficult as they were not always introduced in isolation and were not always recognised specifically as a refinement until after the fact. Nevertheless, after the completion of 54 pigs, six specific refinements have been identified, and are believed to have contributed to greater efficiencies during anaesthesia and lower morbidity and mortality.
Intra-operative hypothermia is a common contributor to post-operative morbidity and active warming is required to prevent the expected decrease in temperature associated with anaesthesia.7,8 The decision to change from a heat pad to a warm air blanket was made when the safety of the heat pad was questioned after one pig was noticed to have a pattern of erythema on the skin in the shape of the heat coils within the pad at the end of the procedure. Purchasing a warm air blanket mitigated the safety risk of using a heat pad for the long procedure. Subsequent to this purchase intra-operative severe hypothermia was avoided, without adverse events.
Neuromuscular blockade during laparoscopy is used to improve the quality of the surgical field. 9 The introduction of TOF monitoring with a peripheral nerve stimulator was acknowledged to provide limited information on the depth of neuromuscular blockade, but was ultimately used to ensure that the reversal agent was administered when there was evidence of spontaneous recovery based on observations following TOF stimulation. 10 Routine reversal of neuromuscular blockade with intravenous neostigmine (0.04 mg/kg) and glycopyrrolate (0.01 mg/kg) was implemented after the 22nd pig as recommended. 10 Prior to this standardised approach, neuromuscular blockade was not always reversed, especially if the most recent dose was administered more than 1 h prior to the end of anaesthesia. The risk of residual neuromuscular blockade has therefore been mitigated.
Catheterisation of the urethra was necessary in this study to allow drainage of the bladder during the long procedure and intra-operative assessment of urine output. Initially the procedure for insertion of the catheter took up to an hour. At the time, reference to the literature for a description of the technique in pigs between 40 and 50 kg was largely unrewarding. Hence, the vagina and urethra were dissected from six pigs and the urethral diverticulum was identified. Following these dissections an eight-point list of tips to aid placement of a urethral catheter in female pigs was published. 6 The presence of a urethral diverticulum was identified, and implementation of these tips substantially reduced soft tissue trauma to the vagina and the time for placement of the urethral catheter.
The placement of a central venous line using the Seldinger technique was initially performed by surgically exposing the internal jugular vein in a similar approach to that described previously. 11 Access to an ultrasound machine enabled the veterinary anaesthetist (GCM) to use ultrasound guidance to identify the internal jugular vein and place a central venous catheter within 30 minutes. The technique was similar to that described elsewhere, 12 although it did not require two veterinarians as Izer et al. report. This refinement has negated the requirement for an additional surgical procedure and decreases the time involved in instrumentation of the pig prior to surgery.
Supplemental intravenous anaesthesia or analgesia is used to describe a combination of intravenous and inhalational anaesthesia as a technique of ‘balanced anaesthesia’. 13 Intermittent bolus injection of drugs produces peaks and troughs of plasma concentration and clinical effect so a constant or variable rate infusion of drugs to supplement inhalational anaesthesia was introduced from the 37th pig. The drugs were morphine (0.2 mg/kg/h) and ketamine (0.3 mg/kg/h) and the intravenous infusions commenced prior to surgery. A bolus of morphine was given initially (0.1 mg/kg). Prior to this refinement intermittent boluses of morphine were administered as deemed necessary and the isoflurane vapouriser setting was altered more frequently. In dogs the infusion of morphine and ketamine will decrease isoflurane requirements, 14 although this effect is not reported in pigs the requirement for isoflurane was lower once this regime was introduced.
Initially the immediate post-procedural monitoring was performed by a single person, which limited their capacity to manage any issues that arose. The aim at that point was to simply ensure the pig was comfortable. From the 47th pig two people were responsible for the post-procedural care, which enabled the continuation of intravenous fluid therapy and a greater capacity to provide comfort and nursing care. This refinement ensures that the provision of post-operative analgesia is more targeted to the individual animal’s requirements and the morning after surgery the pigs appeared more comfortable and were more active.
There are limitations to this retrospective account of multiple refinements introduced to a pig model. Objective assessment of the impact of some of the refinements is very difficult, even in a prospective study design, so the commentary is somewhat speculative. Furthermore, in some instances, measurements, for example the time taken to place a urethral catheter, were not taken so comparisons pre- and post-refinement are not possible. Finally, the sample size for assessment of refinements is small in such a complex model with numerous variables. Targeted studies to determine the positive or negative effects of refinements are warranted in pigs, until which time extrapolation from other species with a basis in evidence to the refinements is required.
The refinements to the anaesthesia protocol for laparoscopic orthotopic kidney auto-transplantation over the first 54 pigs have translated to improved outcomes for the pigs in the post-procedural period. This conclusion is somewhat subjective as accurate measurement of the impact of each refinement is impossible. Nevertheless, refinement is a continuous process and should be applied to the entire range of procedures involved in animal use for scientific purposes. Engagement and consistency of personnel involved in such projects is essential to ensure meaningful refinements can be identified, introduced and evaluated.
Footnotes
Acknowledgements
The authors would like to thank the technical staff in Animal Care Services at the University of Western Australia for the expert care and husbandry of the animals. In addition, the input and engagement of medical and veterinary experts in critical care (Dr Adrian Regli), surgery (Dr Mark Newman) and animal welfare (Dr Melissa Lindeman) was vital to the development and application of refinements to this model. The contribution from veterinary anaesthesia trainees and specialists from Murdoch University to the care of the pigs in these projects is also appreciated.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
