Abstract
We investigated if there were any negative effects on the behaviour and physiology of rats housed in groups of five in two types of enriched cages and compared them with paired-housed rats housed in traditional cages. Eighty-four male Wistar and Sprague-Dawley rats were housed in an enriched rat cage (ERC), a rebuilt rabbit cage (RRC) or a Makrolon III cage (MC) system from 5–16 weeks of age with access to different enrichments. Recordings of behaviour and cage use (3 × 24 h video recording), weekly weighing, measuring food consumption four days/week and water consumption two days/week, were carried out. The rats’ muscle strength was assessed using the ‘inclined plane’ at the end of the study, and after euthanasia the adrenal glands were removed and weighed. Being in the shelter was the most common behaviour in the ERC and RRC groups. In the MC group, which lacked a shelter, rats performed the highest percentage of lying, grooming, rearing, play fighting and manipulating paper shreds. Rats in the RRC had the highest percentage of standing and manipulating gnawing sticks. Water consumption was higher in MC than in ERC and RRC rats. Rats from the RRC managed to remain at a steeper angle on the ‘inclined plane’ than rats from the MC. There were no significant effects of cage type on weight gain, food consumption or relative weights of adrenal glands. In conclusion, male rats kept in groups of five in larger enriched cages benefited from the enrichments, and no negative effects were found in the larger groups.
During the early 1990s, the cage system for rabbits at AstraZeneca (R&D Mölndal, Sweden) no longer met the company’s requirements and had to be replaced. We wanted to find out if the caging system could be made suitable after some rebuilding for other species in our laboratory. Keeping rats in groups with enrichment seemed to be a good alternative to the current single or pair-housing in Makrolon cages. Additionally Makrolon III cages (MCs) are difficult to enrich with shelters and other enrichments. A pilot study showed promise and the decision was taken to make this caging system available for rats in research studies (henceforth called ‘rebuilt rabbit cage’, RRC). As we only had a limited amount of these old rabbit cages, a new cage for group-housing rats was developed together with Scanbur A/S (Karlslunde, Denmark), taking into account our experiences with the RRC. This new cage system was called the enriched rat cage (ERC) and 150 racks containing three cages per rack have been sold between 2000 to 2012 in 10 countries (USA, Finland, Switzerland, UK, Denmark, Sweden, Norway, Germany, Australia and Kuwait). The following study was carried out to investigate more thoroughly the effects on the rats of being housed in larger groups in the enriched cages compared with the traditional Makrolon cages.
The effects of housing rats in different social and enriched environments have been investigated in several laboratory studies (see Young 2003 1 for a review). It has been suggested that adding structures to a standard sized cage is not as effective as providing the animals with larger cages. 2 The type of enrichment provided is also very important since just supplying rats with bigger cages does not necessarily improve their welfare. 3
In preference tests, where rats were able to choose between different types of cages and enrichment, it was found that they preferred cages with a rectangular shape,4,5 increased surface area, 5 and cage heights greater than 18 cm.4,5 Bedding materials were preferred to grid floors, and bedding materials with big pieces that could be manipulated were preferred to small particles. 5 Wood shavings were preferred over grid floors for resting, but if the rats were active then they used both kinds of cage floor for a similar time. 6 Rats kept singly preferred cages with nest boxes (where they mainly rested), to those with no nest, and with nests they showed more exploratory behaviour and less fear response. 7 Further evidence that access to nest boxes is important for rats was confirmed as pair-housed rats were willing to work harder to gain access to a nest box, as shown by their willingness to lift doors more than double their weight. 8 The motivation of the rats to seek a nest box may be to avoid light as it has been shown that both pigmented and albino rats avoid light intensities ≥100 lux, 5 and furthermore, that rats given a choice of cages with different light intensities spent 78% of their time in the darker cages. 9
Rats have been shown to interact with paper tissues, but are not interested in gnawing sticks. 10 In other studies rats used gnawing blocks more when kept in stainless steel cages with a grid floor without bedding than when kept on solid bottom floors with bedding.11,12 In most of these studies, enrichment has been assessed on individual or paired rats but we wanted to investigate the effect of enrichment on group-housed rats since it is possible that it may be different with larger groups. In the present study all the rats had aspen wood shavings on the cage floor and we tested three types of enrichments, i.e. nest box, shredded paper and gnawing blocks.
It has been suggested that social contact is the most sought after enrichment for rats. 3 For example, in one study rats kept in groups showed improved function after a focal brain ischaemic injury compared with individually kept rats with access to a running wheel; and rats kept both in groups and an enriched environment improved significantly more than the other groups. 13
Rats kept individually in barren cages (with a lack of stimuli) have a higher food intake compared with animals kept in a more stimulus rich environment. 13 However, one potential problem with group housing male rats of the same age and weight is that it may be more difficult for them to form a hierarchy, which has been found to be based on age for rats kept in semi-natural environments. 14
If animals are stressed by their housing or any management procedures it may lead to an activation of the sympatho-adrenomedullary system (SAS) and hypothalamic–pituitary–adrenal (HPA) axis.15,16 These adrenal glands release steroids (glucocorticoids, mineralocorticoids) from the cortex and catecholamines (adrenalin, noradrenaline) from the medulla. 17 Glucocorticoids regulate the stress response through a feedback mechanism in the brain, 18 and if stressful conditions persist these systems may fail to switch off and this may lead to maladaptive syndromes due to exhaustion of adrenal gland secretions. 19 These responses can increase adrenal size, which can be measured by weight. In a study of the response of two lines of wild-derived rats, which had been artificially selected for tameness or defensive aggression towards humans, the latter had larger adrenal glands and higher levels of serum corticosteroids in response to a human handling test. 20 Adžić et al. 18 found that male Wistar rats exposed to a 30 min acute immobilization stress, or to 21 days of individual housing stress, or to a combination of both had increased weight of both left and right adrenals.
The aim of the present study was to investigate whether a change from MCs towards the bigger new cage systems (RRC and ERC) enhanced the rats’ welfare. Access to different enrichments was also varied to investigate the degree of usage both after short- and long-term access. The outcome measures were changes in behaviours, use of the cage environment, food and water intake, weight gain, muscle strength and adrenal gland weight. These parameters were compared in two strains of male rats. The hypothesis was that keeping rats in larger cages with social contact would stimulate them, as shown by increased activity, reduced weight gain and stronger muscle development.
Materials and methods
The study was carried out at the Animal Department of AstraZeneca R&D in Mölndal, Sweden. The study was approved by the Ethics Committee on Animal Experiments Gothenburg (Dnr 133-2000).
Animals
The study used a total of 84 male rats (Taconic, formerly M&B A/S, Ry, Denmark) purchased at five weeks of age. Two strains of rats were used, Sprague-Dawley (SD) (Mol:SPRD Han, n = 42) and Wistar (Mol:Wist Han, n = 42) (Taconic). The rats had not been provided with enrichment other than bedding and nesting materials at the breeder’s facility. They were placed randomly in one of three cage types directly after arrival at the animal facility and given seven days of acclimatization before the start of the experiment. For individual identification all the rats were implanted subcutaneously with a microchip (Implantable Micro-Identification [IMI]; Bio Medic Data Systems Inc, Seaford, DE, USA) under isoflurane anaesthesia (Forene®; Abbott, Solna, Sweden) one week after arrival at the animal facilities.
Sentinel animals were housed in the same animal room and were tested for pathogens according to the Federation of European Laboratory Animal Science Associations (FELASA) recommendation. Inspection of the animals in their home cage was carried out daily and once per week at the time of weighing by the same two animal technicians who also cleaned and changed the cages. Each rat was picked up and inspected for skin wounds, and any other clinical signs of ill health.
Husbandry during experiment
Three different types of cages were used: (1) ERC (4900 cm2, 44 cm high; Scanbur A/S) (Figure 1a); (2) RRC (4400 cm2, 41 cm high; AstraZeneca R&D) (Figure 1b); and (3) MC (800 cm2, 18 cm high; Tecniplast, Buguggiate, Italy) (Figure 1c). In the ERC there was a shelf on one side (500 cm2, 60 × 25 cm, 19 cm high) and two horizontal ladders on the opposite wall (Figure 1a). In the RRC there was a coloured plastic nest box placed loose on the floor (33 × 20 cm and 14 cm high; Perstorp Plastic Systems 9073, Perstop, Sweden) (Figure 1b).
Photos of (a) enriched rat cage (ERC), (b) rebuilt rabbit cage (RRC) and (c) Makrolon III cage (MC) systems investigated in this study. Black marks on the rats were made with a marker before filming for individual identification. In the RRC, rats had food in a food hopper hanging on the grid door and not in the cage as in the photo.
For the ERC and RRC two racks with three cages per rack were used, giving a total of six cages for each treatment. MCs were placed on different levels of two shelves, and a total of 12 cages were used. There were five rats in each ERC and RRC and one pair of rats in each MC. This resulted in a total of 30 rats for the ERC, 30 rats for the RRC and 24 rats for the MC. All the groups were held in these cages during the 11 weeks of the study. All the cages had a solid floor with aspen wood shavings (Tapvei Oy, Kaavi, Finland).
During the study the following enrichment items were used:
Nest boxes were placed in the ERCs and RRCs. An extra nest box of coloured plastic (33 cm long, 20 cm wide and 14 cm high, Perstorp Plastic Systems 9073, Perstorp, Sweden) was placed in the middle of the cage floor (Figure 1a and b). In the MCs, one short side and one-third of both long sides of the cage were painted black and a black board was placed on the cage roof at the same place as the sides were painted in order to create something comparable to a nest box. In addition a plastic board, meant to be used as a shelf, was placed inside every second MC. Shredded paper (5 g/rat), long white disposable, produced from raw material used in the food industry, e.g. viscose, manila hemp, wood pulp (Datesand Ltd, Manchester via Scanbur A/S) was added to the cage. Aspen gnawing sticks (50 × 10 × 10 mm) (Tapvei Oy), were placed in the cages: one stick in the MCs and two in the ERCs and RRCs.
The three enrichment items were provided simultaneously when the rats were 10–15 weeks old. The items were put into the cage immediately after the video recording was started for each cage at 10–12 weeks. Gnawing sticks and paper shredding were replaced at the weekly cage cleaning.
All the rats had ad libitum access to food (R3 breeding feed for rats and mice; Lantmännen, formerly Lactamin, Vadstena, Sweden). In ERCs a food hopper was placed on the outside of the cage door. In RRCs a special food hopper made of stainless steel with a lid was hung up on the inside of the cage door. In MCs the food was placed in the lid. All the rats had ad libitum access to tap water, which was provided in plastic bottles with a non-drip nipple.
All the rats were kept in the same animal room which had a temperature of 22℃ (±1℃) and a humidity of 50% (−5, +10%). The air was changed around 20 times per hour, and lights were on a 12 : 12 light:dark schedule with 30 min of dawn and dusk. The light level was kept at 100 lux, but levels inside the cages were below this range as there were roofs on all cage racks and the RRCs and ERCs had shelters. During the weekly cleaning, it was increased to 200 lux.
Experimental procedure
Behavioural observations
Each cage was filmed for 24 h when the rats were: between 7–9 weeks without enrichments; between 10–12 weeks when enrichments were new; and between 13–15 weeks when enrichments had been in the cages for three weeks (see Figure 2 for an overview). Filming was carried out with a Panasonic time-lapse recorder connected to a camera (Elbex, Tokyo, Japan) which was mounted above the top cage of the RRC and ERC racks and pointed straight down. Infrared light lamps were mounted beside the cameras to facilitate recordings during the dark hours. The roof was removed and cameras and lamps were mounted before the rats were placed in the cages for these video recordings. Since only the top cage could be filmed, groups of rats changed cage level once a week and were always placed in clean cages according to the filming schedule. Six MC cages were included in the behavioural study to be the same number as the other two cage treatments. These cages were filmed from the side, two cages with one camera. Before video recording, the rats were marked with different black patterns so that they could be individually recognized on the film (Sanford Uni-Paint Px-20 Black Marker, Oak Brook, USA). The rats were painted three times at three-week intervals. The marker contained xylene, which may be harmful or fatal if swallowed and direct contact can cause skin or eye irritation according to safety testing (information from material safety data-sheet from Sanford Brands, 1 February 2007). This marker was the only one out of several tested markers that was visible in infrared light, and we did not know at the time that the marker could be toxic; however it did not change any of the measures compared with previous studies (see Results and Discussion).
Time line of experiment.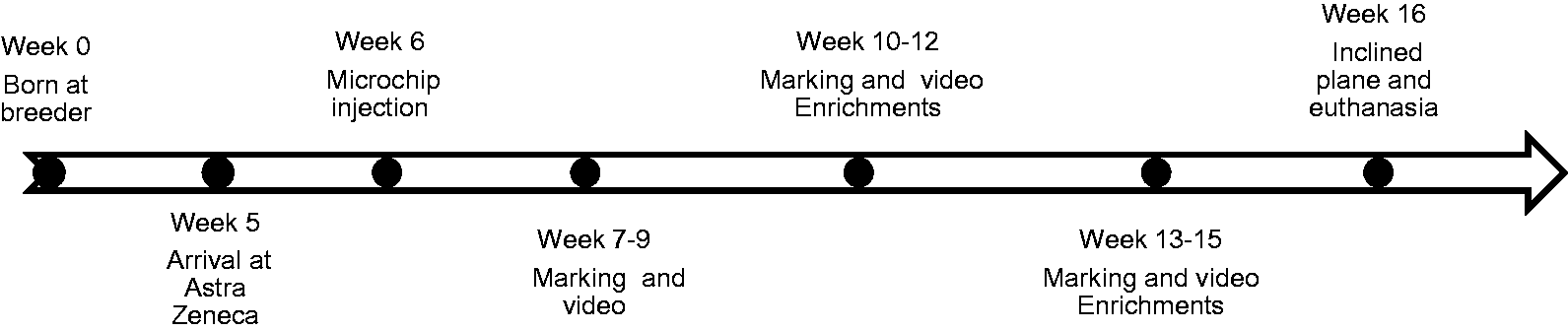
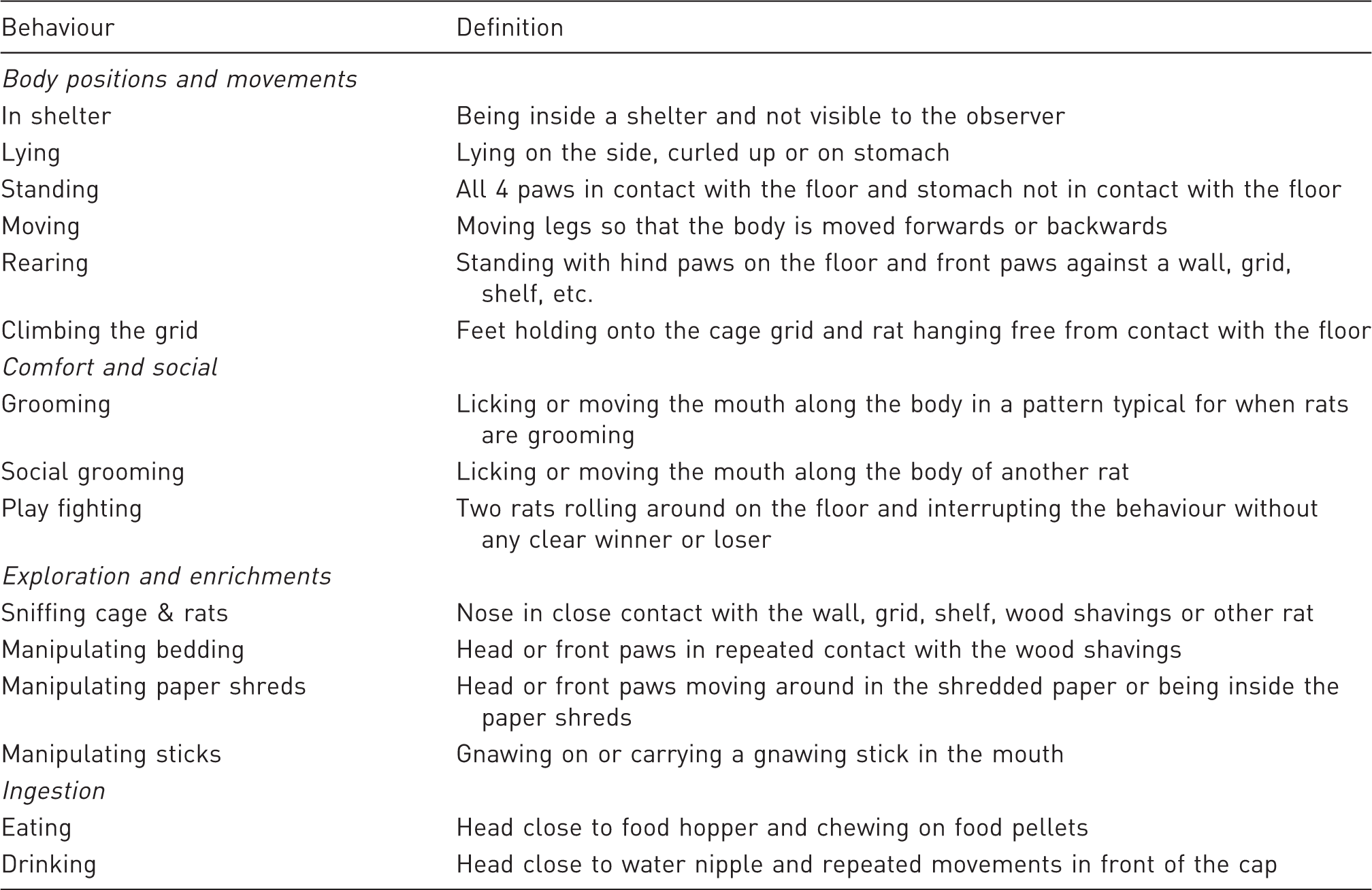
Behaviours of rats recorded in the study and their definitions.
Location in the cage
Recordings of the location in the cage of individual rats were carried out with instantaneous recordings every 10th minute directly after their behaviour had been recorded. In the ERC and RRC the specified locations were: on the floor, on the left nest box, on the right nest box, inside the left nest box, inside the right nest box, on the food trough (RRC), on the ladder (ERC), other place, or could not be seen. In the MC the specified locations were: in the middle part, underneath the food trough or on the short side, on top of the shelf or underneath the shelf. The right nest box was only available for the ERC and RRC during weeks 10–12 and 13–15. The six MCs that were filmed had a shelf during weeks 10–12, but then it was removed as one rat got a tail tip lesion that we suspected had been caused by the shelf tipping and squeezing the tip of the tail against the cage wall. The available space for the two rats was much reduced with the added shelf.
Weight gain, food and water intake
On arrival the rats were weighed, and then weekly weighing was carried out on Tuesday mornings when individual rats were moved to a Makrolon II cage and carried to an adjoining room where the balance was placed. All weighing procedures (rats, food and water) were done with the same balance (PM6000; Mettler Toledo, Stockholm, Sweden) by the same two animal technicians who also cleaned and changed cages, changed enrichments and marked rats for the video recordings. Every week during the experiment the food hopper with the dry food pellets was weighed on Mondays and then weighed again on Fridays to measure food consumption per four days on group level. At the same time the water bottle was weighed over a 2-day period every week to get a measure of water consumption per two days. The food and water consumption was recorded at group level, and thereafter a mean value per rat per day was calculated, so that comparisons between cages with five respectively two rats could be done.
Inclined plane
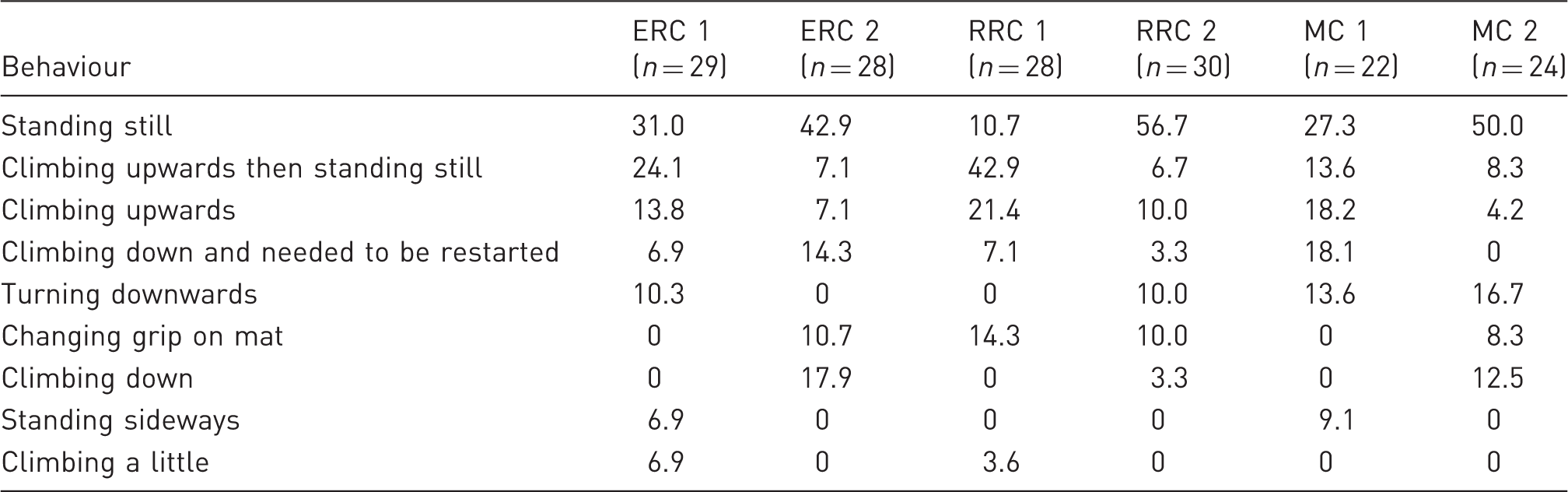
To evaluate if the muscle strength and the ability to maintain balance differed between rats which had been housed in the three cage types for 11 weeks all the rats were tested on an ‘inclined plane’ during the last days of the experiment. The inclined plane was originally developed as a clinical assessment of rats’ motor function after an experimental spinal cord injury. 21 The inclined plane consisted of a container made of polyvinylchloride with solid walls on the short sides and transparent walls on the long sides (100 × 50 cm, 25 cm high). It was equipped with a rubber mat on the bottom to avoid slipping. A calibrated protractor spirit level (Bosch) was placed on the outside of the box to measure the angle of the box when it was lifted. One person held the bottom board of the container at an angle of 45° and a second person placed the rat on the middle of the rubber mat with its head upwards. The angle of the plane was then slowly increased manually until the rat fell/slid down. The maximum angle the rat could still hold on to the rubber mat was recorded by reading the digital water level that was attached to the side of the container. Recordings were made on how the rat finished the inclined plane test, e.g. if it was falling down, sliding down or walking down. Recordings were also done on the behaviour of the rat when the inclined plane was slowly raised as: standing still, climbing upwards then standing still, climbing upwards, climbing down and needed to be restarted, turning downwards, changing grip on the mat, climbing down, standing sideways, and climbing a little. The test was performed on each rat twice on consecutive days.
Adrenal glands
At the end of the experiment the rats were placed individually in a plastic container (Makrolon II cage) where gas could be inserted. Firstly, they were anesthetized with isoflurane (Forene®, Abbott) and subsequently euthanized with an overdose of carbon dioxide. A necropsy was done and the adrenal glands were taken out and weighed (PR503, Mettler Toledo). The adrenal glands were cleaned of other tissues and weighed. The adrenal weight of the glands was then divided by the weight of the rat to give the so-called ‘relative weight’, and these data were used for later analyses.
Statistical procedures
Statistical analyses were carried out using SAS version 8.2 (Statistical Analysis System Inc, Cary, NC, USA). The cage was used as the statistical unit in the tests for food and water intake. For the tests of weight gain, adrenal gland weight and response on the inclined plane, the rat was the statistical unit. Behavioural data were recorded for only three 24 h cycles per cage and could not be assumed to be normally distributed. Due to a few statistical units per cage type (n = 6) and a few repetitions of each cage (three 24 h cycles without and with enrichments) transformation of data was not regarded as appropriate. Hence only arithmetic means and SE are shown for these data. All other data had been checked and had normal distribution. Statistical analysis of food and water intake, weight gain, adrenal gland weight, relative adrenal gland weight and the angle at which rats lost their foothold on the inclined plane was done by an analysis of variance with the general linear model which tested for cage type (three types), strain (two strains), age (three age groups), cage type*strain, cage type*age, and strain*age. The F value and the P value from the Type III SS have been used. As a post hoc test, Tukey’s Student’s test was used to compare differences between cage types and ages. The results are shown as arithmetic means ± standard deviation (SD).
Results
Behaviour in different cage types
Mean percentage of recordings per 24 h (±SE) of behaviours in male rats at 7–15 weeks of age when kept in an enriched rat cage (ERC, five rats/cage), a rebuilt rabbit cage (RRC, five rats/cage) or a Makrolon III cage (MC, two rats/cage) (n = 6 cages/cage type).
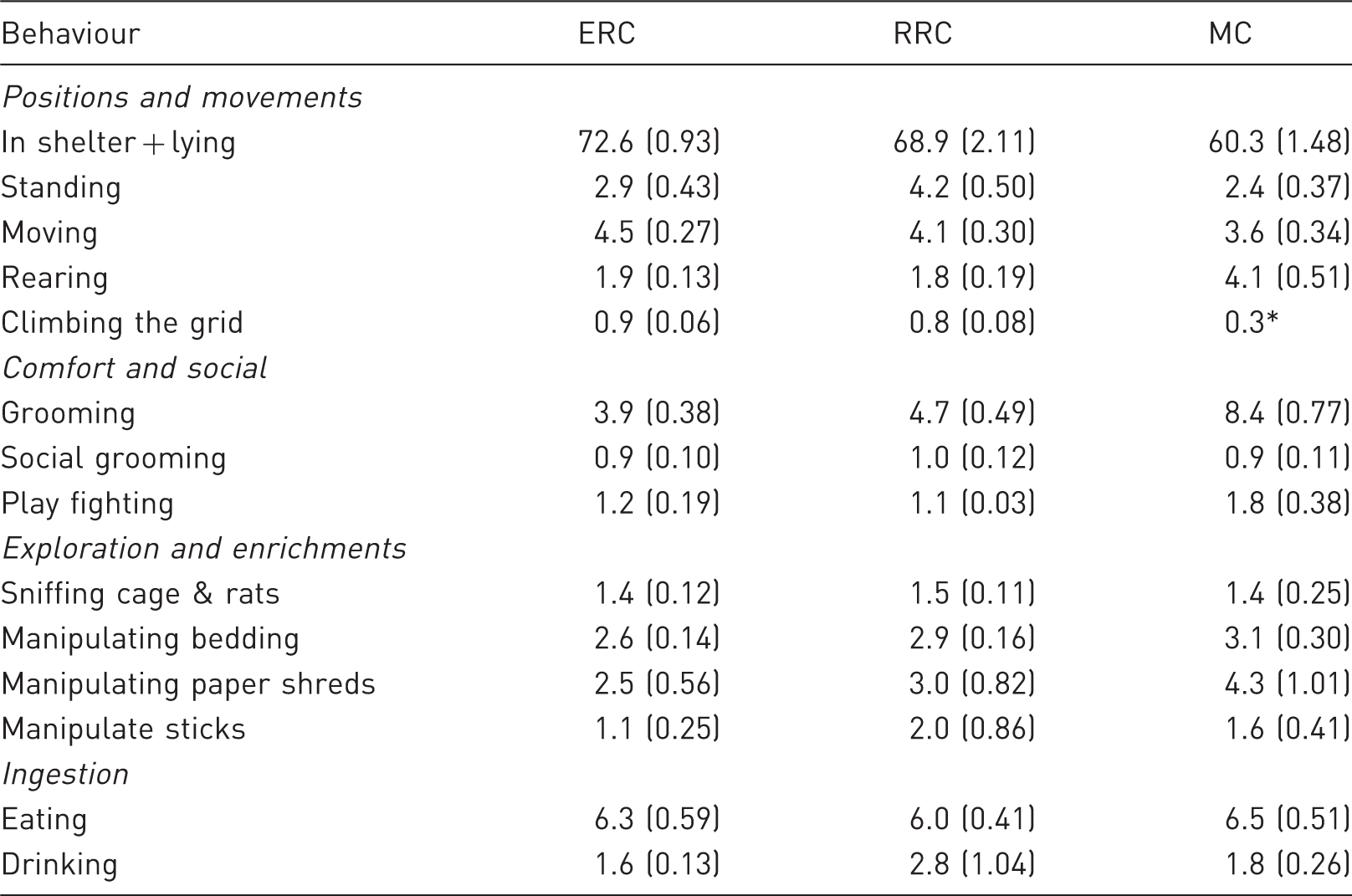
The rats in the RRC had a higher percentage of recording of ‘standing’ than the rats in the ERC or in the MC, whereas rats in the MC had a higher percentage of recording of ‘rearing’ than the rats in the ERC and RRC (Table 2). The rats in the ERC had a slightly higher percentage of recording of ‘moving’ than the rats in the RRC and MC (Table 2). The behaviour ‘climbing the grid’ had a low occurrence in the ERC and RRC rats, and was only recorded once in the MC rats (Table 2).
The rats in the MC performed a higher percentage of recording of ‘grooming’ and slightly higher percentage of recording of ‘play fighting’ than the rats in the ERC and RRC (Table 2). ‘Social grooming’ had a low percentage of recording and did not differ between cage types (Table 2).
‘Manipulate bedding’ had a higher percentage of recording than ‘sniffing cage & rats’, but both behaviours had similar percentage of recording for all three cage types (Table 2). The rats in the MC had a higher percentage of recording of ‘manipulate paper shreds’ than the rats in the ERC, whereas the rats in the RRC had a higher percentage of recording of ‘manipulate sticks’ than the rats in the ERC (Table 2). ‘Eating’ and ‘drinking’ had a similar percentage of recording in all cage types (Table 2).
Behaviour at three ages without and with enrichments
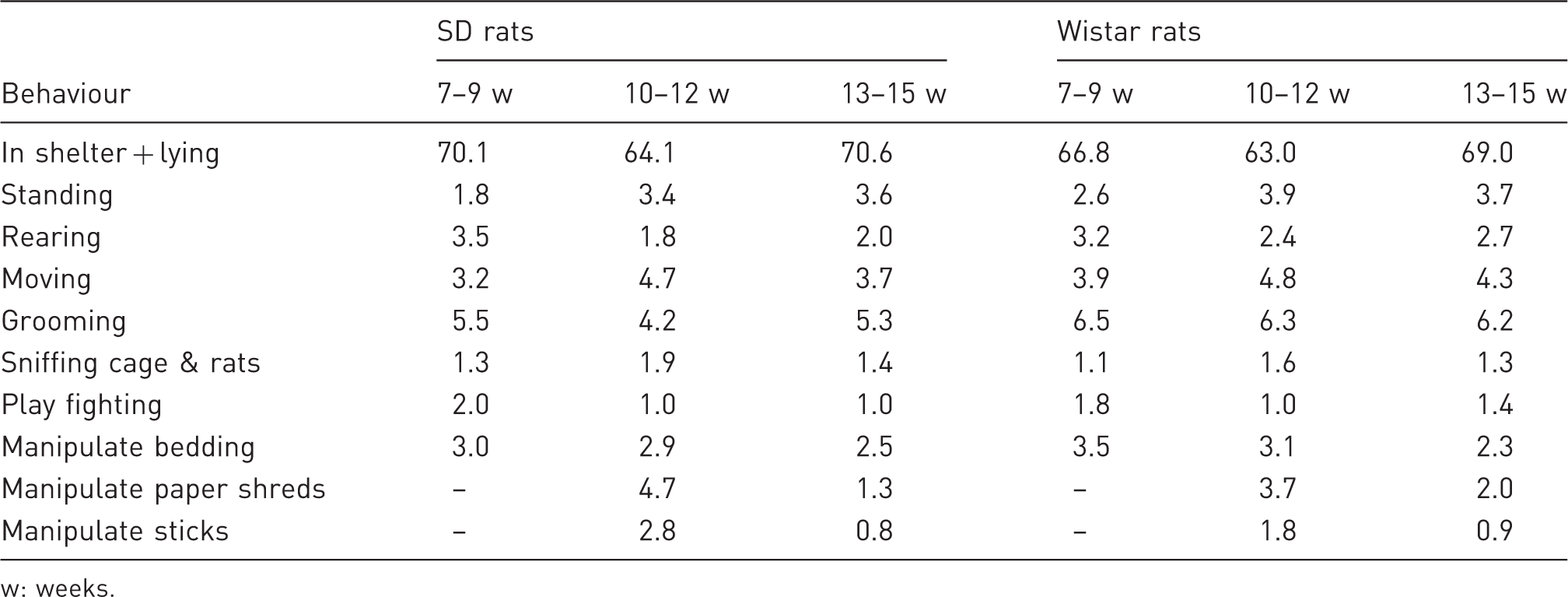
Mean percentage of recordings per 24 h (±SE) of some behaviours in male Sprague-Dawley (SD) or Wistar rats at three ages when kept in three different cage type (n = 9 cages/strain).
w: weeks.
At 7–9 weeks the highest percentage of recordings per 24 h were found for the behaviours ‘in shelter’, ‘rearing’, ‘play fighting’ and ‘manipulate bedding’ (Table 3). The lowest percentage of recording was found at weeks 7–9 for the behaviours ‘standing’ and ‘sniffing cage & rats’ (Table 3). There were no differences between ages in the percentage of recording of ‘moving’, ‘climbing grid’, ‘grooming’, ‘social grooming’ ‘eating’ and ‘drinking’.
Behaviour in Wistar versus SD rats
Wistar rats had a higher mean percentage of recordings of ‘in shelter + lying’, ‘rearing’, ‘moving’ and ‘grooming’ than SD rats (Table 3). During the two ages with enrichments SD rats had a higher percentage of recordings of ‘manipulate sticks’, but there were no strain differences in the percentage of recording of ‘manipulate paper shreds’ (Table 3). The rat strains had a similar percentage of recordings of ‘standing’, ‘play fighting’, ‘social grooming’, ‘sniffing cage & rats’, ‘manipulate bedding’, ‘climbing the grid’, ‘eating’ and ‘drinking’.
Use of the cage environment
The rats differed in the extent to which they used the cage area depending on the cage type. In the ERC they spent 61.2% of their time in a shelter, which is more than the rats in the RRC, which spent 33% of the time in a shelter (Figure 3). The rats in the RRC never spent as much time in the shelter as the rats in the ERC, independently of whether there were one (7–9 weeks) or two (10–12 and 13–15 weeks) nest boxes in the RRC. The rats in the RRC spent more time lying underneath the paper shreds outside the shelter and lay on the floor more than the ERC rats.
Percentage of observations of rats in different areas of an enriched rat cage (ERC), a rebuilt rabbit cage (RRC) and a Makrolon III cage (MC) system when rats were 10–12 weeks old and had just received cage enrichment.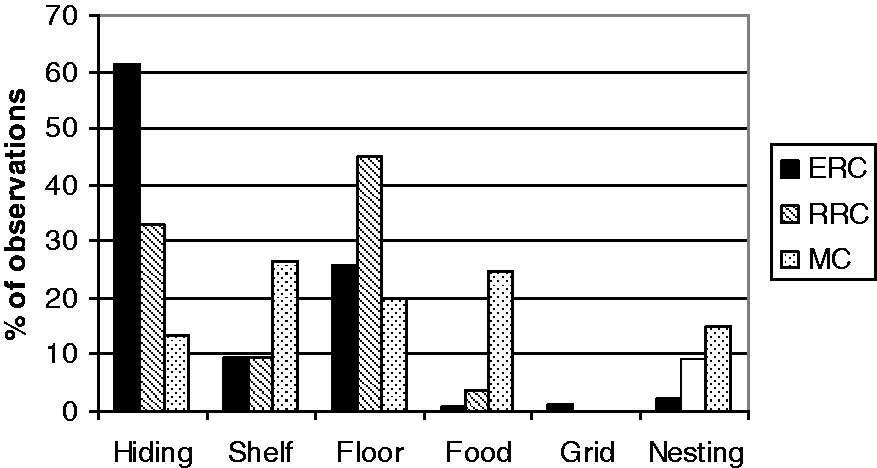
In the MC the rats preferred to rest under the feed hopper (about 58%) at 7–9 weeks, but at 10–12 weeks when half of the MC cages had got a shelf, they used the shelf (mean 26.5%) and also the shredded paper to lie on or within. At 13–15 weeks, when the shelf was removed they used the back of the cage more and the shredded paper was used as much as at 10–12 weeks.
Food intake, water intake and weight gain
Food consumption was not affected by cage type (not significant, F = 1.04), but water consumption was significantly affected by cage type (P < 0.001, F = 30.41), being higher in the MC than in the ERC or RRC rats (Figure 4). There was a significant effect of age on food consumption (P < 0.001, F = 7.29), and the amount of food consumed was higher at 10–12 weeks than at 7–9 and 13–15 weeks (Figure 4). For food consumption there were significant interactions between cage type and strain (P < 0.001, F = 22.94) and between cage type and age (P < 0.01, F = 3.61) (Figure 4).
Mean food consumption (a) and water consumption (b) (g/rat/day, ± SD) for Sprague-Dawley and Wistar rats at the ages of 7–9, 10–12 and 13–15 weeks in an enriched rat cage (ERC), rebuilt rabbit cage (RRC) and a Makrolon III cage (MC) system. Food consumption had significant effects on age (P < 0.001, F = 7.29) and interaction between cage type and age (P < 0.01, F = 3.61). Water consumption had significant effect on age (P < 0.05, F = 3.62), but not between cage type and age.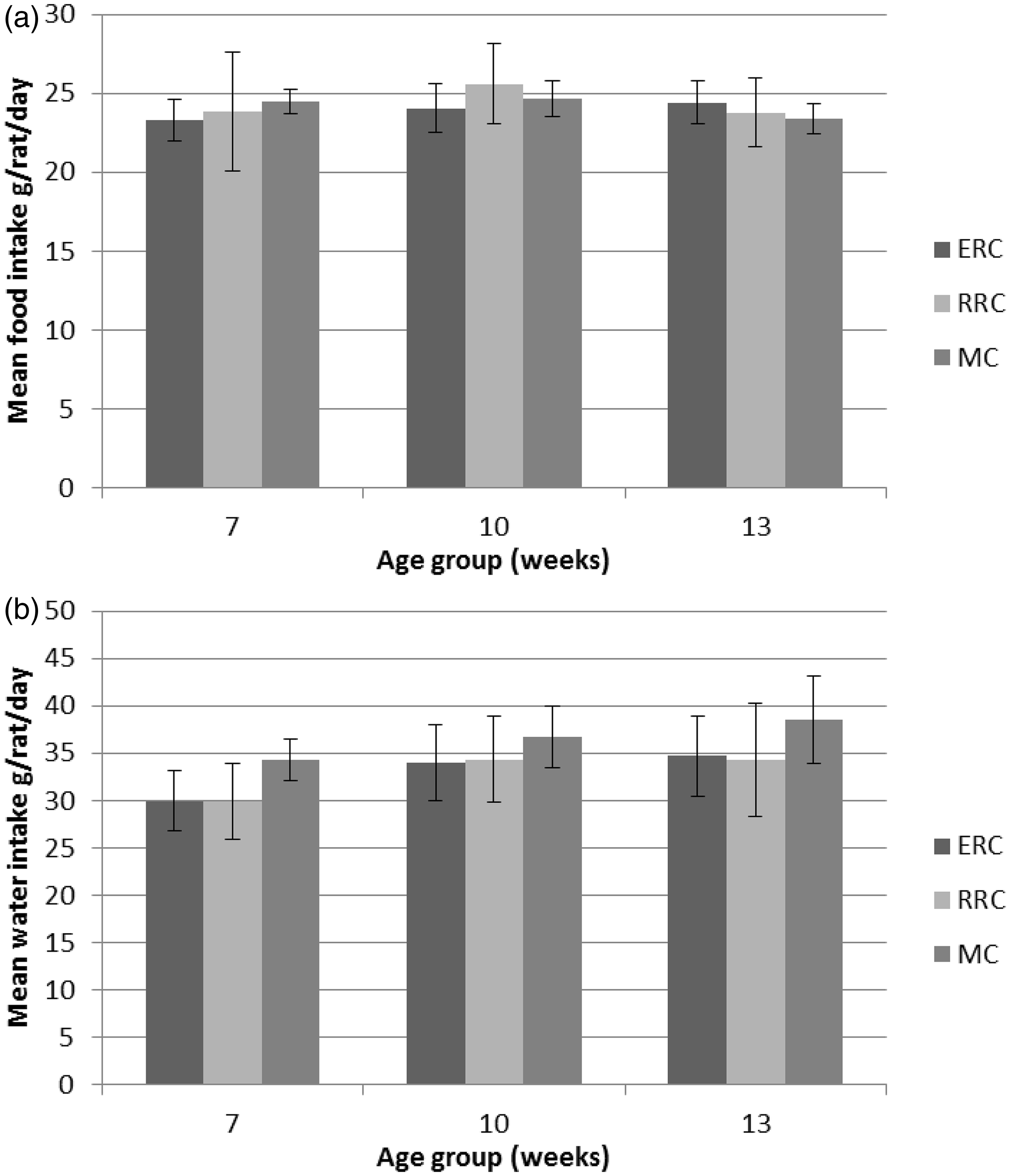
There was a significant effect of age (P < 0.05, F = 3.62) on water consumption, which was higher at 13–15 and 10–12 weeks than at 7–9 weeks (Figure 4). Water consumption had significant interactions between cage type and strain (P < 0.001, F = 12.25) and between strain and age (P < 0.001, F = 7.95).
Mean (±SD) food intake (n = 12 cages/strain), water intake (n = 12 cages/strain), growth rate (n = 42 rats/strain), weight at end of study (n = 41 Sprague-Dawley, n = 42 Wistar), adrenal gland weight (n = 41 rats/strain) and relative adrenal gland weight (gland weight/rat weight, n = 41 rats/strain) after euthanasia for two strains of rats.
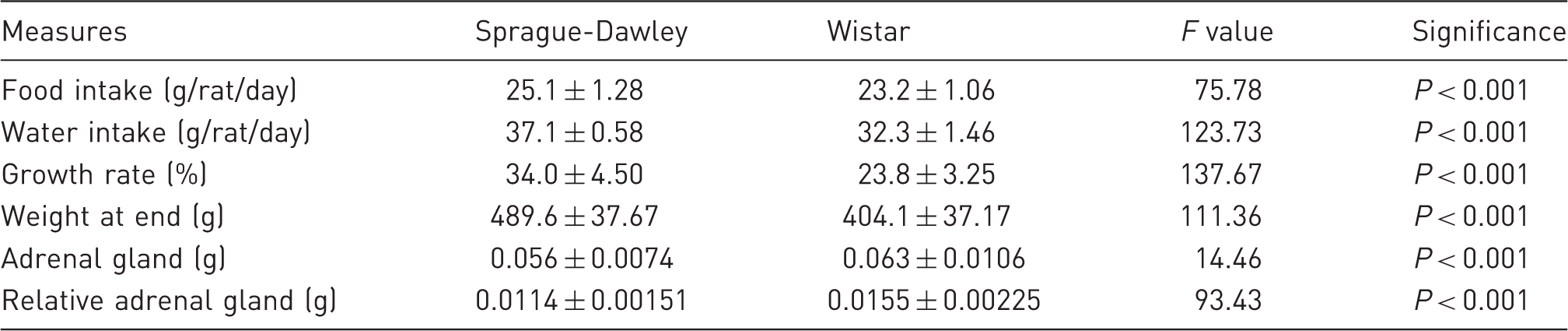
There were no significant effects of cage type on rates of growth (not significant, F = 0.16) or on rat weights at the end of the study (not significant, F = 0.03). The SD rats had a significantly higher growth rate and so were heavier at the end of the study than the Wistar rats (Table 4). There was a significant interaction between cage type and strain on rat weights at the end of the study (P < 0.05, F = 4.74), but there were no significant interactions between cage type and strain (not significant, F = 1.55) on growth rates.
Inclined plane
There was a significant effect of cage type on how steep the angle of the inclined plane was when the rats lost their foothold in both the 1st test (P < 0.01, F = 5.14) and the 2nd test (P < 0.01, F = 6.53). The rats stayed on the inclined plane at a significantly steeper angle when they had been housed in the RRC (81.0° ± 0.7) than when they had been housed in the MC (76.9° ± 0.88, P < 0.05), but there were no differences between the ERC (79.2° ± 0.77) and the other cage types. The Wistar rats stayed on the inclined plane at a steeper angle (79.5° ± 0.76) than the SD rats (77.5° ± 0.90) during the 1st test (P < 0.05, F = 6.12) but not the 2nd test (not significant, F = 1.81). There was no interaction between cage type and strain (1st test: not significant, F = 2.04; 2nd test: not significant, F = 1.77).
The inclined plane test most often ended with the rats falling down or sliding down (Figure 5), and only in three tests, one per cage type, did the rats finish the test by walking down. The initial behaviour of the rats (at both test times) when they were placed on the inclined plane and this was raised is shown in Table 5. A higher percentage of the rats climbed upwards the first test time whereas a higher percentage were standing still the 2nd time (Table 5), although this was not tested statistically.
Mean percentage of rats falling down (a) or sliding down (b) at the end of raising an inclined plane to test muscle strength in rats that were kept for 11 weeks in an enriched rat cage (ERC), rebuilt rabbit cage (RRC) and a Makrolon III cage (MC) system. Percentage of rats performing different behaviours on an inclined plane when being placed there individually two days after each other (1, 2) after having been kept in an enriched rat cage (ERC), rebuilt rabbit cage (RRC) or Makrolon III cage (MC) for 10 weeks.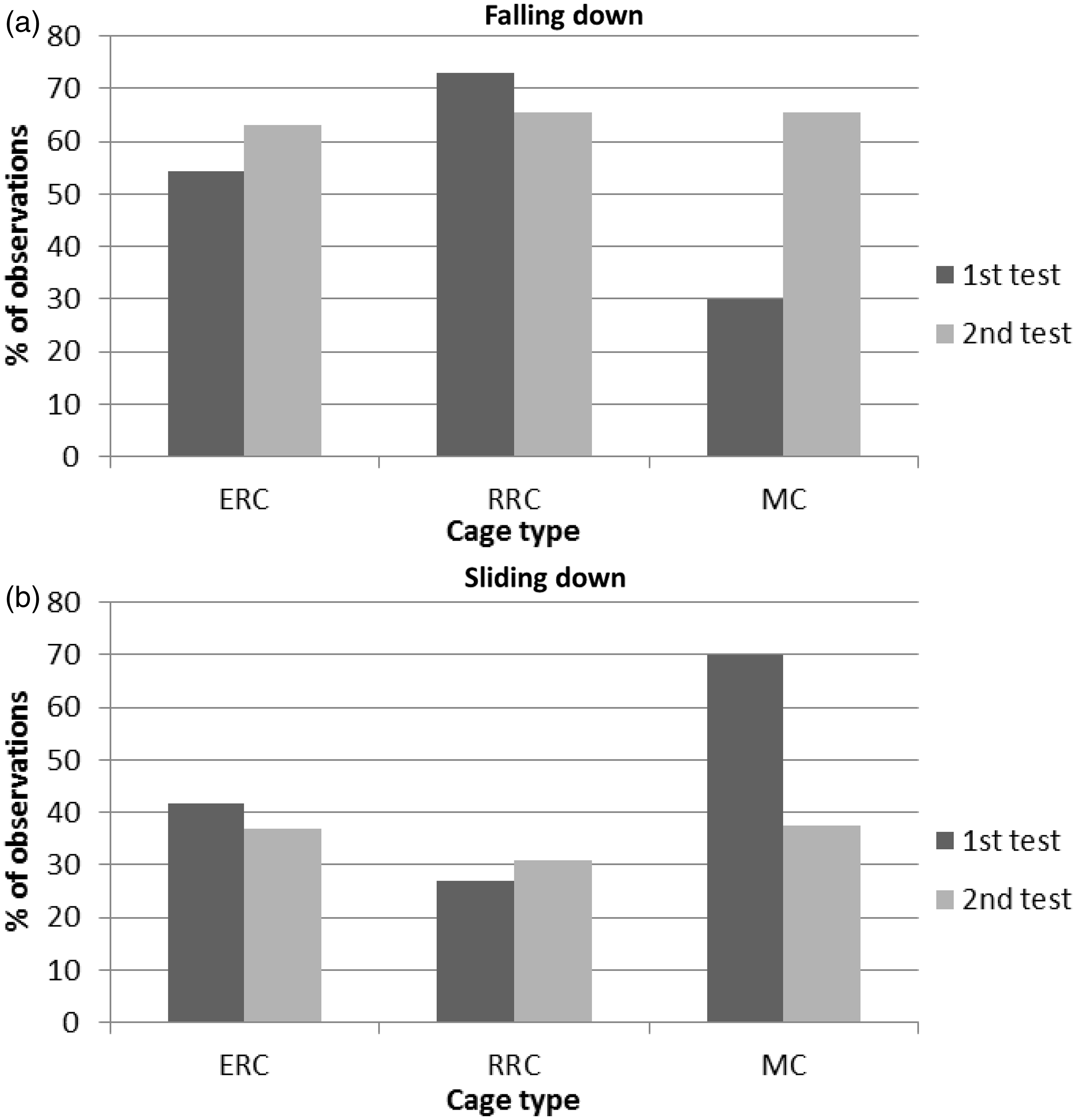
Adrenal glands and health
There were no differences in the weight or relative weights of the adrenal glands with cage type (not significant, F = 1.50; not significant, F = 1.19 respectively). However, there was a significant strain difference with lower adrenal weight and relative adrenal weight in the SD rats than in the Wistar rats (P < 0.001, F = 14.46; P < 0.001, F = 93.43 respectively, see Table 4). Interactions between cage type and strain were significant (P < 0.01, F = 5.92) for adrenal weight, but not for the relative weight of the adrenal glands (not significant, F = 1.54).
For the ERC and RRC rats there was a significant negative correlation between weight gain and the relative weight of the adrenal glands (P < 0.0001, r = −0.77, n = 59). When this was analysed by strain there was a significant negative correlation for the SD rats (P < 0.01, r = −0.52, n = 29), but not for the Wistar rats (not significant, r = −0.002, n = 30).
The health monitoring tests were negative for rodent pathogens according to FELASA recommendations. During the study one rat had to be euthanized because of a skin lesion, and another rat in the MC got a superficial tail tip lesion but was able to continue until the end. This rat appeared healthy in all other aspects and behaved normally, hence the decision to keep it in the study. At euthanasia a second rat was found to have a tail tip lesion and one rat only had one adrenal gland. All the other rats in the study were healthy and without injuries (bite marks, wounds, etc.).
Discussion
The main findings in this study were that male rats housed in groups of five in enriched cages (ERC and RRC) performed less lying in the open space and rearing against the wall, less time grooming themselves, play fighting and manipulating paper shreds, and they consumed less water than pair-housed rats in traditional MCs. Rats from the RRC stayed on the inclined plane at a steeper angle than rats from the MC, and the ERC rats were in between. However, for most of the measures there were no differences between the cage types, i.e. weight gain, weight at the end of the study, relative weight of adrenal glands, food consumption, manipulating the litter, moving, climbing cage bars and allogrooming. There were more differences between strains and at the different ages with and without enrichment, than between the cage types. This indicates that housing rats in groups of five in larger enriched cages did not lead to increased stress as measured by weight gain, adrenal gland weight and food intake, but instead had a positive effect since the rats used the provided enrichment, especially the nest boxes, to a greater extent. However, play fighting, which is often used as an indicator of good welfare in animals, was observed more in the MC rats. Some of the play fighting may have been missed if it occurred in the nest boxes in the ERC and RRC. The larger space and possibility for the rats to climb in the RRC could be an indication of greater muscle strength shown in the inclined plane test compared with pair-housed rats in traditional MCs.
The rats in this study spent most time either lying or being in a shelter where they were also likely to lie down. This corresponds well with previous studies where it was found that group-housed male SD rats were inactive for 74% of the observations,22,23 and that male and female SD rats kept individually in different metabolism cages were inactive about 70% of the observations. 24 Rock et al. 25 found similar results for rats kept both in groups and individually.
When the rats were in a shelter, i.e. under the shelf in the ERC or inside the box in the RRC and ERC, they probably spent most of the time lying down. Townsend 7 found that rats which had access to a nest box spent most of their resting time inside it. Since the rats in the RRC spent more time outside the shelter than the rats in the ERC one might expect it to be a consequence of the smaller area under the box in the RRC compared with the larger area under the shelf in the ERC. The nest boxes probably became too crowded when all the rats tried to enter them thus it is important to offer the animals enough space in nest boxes. The RRC rats chose to lie outside the shelter in the shredded paper, but this was not observed in the ERC rats, mainly because they had moved the paper under the shelter and had a bigger shelter. Comparison of some of the behaviours between the cage types is therefore difficult since the rats in the ERC and RRC were able to perform behaviours under the shelter, such as grooming and manipulating paper shreds, which could not be observed on the video recordings, by contrast with the MC rats where everything could be observed.
MC rats spent more time on the open floor, under the food hopper and at 10–12 and 13–15 weeks of age in the nesting material, as they did not have a shelter. It is interesting that even though the rats in the MC had only a shelf at 10–12 weeks, the proportion of time on a shelf was higher for this cage type than for the ERC or RRC rats. This is probably because the shelf added an important dimension to the cage when it was placed there. Unfortunately the shelf could move when a rat was on it, and when it went back in place the tail tip of the rat could be caught between the shelf and the cage wall. We therefore decided to remove it at 13–15 weeks of age to avoid further injuries. Construction of a better shelf in the MC would be important in the future. However, there is still the problem of the MC being not big enough according to Swedish regulations to house two adult rats. 26 At the end of the study there was not much space for the two rats in the MC, so the dominant rat probably took the place he favoured and then the other one had to lie somewhere else.
The rats in the MC carried out more rearing than the rats in the ERC and RRC. As the walls of the MC were not easy to see through, rearing could be important for the rats to sense their environment by inhaling smells in the room. In that respect the ERC and RRC make sniffing and seeing easier for rats when they try to orientate themselves in an animal room. It has been found that rats initially explore a new surrounding very intensively, but this behaviour decreases quickly with time. 27 In this study we could only film the top cages from above in the ERC and RRC and therefore we changed cage level once a week throughout the study. Additionally, we always filmed rats when the enrichments – shredded paper, gnawing sticks and extra nest box or shelf – were introduced into the cage at 10–12 weeks of age. This could be a reason why there were significant effects between ages in several behaviours.
Play fighting was recorded more often in the MC than in the RRC, but the ERC did not differ from the other cage types. One may question if all of the recorded play fighting was only play or if some of it was also aggressive fighting. Pellis and Pellis 28 describe that ‘during play fighting, snout or oral contact is directed at the opponent's nape of the neck, whereas during serious fighting, male residents mostly direct their bites at the intruder's rump’. Our definition of play fighting was ‘two rats rolling around on the floor and interrupting the behaviour without any clear winner or loser’. As recordings were made at 10 min intervals it was difficult to record how the play fighting ended. Play fighting was more evident in rats at 7–9 weeks than at 10–15 weeks. Foroud and Pellis 29 showed that juvenile rats (30 days) more often stood with all four paws on their partner during a play fight whereas postpubertal rats (70 days) placed their hind paws on the ground more often and thus got a better postural stability during play fighting. Due to the method of recording in this study we could not distinguish between the different types of play behaviours in juvenile and postpubertal rats. However, rats are growing until at least 16 weeks of age 30 and playful behaviour can persist well into adulthood. 28
We had expected the rats in the ERC and RRC to consume more food, as we thought they would be more active and thus use more energy, but food intake did not differ between cage types, either in amount consumed per five days or eating behaviour per 24 h. Spangenberg et al. 31 also did not find any difference in food intake between male SD rats when these were housed in groups of two, four or eight in cages adjusted for group size. As activity levels were quite similar between the rats in the different cages in our study and the rats were housed in the same room this was probably more important for food consumption than the different cage types per se. When individually-housed male rats of 15 inbred and one outbred strains were compared, their food intake ranged from 19.3–24.6 g/day. 32 In this study it was 23.2 for the Wistar and 25.1 for the SD rats, which is in the upper range and above compared with the previous study.
There was a difference in food consumption between the ages, with the highest in rats at 10–12 weeks and the lowest at 13–15 weeks of age. The higher food consumption at 10–12 weeks of age might be explained by their faster growth rate, i.e. they were bigger than at 7–9 weeks and therefore ate more
There was a decrease in manipulating wood shavings from 7–9 to 13–15 weeks of age which could be due to the provision of gnawing sticks as their attention was diverted.
The higher water intake in the MC rats compared with the ERC and RRC rats was not expected, but they did not drink water more often, and so the higher water consumption might have been due to the animals’ movements causing the water to leak due, for example, to the vibrations or body contact with the water nipple. However, the amount of water consumed in the MC in this study was slightly lower than the 40 g/24 h/rat found in males kept in pairs in the MC before being placed in metabolism cages. 24 In this study the SD rats consumed a higher amount of water than the Wistar rats, and differences in water intake between rat strains has been previously recorded. 32
We expected that rats in both the ERC and RRC would develop better muscle strength than the MC rats as they had the opportunity to use the enrichment and exercise more, e.g. climbing around in the cage. However, the only significant difference in ability to withstand an incline was between the RRC and MC rats. At the front of the RRC there was a grid that the rats could climb up which was not present in the ERC (only a horizontal ladder) and this might explain why there was a difference between the RRC and MC but not between the ERC and MC. This is the most plausible reason since there was no difference in how much the rats moved or used the ladder or climbed.
The average incline degree when rats lost their grip was 81° for RRC rats, which was the same found with control rats in a study by Rivlin and Tator. 21 Spangenberg 23 did not find any difference when rats descended an inclined plane after having been housed in the ERC, where rats could run between cages and a Makrolon IV cage. Further she did not find any differences between SD rats and spontaneously hypertensive rats in descending an incline at 61° which is lower than the 79° in this study. 23 However, since the inclined plane tests probably differed somewhat (e.g. surfaces) between the studies 23 the exact steepness of the incline might not be strictly comparable.
We did not find any differences in adrenal gland weights between cage types. Differences in adrenal gland weight have been found previously for rats subjected to different types of stressors, i.e. isolation, handling, crowding and high noise. 19 Kaliste-Korhonen et al. 11 found that male Wistar rats that had access to wooden gnawing blocks when being transferred to stainless steel wire mesh cages had lower adrenal gland weights than rats that did not get access to blocks. In a follow-up study no differences in adrenal gland weight between rats that had access to gnawing blocks or not were found, but rats kept in grid floor cages had larger adrenals than rats kept in solid bottom cages. 12 Altogether our results, with no difference between rats from the different cages in adrenal gland weight, performance of social behaviours and no observed socially related injuries, indicate that it was not more stressful for the rats to be housed in groups than in pairs.
Conclusion
It is concluded that group housing of five male rats of the strains SD and Wistar in ERCs and RRCs did not affect their growth rate, food and water intake, adrenal gland weight and some behaviours compared with pair-housing in MCs. The five rats in the enriched housing spent a large part of the time under a shelter whereas pair-housed rats in the MC were performing grooming, rearing, play fighting and interacting with the shredded paper substrate more often. Muscle strength was slightly higher in the RRC rats compared with the MC rats, but not compared with the ERC rats. The enrichment items (i.e. paper shreds and gnawing sticks) were used particularly when they had just been introduced into the cages. The SD rats had higher food and water intake, weight gain, relative adrenal gland weights and manipulated sticks more, but performed less rearing, moving and grooming than the Wistar rats. Altogether housing rats in larger cages in groups had no negative effects but increased the possibility of giving them access to valuable resources such as nests (which were well used) and access to more exercise which led, at least for the RRC rats, to increased muscle strength.
Footnotes
Acknowledgements
We would like to thank Scanbur A/S in Denmark for considering our ideas and the animal technicians at AstraZeneca in Mölndal for developing the ERC system. We also want to thank Elin Spangenberg and Emma Thorén for taking care of the rats and doing most of the recordings in this study. We thank Tomas Morsing for his advice on the statistical analysis and David Morton for language checking. The analysis of videos and data analysis was funded by Centrala Försöksdjursnämnden (CFN) in Sweden.
