Abstract
This case report addresses the problem of underreporting negative results and adverse side effects in animal testing. We present our findings regarding a hyperphagic mouse model associated with unforeseen high mortality. The results outline the necessity of reporting detailed information in the literature to avoid duplication.
Obese mouse models are essential in the study of obesity, metabolic syndrome and diabetes mellitus. An experimental model of obesity can be induced by the administration of gold thioglucose (GTG). After transcending the blood-brain barrier, the GTG molecule interacts with regions of the ventromedial hypothalamus, thereby primarily targeting glucose-sensitive neurons. When these neurons are impaired, mice become insensitive to the satiety effects of glucose and develop hyperphagia. In a pilot study for optimising dosage and body weight development, C57BL/6 mice were treated with GTG (0.5 mg/g body weight) or saline, respectively. Animals were provided a physiological amount of standard diet (5 g per animal) for the first 24 hours after treatment to prevent gastric dilatation. Within 24 hours after GTG injection, all GTG-treated animals died of gastric overload and subsequent circulatory shock. Animals developed severe attacks of hyperphagia, and as the amount of provided chow was restricted, mice exhibited unforeseen pica and ingested bedding material. These observations strongly suggest that restricted feeding is contraindicated concerning GTG application. Presumably, the impulse of excessive food intake was a strong driving force. Therefore, the actual degree of suffering in the GTG-induced model of hyperphagia should be revised from moderate to severe.
Introduction
An acknowledged experimental technique aiming for massive weight gain by inducing a hyperphagic state is the administration of gold thioglucose (GTG).1,2 Intraperitoneal injection of GTG induces an area of necrosis of ventromedial hypothalamic (VMH) regions, presumably by primarily targeting glucose-sensitive neurons.3–5 The onset and extent of VMH lesions is dose dependent, and lesions become histologically apparent after 12 hours. 6
Physiologically, the calorie intake in mice is dependent on energy expenditure, and under standardised experimental husbandry conditions and ad libitum chow supply, most mice strains develop no clinical signs of obesity. 5 However, GTG-treated mice develop hyperphagia, in which glucose administration induces no satiety effect. 4 In contrast, the administration of adrenal glucocorticoids can resolve hyperphagia and obesity, suggesting that GTG-induced hypothalamic lesions are at least in part dependent on adrenal glands. 6
The GTG mouse model is still used, for example, to discover the mechanism of agents improving insulin resistance and preventing obesity 7 and to study the role of microRNA in insulin-resistant conditions, such as obesity or type 2 diabetes. 8
Case presentation
The intention of this study was to investigate the impact of obesity on specific recombinant congenic mice. As expected, without treatment, mice showed normal eating behaviour and stayed slim. To induce obesity, hyperphagia was induced by GTG administration. It is well known that metabolic responses in mice are prone to genetic factors and certain subtle environmental factors. 9 Intensive literature research revealed that varying doses of GTG were used in previous studies: 0.2 mg/g, 8 0.3 mg/g, 10 0.4–0.8 mg/g,11–14 1.5 mg/g 15 and 2 mg/g.8,16 To determine a suitable GTG dose (i.e. induction of hyperphagia, associated with an increase in body weight (BW), plus good compatibility), a dose-finding experiment was conducted in C57BL/6 mice. C57BL/6 is a standard experimental mouse strain that is most frequently used for diabetes and obesity research.9,13,14,16,17 In the experimental setting, C57BL/6 mice represent the genetic background of mutant mice. C57BL/6 mice were bred and maintained in the German Institute of Human Nutrition (Nuthetal, Germany) under specific pathogen-free (SPF) conditions and fed a standard chow (prod. no. 1310, Altromin, Lage, Germany). Animals had access to water ad libitum. Mice were kept under standard laboratory housing conditions with a diurnal 12:12-hour light and dark cycle with lights off after a dimming period of 30 minutes at 6:30 p.m. To avoid potential conflicts over food resources due to restricted feeding, animals were singly housed for the duration of the pilot study (24 hours). Individually ventilated cages were equipped with aspen wood chips (LTE E-001; ABEDD Vertrieb GmbH, Wien, Austria), and a paper towel and cotton fibre nestlet (Alpha Nest; LBS Serving Biotechnology, Horley, UK) were provided weekly as nesting material. BW, food and water intake were monitored. All animal experiments were performed in compliance with the German animal protection law (TierSchG). Mice were housed and handled in accordance with good animal practice as defined by the Federation for Laboratory Animal Science Associations (www.felasa.eu/guidelines.php) and the national animal welfare body GV-SOLAS (www.gv-solas.de/index.html). The animal welfare committees approved all animal experiments (LUGV Brandenburg, permit number: 23-2347-1-2010).
Since male mice were the targeted models for the designated study, only males were used for this pilot study. Five-week-old male C57BL/6 mice (n = 8) received an intraperitoneal injection of GTG dissolved in saline (AuSC6H11O5; Sigma Aldrich, Germany). Because GTG concentrations were highly variable in previously published studies, we began the dose-finding study with a relatively low GTG dose. If this dose had not resulted in a hyperphagic state, the study was planned to continue with a medium or higher dose, respectively. Therefore, the following doses used in previous studies were also used in this study: 0.5 mg,11,12,14 0.8 mg4,14,18 and 1.15 mg 15 per g BW. Control animals received saline only. To prevent gastric dilatation through the onset of hyperphagia, animals were subjected to restricted feeding in physiological amounts (5 g/d) with the intention of successively augmenting chow quantity. Animals were treated with GTG (0.5 mg/g BW, n = 8; controls, n = 4) at 10:00 a.m. (light phase) and monitored until 10:00 p.m. (dark phase). Monitoring was conducted according to a score sheet that is routinely used in the animal facility and approved by local authorities. This regimen includes observing behaviour, gait, posture, fur coat, potential eye or nose effusions, breathing and structure of faeces. Animals were monitored each hour for 10 minutes in the first 12 hours after GTG administration.
In that time lapse, no changes in behaviour, signs indicating suffering or pain, or hyperphagia were observed. At 7:00 a.m. the next day, all eight GTG animals had died, while all control mice survived. In GTG-treated mice, a massive bulge in the abdominal region could be observed. Sectioning revealed severe distension of the stomach, while the duodenal and jejunal sectors were not distended, and the ileum was only slightly filled with ingesta. In the colon, physiologically normally shaped faecal pellets were evident (Figure 1(a) and (b)). On the outer gastric wall, blood vessels were marked intensively. Stomach dissection revealed a mass (weight 3.03 g) consisting of imbrued and macerated bedding material (Figure 1(c)).
Macroscopic findings. (a) A representative photo of a gold thioglucose (GTG)-treated male C57BL/6 mouse after opening the abdominal cavity. The picture shows the enlarged stomach filled with ingesta. (b) The complete gastrointestinal tract. The stomach was surgically opened, and gastric content was explored. (c) Dimensions of the gastric content (cm) and the anatomical structures of a saline-treated control mouse.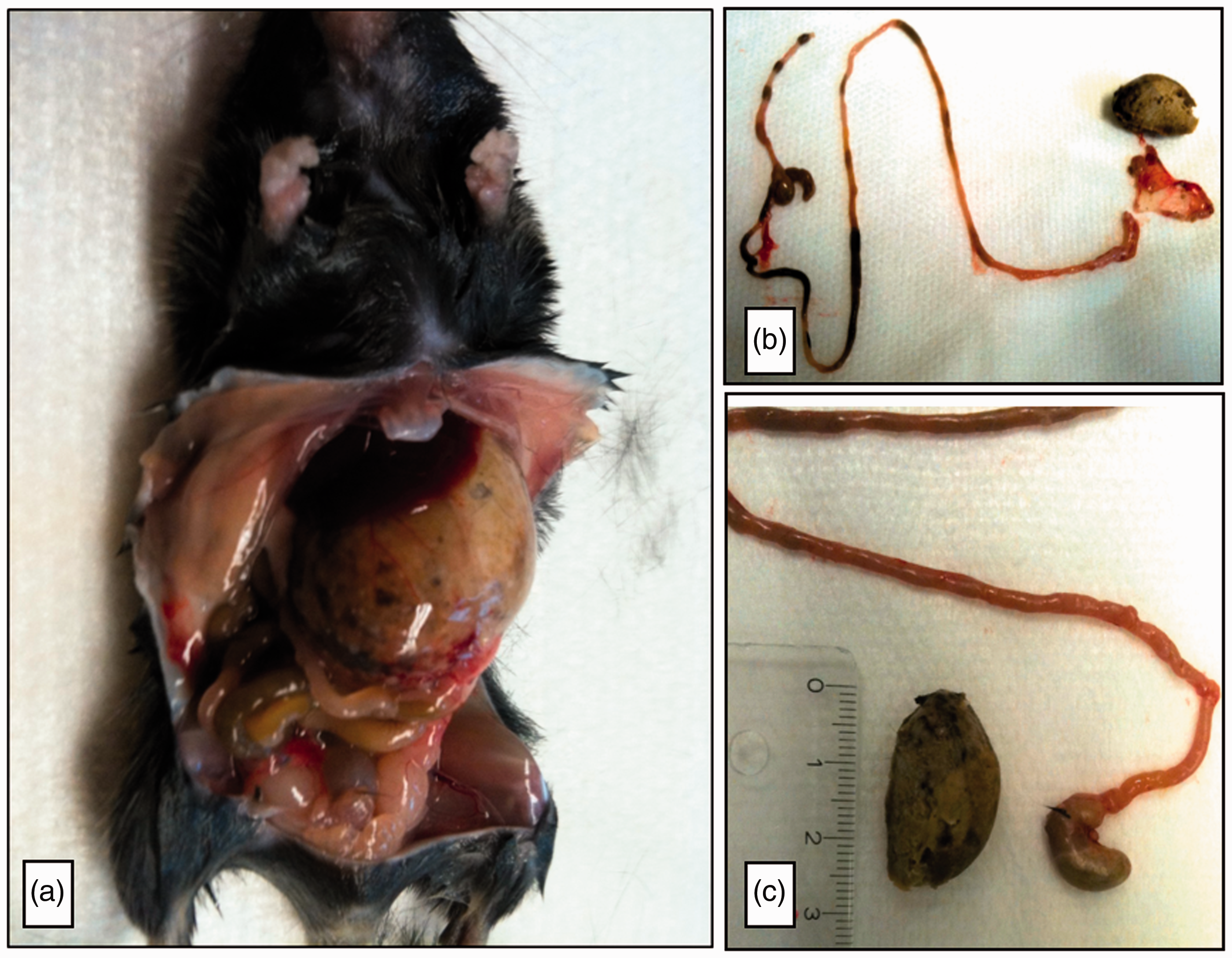
Discussion
Unexpectedly, in this study, the hyperphagic onset after GTG administration (0.5 mg/g BW) occurred later than 12 hours (13–22 hours after injection). The strategy of restricted feeding to prevent gastric dilatation seems to be contraindicated. It is clear that dysregulated appetite sensation converted hyperphagia into pica behaviour, resulting in intake of non-physiological amounts of bedding material. The wooden shavings soaked up fluids and impeded water intake, as the alimentary canal was congested. Gastric distension is associated with high mortality. Owing to volume expansion, the caudal vena cava and portal vein can be obstructed, resulting in venous stasis. This stasis results in a decrease in circulatory blood flow (venous return) with effects on cardiac output and blood pressure regulation. Under severe conditions, symptoms might culminate in hypovolemic shock. From our observations, we can recommend providing animals with semi-liquid diets to mitigate appetite attacks and ensure physiological consistency of the alimentary tract. Scientists working in the field of animal experimentation are fully aware of the sensitivity of animal models. Therefore, extensive planning and literature studies are conducted to fulfil the requirements of ethical considerations and Russell and Burch’s 3R principle (Replacement, Reduction and Refinement). Interestingly, our thorough literature research revealed different doses of GTG administration for inducing a hyperphagic state.4,11,12,14,15,18 Other studies reporting this model indicated serious complications (including high mortality) 14 but no evidence of pica behaviour or other abnormalities. Thus, it was considered appropriate to provide animals with physiological feed rations and to single-house male mice to prevent ranking-order fights and aggression due to limited access to chow. In prior studies, we have experienced that metabolic responses in mice are prone to genetic and various environmental factors, which is why a potential impact of these factors is occasionally difficult to predict. Implementation of pilot studies is a useful tool to introduce a novel model and to identify and/or minimise possible side effects. 18 GTG dosage ranged from 0.5 mg4,11,12,14,17 to 1.15 mg 15 per g BW. A possible explanation for this range might be strain differences or variations in age, sex and BW. Additionally, aspects of husbandry can have a substantial impact on behaviour and metabolism in terms of stress response. Even meal patterns applied in the dark or light cycles have an impact on metabolic conditions. 19 Our study of the literature did not reveal any apparent deviations in terms of standard housing for laboratory mice or hygienic state (in all cases animals were raised under SPF conditions). Moreover, no reference indicated either an impact of application time points or any information concerning a possible daytime-dependent mode of action (concerning manifestation of hyperphagia).
Consequently, we would like to encourage a revision of the degree of suffering associated with GTG administration, as the application of GTG is currently graded as being of moderate intensity. GTG-induced hyperphagia is likely an immense centrally regulated stimulus resulting in abnormal eating behaviour and thereby enhancing psychological and physiological stress. Given that the animals died within a short timeframe after injection of GTG, the application of GTG should be graded as severe. This study and the apparent high rate of mortality indicate that the GTG model must be used only when there is an extremely strong ethical justification. Pica behaviour is defined as a pathological tendency to eat or drink unusual substance(s) (e.g. bedding material) and might be a consequence of stress, pain or the application of emetic stimuli. 20 In fact, we cannot unambiguously reconstruct the intrinsic cause of the crucial trigger for pica behaviour in this study, but pica behaviour itself is categorised as a humane endpoint, becoming relevant when moral, scientific or legal considerations must be taken into account. 21
In conclusion, we would like to share this unfortunate experience with the broader community of experts in laboratory animal science to avoid unnecessary duplication. We recommend (i) conducting a pilot study and expanding supervision for at least 24 hours after GTG application; (ii) avoiding restricted feeding; (iii) providing animals with semi-liquid food to ensure digestive processes, starting with an adaption period a few days prior to GTG administration; and (iv) verifying the degree of suffering in the model of GTG-induced hyperphagia. Furthermore, the outcome of this study points at the common problem of underreporting findings. There remains a lack of detailed information provided by published studies. Thus, we can only suspect that other scientists also experienced negative effects of GTG administration but did not publish the data. Therefore, this study is representative of investigations associated with unforeseen events. Our observations emphasise the importance of reporting negative outcomes to assist in future refinement measures and may help to avoid unnecessary duplication.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
