Abstract
Fibrosis, as a common final pathway in many renal diseases, contributes significantly to the decline of organ function and to progression to end-stage renal disease. To establish therapeutic interventions that target fibrosis, animal models are essential. The most widely used model of renal fibrosis is the unilateral ureteral obstruction (UUO) model. Typically, the control for this model is a sham-operated animal. Sham surgery causes pain and distress to these control animals, and here we aim to show that there is no difference in the main read-outs of this model between sham-operated animals and non-operated C57BL/6J mice. In five experiments, quantification of Picro Sirius Red stained collagen in the renal cortex did not show any difference between 15 sham and 25 non-operated individuals. A comparison of the regulation of genes involved with fibrosis did not show any difference between sham and non-operated groups at 21 days post surgery either. We conclude that there are no significant differences between non-operated controls and sham animals with respect to collagen deposition and fibrosis pathways in the UUO mouse model.
Introduction
Fibrotic remodelling is implicated in 45% of deaths in the Western world, 1 and therefore represents a significant global health burden. There are currently only two approved therapeutics for fibrosis (pirfenidone and nintedanib), and both are only approved for idiopathic pulmonary fibrosis.2,3 There is therefore significant unmet medical need in fibrotic diseases of other organs, including the kidney. In order to develop drugs to treat fibrosis, it is essential first to profile the efficacy of these drugs in both in vitro and in vivo models.
The most commonly used in vivo model of kidney fibrosis is the unilateral ureteral obstruction (UUO) model, as it has similar pathological hallmarks to human chronic kidney disease, such as tubular atrophy, interstitial fibrosis development and matrix deposition. 4 In terms of human disease, the UUO model is most similar to obstructive nephropathy, which is the leading cause of end-stage kidney disease in children. 5 The surgical procedure required for this model is relatively simple, with a small incision being made in the left flank of the animal, the ureter located and ligated. 6 After a complete UUO, there is a progressive decline in renal blood flow, a decline of glomerular filtration rate in the kidney with the ligated ureter and a progressive loss of renal medullary parenchyma. 7 Most of the renal changes in this model occur after 7 days. An increase in tubular pressure, leading to distended tubules, 8 damage to the tubular epithelial cells, 9 infiltration of macrophages 10 and myofibroblasts 9 all begin to increase 7 days after ligation. These cells release inflammatory and pro-fibrotic mediators such as transforming growth factor beta (TGF-β) which drive the deposition of extracellular matrix (ECM). 11 ECM typically begins to accumulate in the interstitial space seven days after ligation and increases until 21 days, after which there is no progressive increase in ECM.
The typical experimental control for induction of disease in the UUO model is a sham-operated animal. 6 This is to correct the original use of the contralateral (untied) kidney as a control, as there are considerable systemic changes in a UUO animal. 12 However, despite appropriate analgesia, sham surgery causes pain and distress to animals. In the interest of the 3Rs, we sought to determine if non-operated animals could be used as an adequate control instead, as has recently been shown in the ovariectomised rat model of osteoporosis. 13
Methods
Animals and husbandry
Housing conditions, husbandry and procedures were carried out under license according to regulations laid down by Her Majesty’s Government, UK (Animals Scientific Procedures Act 1986) and with approval of the Animal Welfare and Ethics Review Body of UCB, Slough, UK. The UUO procedure was performed in male C57Bl6/J mice (8–10 weeks old, weighing 20–30 g) from Charles River (Edinburgh, UK). Animals were housed in open-top cages, in a specific-pathogen-free facility according to the Federation of European Laboratory Animal Science Associations recommendations. 14 Typically, animals were housed in groups of four or five. Food (R05-10; SAFE, Paris, France) and water (untreated mains tap water) were available ad libitum, and the light cycle was 12/12 hours light and dark. Room temperature and humidity were 21℃ and 45%, respectively. Nest building material was provided, and all cages contained enrichment such as a red translucent house, a wheel and a tunnel.
Animals were the experimental unit and were randomly allocated to the groups non-operated, sham or UUO in a computer-generated manner. Animal numbers per group were determined by power calculations using an online tool from the AEEC (www.lasec.cuhk.edu.hk/sample-size-calculation.html), with five animals deemed optimum to attain a statistical significance of
Surgery
Animals were administered preoperative analgesia subcutaneously (buprenorphine 0.05 mg/kg and carprofen 5 mg/kg) and anaesthetised using 5% isoflurane and a flow rate of 2 L/min. Following induction, anaesthesia was maintained with 2–3% isoflurane and a flow rate of 2 L/min. This is the most appropriate form of anaesthesia due to the short surgery time, the lack of renal elimination and the reduced risk of damage from intraperitoneal injections to the internal organs. The animal was then laid on its right side on a heated surgery table (Vet Tech, Congleton, UK), and the left flank was shaved and disinfected with diluted chlorhexidine. The animal was covered with a sterile drape with a small window for surgery. An incision was made with a scalpel on the left flank beneath the ribcage, proceeding vertically downwards for ∼5 mm. The fat was cleared and abdominal muscles parted with blunt dissection. The ureter was then located and ligated with a double ligature of 5-0 Mersilk (Ethicon, Reading, UK) and placed back. For sham animals, the ureter was located and then placed back without ligation. The muscle wall was repaired using a continuous suture of 5-0 Vicryl (Ethicon), and the skin wound was closed using a subcuticular suture of 5-0 Vicryl (Ethicon). Typically, sham operations were performed before UUO operations. Levobupivicaine (0.5 mg/kg) was injected subcutaneously at the wound site. All surgery was performed between 9:00am and 1:00 pm. Animals were allowed to recover in a warmed recovery box before being returned to the home cage. Non-operated animals were not anaesthetised or manipulated.
Postoperative care
Mouse UUO scoring sheet detailing body weight scoring, pain scoring, appearance, termination criteria and actions required based on grimace scoring.
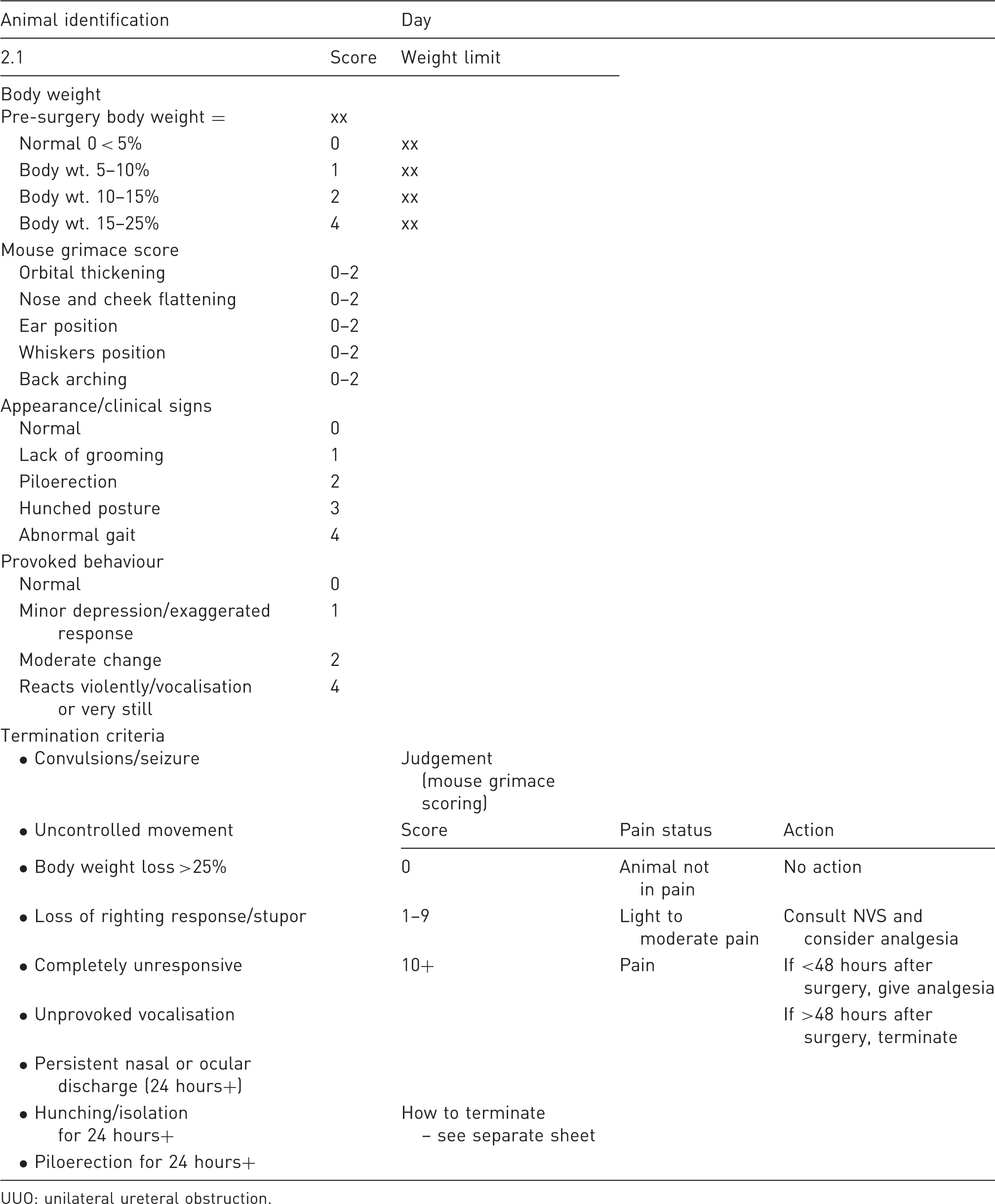
UUO: unilateral ureteral obstruction.
Pre- and postoperative analgesia regimen following UUO surgery.
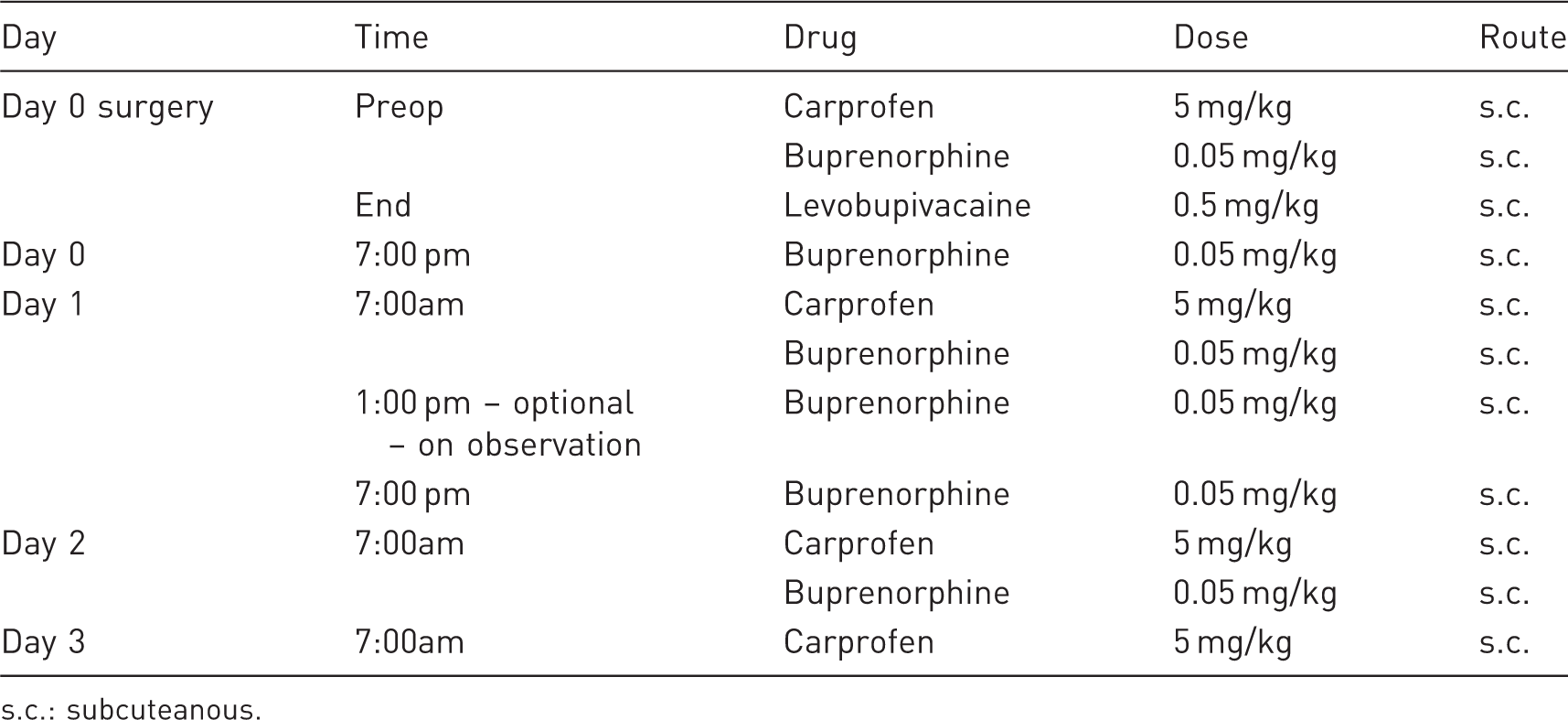
s.c.: subcuteanous.
Fibrosis quantification
Kidneys were halved longitudinally pole to pole through the papilla, with equal cortex and medullary mass in each. Half kidney samples for histology were fixed in 4% paraformaldehyde, embedded in paraffin wax and sectioned at 5 µm thickness. The extent of scarring was assessed in kidneys using Picro Sirius Red (PSR) staining (Sigma–Aldrich, St Louis, MO). Quantification of PSR staining was determined by Definiens image analysis (Definiens, Munich, Germany) in a blinded manner. Briefly, whole-slide scans of PSR-stained kidneys were obtained using a NanoZoomer slide scanner (Hamamatsu Photonics, Hamamatsu, Japan). Next, these scans were uploaded into Definiens Tissue Studio. The Stain Picker tool was then used to train the software to detect nuclei, with the red PSR stain as ‘marker 1’. The cortex was manually annotated, and the software then automatically quantified the amount of ‘marker 1’ staining in the cortex. This was then calculated as the percentage area collagen stain.
Transcriptome analysis
RNA extraction and microarray analyses were performed by Almac Diagnostics, Craigavon, UK. In brief, RNA was extracted from snap-frozen tissue following the RNA-Stat 60 solvent (AMSBIO, Abingdon, UK) extraction protocol. Further purification and elution were carried out using RNeasy MiniElute (Qiagen, Hilden, Germany) spin columns. Purified RNA was amplified using the NuGENTM OvationTM RNA Amplification System V2 (NuGEN, Redwood City, CA), cDNA was subsequently fragmented and labelled with the Encore Biotin Module (NuGEN). Hybridisations were carried out using Affymetrix GeneChip® Human Genome U133 Plus 2.0 arrays (Thermo Fisher Scientific, Altrincham, UK) for 18 hours at 45℃, washed, stained and scanned with a GeneChip® Scanner 3000 (Thermo Fisher Scientific). Raw data (Cel) files were processed with Genedata Expression-ist® 9.1 Refiner Array (Genedata, Basel, Switzerland) using robust multi-array average subtraction and quantile normalisation. Analysis of data was then performed using Genedata Analyst (Genedata) and exported to GraphPad Prism (GraphPad Software, La Jolla, CA) for histograms.
Statistics
A biostatistician was consulted for statistical tests. For PSR analysis, a Kruskal–Wallis test was used, and statistical significance assumed at the 5% level. For transcriptomics analysis, data were log transformed (
Results
Observations
UUO surgery is typically well tolerated. All animals in this study lost 1–2 g post cessation of opioid analgesia, but regained this weight within three days. As this model does not progress to renal failure, animals all presented as normal in appearance and weight.
Collagen quantification
Renal collagen accumulation was determined by PSR staining. A broad overview of these images clearly shows increased PSR red staining in the UUO animals compared to either control, which is readily apparent when focusing on the tubulo-interstitial region (Figure 1(a) and (b)). The percentage collagen staining was significantly elevated in the UUO group (22.8-fold increase). However, there was no statistical difference between sham-operated and non-operated animals (Figure 2).
Picro Sirius Red (PSR) staining of the sham, non-operated and unilateral ureteral obstruction (UUO) kidneys at day 21 reveals broader red staining in the whole-slide overviews (a), indicating increased extracellular matrix (ECM) deposition throughout the cortex and the medulla, and in the tubulo-interstitial regions of the cortex (b), with increased cytoplasmic staining, suggesting upregulated collagen production by these cells, and thickened tubular basement membranes, suggesting increased collagen deposition or reduced collagen breakdown. Quantification of PSR staining in the renal cortex. UUO mice shows significant upregulation of collagen staining in the UUO kidneys compared to both sham and non-operated animals. Statistically, there is no difference between non-operated and sham animals. 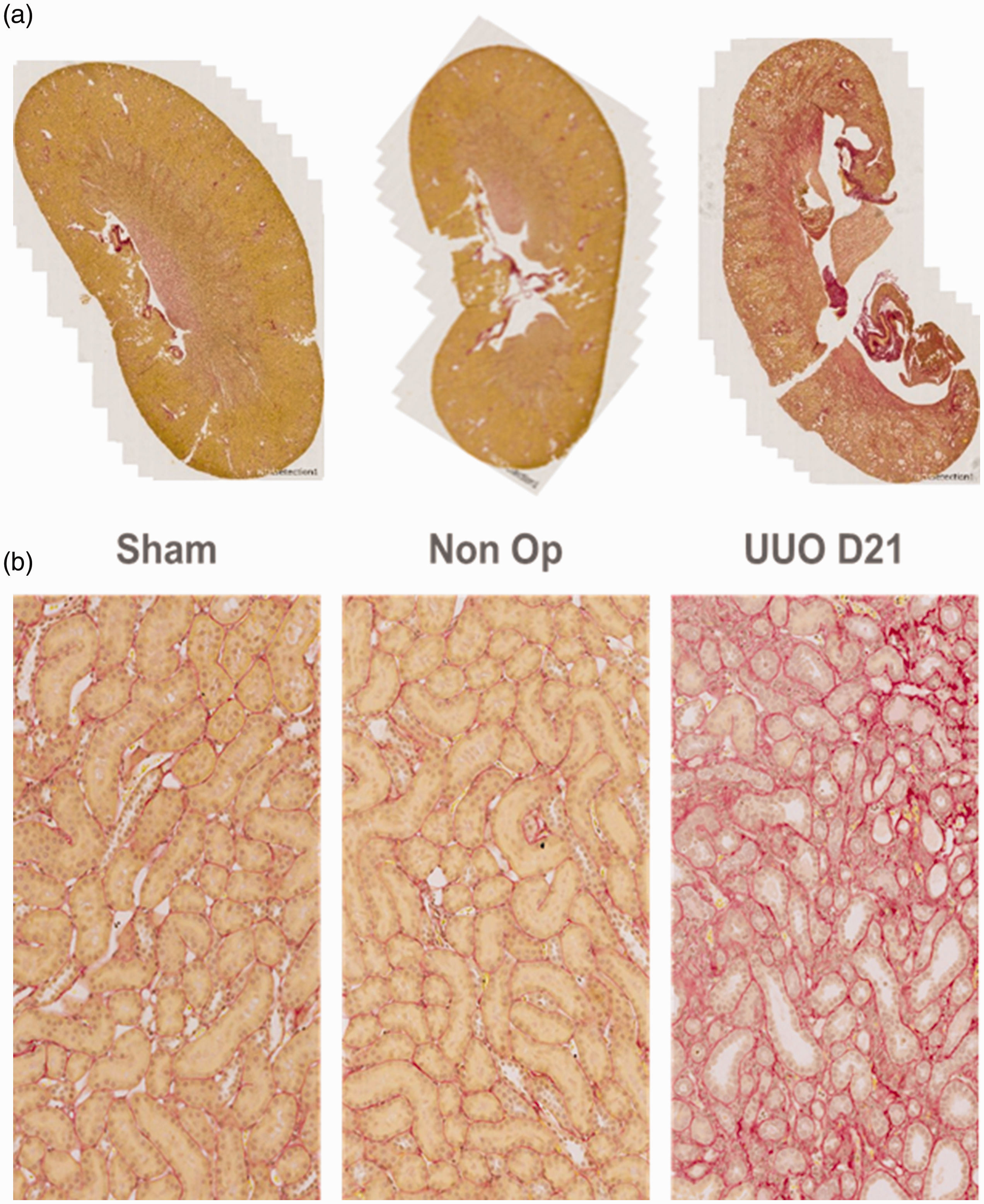
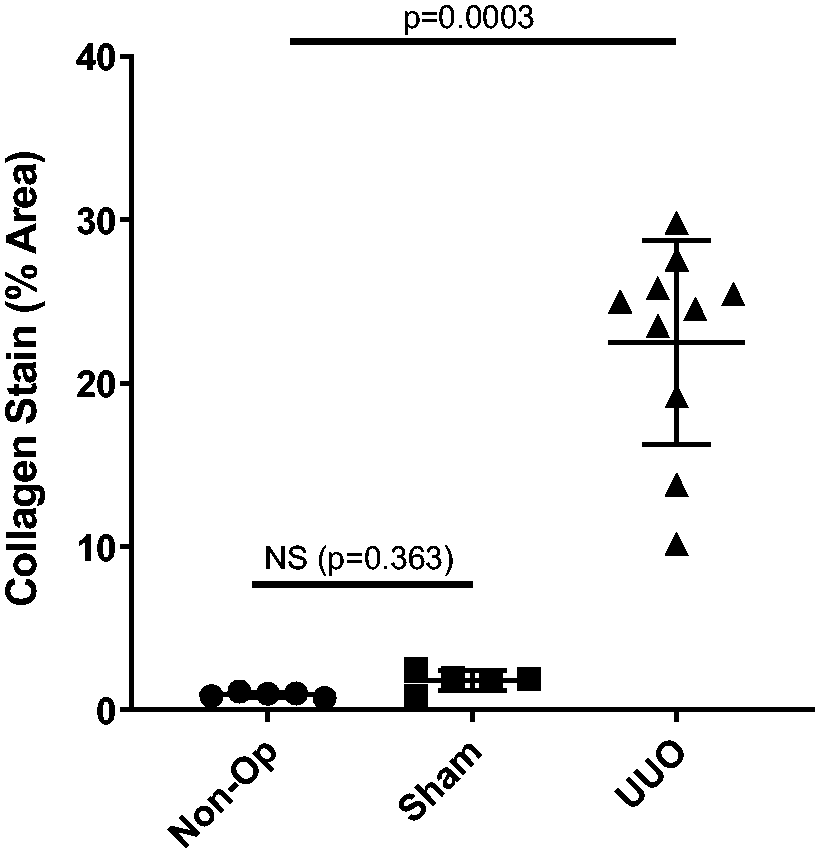
Encouraged by these results, we included non-operated and sham-operated groups in future UUO studies. The combined data from these studies are summarised in Figure 3, showing that across a further four studies, there was no significant difference between non-operated and sham-operated animals in the PSR read-out.
Quantification of PSR staining in the renal cortex in four further studies. In UUO mice, there is significant upregulation of collagen staining in the UUO kidneys compared to both sham and non-operated animals. Statistically, there is no difference between non-operated and sham animals. 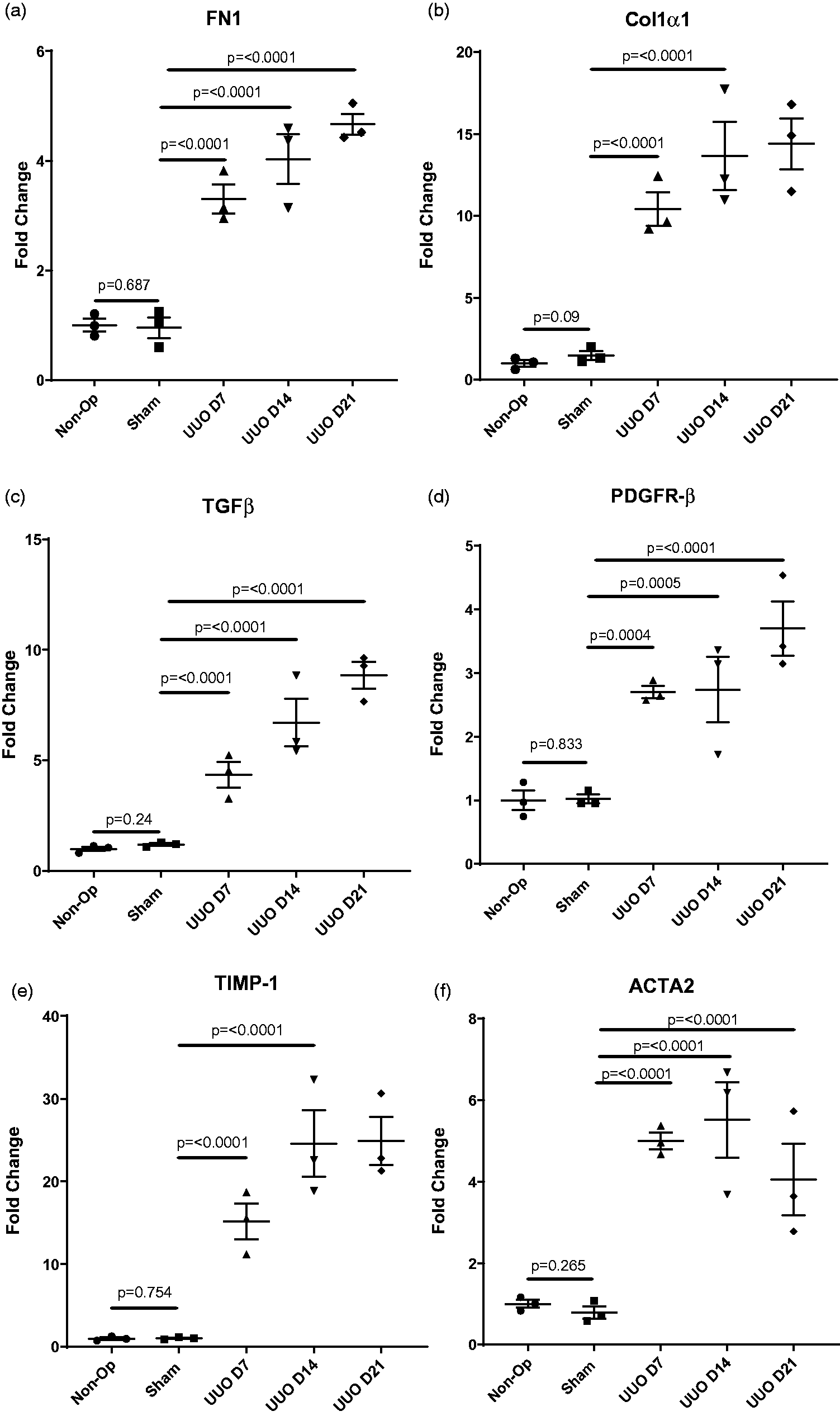
Transcriptome analysis
Transcriptome analysis from snap-frozen half kidneys was determined by Affymetrix microarray analysis (Affymetrix, Santa Clara, CA). We saw significant upregulation of fibronectin (Figure 4(a); ×3.3-fold at day 7, ×4-fold at day 14 and ×4.5-fold at day 21), collagen 1α1 (Figure 4(b); ×10-fold at day 7, ×13-fold at day 14 and ×14-fold at day 21). We saw significant upregulation of TGF-β (Figure 4(c); ×4.5-fold at day 7, ×6.6-fold at day 14 and ×8.8-fold at day 21), along with platelet-derived growth factor receptor beta (PDGFR-β; Figure 4(d); ×2.7-fold at day 7, ×2.7-fold at day 14 and ×3.6-fold at day 21). We also saw significant upregulation of TIMP1 (Figure 4(e); ×15-fold at day 7, ×25-fold at day 14 and ×25-fold at day 21) and ACTA2 (Figure 4(f); ×5-fold at day 7, ×5.5-fold at day 14 and ×4-fold at day 21). None of these gene regulations were significantly different between non-operated animals and sham-operated animals (Figure 4).
Transcription analysis of UUO kidneys showed upregulation of genes coding for structural components of the ECM ((a) and (b)), pro-fibrotic signalling molecules ((c) and (d)), matrix processing inhibitors (e) and myofibroblast differentiation (f). 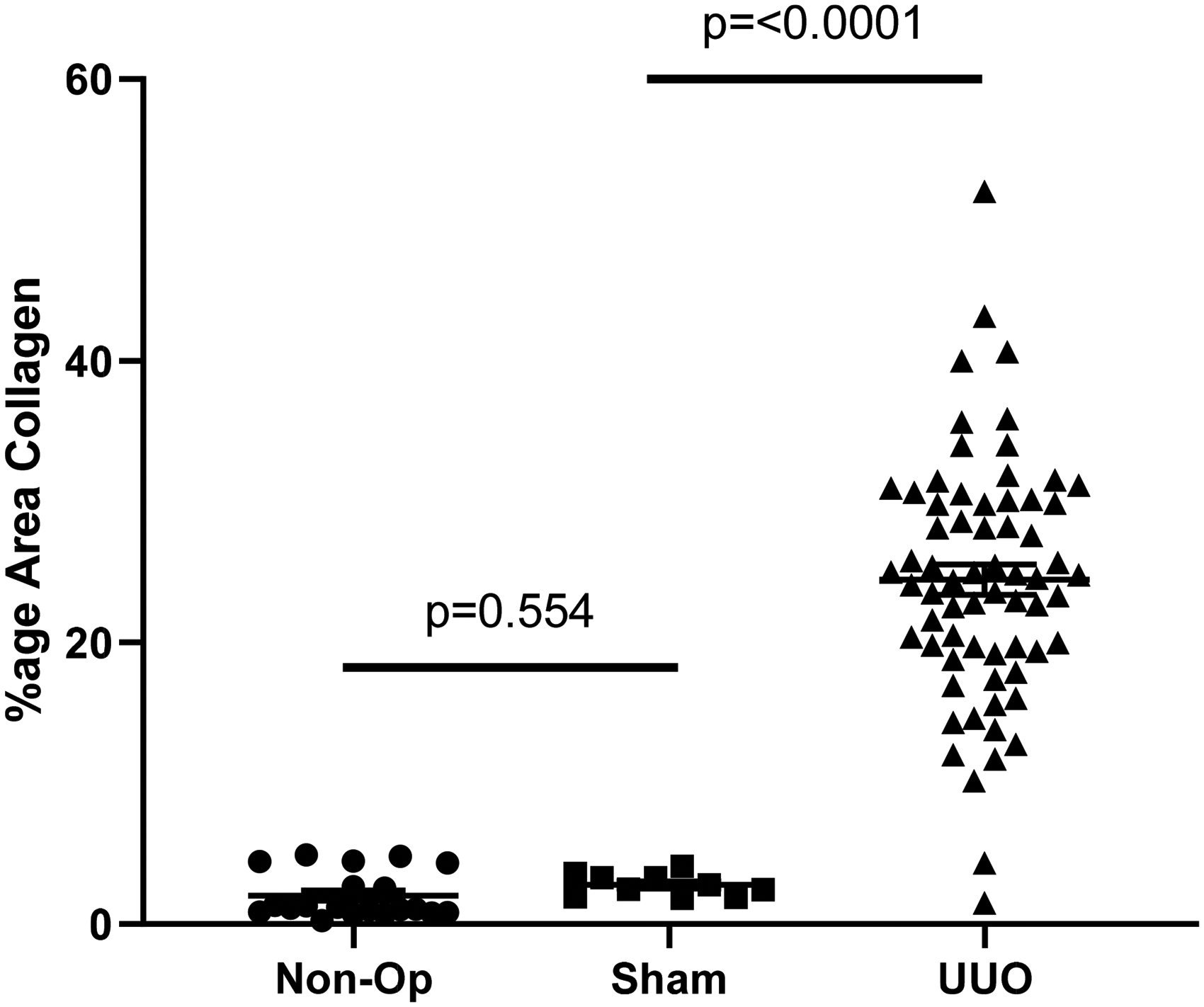
Discussion
The UUO model is the most widely used model of renal fibrosis. This is because it displays the major hallmark of renal fibrosis: tubulo-interstitial accumulation of ECM composed primarily of collagen. Here, we have shown that the kidneys of mice accumulate significant amounts of collagen in response to UUO surgery. We have also shown that there is no ECM accumulation in the kidneys of sham-operated animals or non-operated animals, and therefore that the use of non-operated animals to replace sham-operated animals would be justified in the read-out of collagen accumulation.
However, several other mechanisms contribute to renal fibrosis. To discover if non-operated animals could truly replace sham-operated animals as a standard control for this model, we sought to determine if there was any difference in the mRNA expression of key proteins involved in the mechanisms of fibrosis. These mechanisms were ECM accumulation, pro-fibrotic signalling, inhibition of matrix processing and myofibroblast activation. Here, we showed there was significant upregulation at three time points (day 7, day 14 and day 21) in the UUO model in the expression of key ECM protein fibronectin (FN1). There was no difference in the expression of fibronectin mRNA between non-operated animals and sham-operated animals. Consistent with the PSR data, we showed that there was significant upregulation of the gene coding for the α1 chain of collagen 1, Col1α1, again at days 7, 14 and 21, with no difference between non-operated animals and sham-operated animals.
As well as components of the ECM, we showed that two key regulators of pro-fibrotic signalling, TGF-β and PDGFR-β, had significantly elevated expression at days 7, 14 and 21, with no difference in expression between sham-operated animals and non-operated animals. Tissue inhibitor of metalloprotease 1 (TIMP1) plays a key role in fibrosis by preventing ECM breakdown by matrix metalloproteases. Here, we showed that TIMP1 had upregulated expression at days 7, 14 and 21 after UUO. Again, there was no difference in TIMP1 expression between sham-operated animals and non-operated animals. Finally, we examined a key marker of myofibroblast activation, αSMA, which is coded for by the gene
Conclusion
We have clearly shown there is no confounding collagen accumulation in the kidneys of mice subjected to sham operations. There is also no upregulation of genes involved in any of the major pathways of fibrosis 21 days post surgery: pro-fibrotic signalling, blockade of matrix degradation, myofibroblast activation or ECM protein production. There is clearly no difference between the kidneys of sham-operated animals and non-operated animals in terms of ECM accumulation or mRNA analysis. It is possible that a proteomics approach could detect differences. We therefore conclude that non-operated animals are appropriate controls for collagen accumulation read-outs and transcriptomic profiles in UUO surgery and, in the interest of the 3Rs, should replace sham operations immediately.
Footnotes
Acknowledgements
The authors would like to thank Gareth Davies, Mark Hasler, Jen Needham, Helen Brodigan, Linghong Huang, Remi Okoye and Tanika Duncan for their assistance in generating these data.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship and/or publication of this article.
