Abstract
Sodium pentobarbital is a commonly used agent for euthanizing laboratory rats, however its high pH can cause abdominal discomfort after intraperitoneal injection. Previous studies suggest that the addition of a local anaesthetic may alleviate this discomfort, but the practice has not been widely adopted. We examined the effect of combining lidocaine with pentobarbital on abdominal writhing, defecation, ultrasonic vocalizations, the rat grimace scale and immunohistochemical staining for c-Fos in the nucleus accumbens and basolateral amygdala of the brain. We also compared the amount of abdominal writhing following intraperitoneal administration of pentobarbital–lidocaine with that of pentobarbital–bupivacaine. Our results show that lidocaine reduces abdominal writhing and defecation without affecting immunohistochemistry for c-Fos or latency to loss of posture. However, scores on the rat grimace scale were low in both situations and almost no ultrasonic vocalizations were recorded. Additionally, we found that the amount of abdominal writhing was not significantly different when bupivacaine was used rather than lidocaine. Our results suggest that pentobarbital-induced euthanasia can be refined with the addition of lidocaine or other local anaesthetics.
Sodium pentobarbital may be the least controversial method for the euthanasia of laboratory rodents. It is an injectable, fast-acting, central nervous system depressant which acts via type A gamma-aminobutyric acid (GABAA) receptors to cause a loss of consciousness and cardiovascular depression.1–3 There is effectively a global consensus that it is acceptable, with guidelines recommending its use in Australia, 4 Canada, 5 Europe, 6 India 7 and the USA. 8 This gives it an advantage over other euthanasia agents, such as carbon dioxide which is considered to be acceptable by some 8 but not others 6 because it can cause distress in rodents depending upon the method of administration.9,10
Pentobarbital is a very frequently used euthanasia agent, and it is also sometimes used as an anaesthetic agent. It is by far the most commonly used injectable anaesthetic for euthanasia, although some laboratories use urethane for its long half-life in electrophysiology studies while others use a combination of ketamine and xylazine for euthanasia as well as for anaesthesia. A small number of laboratories continue to use chloral hydrate or chloroform despite guidelines explicitly recommending against its use.8,11 Gaseous methods using carbon dioxide or isoflurane are also used frequently, while physical methods such as decapitation (with or without anaesthesia) or exsanguination under anaesthesia are used more rarely, typically for specific experiments involving electrophysiology or cell culture. While pentobarbital cannot be deployed on the scale of gases such as carbon dioxide, it remains the best available and most commonly used injectable euthanasia agent.
However, pentobarbital and other barbiturates have a high pH which causes irritation and pain during intraperitoneal injection.5,12 Unfortunately, this high pH is necessary for the solubility of pentobarbital and attempts to reduce the pH rapidly cause the precipitation of a sticky white solid. The use of the local anaesthetic lidocaine to refine a pentobarbital solution was first reported in a conference paper which showed a decrease in abdominal writhing in female Wistar rats without effects on latency to loss of posture. 13 A follow-up study examined spinal c-Fos expression 2 h and 45 min after pentobarbital administration in male Wistar rats. 14 It was found that there was approximately 35% less c-Fos immunoreactivity in the spinal dorsal horn of rats treated with a non-lethal dose of pentobarbital/lidocaine than rats treated with pentobarbital alone. However, this was a preliminary study with just four rats per group and there has been no detectable change in practice since it was published in 2007. Given that pentobarbital is the most common injectable euthanasia agent, refinements to pentobarbital euthanasia would provide an improvement to the welfare of a significant number of animals.
More evidence on the efficacy of this refinement may be required. There is currently no evidence indicating whether altering the anaesthetic mixture will affect experimental results, such as immunohistochemistry, and it has not been shown whether the effect is specific to lidocaine. The best available methods of assessing discrete emotions in rats during euthanasia include embodied emotion, as measured by the rat grimace scale 15 which quantifies the nociceptive response, and ultrasonic vocalizations which are associated with anxiety. 16 The aim of the present study was therefore to replicate previous findings13,14 using the rat grimace scale, 15 abdominal writhing, defecation and ultrasonic vocalizations. Moreover, since the efficacy of lidocaine refinement was first shown in female rats 13 and there has been no more than a preliminary demonstration involving male rats, 14 the present study used male rats. Scientists may also have concerns about any possible impact on experimental results, and these were addressed by assessing whether adding local anaesthetic will affect c-Fos staining in the nucleus accumbens and basolateral amygdala following exposure to a novel context containing palatable foods. Examination of c-Fos staining after a behavioural or experimental manipulation is a common design in neuroscience studies that has been used to examine patterns of neural activation, for example, during addiction or stress-related behaviour.17–20 Importantly, c-Fos expression occurs in response to strong neuronal activation, and since its expression is inhibited during pentobarbital anaesthesia 21 it could be more vulnerable to disruption if additional agents are administered during euthanasia than other proteins that have already been expressed. It also demonstrates whether the presence of lidocaine before perfusion affects antibody binding or imaging during the staining process. In the present study, rats were exposed to a novel context containing palatable food to model a c-Fos study using an appetitive stimulus.
Finally, we aimed to show whether this effect could be generalized to another local anaesthetic by testing the effect of bupivacaine on abdominal writhing during pentobarbital euthanasia. Bupivacaine has a slower onset of action and a longer duration than lidocaine and so could be expected to have a smaller effect or to be ineffective when compared with lidocaine. However, it also serves as a useful control because it targets voltage-gated potassium channels.22,23 Thus, a comparison between lidocaine and bupivacaine could demonstrate whether the fast action of lidocaine is specifically required or whether this effect generalizes to other local anaesthetics with different pharmacodynamic properties.
Materials and methods
Animals
Male Sprague–Dawley rats (n = 70) weighing 350–550 g were transferred to this study following the completion of a behavioural experiment involving self-administration of 4% alcoholic beer 24 or food or fear learning, 25 but with no prior history of injections, anaesthetics or surgery. Male Sprague–Dawley rats were chosen because they are a common animal model readily available in our laboratory and were evenly distributed between experimental conditions. They were originally obtained from the Animal Resource Centre (Perth, WA, Australia). Standard housing was in groups of four in plastic cages (309 mm wide, 617 mm long and 284 mm high, Airlaw, Golden Grove, SA, Australia) in ventilated racks with corncob bedding and an alloy mezzanine. The rats were housed in an environment with a 12:12 h light/dark cycle (lights on at 07:00 h), with ad libitum access to food and water throughout the experiment. The rats were handled over the course of one week to acclimatize them to their new experimenter before undergoing behavioural experiments, which were conducted during the light cycle. All procedures were approved by the University of New South Wales Animal Care and Ethics Committee, and were conducted in accordance with the National Health and Medical Research Council’s Australian Code for the Care and Use of Animals for Scientific Purposes.
Rat grimace scale
To habituate the rats (n = 24) to the testing apparatus, on day 1 of the experiment, they were placed in a small perspex chamber (21 × 10.5 × 9 cm high) with two transparent sides, two opaque black sides and a transparent top, as previously described. 15 The rats were videotaped in high definition (1080p) using Sony HD Action Cams (Sony, Tokyo, Japan) from the front, side and top. Ultrasonic vocalizations were recorded in the 22 kHz range using a Mini-3 Bat Detector (Ultra Sound Advice, London, UK). After 5 min in the chamber, the rats were removed, given a 1.5 mL injection of 0.9% saline (Pfizer, West Ryde, NSW, Australia) and returned to the chamber for 5 min. After which they were returned to their home cage.
On test day 2, a behavioural experiment was simulated by placing the rats in a novel context which consisted of a rectangular arena (71.1 × 86.4 × 55.9 cm) with a Petri dish containing a spoonful of peanut butter and a small handful of roasted cashew nuts for 25 min. This was undertaken to model a common design in neuroscience where pentobarbital euthanasia is often used and to address possible concerns that refinement may affect immunohistochemical results. The rats were then placed in a holding cage for 90 min. After which they were placed in the small perspex chamber for 5 min, given an overdose of sodium pentobarbital (2 mL) and then returned to the chamber until they lost posture. Fecal boli excreted during or after pentobarbital injection and before transcardial perfusion were counted. Rats were randomly allocated to receive either sodium pentobarbital (Virbac Animal Health, Milperra, NSW, Australia) diluted with saline to a final concentration of 162.5 mg/mL (pH = 10.5) or pentobarbital diluted with lidocaine (Ilium, Smithfield, NSW, Australia) to a final concentration of 162.5 mg/mL pentobarbital and 10 mg/mL lidocaine (pH = 10.1). A sample size of 12 per group was chosen because an a priori power analysis for a two-tailed independent t-test using G*Power 3.1.9.2 showed that it would provide a 90% chance of detecting a large effect (d = 1.5) with α = 0.05. 26
Scoring
All scoring was done manually on video recordings blinded to the experimental condition. Abdominal writhing involves contraction of lateral abdominal muscles (Figures 1a–c) and this was coded for duration.13,27 Immobility was defined as the point when the rat ceased movement for at least 20 s. Scoring of the rat grimace scale was performed as previously described
15
with adjustments made for the short time available to monitor for facial grimaces during euthanasia. Ten still images were captured during the baseline period at approximately 30 s intervals, and 5–10 images were captured during the post-injection period at intervals of approximately 3–5 s when a full view of the face was available. Attempts were made to acquire an even number of images from both front and side views, but this was not always possible. Grimaces were scored on a scale of 0–2 for four action units (orbital tightening, nose/cheek flattening, ear and whisker changes; Figures 1d–f) which were averaged for each still image. The final grimace score was the difference between the mean of the post-injection period and the mean of the baseline period.
Scoring abdominal writhing and the rat grimace scale. Abdominal writhing was scored for duration. These images show animals where writhing was (a) absent, (b) evident in lateral contractions of the abdomen and (c) evident in contractions, stretching and twisting. The rat grimace scale was coded according to the facial response of the animal. These representative still images were coded with a rat showing (d) no grimace response, (e) a moderate response including a change in ear position and some tightening of the whiskers and (f) a pronounced response including orbital tightening, nose flattening, change in ear position and whiskers standing on end. The pronounced response was rarely seen.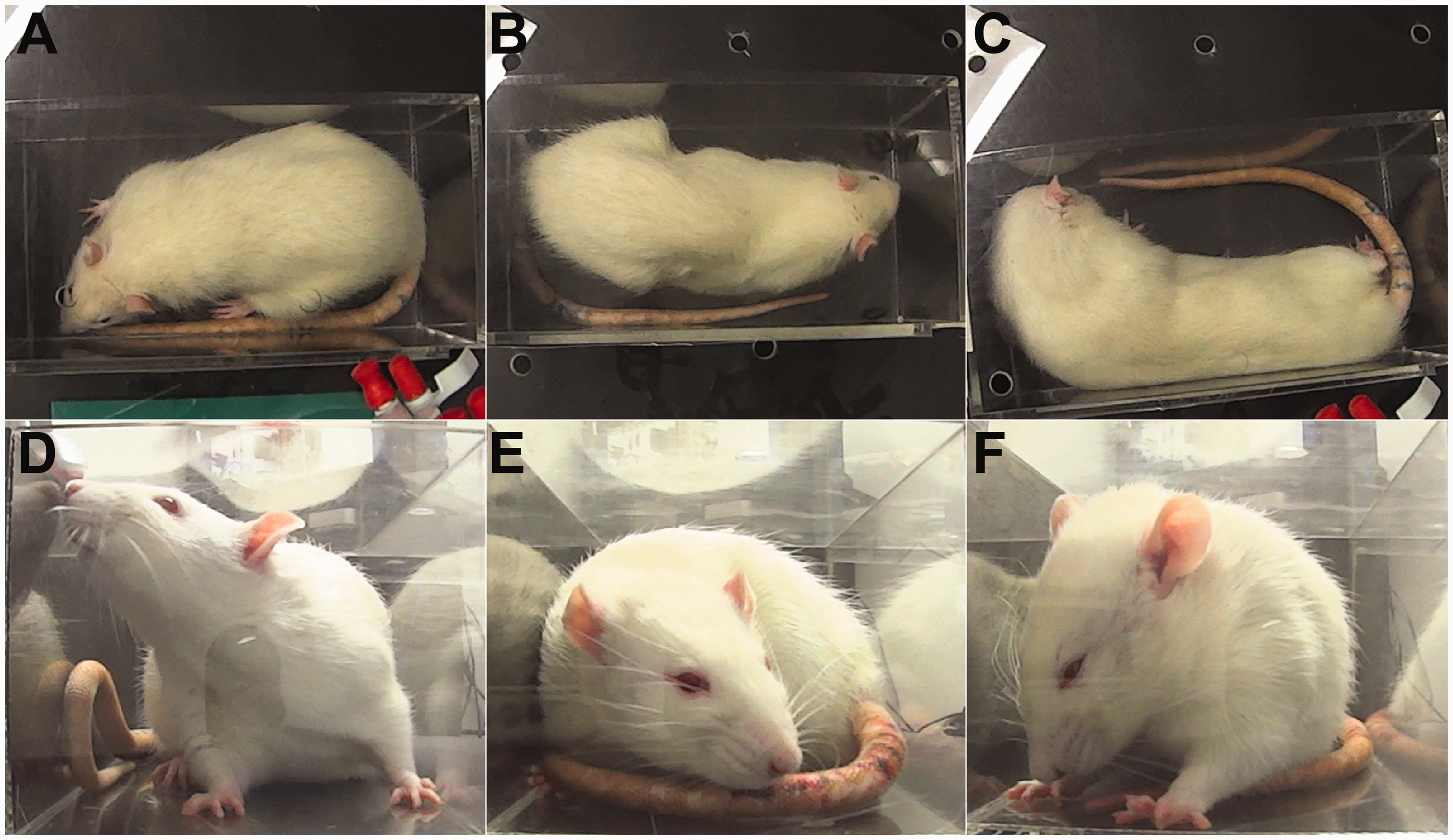
Perfusion
After the rats had lost posture they were transported to a fume hood for transcardial perfusion. Anaesthesia was verified by a paw pinch, and the rats were transcardially perfused with 150 mL of a heparinized 0.9% saline/0.0125% sodium nitrite solution. The tissue was then fixed using 200–300 mL of a 4% paraformaldehyde/0.1 M phosphate buffer solution (pH 7.2). Brains were dissected and post-fixed overnight before being placed in a 0.1 M phosphate buffer/20% sucrose solution overnight. They were then coronally sectioned in a 1:4 series at 40 µm in a cryostat (CM1950; Leica Microsystems, North Ryde, NSW, Australia) and stored in a 0.1 M phosphate buffered saline/1% sodium azide solution until ready for immunohistochemistry.
Immunohistochemistry
Brain slices were washed for 30 min in 0.1 M phosphate buffer, 50% ethanol, 50% ethanol/1% hydrogen peroxide and 0.1 M phosphate buffer/5% normal horse serum until they were incubated for 48 h in a 0.1 M phosphate buffered saline/1% sodium azide solution containing 2% normal horse serum, 0.2% triton-X and rabbit anti-c-Fos antibody (Santa Cruz Biotechnology, Dallas, TX, USA) diluted 1/2000. Brain slices were then washed 3 × 20 min in 0.1 M phosphate buffer and incubated in a 0.1 M phosphate buffer, 2% normal horse serum, 0.2% triton-X and biotinylated donkey anti-rabbit solution (diluted 1/2000; Jackson ImmunoResearch Laboratories, West Grove, PA, USA) overnight. Brain slices were then washed 3 × 20 min in 0.1 M phosphate buffer and incubated for 2 h in a 0.1 M phosphate buffer, 2% normal horse serum, 0.2% triton-X and ABC solution (Vectastain®; Vector Labs, Burlingame, CA, USA). Then brain slices were washed in 2 × 20 min in 0.1 M phosphate buffer, 1 × 20 min in 0.1 M sodium acetate (pH = 6) and incubated for 15 min in diaminobenzidine (0.2% D-glucose, 0.04% ammonium chloride and 2% nickel sulfate hexahydrate). The reaction was catalysed by glucose oxidase aspergillus for 8 min before the brain slices were washed in 0.1 M sodium acetate, 0.1 M phosphate buffer and returned to sodium azide before being mounted on 4% gelatinized slides. Cell counting was performed in the nucleus accumbens shell and basolateral amygdala using cellSens Dimensions (Olympus Life Science, Tokyo, Japan) based on anteroposterior atlas coordinates of +2.2 to +1 mm from the Bregma for the nucleus accumbens and –1.8 mm to –2.8 mm for the basolateral amygdala. 28 These regions were chosen because they are both important parts of the reward neurocircuitry that also respond to novel contexts.29–31 They would therefore have high levels of c-Fos expression which could be vulnerable to disruption if lidocaine has an adverse effect on immunohistochemistry. Bilateral images of three sections per region were taken at 10 × magnification on an Olympus BX50 microscope (Olympus). The boundaries of the region were then defined on each image by a person blinded to the experimental conditions for automatic counting.
Bupivacaine test
A separate cohort of rats (n = 46) were randomly allocated to receive sodium pentobarbital (162.5 mg/mL) with saline (0.45%), lidocaine (10 mg/mL) or bupivacaine (final concentration of 2.5 mg/mL, pH = 10.2; AstraZeneca, West Ryde, NSW, Australia). Rats in this experiment received an overdose of pentobarbital solution (2 mL) and were placed in an opaque cylindrical chamber (19 cm radius, 40 cm height) with corncob bedding. Video recordings were taken from above and scored for abdominal writhing as described above. A sample size of 14–16 per group was chosen because a priori power analysis for a three-group, one-way omnibus analysis of variance (ANOVA) showed that this would provide at least a 90% chance of detecting a large effect size (η2p = 0.25) with α = 0.05.
Data analysis
Data were analysed using SPSS 22.0 software (IBM, Armonk, NY, USA) using independent t-tests and a one-way ANOVA using Helmert contrasts 32 to compare the control group with the treatment groups, and the treatment groups with each other. Degrees of freedom were adjusted as required following a significant Levene’s test for homogeneity of variance. Data are presented as means ± SEM. As a null result was expected for latency to loss of posture and for immunohistochemical results, inverse Bayes factors (BF01) were calculated using JASP 0.7.5.6. Raw data are available on Figshare (doi: 10.6084/m9.figshare.4551682).
Results
Lidocaine reduces defecation and abdominal writhing but not grimace scores or latency to immobility
Rats were randomly assigned to receive either 162.5 mg/mL pentobarbital (Pento–Sal, n = 12) or 162.5 mg/mL pentobarbital/10 mg/mL lidocaine (Pento–Li, n = 12). The duration of abdominal writhing and latency to immobility was scored blind based on the top-down video. The mean duration ( ± SEM) of abdominal writhing in the Pento–Sal group was 34.2 ± 4.22 s, and was significantly lower for the Pento–Li group at 22.1 ± 3.82 s (t(22) = 2.14, P = 0.044; Figure 2a). There were no significant differences in latency to immobility (t(22) = 0.264, P = 0.794, BF01 = 2.61; Figure 2b), which were 66.5 ± 7.02 s for the Pento–Sal group and 63.5 ± 8.95 s for the Pento–Li group. The number of fecal boli excreted by rats in the Pento–Li group was 0.75 ± 0.22 boli which was significantly lower than the 0.08 ± 0.08 boli excreted by the Pento–Sal group (t(14.16) = 2.86, P = 0.012; Figure 2c). Grimace scores (grimace score post-injection – grimace score pre-injection) were 0.44 ± 0.14 for the Pento–Sal group and 0.37 ± 0.11 for the Pento–Li group, and these were not significantly different (t(22) = 0.381, P = 0.707; Figure 2d). Too few ultrasonic vocalizations were detected for analysis. Only two rats emitted any ultrasonic vocalizations, of which one emitted ultrasonic vocalizations during both the baseline and post-injection periods.
Lidocaine reduced the duration of abdominal writhing and defecation without affecting latency to immobility or grimace score. (a) Lidocaine reduced the duration of abdominal writhing. (b) Lidocaine did not reduce the latency to immobility. (c) Lidocaine reduced defecation. (d) Low levels of grimace were observed in both conditions but this was not significantly different in either group. Data are means ± SEM. *P < 0.05.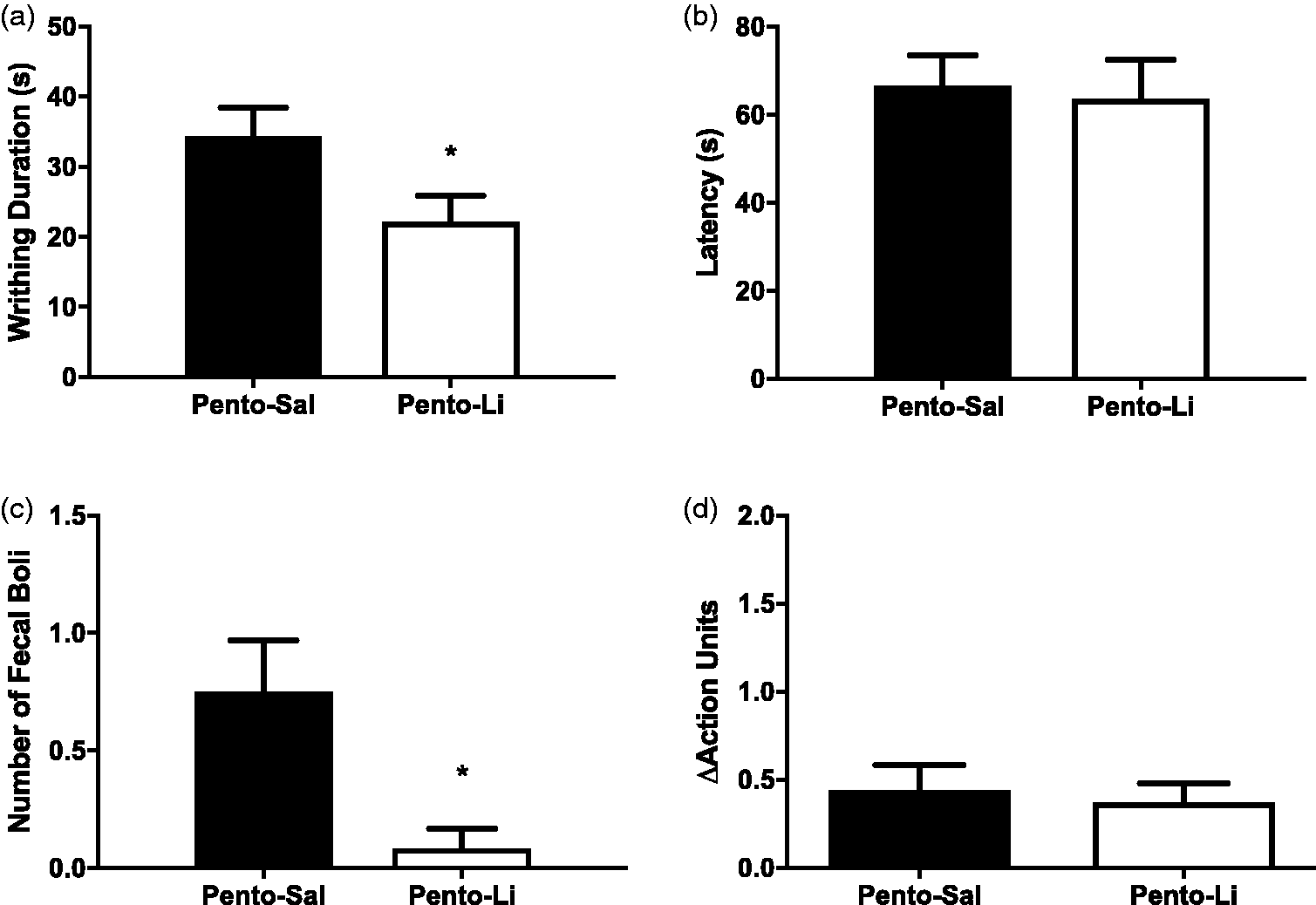
Lidocaine has no effect on c-Fos immunoreactivity
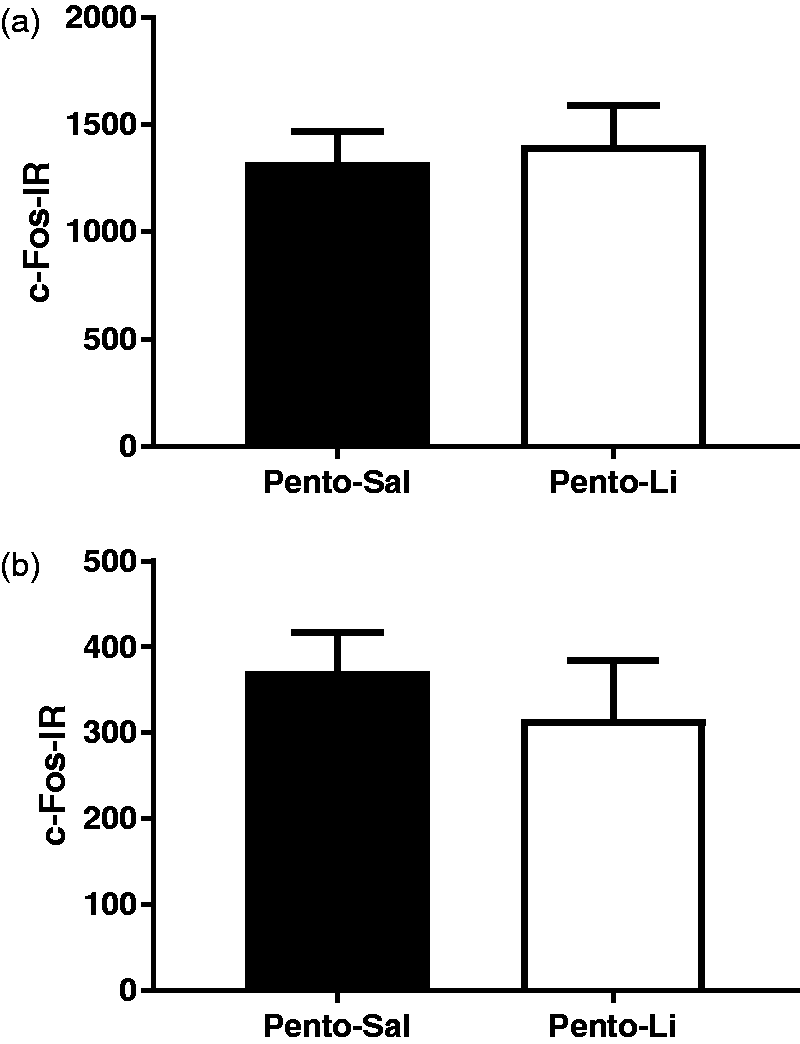
The nucleus accumbens shell and the basolateral amygdala were examined for c-Fos immunoreactivity (c-Fos-IR). As shown in Figure 3, there were no qualitative differences between the rats in the Pento–Sal or Pento–Li groups as evidenced by identical size, shape, colour and intensity of cell staining in both the Pento–Sal and Pento–Li groups. Quantitative analysis, presented in Figure 4, also shows that the euthanasia solution made no significant differences to c-Fos-IR. The Pento–Sal group had 1323 ± 144 c-Fos-IR cells while the Pento–Li group had 1403 ± 187 c-Fos-IR cells in the accumbens (t(22) = –0.338, P = 0.739, BF01 = 2.57). Similarly the Pento–Sal group had 371 ± 45 c-Fos-IR cells while the Pento–Li group had 316 ± 68.4 c-Fos-IR cells in the basolateral amygdala (t(22) = 0.68, P = 0.503, BF01 = 2.26).
Representative photomicrographs showing c-Fos immunoreactivity. Images from the nucleus accumbens of rats in the Pento–Sal group (a) were not qualitatively different in stain size, shape, colour or intensity from rats in the Pento–Li group (b). Similarly, images from the basolateral amygdala of rats in the Pento–Sal group (c) were not qualitatively different from rats in the Pento–Li group (d). Scale bar represents 250 µm. Cell counting results show no differences in c-Fos-IR in either (a) the nucleus accumbens or (b) the basolateral amygdala. Data are means ± SEM.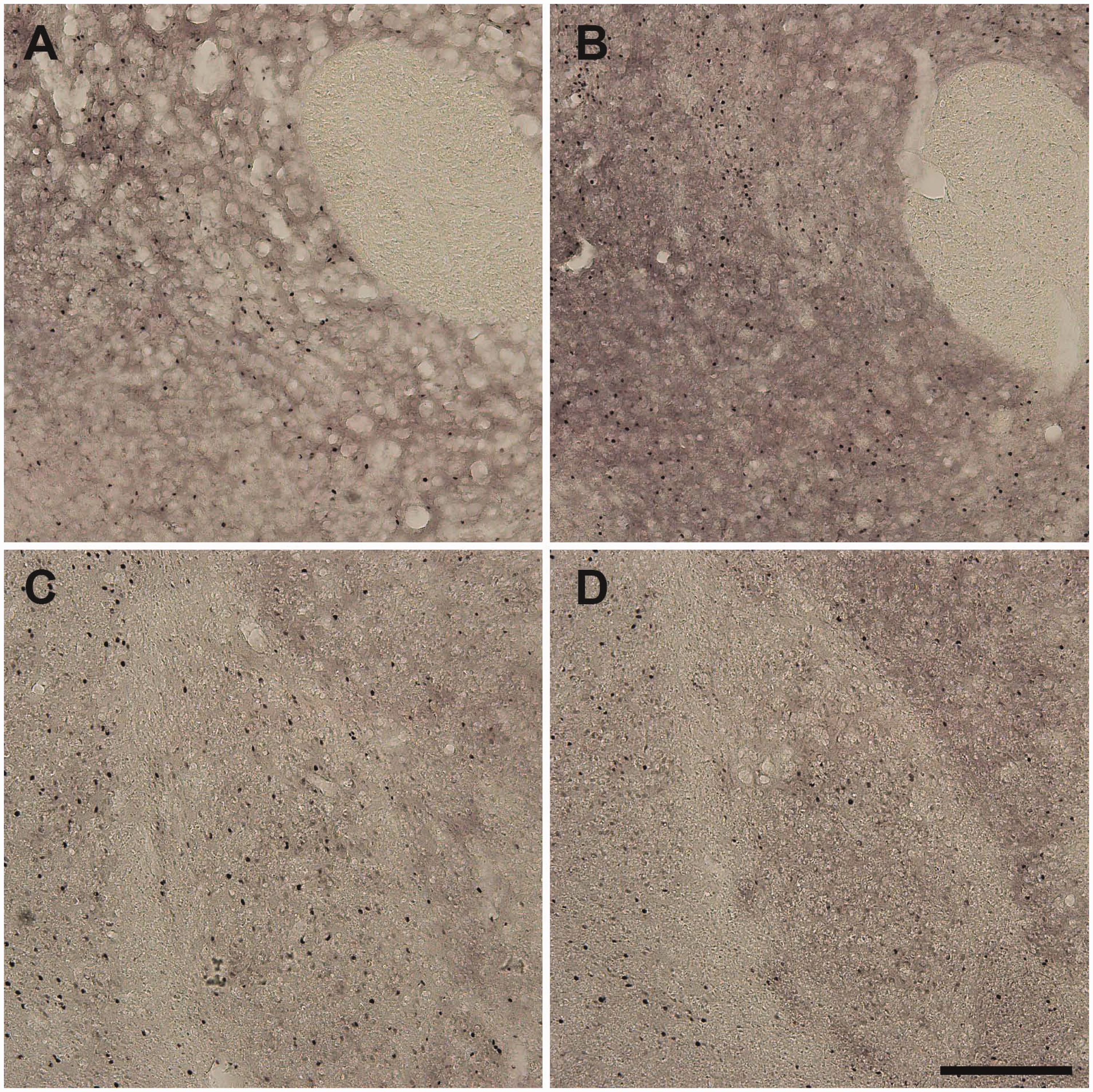
Bupivacaine also reduces abdominal writhing
Rats were randomly assigned to receive either 162.5 mg/mL pentobarbital (Pento–Sal, n = 16) or 162.5 mg/mL pentobarbital/10 mg/mL lidocaine (Pento–Li, n = 16) or 162.5 mg/mL pentobarbital/2.5 mg/mL bupivacaine (Pento–Bup, n = 14). As shown in Figure 5, the Pento–Sal group had a significantly longer duration of abdominal writhing of 20.8 ± 3.8 s than the Pento–Li and Pento–Bup groups (F(1,20.74) = 8.39, P = 0.009), which had durations of 5.94 ± 2.26 s and 11.6 ± 2.39 s, respectively. The Pento–Li and Pento–Bup groups were not significantly different from each other (F(1,27.54) = 2.93, P = 0.098). There were no differences between groups in latency to loss of posture (F(2,43) = 0.198, P = 0.821, BF01 = 5.38) which were 68.3 ± 4.92 s for the Pento–Sal group, 67.9 ± 6.16 s for the Pento–Li group and 72.8 ± 6.73 s for the Pento–Bup group.
Lidocaine and bupivacaine reduced the duration of abdominal writhing without affecting latency to immobility. (a) Abdominal writhing was reduced in both the Pento–Li and Pento–Bup groups, but these groups were not significantly different to each other. (b) There were no differences in the latency to immobility. Data are means ± SEM. *P < 0.05 compared with Pento–Sal.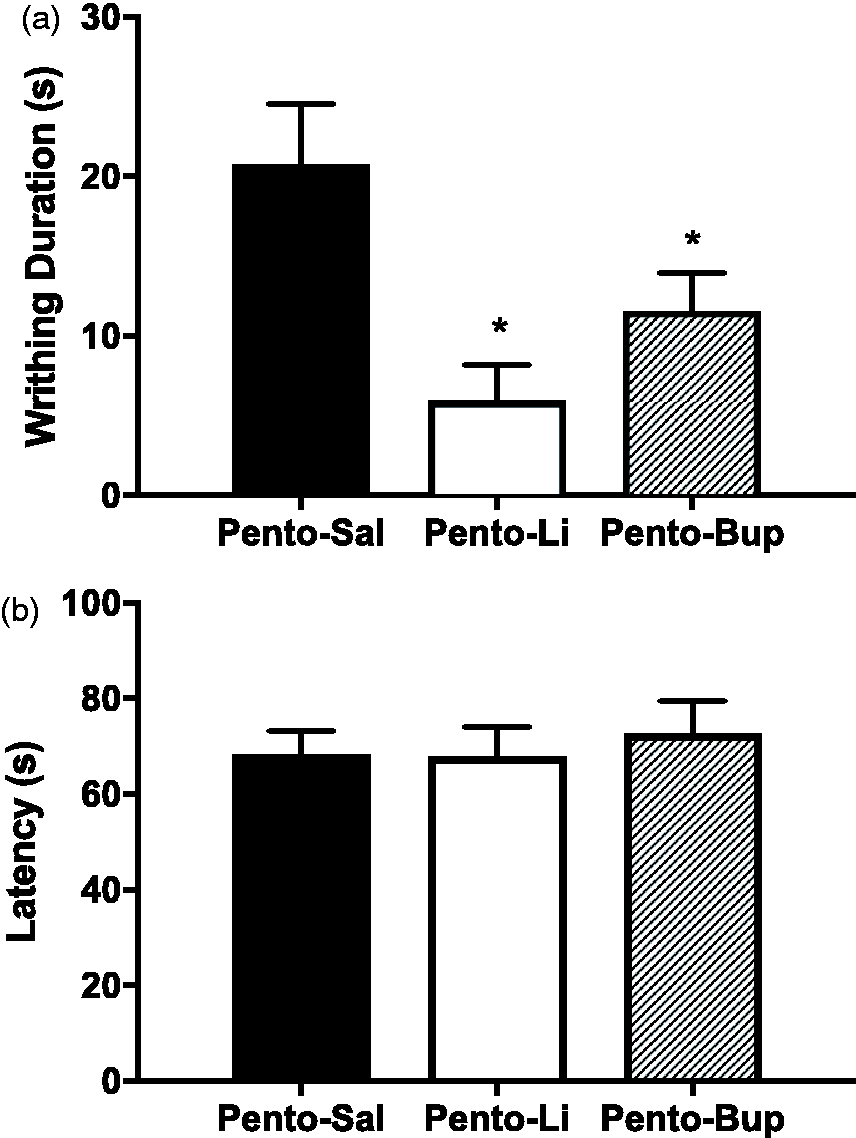
Discussion
The present study has replicated and extended previous findings 13 by showing that lidocaine reduces abdominal writhing and defecation during pentobarbital euthanasia without affecting pentobarbital's efficacy or immunohistochemical staining for c-Fos. The effect on abdominal writhing also generalizes to bupivacaine. Additionally, we have shown that pentobarbital euthanasia does not cause a high level of distress because no significant anxiety-associated 22 kHz vocalizations were detected; and the nociceptive response, which while present, was only evident in modest scores on the rat grimace scale.
The present finding that lidocaine reduces abdominal writhing is most consistent with the local anaesthetic properties of lidocaine. Lidocaine and other local anaesthetics work by blocking voltage-gated sodium channels which prevent action potentials and the transmission of pain signals back to the brain.33–35 While lidocaine has effects on neuromuscular function during clinical procedures, 36 it does not reduce muscle artefacts observed during non-invasive electroencephalography, 37 suggesting that this is due to reduced sensory feedback rather than a muscle relaxant or paralytic effect. Moreover, a muscle relaxant effect does not explain a reduction in defecation nor does it explain the similar effect of bupivacaine, which has been shown to have opposite effects to lidocaine on umbilical smooth muscle. 38 It is interesting that both lidocaine and bupivacaine had similar effects even though bupivacaine is a slower acting anaesthetic. However, there were no meaningful differences in pH between the Pento–Li (pH = 10.1), Pento–Bup (pH = 10.2) and Pento–Sal (pH = 10.5) solutions. Since bupivacaine and lidocaine have different molecular targets, with bupivacaine acting on voltage-gated potassium channels,22,23 these data suggest that the reduction in abdominal writhing was due to the local anaesthetic effect rather than the specific molecular target of lidocaine or bupivacaine. While lidocaine remains the local anaesthetic of choice for its faster onset, bupivacaine may also be beneficial if lidocaine is unavailable. It is therefore the local anaesthetic properties of lidocaine and bupivacaine that are the most likely cause of the observed reduction in abdominal writhing.
Immunohistochemical staining for c-Fos was also unaffected, suggesting that there is unlikely to be any impediment to the addition of local anaesthetics to the euthanasia solution for studies where postmortem immunohistochemistry is to be performed. A previous study has used c-Fos staining in the dorsal horn to examine the effects of lidocaine on nociception, 14 but there has been no previous attempt to examine whether lidocaine during euthanasia could affect experimental results. In order to examine whether adding lidocaine to the euthanasia solution might affect an immunohistochemical endpoint, the present study examined c-Fos-IR after exposure to a novel context containing palatable foods in two brain regions which respond to reward and novelty, the nucleus accumbens and basolateral amygdala.17–20 The c-Fos marker is an immediate early gene that is expressed during strong neuronal activation. 39 It was chosen because as newly expressed protein it might be more easily disrupted, and since its expression is inhibited during pentobarbital euthanasia 21 scientists may be more concerned that it may be affected by adding a local anaesthetic during euthanasia. However, this was not the case. There was strong c-Fos expression in both the nucleus accumbens and basolateral amygdala, regions which are involved in responding to rewards. 30 No qualitative (i.e. size, shape, colour or intensity) or quantitative differences were found in c-Fos-IR in these regions. This confirms that there is no interference with the general process of antibody binding and labelling, at least with a diaminobenzidine-based protocol. It suggests that scientists using immunohistochemical endpoints may be able to adopt refined euthanasia protocols without affecting these results. We have adopted this refinement in experiments involving immunohistochemistry for other biomarkers without any apparent adverse effects on results. 40 However, effects on other immunohistochemical targets cannot be ruled out, especially if they have binding sites for local anaesthetics.
The behavioural data suggest that pentobarbital euthanasia does not cause high levels of distress. Both groups showed a grimace score of less than 0.5, which is equivalent to an average increase of just one point on one of the four action units that make up the scale. This indicates that the levels of pain were fairly modest. Additionally, the rats made so few 22 kHz ultrasonic vocalizations that the data were insufficient for analysis, with only two out of 24 rats making any 22 kHz calls. The 22 kHz calls could be in response to discomfort or distress to warn conspecifics and have been associated with aversive stimuli, such as predators or diazepam-withdrawal, in a variety of experimental conditions.41–45 One possible explanation for this is that the pentobarbital's anxiolytic effects counteract any distress that might be caused during euthanasia. These results suggest that pentobarbital euthanasia in rats does not cause anxiety, as measured by ultrasonic vocalizations, and causes only a modest level of pain as assessed by the rat grimace scale.
Limitations
The present study provides behavioural evidence that local anaesthetic refines pentobarbital euthanasia, but it has several limitations. The experiments in this study were powered to detect large effect sizes, which is standard for basic preclinical research. However there may have been smaller effects that may be real but may not have reached statistical significance. For example, in the bupivacaine test, visual inspection might suggest that bupivacaine may be less effective than lidocaine, but that would be a relatively small effect that the present study was not sufficiently powered to detect. If there was a small difference, it would be consistent with the slower onset of bupivacaine relative to lidocaine. The rat grimace scale may also not have the sensitivity required to properly assess nociception during euthanasia. While it did show that rats experienced pain post-injection compared with pre-injection, consistent with the abdominal writhing results, it was not sensitive enough to show the reduction caused by the addition of lidocaine. This may be because the pain experienced during euthanasia is very temporary and the animal rapidly loses consciousness or because pain levels are not high enough for differences to become evident.
Conclusion
Euthanasia by pentobarbital does not cause high levels of distress to rats as assessed by the rat grimace scale but it does cause abdominal discomfort which can be reduced by adding a local anaesthetic. Lidocaine is the anaesthetic of choice for this purpose because it is fast-acting and readily available. Bupivacaine may also be an effective substitute if lidocaine is unavailable. Importantly, lidocaine does not affect immunohistochemical staining for c-Fos; and while validation for additional biomarkers is necessary its use can be encouraged as it is unlikely to affect experimental results.
Footnotes
Acknowledgements
The authors gratefully acknowledge Katrina Blazek for advice on animal care and ethics committee (ACEC) procedures, Fred Westbrook for comments on the study design, Gabrielle Gibson for technical assistance and Joanna Makowska and Gilly Griffin for assistance locating the original Ambrose study.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The preparation of this manuscript was supported by grants from the National Health and Medical Research Council (GNT1077804 and GNT1098436) and by a Future Fellowship from the Australian Research Council (FT120100250) to GPM. SY-SK was supported by an Australian Postgraduate Award.
