Abstract
Young female Wistar rats from a specific pathogen free breeding colony presented an outbreak of infertility along with neurological symptoms and malignant lymphomas. We evaluated the presence and the potential role of the rat leukemia virus (RaLV) in the disease because these clinical signs could be compatible with a retrovirus. RaLV is a mammalian type C endogenous retrovirus initially isolated from in vitro Sprague–Dawley rat embryo cultures. There are no reports of clinical disease in rats associated with this virus, and little is known about its interaction with the host. Using reverse transcription polymerase chain reaction and enzyme-linked immunosorbent assay, we studied the synthesis of the viral particles and the development of an immune response against the virus in this rat colony. The results showed that healthy and diseased Wistar rats synthetized viral RNA but only diseased animals developed a detectable immune response against RaLV envelop protein. Furthermore, rats with lymphomas tended to have higher titers of antibodies against RaLV epitopes than those with infertility or neurological symptoms. The results suggest that increases in the RaLV infectious particle loads could be involved in the development of lymphomas in young rats. The potential causes of RaLV reactivation are discussed.
Introduction
Rat leukemia virus (RaLV) is a mammalian type C endogenous retrovirus (ERV) present exclusively in the rat genome. 1 Germ-line integrated ERV sequences usually degenerate via the accumulation of mutations or recombination events that impair the ability to produce infectious viruses. 2 However, RaLV can spontaneously release competent viral particles in cultured Sprague–Dawley embryo cells and exogenously infect new embryo rat cells. 3 This ability to produce competent endogenous copies was also reported in many strains of laboratory mice infected with murine leukemia virus and mouse mammary tumor virus. 4 Their similarity in their genomic structure recognizes ERVs and typically consists of three long open reading frames (pol, gag, and env genes) flanked by two long terminal repeats. 5 Expression of ERVs can be induced in various conditions, including immunosuppression, infections with other viruses, exposure to drugs, or recombination between endogenous retrovirus-producing viral strains with novel biological or pathological properties. 6
In vitro studies in cancer cells showed that RaLV could cause an activated c-Ras to form an acutely transforming virus named Rasheed rat sarcoma virus. 1 Other acutely transforming viruses have been reported in rats, including Kirsten sarcoma virus and Harvey sarcoma virus. These are mouse–rat recombinant viruses with viral genomes. 7 In most cases, these transforming viruses are recombinant between portions of the genomes of helper independent type C viruses and other genetic information derived from the host species from which the transforming virus was originally isolated. 7
Retroviral infections have been associated with leukemias, lymphomas, anemia, immunodeficiency, and neurological disorders. 5 Retroviral infections increase the incidence of lymphomas in different species. Indeed, 1–21% of cats positive for feline immunodeficiency virus develop neoplasia; 8 0.1–10% of cattle infected with bovine leukemia virus have virus-induced lymphomas. 9 People with HIV/AIDS have a higher risk of developing non-Hodgkin’s lymphoma than the normal population. 10 A variety of human and animal retroviruses can also cause central nervous system neurodegeneration as manifested by motor and cognitive deficits. 11 The pathogenesis of retroviral neurodegeneration is caused by an indirectly mediated mechanism of injured neuronal tissue mainly associated with microglial retroviral infections. 12
Details about the biology of the RaLV and its interaction with the host are very limited. There are no reports of clinical disease in rats associated with RaLV. This virus was initially isolated in Sprague–Dawley rats, but there are no reports of this virus naturally infecting laboratory rat strains. Even though RaLV is considered a replication-competent retrovirus, it is unknown whether viral proteins are continuously synthesized or ensembled or whether the rats develop an adaptative response against the virus.
In 2019, a Wistar rat colony of the School of Medicine, Universidad de la República presented an outbreak of infertility, neurological signs, and lymphomas. A group of 60 females out of a total of approximately 80 showed an abrupt reduction in fertility rates. The conception rates were reduced, but the mean number of neonates at delivery and weaning dropped by nearly 50%. The maternal behavior was also altered: one-third of the mothers did not build nests nor did they nurse their litters adequately. Eight adult female rats between six and eight weeks old showed neurological disorders, including abnormal gait, tremors, and seizures. Concurrently, another six female Wistar rats (between four and eight months old) presented lymphomas in the thymus, spleen, and mesenteric lymph nodes. Neurological disorders and lymphomas appeared a few weeks after the decline in reproduction rates.
Thus, based on the epidemiology, the neurologic features, and the development of lymphomas in affected rats, we studied whether RaLV could be directly or indirectly involved in the development of the pathologies described in that rat colony.
Animals, materials, and methods
Animals
Outbred Wistar rats (Crl:WI) acquired in 2019 from Charles River Laboratories (Wilmington, MA, USA) were bred at Unidad de Reactivos y Biomodelos de Experimentación, Facultad de Medicina, Universidad de la República. They were reared in a barrier vivarium maintained at 24 ± 2°C with a relative humidity of 60% ± 20% with 12-h light/dark cycles; the room was ventilated at least 12 times/h with HEPA-filtered air.
Rats were housed in ventilated racks (Lab Products, USA) at two to four animals per cage, depending on their age and sex. The cages were changed at least once a week. The rats were given an autoclaved pellet diet (5K67, PMI Nutrition International, USA) and autoclaved water ad libitum. All procedures for animal handling and experimental procedures were approved by the Institutional Animal Care and Use Committee (CEUA, Facultad de Medicina, Universidad de la República, protocol number 070153-000881-20).
Female Wistar rats of four to eight months old showing severe clinical signs (i.e. neurological signs, altered maternal behavior, weight loss, or dyspnea) were separated from the breeding colony by the veterinary staff. These rats were anesthetized with xylazine ketamine solution in order to obtain blood samples, and later euthanized with CO2 and necropsied. Clinically healthy rats were randomly selected for blood collection to be used as hematological and serological controls. Finally, according to the clinical manifestations and the pathological findings, they were grouped into the following groups: a) asymptomatic group (n = 9): female rats without clinical signs and average reproductive performance; b) ataxia–infertility group (n = 8): females rats with tremors, ataxia, and rats with reduced conception rates or altered maternal behavior; c) lymphoma group (n = 5): female rats clinically observed with severe respiratory distress and weight loss who later had enlargement of thymus and spleen at the necropsy, with histopathological diagnosis of lymphoma.
All rats involved in the present study were identified with a unique number using an ear punch code. Each animal was considered as an experimental unit.
Hematological, serological, and histopathological analysis
Blood samples from healthy and diseased rats were taken for hematological and serological studies. Rats were anesthetized with an intraperitoneal injection of xylazine ketamine solution (10 mg/kg and 75 mg/kg, respectively) and bled from the retroorbital plexus. Whole blood and serum samples were placed in EDTA (K3 plus ethylenediamine tetraacetic acid) and dry tubes, respectively. Hematological analysis was performed by Laboratorio de Análisis Clínicos, Veterinary Faculty, Universidad de la República. Cell blood analysis was performed with a veterinary hematology analyzer (Mythic 18 Vet, Orphée, Geneva, Switzerland) and by microscopic evaluation of air-dried smears stained with May Grunwald-Giemsa. Serum samples were stored at –20°C for enzyme-linked immunosorbent assay (ELISA) determination of RaLV antibody titers. Rats were euthanized by CO2 inhalation, and a full pathologic examination was performed. All organs were collected, weighed, and fixed in 10% neutral buffered formalin solution. The fixed organs were trimmed and embedded in paraffin. Sections (4 μm thick) of tissue were stained with hematoxylin–eosin for histopathological examination.
Preparation of total RNA and identification of RaLV in cDNA samples
Total RNA from the thymus and the uterus were obtained from clinically affected and healthy rats using RNeasy mini total RNA isolation kit (Qiagen, USA) according to the manufacturer’s protocol. This was later converted into cDNA using the Superscript III Reverse Transcriptase (Invitrogen). Primers were designed from the RaLV cDNA sequence (GenBank access code: M77194.1) using Primer-BLAST bioinformatics tool. These covered regions of the three genes present in the virus: gag (group-specific antigen), pol (polymerase), and [env] (envelope). Four pairs of primers were designed (two for gag, one for pol, and one for env) and are shown in Figure 1.

Primers used for the detection of RaLV gag, env, and pol genes. The sequences were designed using the RaLV genome published by Lee et al.1 in 1998.
The polymerase chain reaction (PCR) conditions were adjusted to a final volume of 25 µl (buffer 2.5 µl; MgCl2 1.5 mM; dNTP 0.4 mM; primers 0.4 µM; Taq Platinum Invitrogen 0.2 µl; cDNA 2 µl). The cycles were 94°C for 1 min; denaturation 94°C for 30 s; alignment 60.5°C for 30 s; and extension 72°C for 1. This was cycled 35 times, and the final extension was 72°C for 7 min. The fragments were then visualized on a 2% agarose gel with ethidium bromide solution (BIORAD, USA). The PCR products were sent to Macrogen (Korea) for sequencing.
Genomic DNA extraction and identification of RaLV endogenous sequences
Genomic DNA was extracted using standard protocols from tail tip biopsies of rats belonging to different animal facilities in Uruguay, Argentina, and USA. The samples included Wistar, Wistar Kyoto, and Sprague–Dawley outbred rats as well as Fischer 344 (F344) and Brown Norway (BN) inbred strains.
Antibody determination anti RaLV by ELISA
A portion of the sequence of RaLV envelope (EnvRaLV) protein was synthesized (China Peptides, China) and used as an antigen for anti RaLV antibody determination (Figure 2). ELISA plates (Greiner Bio-one, Germany) were coated with 10 µg/ml of EnvRaLV protein in PBS (0.01 M, pH 7.4) (100 µl per well); the plates were placed at 4°C overnight. The plates were then washed to remove unbound EnvRaLV protein, and PBS plus 2% soy protein (PBS-S) was added for 45 min at 37°C. Rat sera samples from affected animals and controls (1/25 to 1/200 dilutions in PBS containing 0.05% Tween-20 (PBS-T)) were incubated in the plates for 45 min at 37°C. A pool of rat sera with high titers was used as positive control; the sera samples from non-affected rats were used as negative control. The wells were then washed three times with PBS-T (0.05% Tween-20). Rat immunoglobulin bound recombinant EnvRaLV protein was detected by adding HRP-conjugated goat anti-rat IgG (Sigma) diluted 1/10,000 in PBS-S-T. We then incubated the plates for 45 min at 37°C. After washing the wells with PBS-T, the tetramethylbenzidine substrate (100 µL/well; Sigma) was used for colorimetric reactions. It was stopped with 50 µl of H2SO4 (0.5 M). The optical density was measured at 450 nm on an automatic ELISA plate reader (Multiskan EX, Thermo, China). The sample antibody titers were expressed as arbitrary units (UA/ml) determined relative to a known standard curve.

Envelope protein sequence used for rat antibody determination against RaLV by ELISA. This protein has a high identity with RaLV and M-MuLV envelope proteins.
Statistical analysis
We performed analyses on numeric variables (red and white blood cell counts and antibody titers) to verify the residual distribution’s normality. In addition, means of red and white blood cell counts of each group and antibody titers against RaLV were compared using two-way analysis of variance. All the analyses were performed using GraphPad Prism version 8.2.1 for Windows (GraphPad Software, La Jolla, CA, USA) and differences were considered significant when p < 0.05.
Results
Hematological analysis
Rat samples were grouped according to the clinical and pathological manifestations into three groups: asymptomatic (n = 9), ataxia–infertility (n = 8), or lymphoma (n = 5). No alterations in the red blood cell counts or hemoglobin concentrations were seen between the three groups (data not shown). However, the white blood cell counts (WBC) in ataxia–infertility (4.1 ± 0.85 × 103 cells/µl) and lymphoma groups (33.5 ± 30.1 × 103 cells/µl) differed from asymptomatic group (6.26 ± 0.99 × 103 cells/µl) (p < 0.005) (Figure 3(a)). Also, the ataxia–infertility group differed from the lymphoma group for WBC (p = 0.01). Similar results in the lymphocyte counts were found. The mean value of lymphocytes in the asymptomatic group was 5.2 ± 2 × 103 cells/µl while ataxia–infertility and lymphoma groups showed 2.98 ± 0.67 × 103 cells/µl and 17.81 ± 23 × 103 cells/µl, respectively (p < 0.0003) (Figure 3(b)). All infertile rats and/or those with neurological signs had leukopenia, lymphopenia, and mild thrombocytopenia. In contrast, 80% of the rats with lymphomas showed marked leukocytosis, lymphocytosis (Figure 3(a) and (b)), and thrombocytopenia (data not shown). More than 50% of the lymphomas were leukemic because the blood smears showed a significant amount of lymphoblasts (Figure 3(f)).
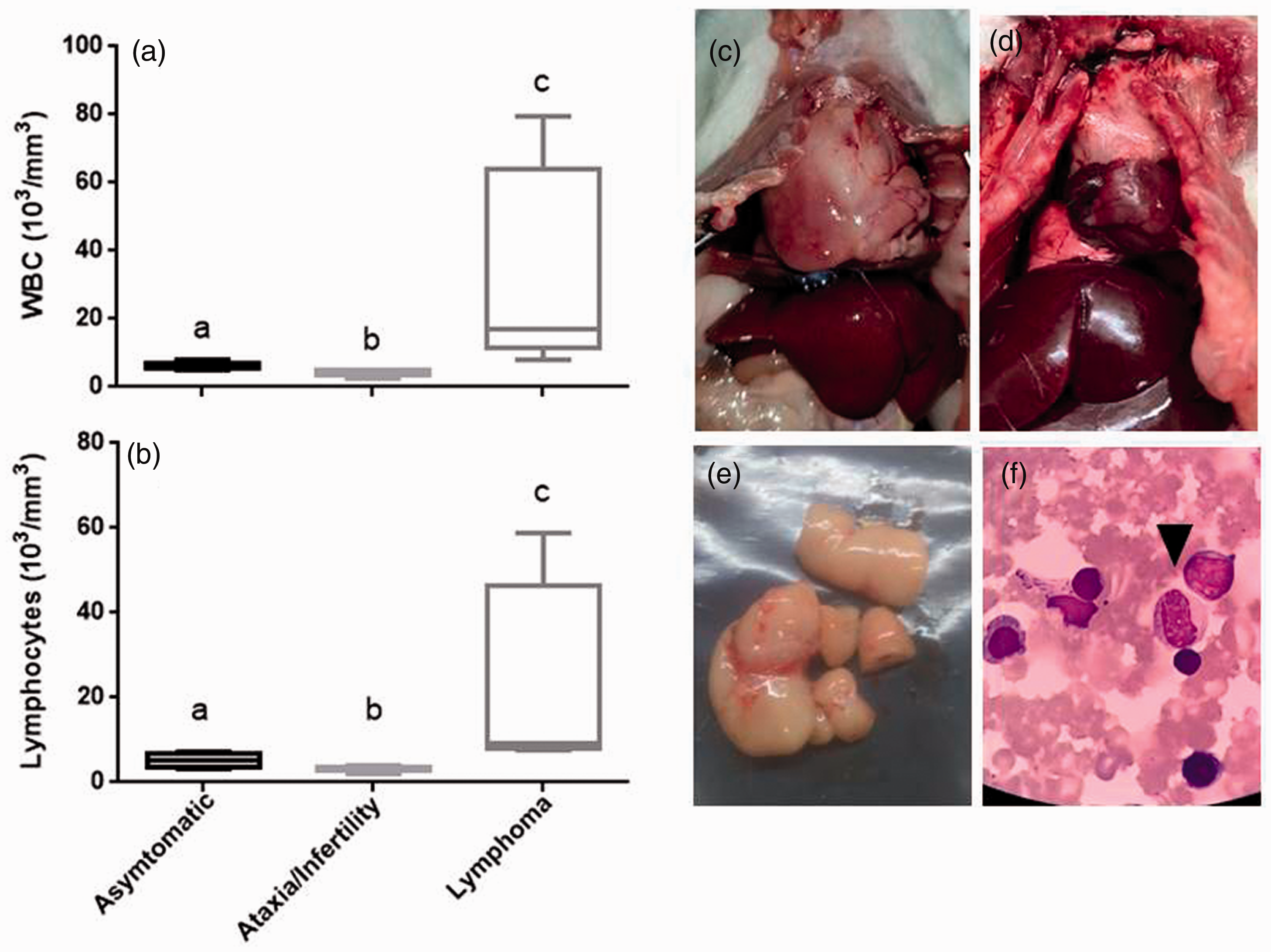
White cell count and pathological changes in diseased Wistar rats. Animals were grouped according to the severity of the clinical signs as well as histopathological findings: asymptomatic (n = 9), ataxia–infertility (n = 8), and lymphoma (n = 5). (a) White blood cell counts. (b) Lymphocyte counts. (c) Thymus lymphoma filling the thoracic cavity of the rat. (d) Normal thymus from asymptomatic group. (e) Mesenteric lymphoma. (f) Blood smear of a leukemic rat stained with May Grundwal-Giemsa. Arrow, lymphoblast cells. Different letters show significant differences between groups (p < 0.05).
Necropsy and histopathological studies
Gross examination of the organs from asymptomatic and ataxia–infertility rats did not show significative macroscopic alterations (Figure 3(d)). At the histopathological examination performed by a veterinary pathology (American College of Veterinary Pathologists) a tumor of the thymus was seen in all of the rats from the lymphoma group (Figure 3(c)), accompanied in most of the cases by an enlargement of spleen and mesenteric lymph nodes (Figure 3(e)). The histopathological analysis of rats with tumors revealed that the affected thymus and lymph nodes had a damaged architecture. The parenchyma was impacted by proliferating cells with prominent nuclei and nucleoli and minimal cytoplasm mixed with plasma cells. The regular architecture was lost in some areas of the spleen, and the liver portal and centrilobular areas had a focal collection of cells similar to that observed on the thymus and lymph node. The final histopathological diagnoses were malignant lymphomas.
Identification of RaLV sequences in total mRNA from thymus and uterus
All Wistar rats produce continuously competent viral particles. Sequences of RaLV genes were amplified by PCR using cDNA (thymus and uterus) from healthy and affected Wistar rats. Figure 4 shows the amplification of 253 bp and 73 bp for the different regions of the gag gene, 204 bp for the pol gene, and 169 bp for the env gene. The bioinformatic analysis confirmed that the amplified fragments belong to RaVL. A preliminary study using reverse transcription PCR showed a higher number of copies of RaLV in the thymus than the uterus, perhaps because of the different number of lymphoid cells in these tissues (data not shown).
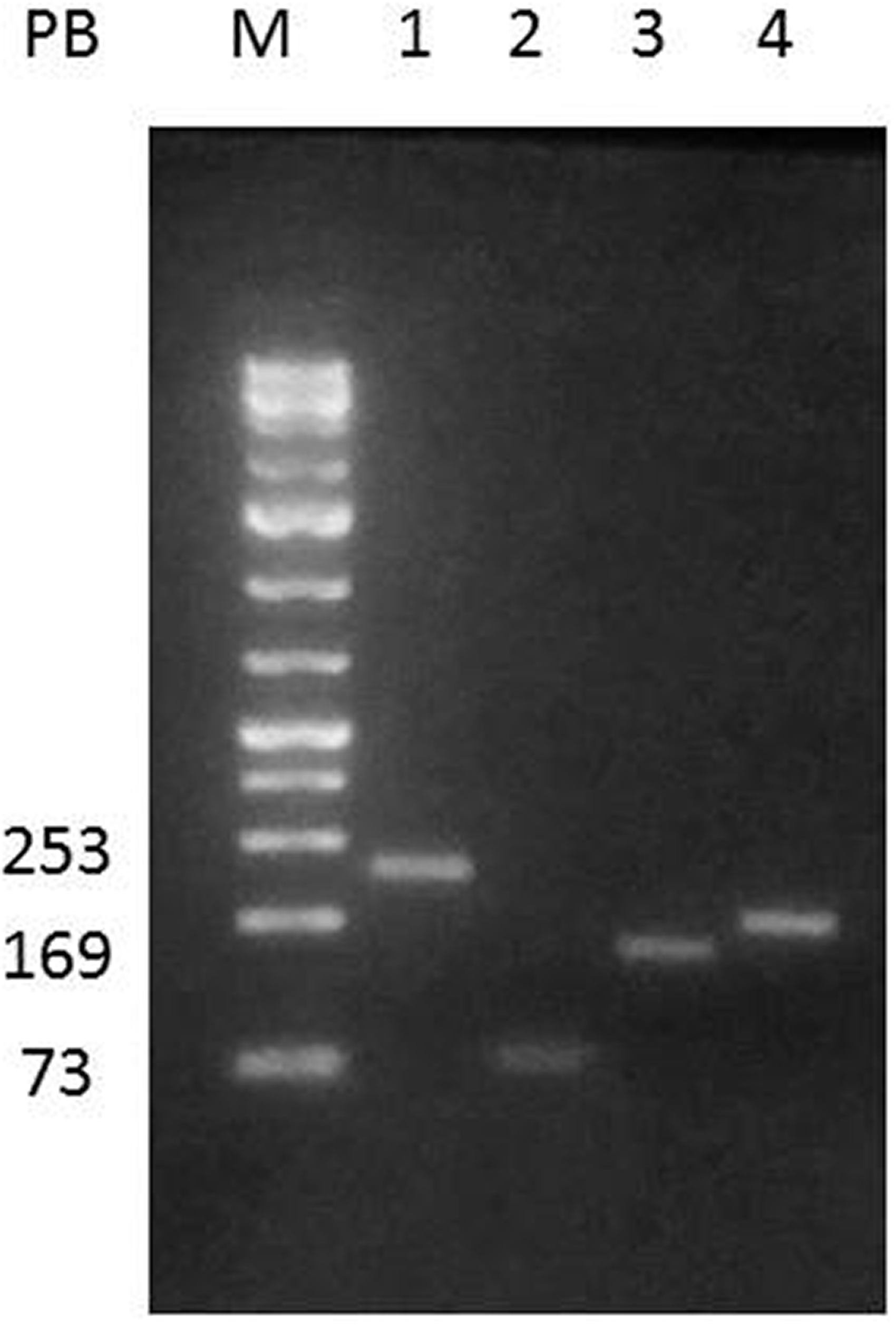
An example of polymerase chain reaction identification of rat leukemia virus from cDNA obtained from Wistar rats.
Confirmation of the presence of RaLV endogenous sequences in genomic DNA from different rat strains
The PCR analysis demonstrated that all rat strains carry RaLV proviral sequences on their genomes as expected for ERV. The five strains belong to seven animal facilities distributed in different regions (Figure 5).
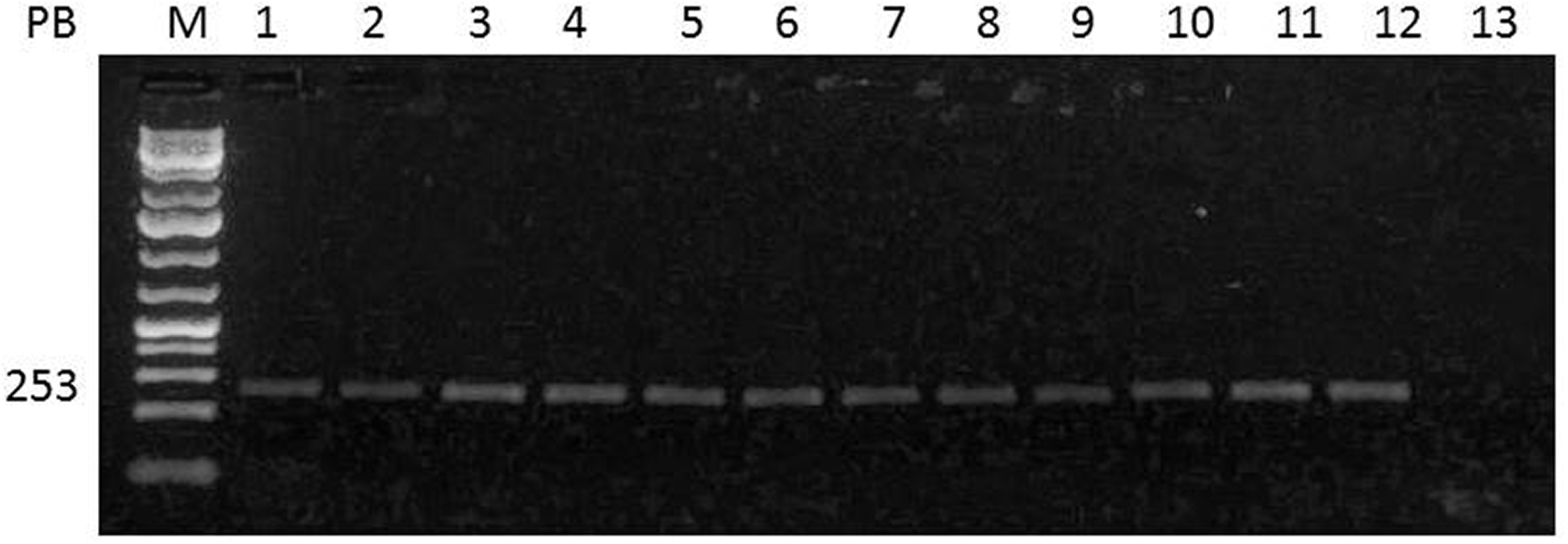
Identification of rat leukemia virus provirus (Gag gene) in stocks and strains of rats belonging to different animal facilities.
Antibody determination anti RaLV by ELISA
The RaLV ELISA developed in our laboratory was used to measure antibody titers against this retrovirus. The EnvRaLV protein was produced from a cDNA sequence of enveloping genes of RaLV described by Lee et al.1 The ELISA assay conditions were optimized for measuring rat antibodies. This assay allowed us to determine for the first time the presence of rat antibodies against RaLV.
All diseased rats had antibodies against RaLV; the sera of clinically healthy rats without macroscopic alterations on necropsy were negative. Interestingly, when the rat samples were grouped according to the clinical signs and manifestations (asymptomatic, ataxia–infertility, or lymphoma), the RaLV antibody titers tended to be higher in the lymphoma group than in the ataxia–infertility group (p = 0.07) (Figure 6).
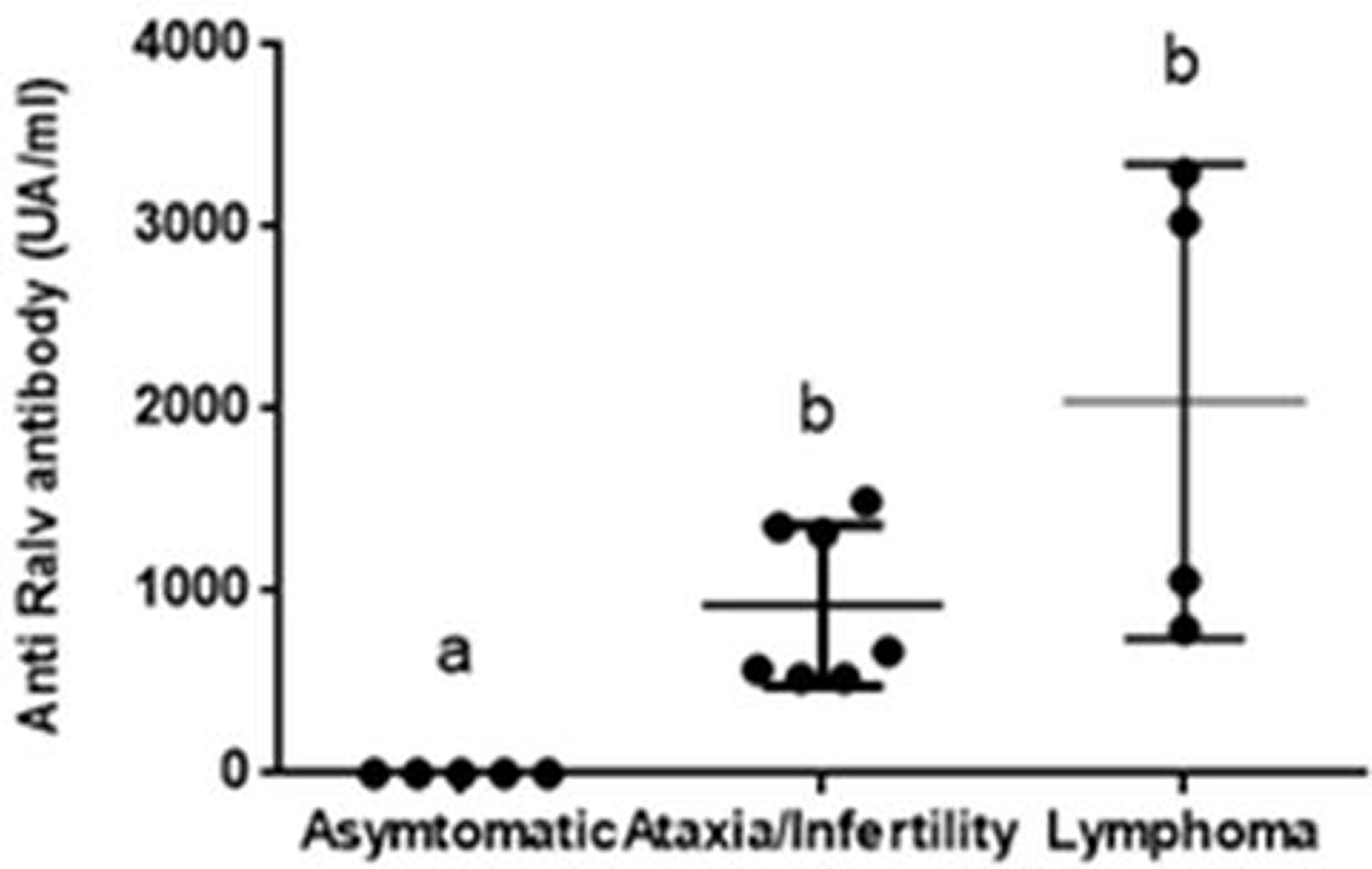
Determination of antibodies against RaLV by enzyme-linked immunosorbent assay. Animals were grouped according to the severity of the clinical signs including asymptomatic (n = 5), ataxia–infertility (n = 7), and lymphoma (n = 4). Different letters show significant differences between groups (p<0.05).
Discussion
Retroviral diseases are usually associated with immunosuppression, tumors, neurological disorders, and infertility. In the face of the outbreak with these main clinical signs, we studied the potential role of RaLV in the pathogenesis of this rat disease. RaLV is an ERV of the rat that produces competent virions and exogenously infects new cells. 1 RaLV can also undergo recombinations and form an acutely transforming virus. 1 ERVs are generally, but not exclusively, replication-defective but can affect multiple aspects of host physiology and pathology, including cancer initiation and progression. 13 Viral loads of ERVs in koalas were suggested to be associated with secondary bacterial infections and neoplasias. 14 Therefore, RaLV could directly or indirectly be responsible for clinical disease.
Proviral sequences (DNA) were found in all analyzed rat strains and stocks. Brown Norway, Fisher, Wistar, and SD strains carry viral sequences in their genomes (Figure 5). PCR analysis detected gag, pol, and env genes. To the best of our knowledge, this has not been reported previously although most laboratory strains have a similar origin. Further studies are needed to confirm whether all lab rats or other species belonging to the Rattus genus carry this retrovirus. Viral loads (RNA) were found in healthy and affected Wistar rats (Figure 4). Viral particles were exclusively reported in Sprague–Dawley rats, 1 but different strains could be producing circulating virions probably with different intensities.
Wistar rats with infertility and/or neurological disorders showed severe leukopenia and lymphopenia; rats carrying lymphoma and/or leukemia showed marked leukocytosis (Figure 3). Immunosuppression is a well-characterized property of retroviruses, and it has been reported in cats and mice with feline leukemia and murine leukemia virus, respectively. 15 The envelope glycoprotein of diverse endogenous and exogenous retroviruses is inherently immunosuppressive, 16 and its effects depend on viral loads. 15 In addition, many lymphomas were seen in young (three to six months old) Wistar rats over a period of six months; at least 5–10% of the diseased rats developed lymphomas. Lymphoma is usually a rare disease in rats that appears isolated and is generally associated with older animals. 17 However, up to 21% of cats that are positive for feline immunodeficiency virus develop neoplasia, 8 and 0.1–10% of cattle infected with bovine leukemia virus have lymphomas. 9
Ataxia, tremors, and weakness of the hind limbs were observed in 10% of the diseased rats. Humans with HIV can develop neurocognitive disorders associated with cognitive deficits in attention, memory, and particularly cerebellum-associated motor symptoms, including gait disturbances, limb weaknesses, and tremor. 18 Cats or mice infected with feline leukopenia virus or murine leukemia virus also develop neurologic disorders.12,19 Finally, the fertility and the weaning were abruptly reduced in this Wistar rat colony; up to 80% of females of the colony had reproductive symptoms. Infertility, aberrant pregnancies, and reproduction failures were reported during feline leukopenia viral infections. 20
Previous reports have suggested that the severity of clinical signs of retroviral diseases could be associated with viral loads.14,15 In this sense, we studied whether diseased and healthy rats developed antibodies against RaLV. There are no previous reports on RaLV antibodies, and thus we developed an ELISA using a RaLV envelope as the antigen. The results showed that clinically affected rats developed antibody titers against RaLV, which were higher than healthy rats (Figure 6). To the best of our knowledge, this is the first report of a rat antibody response against RaLV. Higher antibody titers could correspond to higher RaLV antigen stimuli, suggesting the increase of viral activity in diseased animals.
Multifactorial causes could trigger increases in RaLV activity and clinical signs in the Wistar rats. The environmental conditions did not change dramatically, but a different stressor might have triggered the problem. A spontaneous mutation unique to this rat colony might have left them more susceptible to RaLV or viral recombination. Retroviruses with restored infectivity through recombination have been discovered in rats and mice.7,13 Also, the resurrection of ERVs was reported in transgenic mice carrying immunodeficiencies. 21 The genetic background of Koalas in Australia has been proposed as a predisposing factor for increases in viral loads of KoRV (an ERV) and the resulting development of neoplasia or secondary infections. 14 Further studies are needed to determine the environmental or genetic causes of this disease.
Footnotes
Acknowledgements
We thank Dr. George Kassiotis for advice about the manuscript and Dr. Ortega for sending us rat tissue samples.
Data availability statement
The data that support the findings of this study are available on request from the corresponding author.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
