Abstract
As a step towards implementing non-aversive handling techniques at a big mouse breeding facility in Germany, tunnel handling was introduced in a breeding unit comprising three inbred mouse strains. To assess whether tunnel handling would be feasible for the animal technicians in their everyday work and beneficial for the mice when being handled during weekly cage change only, the behaviour of tunnel- and tail-handled animals of both sexes was examined before, during and after the handling events over a period of nine weeks. Moreover, the time expenditure was compared between both handling techniques. It was possible to use the tunnel in all three mouse strains. However, the impact of the handling techniques on the behavioural parameters investigated in the present study were strain-specific. All behavioural parameters suggested that NZW mice benefited the most from tunnel handling. The results obtained from Hello Kitty and WNK mice were ambiguous, which may suggest that a brief handling session during the cage clean may have not been sufficient to habituate them to the process of handling. It took the animal technicians approximately 3 seconds longer per mouse when using a tunnel. The strain-specific results should encourage researchers to share their experiences with non-aversive handling techniques in different mouse strains, for example, along with their research articles.
Introduction
Non-aversive handling techniques such as the tunnel and cup technique were introduced more than a decade ago to replace the traditional method of picking mice up by their tail. 1 Their benefits on both animal welfare and quality of scientific data were demonstrated in various independent studies. Tunnel and cup handling were shown to reduce anxiety when the mice anticipated being handled. 1 This effect is not reversible by procedures carried out after the mouse was removed from the cage. Independent of whether the mice are scruffed, subcutaneously/intraperitoneally injected or anesthetized with isoflurane for a short term, the voluntary interaction time with the experimenter is higher after tunnel when compared with tail handling.1 –3 Moreover, tunnel and cup handling decrease anxiety-related behaviour in the open field test and elevated plus maze test.1,4,5 Observations in the sucrose preference test confirmed that tail-handled mice responded differently towards a positive stimulus from tunnel-handled mice, indicating that tail handling induced a depressive-like state. 5 The behavioural data are supported by physiological measurements. Higher weights of adrenals and elevated corticosterone concentrations were found in tail-handled mice.4,6 In spite of these clear advantages for animal welfare, tunnel and cup handling have still not been implemented in all animal facilities. 7 The reasons for this vary. The evaluation of our courses on non-aversive mouse handling techniques at the Research Facilities for Experimental Medicine (FEM), Charité – Universitätsmedizin Berlin in 2019 revealed that an additional expenditure of time represented one of the greatest fears. 8 Moreover, there is a lot of uncertainty in whether tunnel and cup handling may be suitable for all mouse strains since most research data on non-aversive mouse handling is based on C57BL/6, BALB/c and CD-1. After courses on non-aversive mouse handling techniques were carried out at the FEM, as a further step towards implementation tunnel handling was introduced in a breeding unit of the FEM comprising the inbred strains WNK 1, 9 NZW10 and Hello Kitty. 11 To assess whether tunnel handling would be feasible for the animal technicians (i.e., could be implemented into their everyday work) and beneficial for the mice when being handled during weekly cage change only, the behaviour of tunnel- and tail-handled animals was examined before, during and after the handling events over a period of nine weeks. Furthermore, the time expenditure was compared between both handling techniques.
Materials and methods
Ethics
Maintenance of the animals was approved by the Berlin State Authority (‘Landesamt für Gesundheit und Soziales’, permit number: ZH158). The mice were handled according to standard care procedures, implementing the non-aversive tunnel technique. The data presented were derived from live observations before, during and after they were handled. Since the animals were not subjected to pain, suffering or harm, this study was not considered as an animal experiment in the sense of European legislation. A protocol was not preregistered.
Animal technicians
Three female professionally trained animal technicians with more than 10 years of experience were responsible for handling the mice and collecting the data. The implementation of the tunnel handling technique as a routine handling procedure started with a theoretical and practical training. The theoretical training of 90 min included videos, pictures and an overview of the effects caused by the different handling techniques on the mice. It was accompanied with a survey on the acceptance and concerns of the participants. 8 During the approximately 120 min of practical training, the participants were able to learn the use of the tunnel as a working tool. In order to also map the learning process of the animal technicians, the early phase of implementation was included in the study. According to self-reports of the animal technicians, they were still learning the practice of tunnel handling at the beginning of the study. Due to obvious reasons, it was not possible to blind the animal technicians to the handling techniques.
Animal facility conditions
The mice were housed in a productive breeding facility with 8000 mice of approximately 100 genetically modified mouse strains for the use in experiments under specifically pathogen free barrier conditions. They were free of pathogens listed in the FELASA recommendation. 12 Regular access for the personnel was limited to animal technicians and veterinarians after wet showers and cloth changes. Material, feed and water were autoclaved or disinfected by peracetic acid. The mice entry was limited to embryo transfer.
Housing conditions
The animal facility had a temperature of 22°C ± 2°C, a relative humidity of 55% ± 10%, and a 14/10-h light/dark cycle. Lights were turned on at 06:00 h. Mice were housed in sex-equal groups of 2–6 in type II long cages (1284L Eurostandard Type II L, Tecniplast, Buggugiate, Italy) with grids and open tops on low dust poplar wood bedding (PG2, LASvendi, Soest, Germany) with ad libitum access to food (Mouse breeding fortified, Ssniff, Soest, Germany) and water. A clear polycarbonate handling tube (130 mm × 50 mm, Datesand Group, Stockport, UK), a red mouse house (Zoonlab, Mouse shelter), 1–3 gnawing sticks (Ssniff, Aspen wood, size “S” Small) and tissue as nesting material were provided in the cages.
Animals
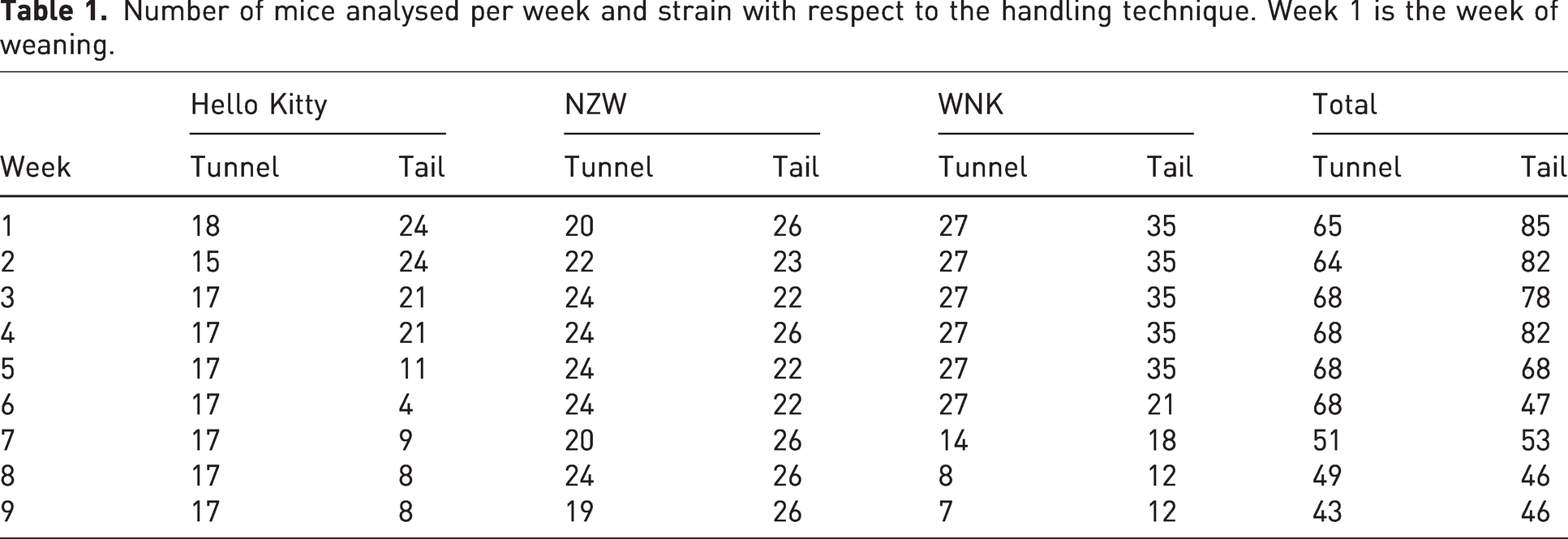
WNK 1 (a Charité substrain of WNK he mice, MGI_C57BL6J_2442092), 9 NZW (a Charité substrain of NZWLac/J RRID:IMSR_JAX:001058, MGI:2159914) 10 and Hello Kitty (a Charité substrain of C57BL6-Cpa-Cre;Mcl-1fl/fl) 11 inbred male and female mice were analysed from weaning at the age of approximately three weeks to a maximum of 12 weeks after weaning. The number of mice analysed during the study period with respect to strain and handling technique is listed in Table 1. The animals were assigned to the handling technique by simple randomization; animals living in the same cage experienced the same handling technique.
Number of mice analysed per week and strain with respect to the handling technique. Week 1 is the week of weaning.
Handling techniques
Mice were handled once a week during cage change. The animal technicians wore latex (Micro touch, Powder-Free Examination Gloves, Ansell, Brussels, Belgium) or nitrile gloves (Vasco, Nitril white, Powder-Free Gloves, B. Braun, Melsungen, Germany). The tail and the tunnel handling techniques were compared. Tail-handled mice were picked up by the tail base with the thumb and the index finger and transferred from the used to the clean cage. In order to transfer the mice from the used to the clean cage with the tunnel, the animal technicians held the clear polycarbonate handling tube (130 mm × 50 mm, Datesand Group, Stockport, UK) with one hand in the middle close to the bedding. Either both ends of the tunnel were open or the hand loosely closed one of the tunnel ends. If the mice did not voluntarily enter the tunnel, they were gently guided with the other hand into the tunnel. The tunnel was lifted either with both ends closed or with open ends. The first option was preferred if there was a risk that the mice may jump out of the tunnel during cage changing.
Cage change, time measurement and observation workflow
The cage change process including the behavioural observation, time measurement and documentation was strictly standardized and took place at the beginning of the working day before 12:00 h
The clean cage was placed on a working table. The animal technicians took the used cage from the rack and opened it next to the clean cage, which was followed by a 15-s observation period and documentation. The mouse house and grid were transferred to the clean cage.
The timer was started and all mice of one cage were transferred to the clean cage by tunnel or tail, whereupon the timer was stopped. While cage changing, the animal technicians observed the mice during capture and after release to investigate their behaviour in anticipation of being handled and after this process. The health inspection was made either in the cage and while lifting the mouse by the tail or in the clear tunnel. After all mice were transferred to the clean cage, the time and observations were immediately documented. The documentation process lasted approximately 20 s. Then all mice were monitored in the clean cage for another 15 s and the observations were documented. Finally, the grid was closed and the clean cage was returned into the rack.
For further analysis, the mean duration for the transfer of one mouse per cage was calculated by dividing the measured transfer time of a mouse group in a cage by the number of mice in the cage.
Documentation
Behavioural observations and time expenditure for the cage change process were documented in a standardized form with defined options to choose at the above-described time points.
Behaviour in the used cage; number of animals, that:
are located in the nest/house; explore the cage attentively; freeze; move fast to the nest/house; move fast in the cage; jump.
Behaviour during capture; number of animals showing:
defecation; urination; vocalization; jumping; voluntary contact to the animal technician’s hand (i.e. the animals approached the tunnel or hand); for tunnel handling only, voluntarily enter the tunnel, more than one animal in the tunnel.
Health condition; number of animals and description of wounds or abnormalities.
Behaviour after all animals were released into the clean cage; number of animals, that:
are located in the house; explore the cage attentively; freeze; move fast in the cage; jump.
The transfer of one mouse from a used cage to a clean cage at one time point is referred to as one cage change event. In terms of one week, the number of cage change events coincides with the number of animals listed in Table 1. Over the observation period 544 tunnel cage change events and 587 tail cage change events were documented, that is,152 tunnel- and 130 tail-events for Hello Kitty, 201 tunnel- and 219 tail-events for NZW, and 191 tunnel- and 238 tail-events for WNK.
Statistics
Data were stored in an MS Excel® sheet (version 2016) and analysed in IBM® SPSS® Statistics (version 27). The influence of week, strain, sex, duration of transfer per animal, animal technician and method on the number of animals exhibiting each behaviour was examined using mixed regression models with Poisson distribution. These models were analysed separately for weeks 1–4 and weeks 5–9. Only behaviours that showed variability between the methods and for which the statistical model converged were investigated. Therefore, the following behavioural parameters were not analysed: move fast in the cage, freeze, jumping, urination, vocalization, voluntarily enter the tunnel, and more than one animal in tunnel. The cage was included in the model as a random factor. All variables, including the interactions between strain and method, as well as between animal technician and strain, were used as influence factors in all models. For the exploration in the used cage, the interaction between week and strain was also included because descriptive data gave reason. For some target variables, a mixed model could not be run because the convergence criteria were not met: nest residence in used cage, defecation during capture, voluntary contact to hand, defecation in clean cage). Here, the random factor ‘cage’ was removed from the model, which may have resulted in an overestimation of the effects. The influence of week and strain on the duration of conversion was examined for the ‘tunnel’ method using a mixed linear model with cage as a random factor. Post-hoc comparisons were corrected using Bonferroni’s method. Model fit of each model was tested with visual inspection of normality and homoscedasticity of residuals. Odds ratios (ORs) as well as 95% confidence intervals (95% CIs) were calculated as well. The global
Results
The study was carried out over 10 months. Over a period of nine weeks, a maximum of 150 mice (week 1) to a minimum of 89 mice (week 9) were kept in 40 (week 1) to 26 (week 9) cages. Fifty-six mice had to be transferred to an experimental animal facility between week 2 and week 9. Four animals were transferred to breeding cages, which were not part of our analysis; one mouse had to be humanely killed because of uncontrolled growth of incisors. Mice were handled once a week during routine cage changes with the respective handling techniques. Time consumption and behavioural observations were documented considering the handling technique. Each transfer of one mouse from a used cage to a clean cage by tunnel or tail was analysed as one cage change event.
Transfer time
The duration to transfer a mouse from the used cage to the clean cage was compared concerning tail handling and tunnel handling in the different mouse strains (Figure 1). Tunnel handling was always more time consuming than tail handling. The duration of tail handling was not significantly different over time in all strains and between strains. The time period needed for the cage transfer by tunnel handling was not significantly different over time in the Hello Kitty and WNK mice. Time consumption decreased over time in NZW mice. Transfer by tunnel handling took longer in Hello Kitty than in NZW and WNK mice.
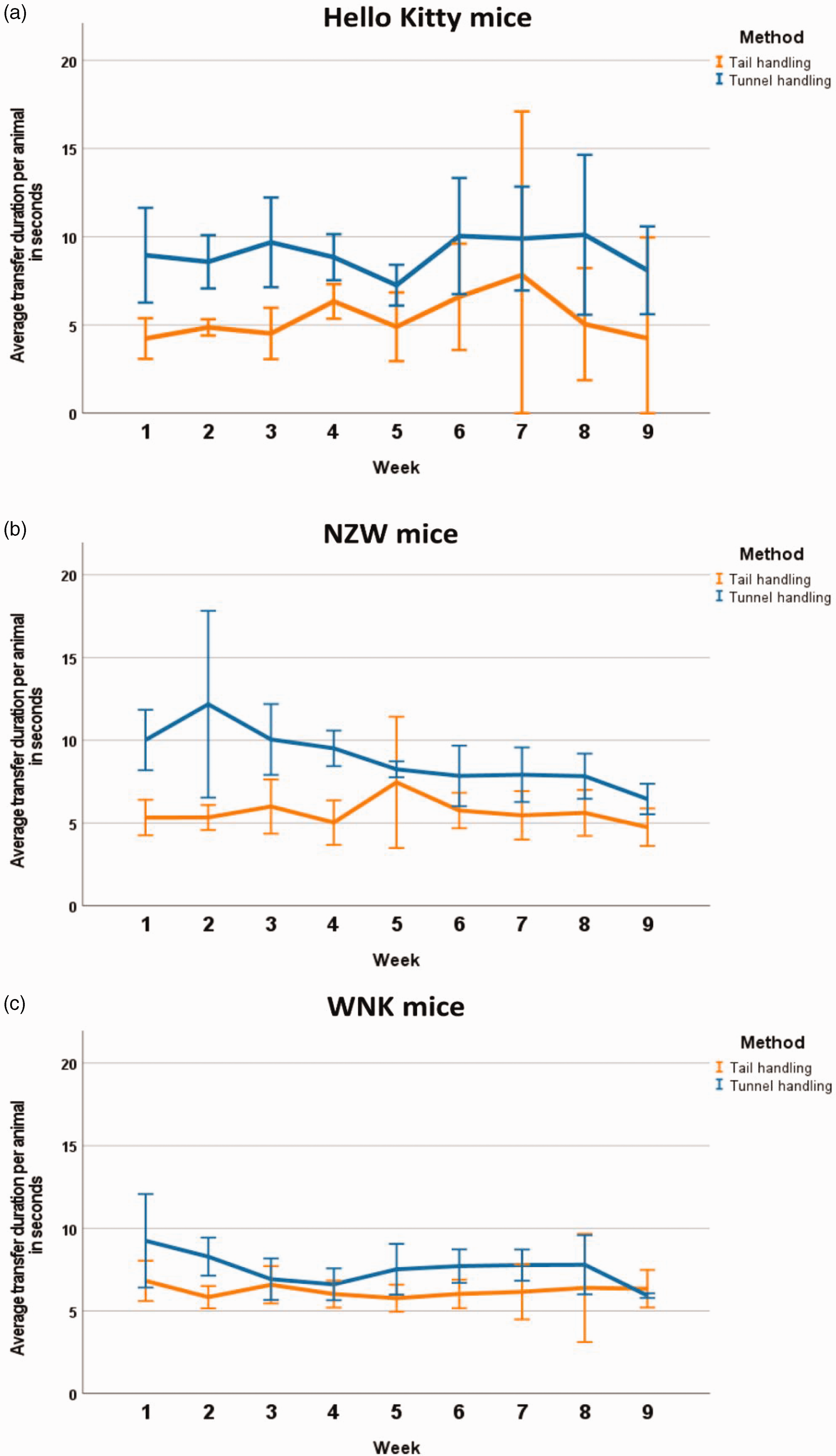
Transfer duration for (a) Hello Kitty, (b) NZW and (c) WNK mice. Data are shown as mean and 95% confidence interval.
Nest residence in used cage
In weeks 1–4, significantly more tunnel-handled NZW mice were sitting in the nest in the used cage before mice were transferred to the clean cage (
From week 5 to week 9, the number of animals in the nest had a significantly increasing effect on the duration of transfer (
In weeks 5–9, fewer animals were in the nest in weeks 5–8 compared with week 9 (
House residence in used cage
For the number of animals sitting in the house in the used cage in the first four weeks, there was a significant effect of week (
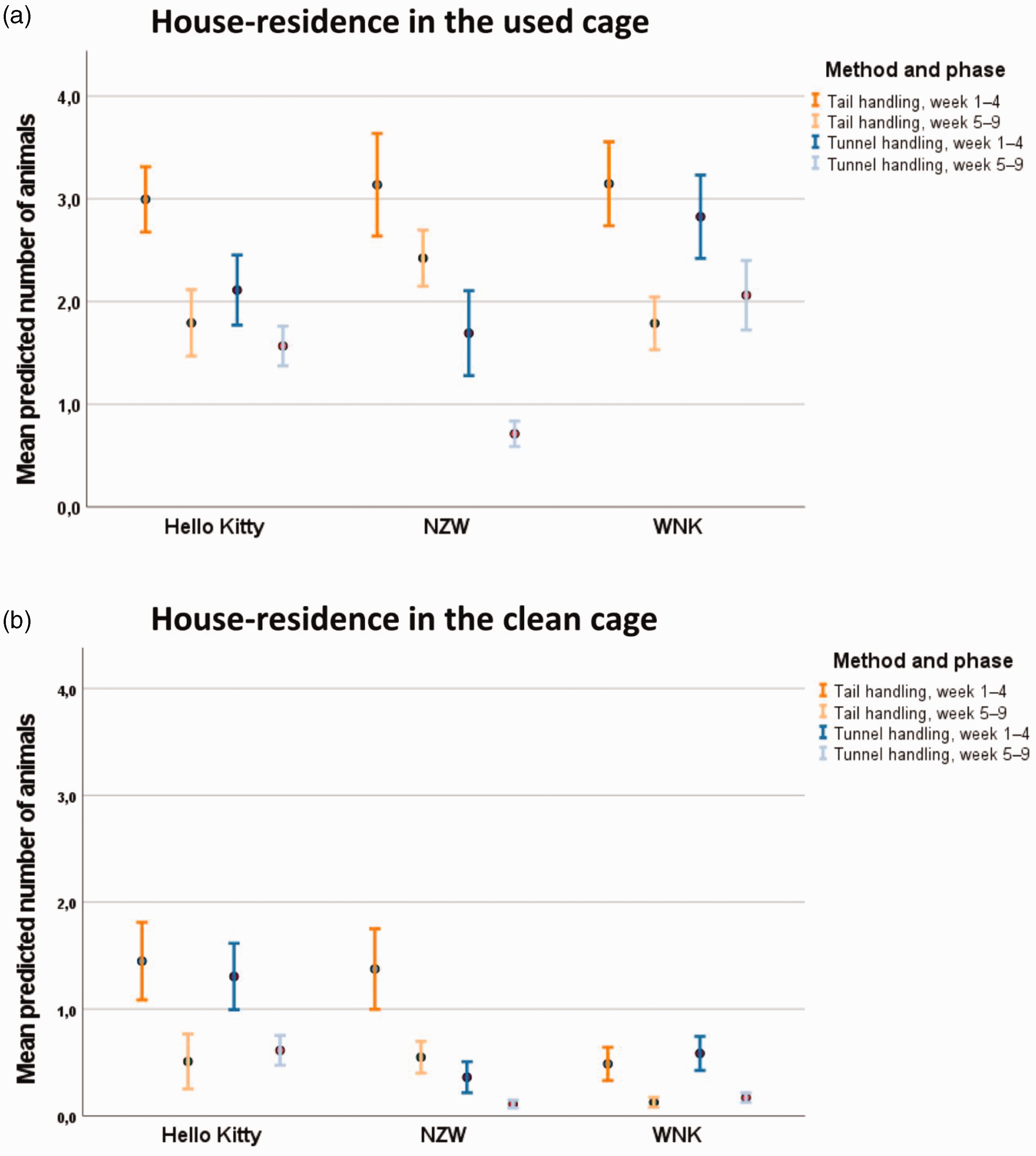
Number of mice located in the house in the (a) used cage and (b) clean cage. Data are shown as mean and 95% confidence interval.
In weeks 5–9, a longer duration of transfer was associated with an increased number of animals in the house. In NZW mice, significantly more animals were in the house when tail-handled compared with tunnel handling (
Exploratory behaviour in the used cage
Figure 3(a) illustrates that the exploratory behaviour, which was documented as ‘explore the cage attentively’, in the used cage increased significantly from week 1 to week 4 (
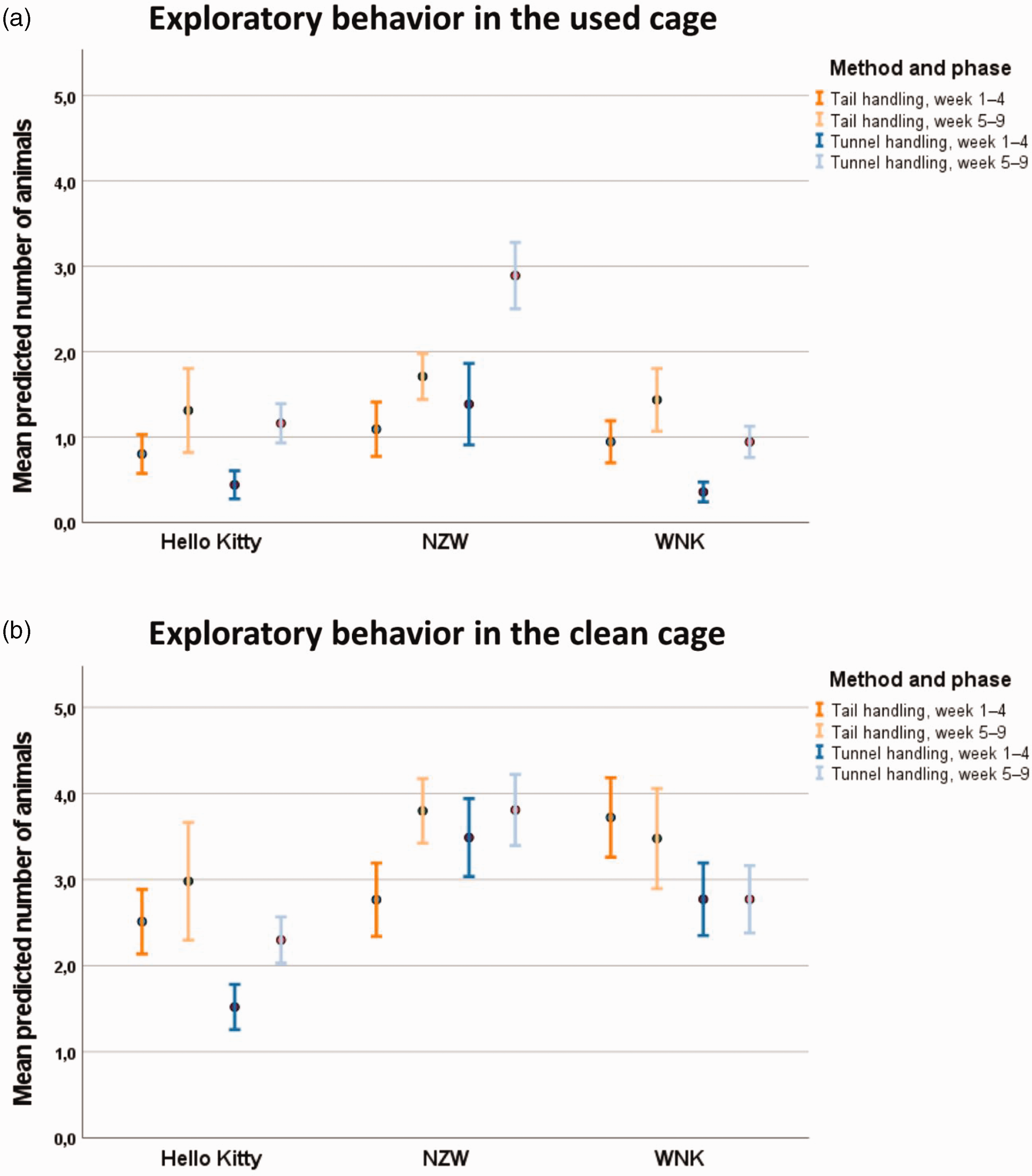
Number of mice showing exploratory behaviour in the (a) used cage and (b) clean cage after all mice of the group had been transferred. Data are shown as mean and 95% confidence interval.
In the weeks 5–9, there were also significant differences between the weeks (
Moving fast into the house or nest in the used cage
There were no significant effects of any investigated factors on the observation that the mice moved fast into the house.
Voluntary contact to hand at capture
In weeks 1–4, only three of 169 tail-handled Hello Kitty mice did voluntarily contact the hand of the animal technicians during capture, while NZW (18% tunnel handling, 2% tail handling) and WNK (0 tunnel handling, 2% tail handling) were seeking contact (Figure 4). Due to the zeros in the data, confidence intervals of statistical tests were not meaningful and, thus, are not presented here.
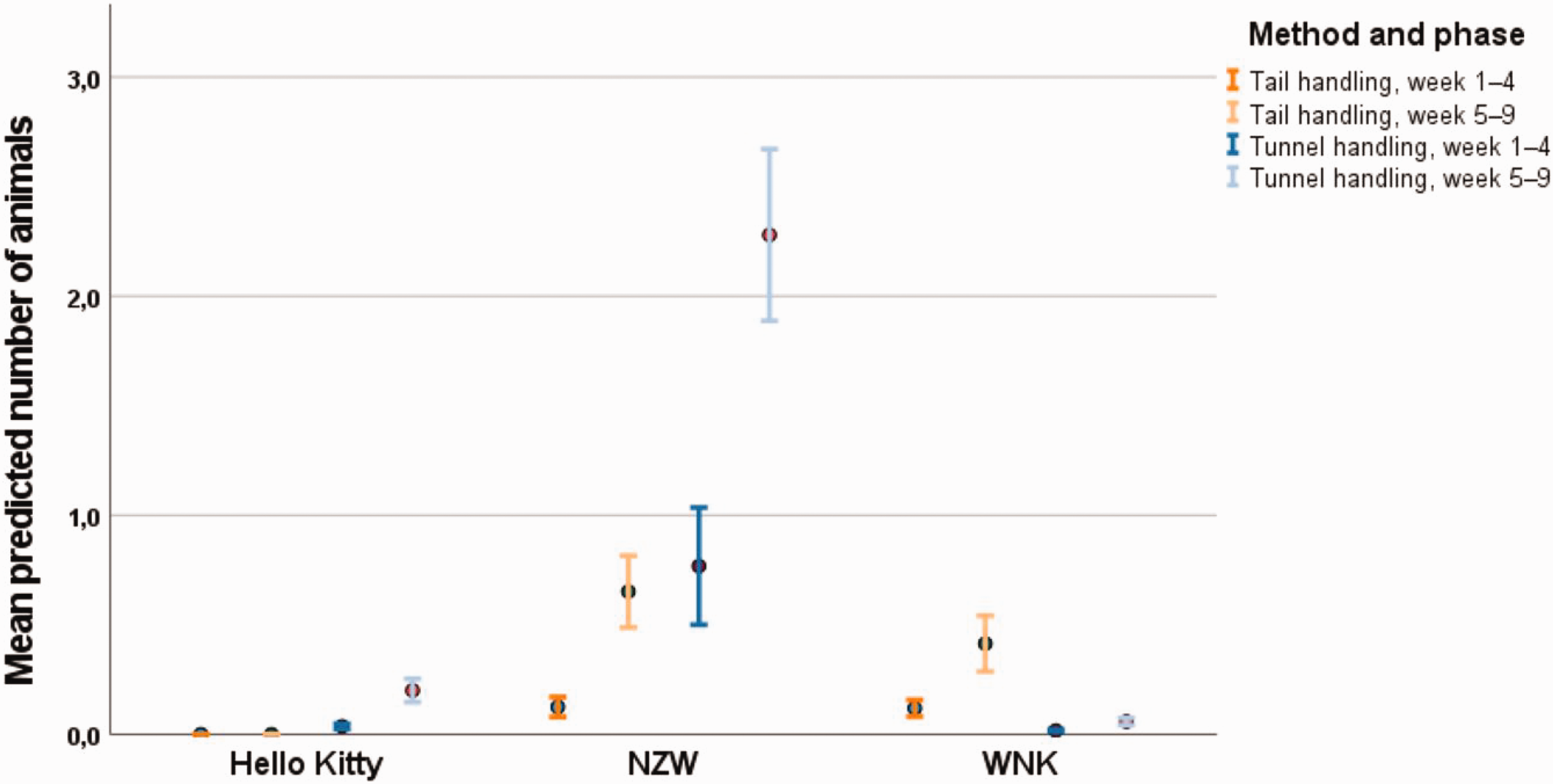
Voluntary contact to the animal technician’s hand when being captured. Data are shown as mean and 95% confidence interval.
In weeks 5–9, method, strain and their interaction were strongly associated with the voluntary contact to the animal technician’s hand at capture (
Defecation during capture
Strain (
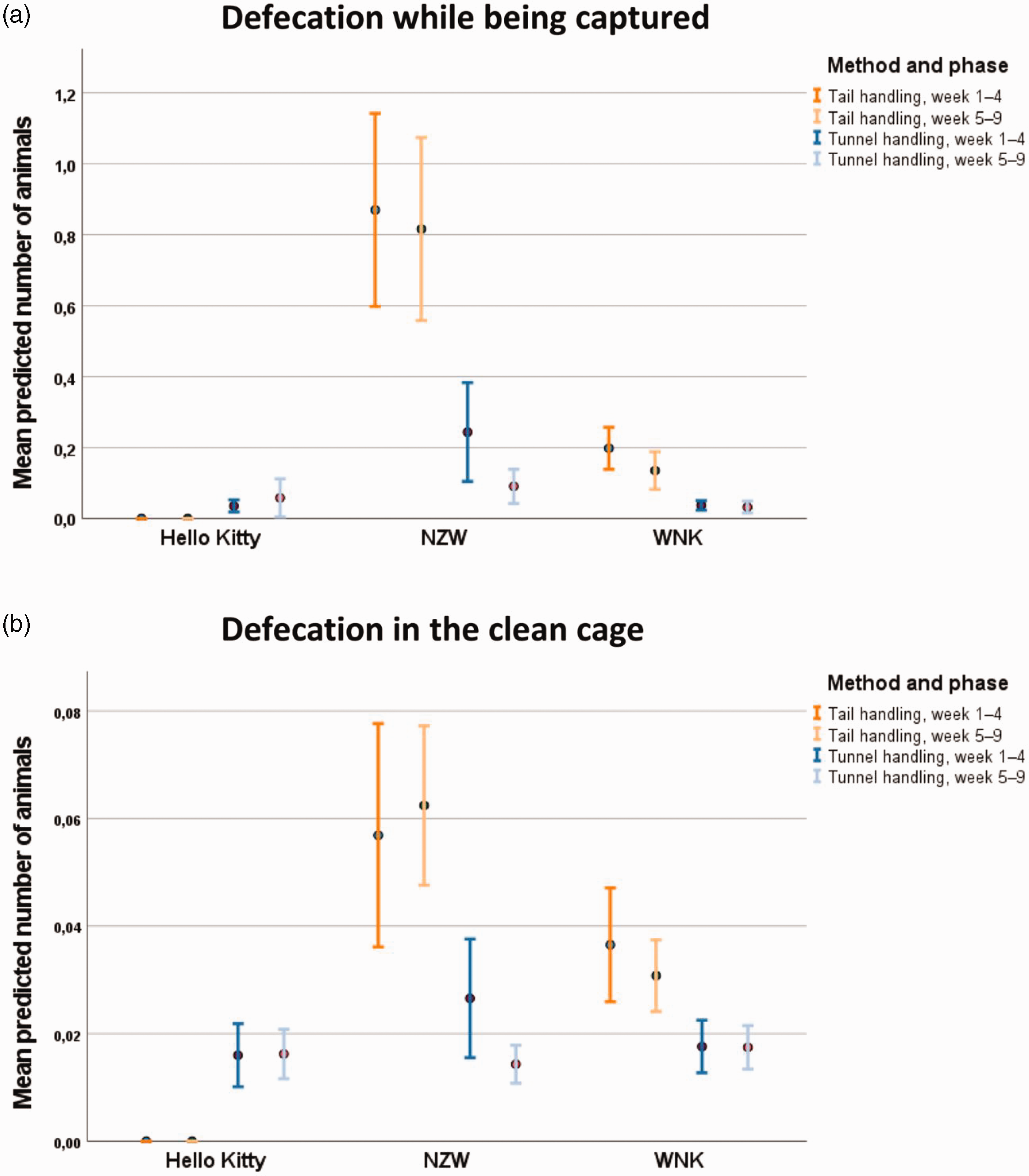
Defecation observed (a) while being captured in the used cage and (b) after release in the clean cage. Data are shown as mean and 95% confidence interval.
In weeks 5–9, it was found that method (
Defecation in clean cage
There were no significant effects of any of the investigated factors to defecation in clean cage (Figure 5(b)).
House residence in the clean cage after all mice from a group were transferred
After all mice of a group were transferred to the clean cage and the transfer process was documented, some mice were in the house in the clean cage. While this was only rarely the case in WNK mice in the first four weeks, more Hello Kitty and NZW mice were in the house, and for NZW mice, tail handling increased this behaviour compared with tunnel handling 17.3 times (
Compared with week 9, significantly more animals were sitting in the house in the clean cage than in weeks 5–8 (
Exploration in clean cage after all mice from a group were transferred
After all animals were transferred to the clean cage and the transfer process was documented, the behaviour of the animals was monitored again. In weeks 1–4, the exploration increased significantly over the weeks (
Even in weeks 5–9, there was a significant increase from week to week concerning the exploration of the clean cage (
Discussion
When introducing tunnel handling in a breeding unit of the FEM where the strains WNK, NZW, and Hello Kitty were kept, the feasibility for the animal technicians and the effects of this technique on the behaviour of the mice were examined over a period of nine weeks.
The behavioural parameters were found to be affected differently in the three strains and were partially associated with the time to transfer the mice from the used to the clean cage.
Over all strains, tunnel handling decreased defecation while being captured when compared with tail handling. Since defecation is an indicator for stress, the data suggested that tunnel-handled mice experienced less stress. Although this study did not involve regular tunnel handling training several times a week as reported in other protocols, this is in line with previous studies demonstrating that defecation was observed in fewer handling sessions when a tunnel was used instead of picking the mice up by the tail.1,13 This effect can even be amplified by positive reinforcement training. When cup-handled mice, that were trained by positive reinforcement, were restrained by the scruff, they defecated less than their untrained counterparts. 14 However, a closer look at the strain Hello Kitty revealed that fewer animals of this strain defecated during capture in comparison with NZW and WNK mice. Therefore, the relevance of this parameter in Hello Kitty mice may be questioned.
The voluntary interaction of the mice with the experimenter’s hand was investigated in various mouse handling-related studies, revealing a higher interaction time after tunnel than tail handling.1 –3 The voluntary interaction can give hints about the ‘anxiety-related behaviour in anticipation of handling’. 1 In the present study, the voluntary interaction was examined as a binary outcome (i.e. contact/no contact) instead of a duration of time because the routine cage change process should not be prolonged. Since the contact rate increased over time in the late study phase from weeks 5 to 9, the mice seemed to be less anxious in anticipation of handling and to habituate to the handling process. Gouveia and Hurst also demonstrated a higher voluntary interaction of tunnel- and cup-handled over several handling sessions, with a relatively high interaction time in mice picked up with a tunnel after the first handling session. 3 A clear effect of the handling method was found only for NZW, with higher voluntary contact in the tunnel-handled in comparison with the tail-handled mice. Results of the other strains were ambiguous because the contact rates generally were low and no significance was found.
The residence in the house was investigated at two points in time, that is, before and after handling. This behaviour was considered as seeking a safe place and hiding from the animal technician when the animal anticipated being handled or after it had the experience of handling. Nevertheless, the animal technicians could not differentiate whether the mice in the house in the used cage were sleeping or awake. In all strains, the predicted number of mice hiding in the house at both time points decreased in the late phase (weeks 5–9) in comparison with the early phase (weeks 1–4). This may indicate that the animals habituated to the process of being handled to a certain extent and perceived it as less aversive over time. In NZW, tunnel-handled mice were less often observed in the house than tail-handled mice at both time points, suggesting that tunnel handling was associated with less negative effects. A lower occurrence of house residence was also found in tunnel-handled Hello Kitty, although this effect was only present before handling in the early phase; in the late phase, both tail- and tunnel-handled mice showed this behaviour less often and did not significantly differ from each other. The same applied to Hello Kitty after handling and WNK at both observation times: fewer mice were found in the house in the late when compared with the early phase, independent of the handling method. Accordingly, both handling methods had a comparable habituation effect in these cases with regard to the parameter ‘house residence’.
The parameter ‘nest residence’ in the used cage was influenced by the handling method in NZW mice and, independent of the strain, the predicted number of mice sitting in the nest was lower in weeks 5–8 compared with week 9. Assuming that the mice were resting in the nest when the animal technician removed the cage from the rack and opened it, mice staying in the nest instead of leaving the nest and hiding in the house may be considered as less anxious in anticipation of being handled. Against this background, the results of this parameter may indicate that, in weeks 1–4, tunnel-handled NZW mice may show less anxiety-related behaviour before being handled than their tail-handled counterparts. Since the mice became increasingly familiar with the handling process, a higher number of mice were observed sitting in the nest in week 9. Exploratory behaviour was also observed before and after handling in the used (familiar) or clean (unfamiliar) cage. In the documentation, exploratory behaviour was distinguished from flight behaviour (i.e. moving fast). In contrast to hiding in the house, exploratory activity was considered to be associated with lower levels of anxiety-related behaviour. Previous studies revealed that the use of tunnels decreased anxiety-related behaviour in the open field test or elevated plus maze test when compared with tail handling.1,4,5 The increase of exploration over time before and after handling may be explained by the habituation effect. A habituation effect was also observed with regard to the time-dependent increase in voluntary contact with the animal technician’s hand and the house residence. The animals became increasingly familiar with the process of handling. In the late phase, this effect was the strongest in tunnel-handled NZW showing higher exploration before handling when compared with their tail-handled counterparts. After handling, the method was not found to have a significant effect on this parameter. However, to draw conclusions on strain-specific effects of the handling methods on anxiety-related behaviour, additional validated behaviour tests should be carried out.
The time factor usually is a critical issue that is discussed when replacing the traditional tail handling technique with tunnel handling. 8 There are concerns that tunnel handling will increase time and financial costs.7,8 Therefore, in the present study, the time to transfer the animals from the used to the clean cage was measured. Indeed, it took the animal technicians approximately 3 s longer per mouse when using the tunnel. Depending on the strain, they became faster over time, at least in NZW mice that may have habituated better to being handled by the tunnel than the other strains. Doerning et al. also reported tunnel handling to take longer time than picking the mice up by their tail. 15 It must be considered that, in the present study, the animals were handled by animal technicians with more than 10 years of experience who may have better skills in handling mice than inexperienced animal technicians. Therefore, it is conceivable that the time difference in the transfer time might be higher if persons with less experience handled the animals.
Moreover, it must be taken into account that, in the present study, the animal technicians were used to tail handling but only basic-trained to tunnel handling at the beginning of the study and, therefore, required more time to pick up the mice with a tunnel. If they had already been familiar with tunnel handling for a longer period, they may have required less time applying this method. Nevertheless, this effect should not be overestimated. The observation of transfer time and behaviour was carried out over a period of several months, that is, the technicians were familiar with tunnel handling during later phases of the study. In general, the learning process of the animal facility and scientific staff would be circumvented if non-aversive handling methods was part of their education. 8 In an international survey on mouse handling techniques, a few participants even stated that tunnel handling was faster and/or more efficient than tail handling 7 . The time difference between the handling methods detected in the present study was a few seconds. However, the staff of animal facilities is concerned that this amount of time could ‘result in a significant overall increase in the time required to process large numbers of cages’. 7 This issue of additional labour, time or costs has to be calculated and discussed for each individual case, considering the benefits of tunnel handling to animal welfare and scientific quality.
The data analysis revealed that the transfer time depended on the strain and was associated with the residence in the nest or house and the exploratory behaviour in the used cage. The more animals were in the nest or house, the longer the transfer time. In contrast, the number of mice exploring the used cage was negatively associated with the time required to transfer the animals. These observations can be attributed to the fact that mice exploring their environment may have a higher tendency to explore and enter the tunnel than those sitting in the nest or house. It must be noted that the house/nest was removed from the cage before the animals were transferred so that the house itself cannot be the reason for the higher transfer time.
Interestingly, a longer transfer time was also associated with higher defecation (i.e. higher stress levels), which may be interpreted in two ways. A longer lasting handling process may increase the stress levels of the animals. Against the background that tail handling generally caused more defecation than tunnel handling, long-lasting tail handling sessions induced the highest defecation rate. Vice versa, it may take the animal technicians longer to handle and transfer mice with elevated stress levels.
The main limitations of this study were that the animal technicians handling the animals and collecting the data were not blinded. Blinding the handler did not appear to be possible. The data collection could have been blinded if the handling process had been video-recorded. Since, however, the study was carried out during routine husbandry procedures, video recordings would have disrupted the animal technicians’ workflow and therefore were not made. For the same reason, the animals were only monitored in their home cage and no additional behavioural tests were performed. Therefore, further advanced behavioural tests would be needed to confirm the present data. Since, to the knowledge of the authors, there are no behavioural phenotyping studies comparing the three mouse strains investigated or providing a detailed behavioural profile of them, the strain-specific findings of the present study cannot be discussed against the background of such studies.
Another limitation of the present study may be the age of the mice, since handling and observations started from weaning at the age of approximately three weeks to a maximum of 12 weeks after weaning. In this period, the physiology and behaviour of the mice can be influenced by the weaning process and the transition from infancy to adolescence. Mice older than 12 weeks may respond differently (or more clearly) to tunnel handling and tail handling with regard to the parameters investigated in this study.
Conclusion
Tunnel handling was implemented in a mouse breeding facility during routine husbandry which included weekly handling of the mice for cage changes. The impact of the handling techniques on the behavioural parameters investigated in the present study was strain-specific. All parameters suggested that NZW mice benefited the most from tunnel handling. The results obtained from Hello Kitty and WNK mice were ambiguous, which may suggest that a brief handling session during the cage clean may have not been sufficient to habituate the mice of these strains to the process of handling. Nevertheless, it was possible to use the tunnel in all three mouse strains. Whether the benefits of non-aversive mouse handling outweigh the amount of additional time expenditure must be considered against the background of related studies on tunnel handling. Since there are hints for slight strain differences in the responses to tunnel handling, it may be worth encouraging researchers to report their experiences with tunnel handling along with their research articles or other platforms such as Norecopa’s Refinement Wiki (https://wiki.norecopa.no).
Supplemental Material
sj-xlsx-1-lan-10.1177_00236772231215077 - Supplemental material for The implementation of tunnel handling in a mouse breeding facility revealed strain-specific behavioural responses
Supplemental material, sj-xlsx-1-lan-10.1177_00236772231215077 for The implementation of tunnel handling in a mouse breeding facility revealed strain-specific behavioural responses by Katharina Hohlbaum, Roswitha Merle, Ramona Warnke, Stefan Nagel-Riedasch, Christa Thöne-Reineke and Kristina Ullmann in Laboratory Animals
Footnotes
Acknowledgements
We thank the animal technicians for being open to new handling techniques and carefully documenting their observations. The funder had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript.
Data availability
All relevant data are provided upon request.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Charité 3R, Charité – Universitätsmedizin Berlin. KH and CTR were funded by the Deutsche Forschungsgemeinschaft (DFG, German Research Foundation) under Germany’s Excellence Strategy – EXC 2002/1 ‘Science of Intelligence’ – project number 390523135.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
