Abstract
Preclinical cardiovascular research is the foundation of our understanding and broad knowledge of heart function and cardiovascular disease. Reliable cardiac imaging modalities are the basis for applicable results. Four-dimensional cardiac magnetic resonance (4D CMR) has been set as the gold standard for in-vivo assessment of ventricular function in rodents. However, technical improvements in echocardiography now allow us to image the whole heart, which makes four-dimensional echocardiography (4DE) a possible alternative to 4D CMR. To date, no study has systematically assessed 4DE in comparison with 4D CMR in rats. In total we studied 26 juvenile Sprague-Dawley rats (Crl: CD (SD) IGS). Twenty rats underwent echocardiographic imaging (2D and 4D) and 4D CMR. Five of those rats underwent a ligation of the superior and inferior vena cava to reduce the cardiac inflow as a disease model. Six additional rats were used to assess reproducibility of echocardiography and underwent three echocardiographic examinations. 4D CMR was performed on a 7T scanner; 2D and 4D echocardiography was conducted using a 40 MHz transducer. Correlation between 4D CMR, 4DE and 2DE for left-ventricular ejection fraction (LVEF) was assessed. An excellent correlation was observed between 4DE and 4D CMR (r = 0.95, p < 0.001). Correlation of 2DE and 4D CMR was weak (r = 0.57, p < 0.01). 4DE provides results that are equally precise as 4D CMR and highly reproducible with less technical effort than 4D CMR.
Introduction
Non-invasive cardiac imaging is an integral part of preclinical cardiovascular research. 1 Rodents in particular are frequently used to evaluate cardiovascular disorders 2 in order to understand physiological, molecular and biochemical mechanisms of cardiovascular diseases. 3 This research requires a reliable imaging technique to gain valid data. Different imaging modalities are available to determine heart function, each having their advantages and disadvantages concerning acquisition time, availability and costs.
Among these tools, the first to be mentioned is four-dimensional cardiac magnetic resonance imaging (4D CMR). At this point in time, 4D CMR is the gold standard for assessing left ventricular (LV) function in humans 4 and in rodents 5 due to its ability to obtain 3D images over time (with time being the fourth dimension) with few geometric assumptions.6,7 The high spatial and moderate temporal resolution of 4D CMR provides highly reproducible images with minimal inter- and intra-observer variations. 8 However, most research institutions do not have small animal MR scanners available. As a result, echocardiography is the method of choice for many researchers,9,10 as it produces images with high resolution at lower cost and shorter scan duration than 4D CMR.
Currently the determination of LV volumes from 2D images is a widely used approach in small animal echocardiography. Due to the circumstance that only one slice is used to calculate volumetric heart function, two-dimensional echocardiography (2DE) is only accurate in normal functioning hearts and highly dependent on the experience of the operator. 11 There are different approaches to determine LV function from transthoracic echocardiography (TTE), relying on either 1D M-mode evaluation, 2D short axis view, or 2D long axis view. M-mode only provides information about two opposing myocardial walls, e.g. the anteroseptum and the posterior wall from parasternal views. Calculation of left ventricular ejection fraction (LVEF) from parasternal short axis or long axis view in B-Mode using LV-Trace can provide a more representative evaluation of LVEF, but it displays only one slice of the heart not capturing the rest of the myocardium. In 2004 Dawson et al 12 developed a method of three-dimensional echocardiography (3DE): 10 to 18 consecutive short axis views were acquired by moving the mouse platform in 0.5 mm steps with the linear array probe being secured in one position. Respiratory gating and ECG were used. Using this technique Dawson at al measured no significant difference concerning LV mass, volumes and EF in comparison with 4D CMR. 12
Arias et al. 13 compared 4D CMR with 2DE that used geometric models (hemisphere cylinders) to calculate LV volumes. In their publication they claimed matrix-array transducers and gated imaging need to be able to acquire 4D imaging by echocardiography. In 2016 a new method of echocardiography became available, one that produces 3D images of the beating heart.
Similar to 4D CMR many slices are acquired; they are then put together to create a 3D model of the beating heart (Figure 1).
Principal geometric figures used to represent the left ventricle. (a) Algorithms for a full volume data set of 4D CMR and 4DE; the volume of each slice gets calculated separately and to get the full volume all slices are added. (b) Volume quantification for 2DE by ellipsoid formula using only one single slice.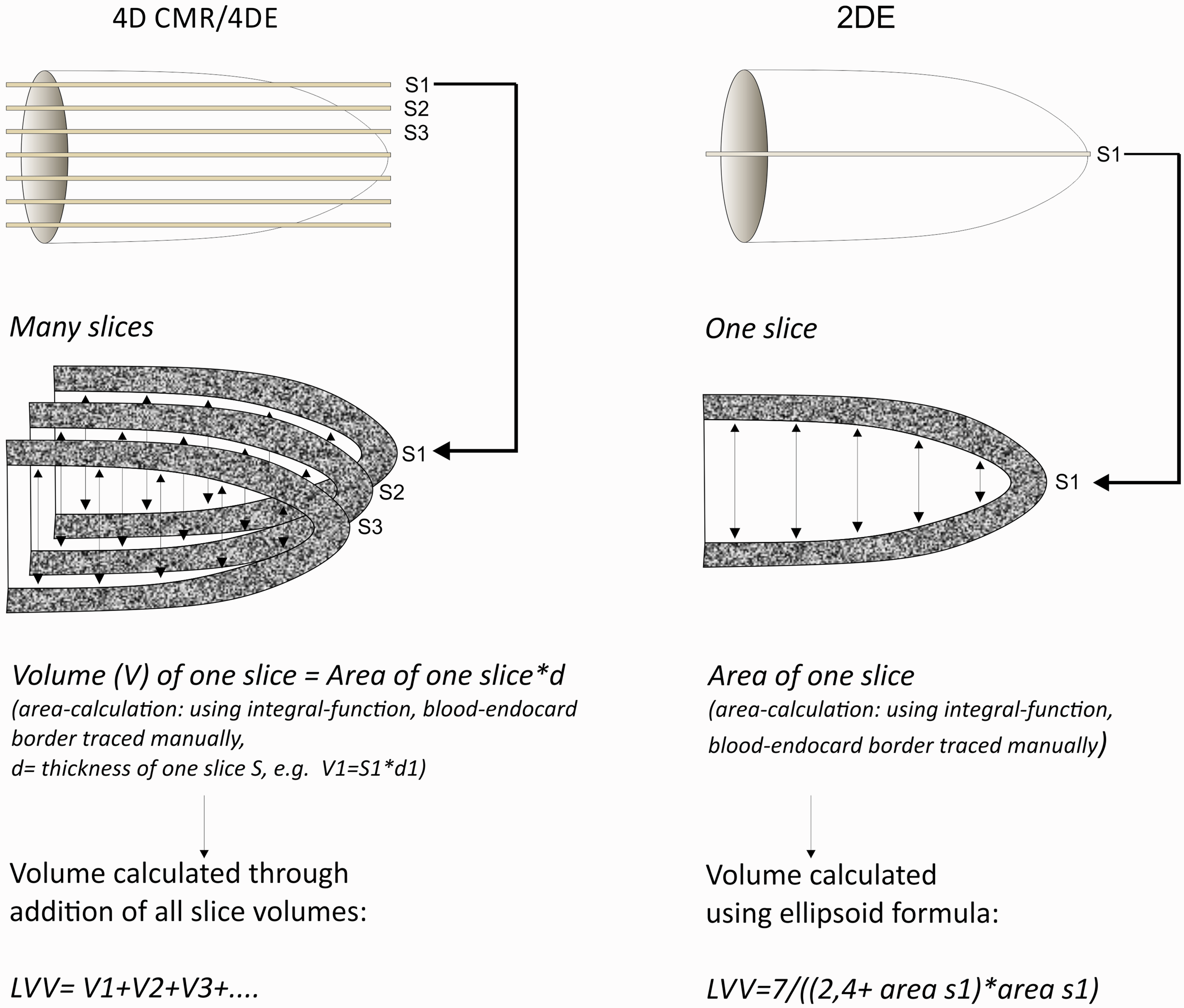
Compared with Dawson et al 12 the number of slices that can be acquired are increased significantly by the technical progress of hardware and software. The probe is now connected to a motor that is able to move through the heart in steps of a few micrometres. The acquired slices are automatically combined to a 3D model of the beating heart. Clinical 4D ultrasound uses convex probes 14 that are only able to create cone-shaped 4D models. Unfortunately, important structures such as the apex and myocardial walls are often cut off due to the geometrical circumstances. 15 To eliminate this error the Vevo® 3100 imaging platform uses linear array transducers creating a cube-like model.
Accordingly, the objective of our study was to compare four-dimensional echocardiography (4DE) with the gold standard 4D CMR in order to test the hypothesis that 4DE is a valid method for cardiac imaging. To the best of our knowledge, no evaluation of 4D CMR, 4DE and 2DE has been undertaken in rats. Therefore, we sought to determine the accuracy, objectivity and reproducibility of 4DE in comparison with 4D CMR.
Animals, material and methods
Study protocol
Animals were obtained and handled as approved by the Franz-Penzoldt Centre of the Friedrich-Alexander University Erlangen-Nürnberg in accordance with the EU Directive 2010/63/EU for animal experiments. The study was confirmed by the government according to the German law for animal protection (Tierschutzgesetz Regierung Unterfranken). In total we studied 26 juvenile Sprague-Dawleys rats (Crl: CD (SD) IGS) weighing 75–100 g from our institutional breeding colony. The original animal supplier was Charles River (Wilmington, MA, USA). We decided to examine juvenile rats because those animals were needed for further experiments at a later age. Both male and female rats were used. Animals were kept in a 12 h:12 h light:dark cycle at room temperature between 21–24℃, food (V1534000, ssniff rat/mouse, Ssniff GmbH, Soest, Germany) and water were provided ad libitum. All rats were not handled before their inclusion in the study except for routine cage cleaning maintenance and had no history of health abnormalities. Echocardiography and 4D CMR were performed in 20 rats at four weeks age. To compare the method of assessment in healthy and diseased animals, five rats underwent ligation of the superior and inferior vena cava to reduce cardiac inflow two weeks prior to imaging (method in publishing process). The time interval between 4D CMR and echocardiography was 0 ± 1 d. The other 6 animals underwent three echocardiographic scans to assess reproducibility. Rats were anaesthetized with 1.5% isoflurane for both imaging modalities. 16
Disease model
Five animals aged 14 ± 1 d underwent surgical banding of the superior (SVC) and inferior (IVC) vena cava. Anaesthesia was induced with 5% isoflurane and maintained with 1.5% isoflurane. The animals were intubated and buprenorphine (0.05–0.1 mg kg−1 bodyweight) was given for analgesia. Chest hair was removed with a small animal clipper.
The thoracic cavity was opened and the SVC and IVC were carefully dissected. The diameters of SVC and IVC were reduced to a standardized 0.6 mm to minimize blood flow to the right heart.
After closing the thorax, isoflurane was switched off and a second dose of buprenorphine was given intra-peritoneal when the animal was breathing sufficiently. The fully awake animal was returned to its mother’s care and closely monitored for the next 24 h. Analgesia was provided with buprenorphine 0.1 mg kg−1 bodyweight every 8 h for the next three days.
Cardiac magnetic resonance imaging
Comparison of acquisition parameters of 4D CMR and 4DE.

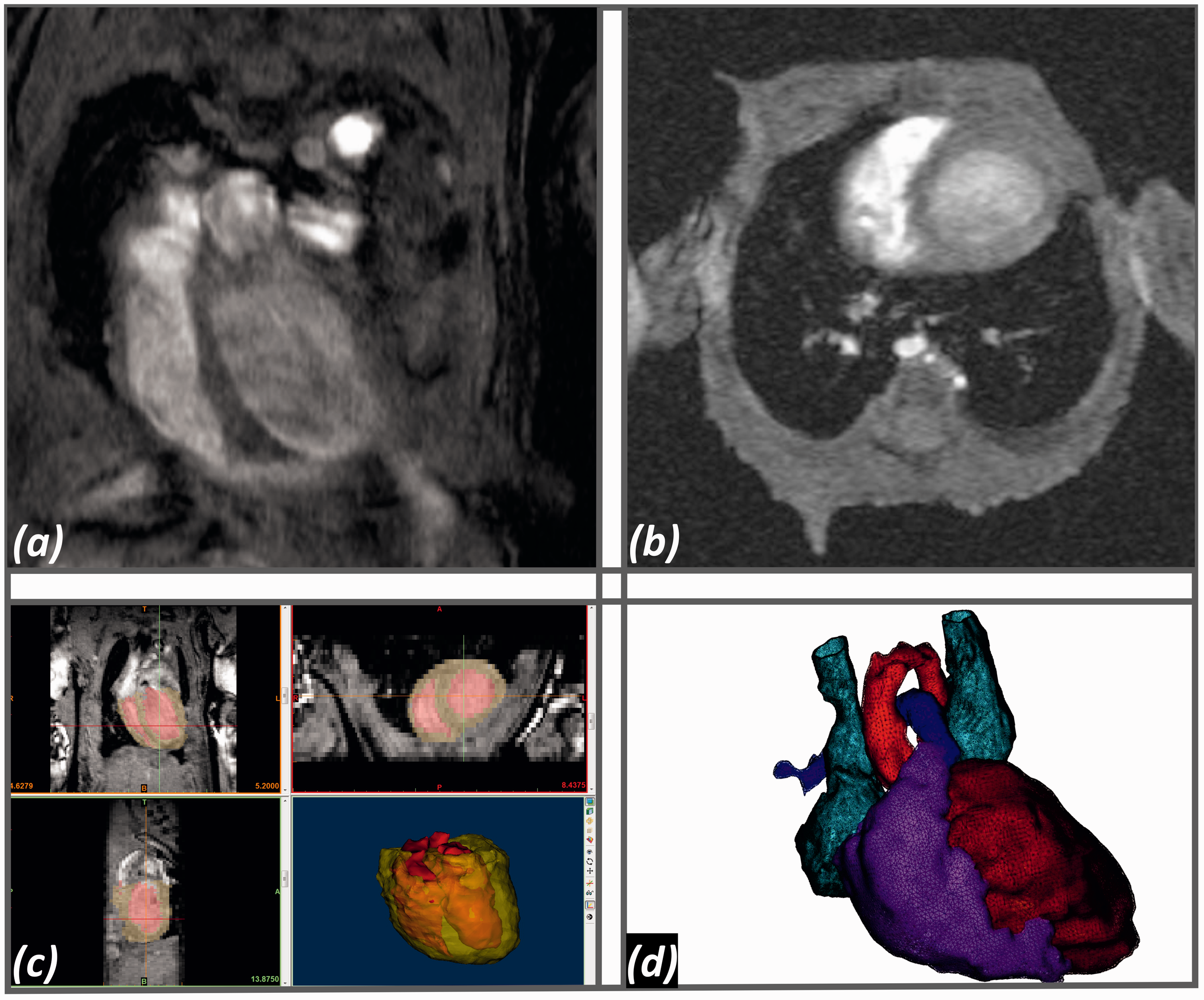
Four-dimensional cardiac magnetic resonance (4D CMR). (a) Long axis in end-diastole. (b) Short axis in end-diastole. (c) Mimics Research 19.0, end-diastolic 4D CMR images including the calculated 3D model of the left and right ventricle (red) and the myocardium (yellow). (d) End-diastolic 3D model of the whole heart for volume measuring using Mimics Research 19.0 based on 4D CMR images. Dark red, left ventricle; light red, aortic arch; purple, right ventricle and pulmonary artery; blue, superior vena cava and left persistent superior vena cava.
Echocardiography
The images were acquired using a 3100 Vevo® Imaging Platform (Fujifilm VisualSonics Inc, Toronto, Canada) with a 40 MHz linear array transducer (MS 400). In 2016 the Vevo® 3100 imaging platform integrated a motor into their system and developed software that can calculate a 3D model of the beating heart (time as fourth dimension, therefore also referred to as real-time 3DE) by combining the imaged slices. In addition, gated imaging allows for the heart cycle to be split into different phases to differentiate between systole and diastole. For 4DE the probe is mounted into a bracket ensuring a horizontal movement of the probe without any vertical deviation. The rats were under anaesthesia with 1.5% isoflurane
16
via mask and placed in a supine position on a heat-regulated table. Chest hair was removed, using a small animal clipper. The experimental set-up is shown in Figure 3(a). Heart rate and respiratory rate were monitored. A rectal probe registered core temperature, which was held stable at 37.0 ± 0.5℃. 4DE (Figure 3(b)) and 2D parasternal long axis (Figure 3(d)) were performed. Parasternal long axis (PSLAX) was chosen as 2DE method, because this was the starting position for 4D scanning and the examination time was not prolonged by short axis image acquisition. The investigator uses adjusting screws, moving the bracket with the probe to get into the position of PSLAX. Once the probe is in the right position, the system is to scan through the whole heart systematically across the orientation of PSLAX. The total study duration including 2DE took between 15–20 min per animal. 4D scanning was performed at the beginning of the examination and took 8 min per animal in the highest definition with a slice thickness of 10 µm and 200 frames per cycle (overview in Table 1). The average number of slices was 480. All data was evaluated offline using the Vevo LAB 3.0 software. Using this software the investigator had to mark the endocardium in an average of six slices, in diastole and systole. The software was then able to trace the endocardium in the other slices. Papillary muscle was not included in the blood volume using either software.
Transthoracic echocardiography (TTE). (a) Photograph illustrating TTE set-up with 40 MHz transducer fixed in bracket that is connected to the motor. (b) Coordinate 4D view of the rat heart showing the left ventricular long axis (apex left, base right). (c) M-Mode sequence. (d) Representative 2D parasternal long axis in end-diastole.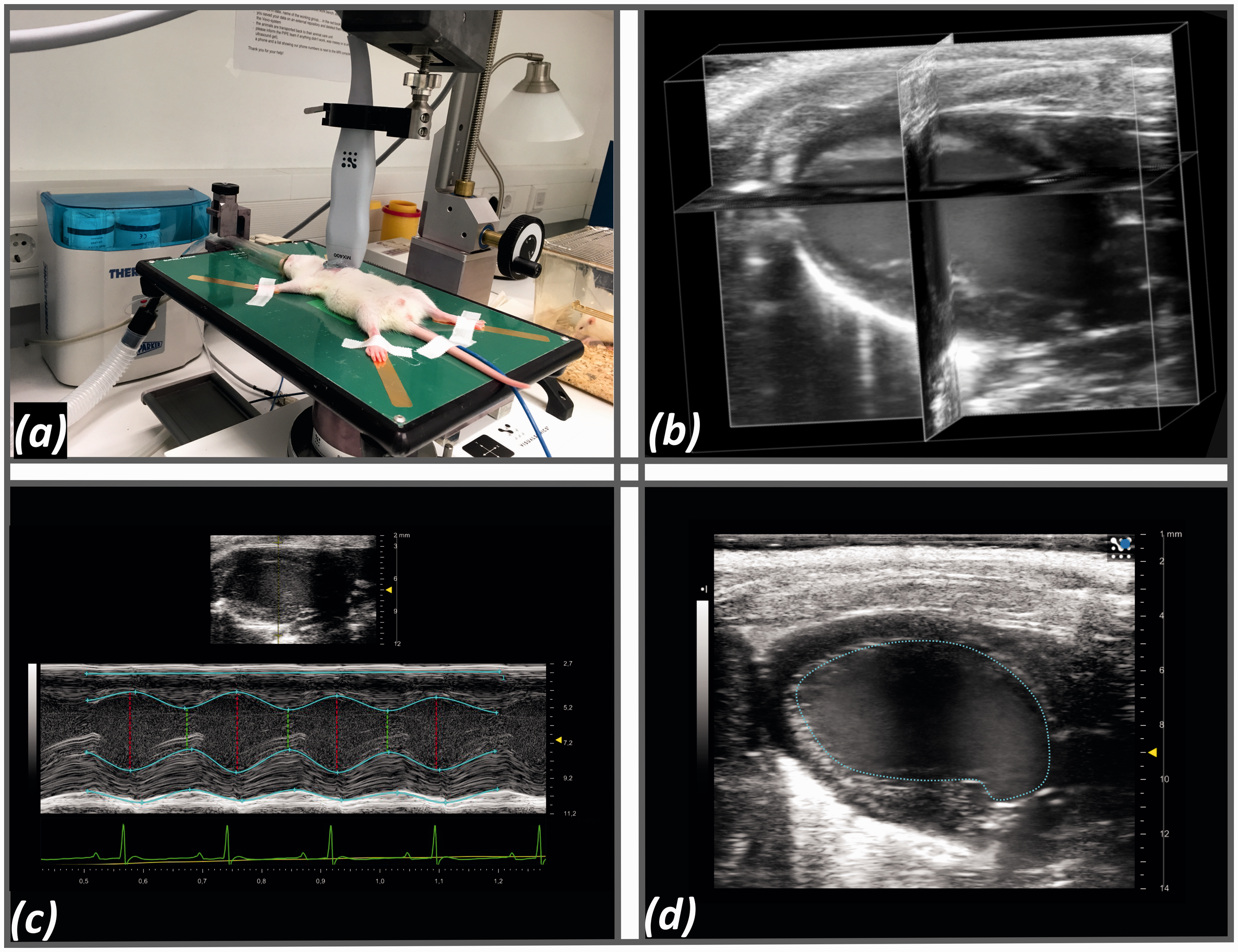
Reproducibility
To assess the intra- and interobserver reproducibility of echocardiography six additional animals were examined twice by the same observer and underwent a further echocardiographic scan by an independent observer. All three scans were performed in different periods of anaesthesia with an interval of at least two hours.
Statistics
IBM SPSS Statistics 21 (IBM Corp. Released 2012. IBM SPSS Statistics for Windows, Version 21.0. Armonk, NY) was used for statistical analyses. LVEF values were expressed as mean ± standard deviation (SD). Bland-Altman analysis was conducted with mean bias, standard deviation and 95% limits of agreement (mean ± 1.96 SD) calculated. Pearson’s correlation analysis was performed between LVEF derived by 4DE, 4D CMR and 2DE. Intra- and interobserver reproducibility was tested by calculating the interclass correlation for two consecutive measurements of 4DE and 2DE. Variabilities were described as relative changes in percent between two datasets of different recordings as a coefficient of variation (CV) of differences and were calculated as following for n animals:

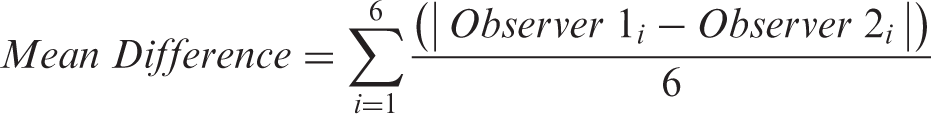
Results
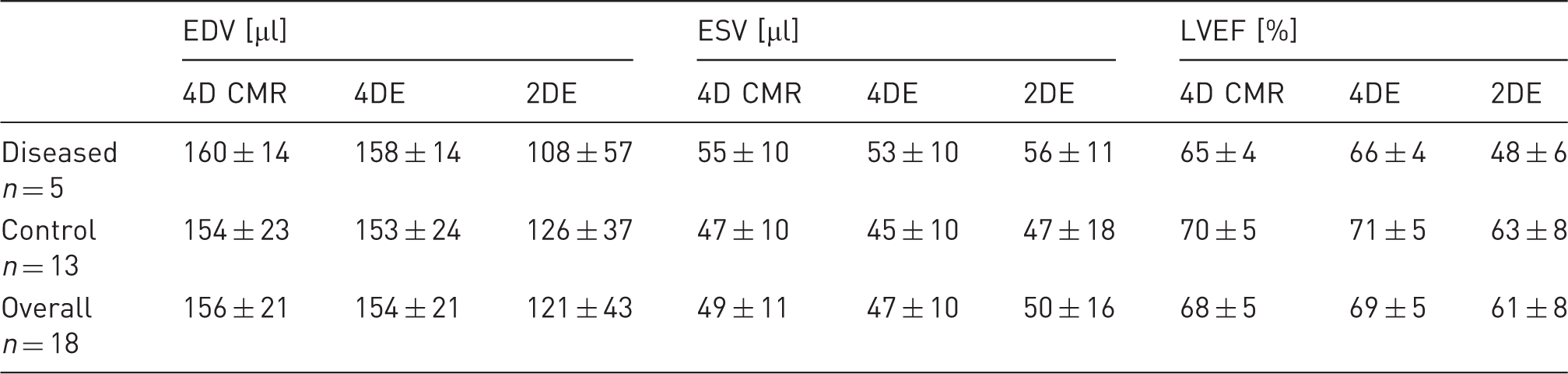
Comparison of 4D CMR, 4DE and 2DE (PSLAX) for EDV, ESV and LVEF in diseased and normal animals, separately and for the whole study sample. Values are expressed as mean ± standard deviation (SD).
Correlation between modalities
Linear regression analysis (Figure 4(a)) and Bland-Altman analysis (Figure 4(b)) showed excellent correlations and strong agreements between 4DE LVEF and 4D CMR LVEF. Furthermore, the correlation coefficient between 4DE LVEF and 4D CMR demonstrated better correlation than that of 2DE LVEF (2DE LVEF v. 4D CMR: r = 0.577, p < 0.01; 4DE LVEF v. 4D CMR: r = 0.952, p < 0.01; Figure 4(b) and (d)). As seen in the Bland-Altman analysis (Figure 4(c)) 4DE and 4D CMR show strong concordance regarding LVEF, whereas 2DE (Figure 4(b) and (d)) has wider distribution and a significant lower LVEF with a mean difference of 8% in comparison with 4D CMR. Overall, 4DE and 4D CMR gave concordant measures of EDV, ESV and LVEF (Table 2).
Correlation between imaging modalities. Dots are used for control animals (n = 13), stars for diseased animals (n = 5). (a), (b) Linear correlation charts with regression lines for each comparison. (c), (d) Corresponding Bland-Altman plots with mean bias and 95% limits of agreement (mean ± 1.96 SD) displayed.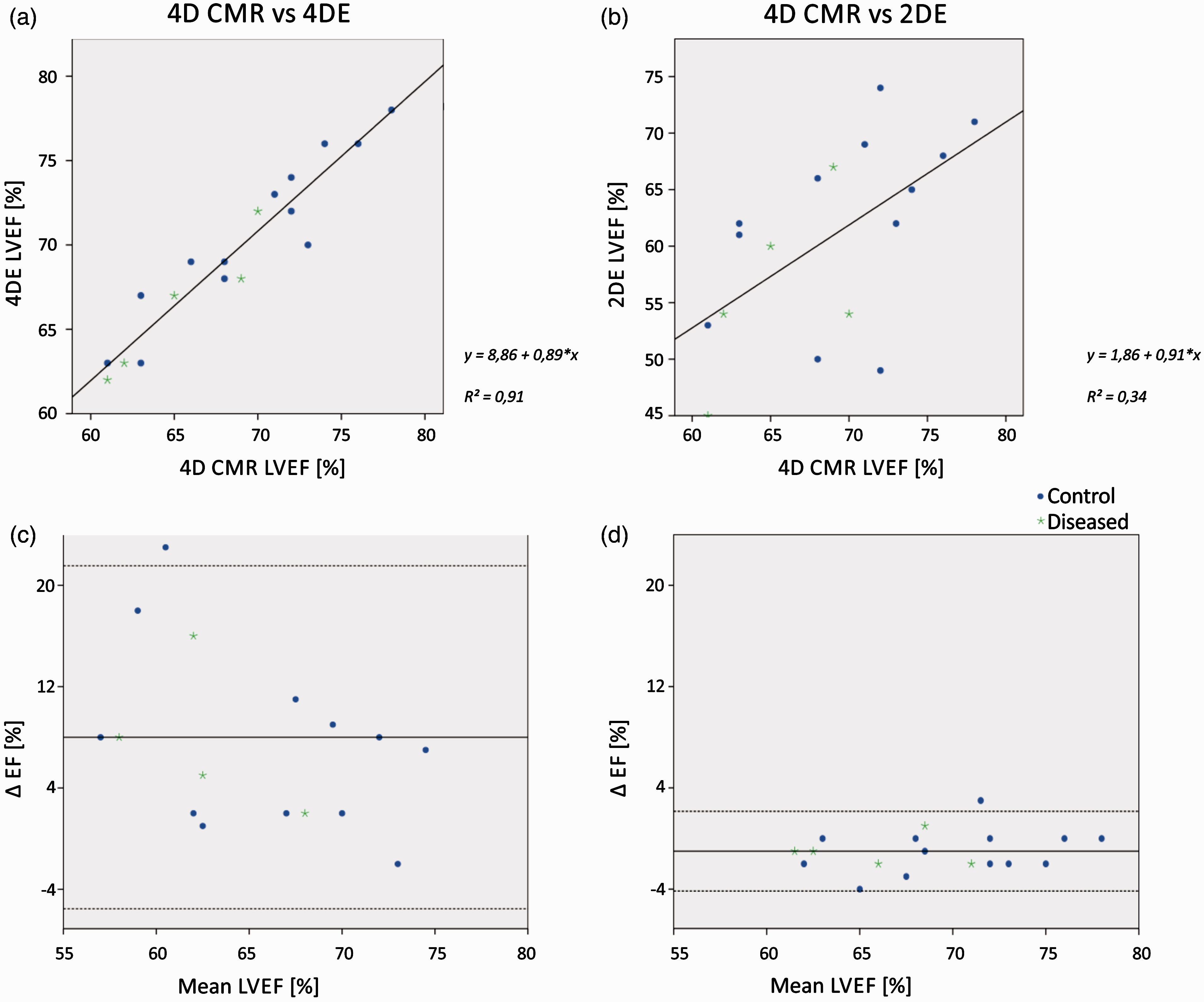
Intra-observer agreement
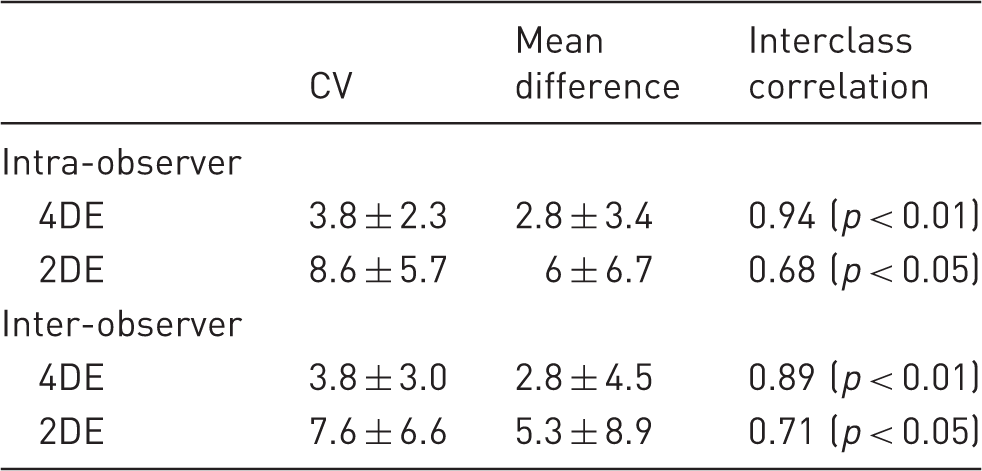
Coefficient of variation (CV), mean difference with a confidence interval of 95%, and interclass correlation are displayed for intra- and inter-observer reproducibility of 4DE and 2DE.
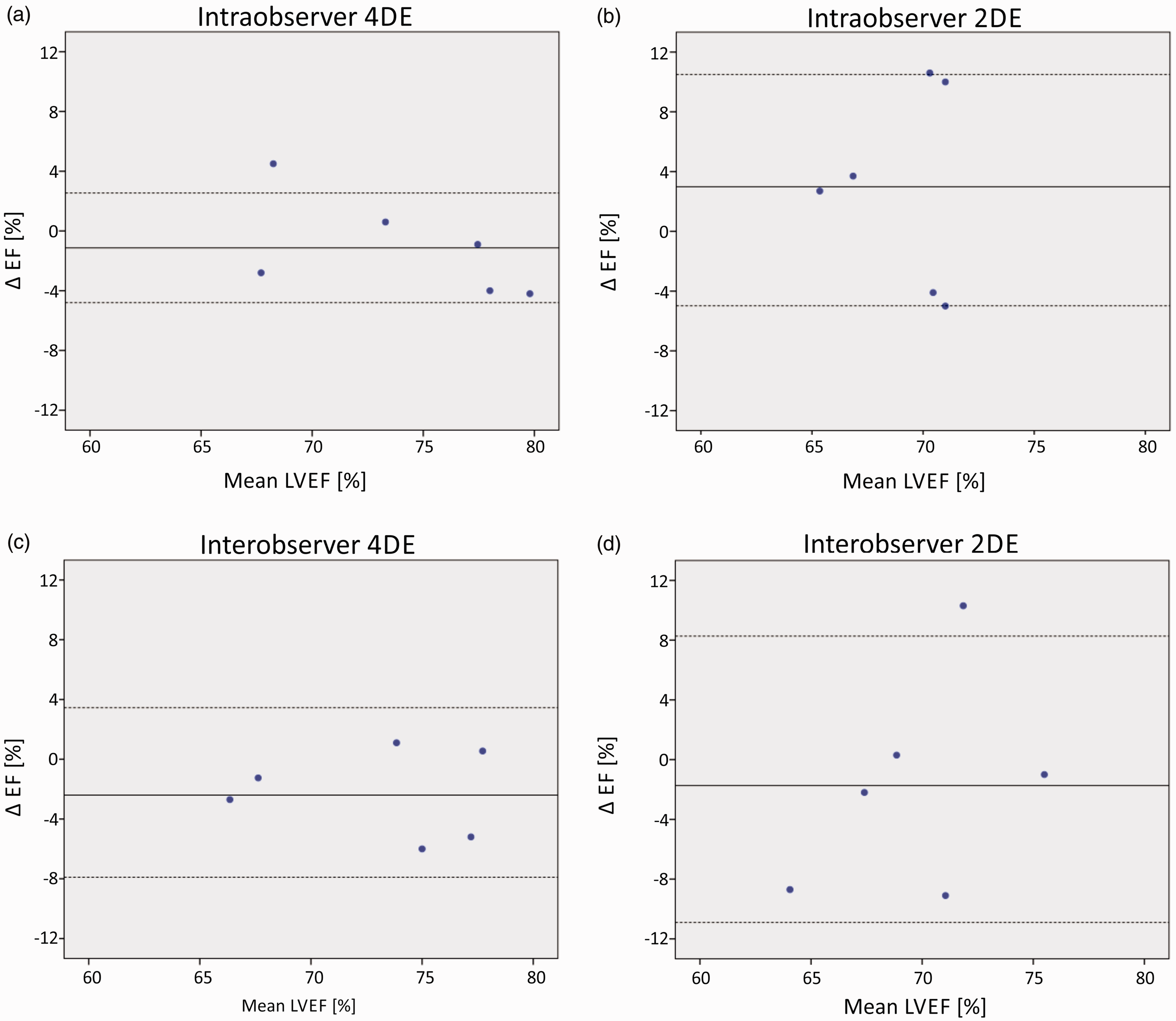
Bland-Altman plots with mean bias, standard deviation and 95% limits of agreement displayed. (a) Intra-observer reproducibility of 4DE; (b) intra-observer reproducibility of 2DE; (c) inter-observer reproducibility of 4DE; (d) inter-observer reproducibility of 2DE.
Inter-observer agreement
Mean differences in EF between two different observers were lower for 4DE (2.8 ± 4.5) than for 2DE (5.3 ± 8.9). Inter-observer agreement was excellent for 4DE (ICC = 0.89, p < 0.01) (Figure 5(c)) whereas 2DE derived EF was only moderate in agreement (ICC = 0.71, p < 0.05) (Figure 5(d)). Bland-Altman analysis revealed a trend for one observer to systematically measure 4DE EF marginally lower (bias 2.5%). For 2DE there was no significant bias, however differences were found between observers at either end of the EF spectrum.
Discussion
The results of our study imply two main messages: (a) 4D CMR and 4DE show excellent correlation in measurements of volumetric heart function, and (b) 4DE is very accurate and highly reproducible. This is the first time that echocardiography has produced results that are comparable with measurements done by 4D CMR.
Previously, HC 2D-echocardiographic models 13 and measurements of LV cross-sectional area derived ejection fraction 7 were compared with CMR-derived full-volume LVEF. Geometric assumptions of HC models led to an overestimation of LV volumes. 13 Another method to calculate LV function is Simpson’s rule. It uses biplane apical discs throughout the heart instead of just one single slice. Simpson’s rule produces accurate results in normal functioning hearts and is very common in clinical echocardiography. 11 However, it is inaccurate when wall motion abnormalities are involved. 19
Van de Weijer 10 described 4D CMR as superior to echocardiography because the volume measurement is based on many acquired slices, therefore crucially minimalizing the geometric assumptions. 4DE is now producing images on the same technological basis as 4D CMR by combining many acquired slices (Figure 1). Evaluating LVEF by 4DE with minimal geometric assumptions leads to results that are consistent with 4D CMR. Another advantage of 4DE is that by displaying the whole heart it becomes very useful for diagnostic imaging. Further studies are needed to confirm that it is able to detect wall motion abnormalities or changes in structures of the myocardium. In addition, we have shown that the intra- and inter-observer reproducibility of 4DE is significantly higher than in 2DE. The impact of the initial position of the probe isn’t as crucial as it is in 2DE, because the whole heart is divided into many slices to calculate heart function. Therefore, the results of longitudinal studies are more reliable when measured by 4DE in comparison with other echocardiographic approaches, even when the scans are carried out by independent observers.
As natural variation in EDV, ESV, and LVEF is fairly large 20 (in this study both 4DE and 4D CMR show that LVEF ranges from 60–80%), this might produce undetectable variation at the group level, when the mean between groups is examined. In these cases, longitudinal studies with 4DE offer the possibility to detect subtle changes in systolic function by individual follow-up. This may allow researchers to use smaller groups in detecting changes in heart function.
A major advantage of 4DE is the high spatiotemporal resolution and the fast scan time compared with 4D CMR (Table 2). A high definition 4DE of the heart takes 8 min, which is less than a quarter of the time needed for 4D CMR. As a result, stress on rodents is reduced since the animals only require approximately 10 min of light anaesthesia. 21 This time can be further reduced when the slice thickness (in our study 10 µm) is increased.
In our experience there were fewer problems with gating for ultrasound than we would expect with 4D CMR, which is more susceptible to disruptions and disturbances concerning cardiac monitoring, because ECG-gating is strongly influenced by the magnetic field. 22 However ultrasound is more vulnerable to gas and bone artefacts, and extra care must be taken when scar tissue is present. The Vevo 3100 imaging platform provides probes for rats of all ages, so we don’t expect other results when 4DE is performed in older animals. Further studies have to confirm this assumption.
Conclusion
To the best of the authors’ knowledge, this is the first study to compare 4DE with 4D CMR in rats. Excellent correlation of LV size and heart function was seen between the newly available method of 4DE and 4D CMR in healthy and diseased animals, showing that 4DE is an improvement compared with older echocardiographic approaches. Availability, expertise and the aim of studies will determine which imaging modality will be used. Our results suggest that researchers can now confidently rely on 4DE as the method of choice for cardiac imaging.
Limitations
This study was done in young Sprague-Dawley rats; further studies for adult rats are needed. Reproducibility analysis was only done for echocardiography not for 4D CMR. Therefore we could only show that measurements by 4DE are highly reproducible, but were not able to compare reproducibility of 4DE and 4D CMR. Simpson analysis was not included in the study.
Footnotes
Acknowledgements
We thank Dipl.-Phys. Jutta Jordan, Dr. rer. nat. Clarissa Gillmann and Dr. rer. nat. Marc Schwarz from the Institute of Radiology of the UK Erlangen for their technical support. The present work was performed in fulfilment of the requirements for obtaining the degree Dr. med for Hedwig Stegmann.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: this research was supported by the Klaus Tschira Stiftung gGmbH.
