Abstract
For studies requiring accurate conception-timing, reliable, efficient methods of detecting oestrus reduce time and costs, whilst improving welfare. Standard methods use vaginal cytology to stage cycle, and breeders are paired-up using approximately five proven females with proven males to achieve at least one conception on a specific day. We describe an alternative, fast, consistent, non-invasive method of timed-mating using detection of lordosis behaviour in Wistar and Lister-Hooded rats that used unproven females with high success rates. Rats under reverse lighting had body masses recorded pre-mating, day (d) 3–4, d8, d10 and d18 of pregnancy. Using only the presence of the oestrus dance to time-mate females for 24 hours, 89% of Wistar and 88% of Lister-Hooded rats successfully conceived. We did not observe behavioural oestrus in Sprague-Dawleys without males being present. Significant body mass increases following mating distinguished pregnant from non-pregnant rats, as early as d4 of pregnancy (10% ± 1.0 increase cf. 3% ± 1.2). The pattern of increases throughout gestation was similar for all pregnant rats until late pregnancy, when there were smaller increases for primi- and multiparous rats (32% ± 2.5; 25% ± 2.4), whereas nulliparous rats had highest gains (38% ± 1.5). This method demonstrated a distinct refinement of the previous timed-mating common practice used, as disturbance of females was minimised. Only the number required of nulli-, primi- or multiparous rats were mated, and body mass increases validated pregnancy status. This new breeding management method is now established practice for two strains of rat and has resulted in a reduction in animal use.
Introduction
Rats are model organisms with a short oestrous cycle that are used in a multitude of research areas. For some studies, accurate timing of pregnancy or stage of offspring development is critical, and it is common practice for multiple dams to be time-mated for each study dam required, which allows for the probability of most dams not conceiving during the required time window. Current practices of non-invasive time-mating involves pairing approximately five proven females with proven males to achieve at least one conception per group of five females mated. This is both time-consuming and an inefficient use of animals, either generating excess litters or requiring monitoring of the additional females for possible pregnancy. Our overall study aim was to improve on current methods and replace them with a simpler method of selecting single females in oestrus for time-mating and then accurately predicting their pregnancy status, as early as possible.
The rat oestrous cycle is 4 to 5 days long and consists of clearly identifiable stages of proestrus, oestrus, metoestrus and dioestrus.1,2 The cycle is divided into several phases of different length. Proestrus lasts approximately 12 hours and corresponds to the onset of mating behaviour, known as behavioural oestrus. The female ovulates at the end of proestrus and enters the oestrus phase, which lasts approximately 26 hours. Metoestrus follows oestrus for approximately 6–8 hours, and then dioestrus ensues, a period of 50 + hours that is subdivided into dioestrus I (first 24 + hours) and dioestrus II (second 24 + hours). After dioestrus, the rat returns to proestrus and starts a new cycle, if she has not conceived. Rats do not form a fully functioning corpus luteum unless they are coitally stimulated, so infertile mating can lead to a state of pseudopregnancy that can persist for approximately 10 days before the cycle resumes. The gold standard of cycle stage determination in rodents is vaginal cytology, with preference towards vaginal lavage rather than a cotton swab due to collection of an increased number of cells with undisturbed morphology. 3 However, it is possible to accidently induce pseudopregnancy when conducting vaginal cytology, particularly amongst untrained users.
Female rats display behavioural signs of oestrus. When receptive to males during the peri-ovulatory phase, females have increased running activity, hopping, darting, freezing and ear quivering. 1 This leads to male mounting, which in turns solicits lordosis from the female due to pelvic stimulation. Lordosis is a distinctive mating behaviour of the female rat that involves dorsiflexion of the vertebral column. 4 Our main aim was to determine if we could make use of this observed behaviour prior to oestrus to predict when females were likely to be receptive to the male to increase the individual chances of conception when pairing them for timed-mating. We also kept the animals under reverse lighting as this is more appropriate for studying natural behaviour in nocturnal animals, 5 so observations were made during their active phase.
Pregnancy confirmation in laboratory rats in modern research facilities can involve a range of different methods, some of which may be invasive, such as taking blood samples to check for raised progesterone concentrations. Other methods involve animal separation and handling, such as ultrasound scanning for conceptus visualisation, or checking for physical changes including enlarged abdomen and eventually noticeable body mass increases, detectable by weighing. Another study aim was to confidently predict pregnancy as soon as possible following conception.
Pregnancy is a time of extensive maternal adaptation to facilitate foetal growth and development and also to build energy reserves in preparation for the demands of lactation. In rats, maternal body mass increases throughout pregnancy, starting from as early as day 3 post-conception, 6 partly resulting from increased accumulation of adipose tissue,7,8 ready to support lactation. Body mass increases in pregnant dams follows increased food intake. 9 Hyperphagia during pregnancy in rodents has been reported to increase food intake by 20% during the first week of pregnancy,10,11 peaking at between 50% 10 and 60% 6 by the end of pregnancy compared to nulliparous controls. Maternal leptin resistance also occurs during pregnancy; leptin increases in proportion to the adipose reserves accumulated and develops from mid-gestation to support hyperphagia and allow further adipose accumulation.
Previous work with Wistars has established body mass increases from day 14 (d14) to day 19 (d19) of pregnancy as a reliable method for recognising pregnancy and determining the exact day of gestation, 12 but this is obviously a retrospective and later stage method. A further aim of this study was to see if we could track body mass increases from mating to conception and during pregnancy, to establish the earliest time point following conception, to confidently predict pregnancy status, rather than having to wait until pregnancy was physically very obvious. Additionally, we aimed to establish if these methods could be used with unproven females who had not previously conceived.
Animals, materials and methods
Ethical approval
This work took place in the Biomedical Research Unit and was licensed under the Home Office Animals (Scientific Procedures) Act 1986 and had approval from The Open University Animal Welfare and Ethical Review Body (AWERB).
Animals and housing
Female rats were housed in groups of four in wiretop ventilated cages (NKP, UK) within larger cabinets (Scantainer, Scanbur, Denmark) and maintained under a 12-hour reverse light cycle (lights off between 11:00 and 23:00 in Wistar room and 8:00 and 20:00 in Lister-Hooded room, for practical reasons) with free access to standard RM3 breeding diet (SDS Diets, http://www.sdsdiets.com/pdfs/RM3-E-FG.pdf) and water. Animals were given time to adjust to the reverse light cycle prior to the study, if needed. Intact stud male rats for breeding were housed singly. All cages of reproductively cycling females of the same strain were kept in the same Scantainer along with one cage of male rats to keep females cycling normally.
Female nulliparous Wistars for ovarian cycle stage determination were obtained from Harlan, UK (n = 53) and an in-house colony was subsequently bred from more purchased animals. Wistar rats that were also used in another pregnancy and lactation study as well as this one were bred in-house (two to three females per cage, n = 54; n = 3 males). To obtain initial litters from the colony, females were introduced to a male (one to two females per male) in his home cage for a period of 5 days and litters (un-manipulated) were kept to maturity for future pregnancy and lactation time points. Nulli, primi and multiparous Lister-Hooded (breeding stock derived from Harlan, UK, n = 91) and Sprague-Dawley rats (in-house strain, n = 15) were included in the study.
Rats were regularly handled to reduce stress during regular weighing, and for the subset of Wistars that underwent daily oestrous monitoring, vaginal lavage (detailed below) was conducted at 24-hourly intervals between 11:00 (lights off) and 13:00, to observe and determine cycle stages (see below and Figure 1) and therefore confirm normal cyclicity prior to studies.All handling and data collection activities were carried out during the dark phase, so they were obtained when most physiologically relevant for natural activity and reproductive behaviour. Body masses were recorded at approximately the same time each day at the following time points: pre-mating, day 3–4 pregnant and days 8, 10 and 18 pregnant. Rats were checked for lordosis behaviour at pre-mating and then again at each weighing time point; checking at the later time points was included to confirm that the behaviour seen was mating-specific, also that the pregnancy was still viable and the dam had not lost the conceptus and returned to ovarian cyclicity and proestrus behaviour. The body mass data for one Lister-Hooded rat are missing.
Representative images for each stage of the Wistar rat oestrous cycle (×100 magnification). (a) Proestrus; (b) oestrus; (c) metoestrus; and (d) dioestrus.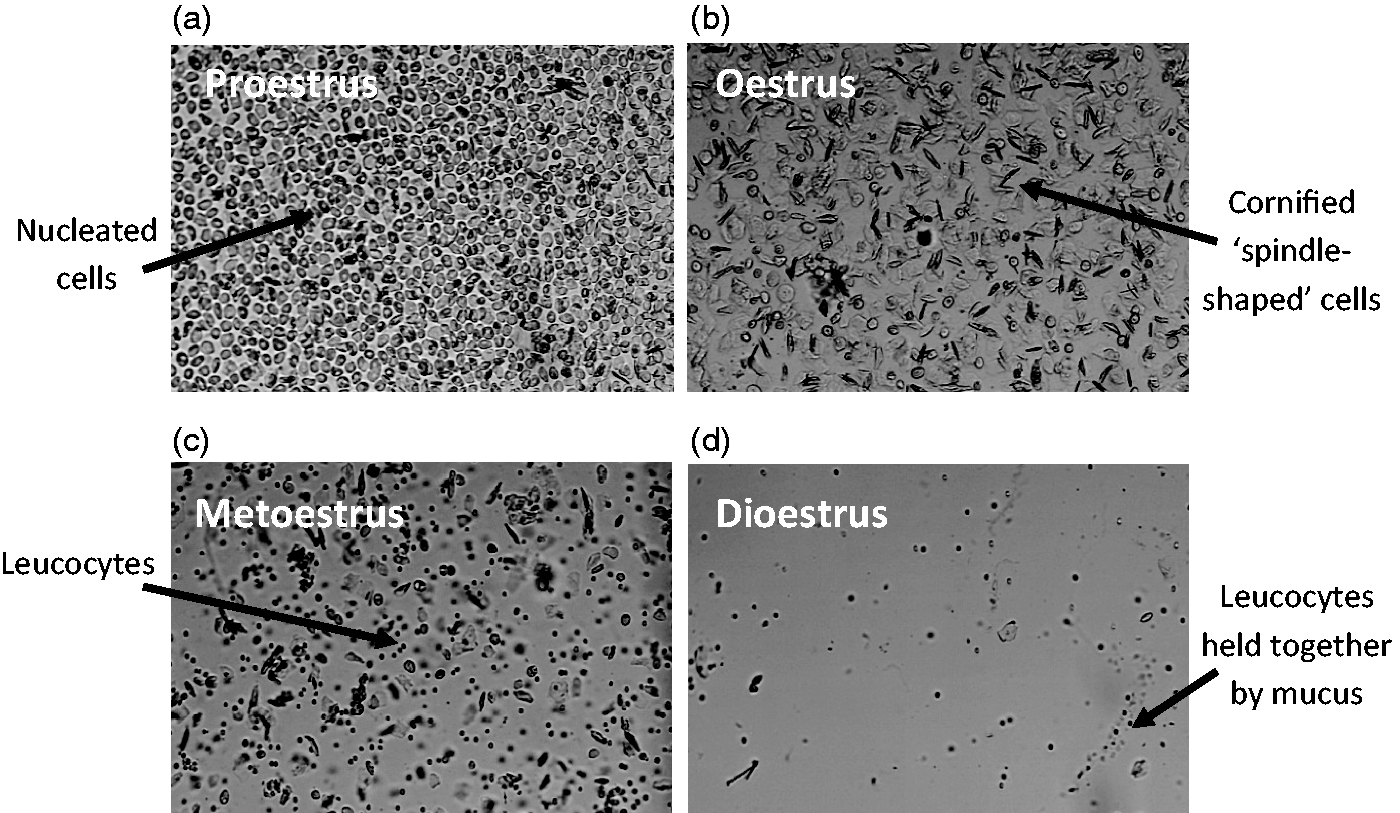
Whilst handling the Wistar animals to reduce stress prior to the commencement of oestrous monitoring (details below), a distinct behaviour was observed, which was identified as an ‘oestrus dance’ (Figure 2). When in proestrus (confirmed by vaginal cytology and uterine masses (Figure 3) for Wistar rats subgroup/in pilot studies), gentle physical manipulation with stimulation of the back of the rat near the hind legs would result in initiation and presentation of lordosis with ear wiggling, hopping, darting and freezing, without a male rat being present or even visible, as described by Feder.
1
This behavioural stimulation was repeated in Lister-Hooded rats, without vaginal cytology; the check for the ‘dance’ was carried out between 11:00 and 12:00 and took approximately 1 min per animal (see supplementary video, which is available online with this article). The oestrus dance was then used on its own, without cytology, for accurately time-mating Wistar females for the pregnancy time points of days 4 (n = 7), 12 (n = 7) and 18 (n = 7) of pregnancy, then in a much larger group of Lister-Hooded rats for work requiring specific embryonic time points and early postnatal studies.
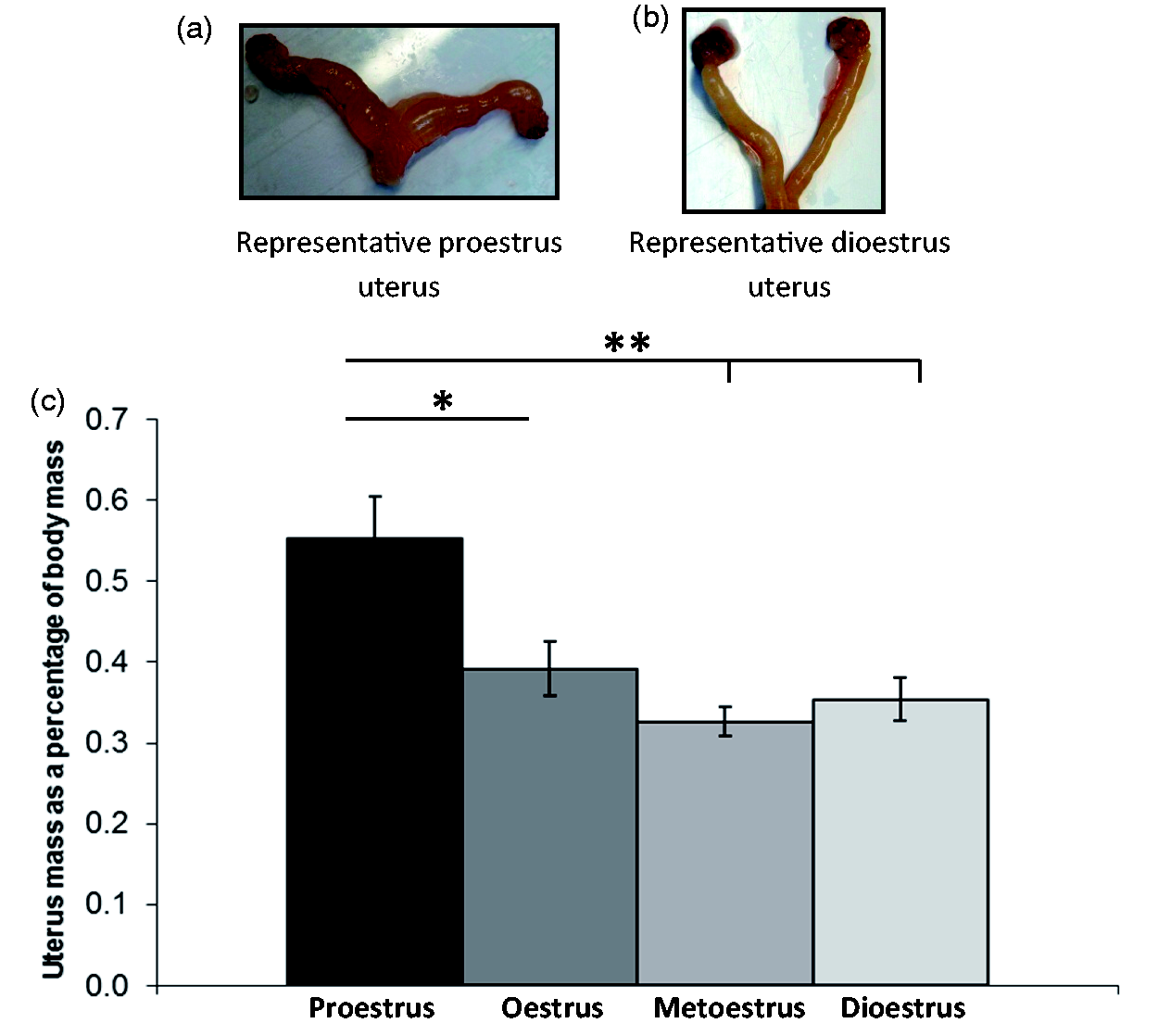
The oestrus dance in Wistar females. (a and b) The oestrus dance occurred in proestrus during the dark phase of the lighting cycle (lights turned on for photography only). See Video 1 for Lister-Hooded rat showing behavioural oestrus – note the hopping and ear twitching. Uteri (not to scale) became fluid filled at proestrus (a), but not at other cycle stages (b). (c) Intact uterus mass corrected for individual body mass (proestrus, n = 12; oestrus, n = 11; metoestrus, n = 9; dioestrus, n = 11; *P < 0.05; **P < 0.01). Uteri were significantly heavier at proestrus in Wistar rats.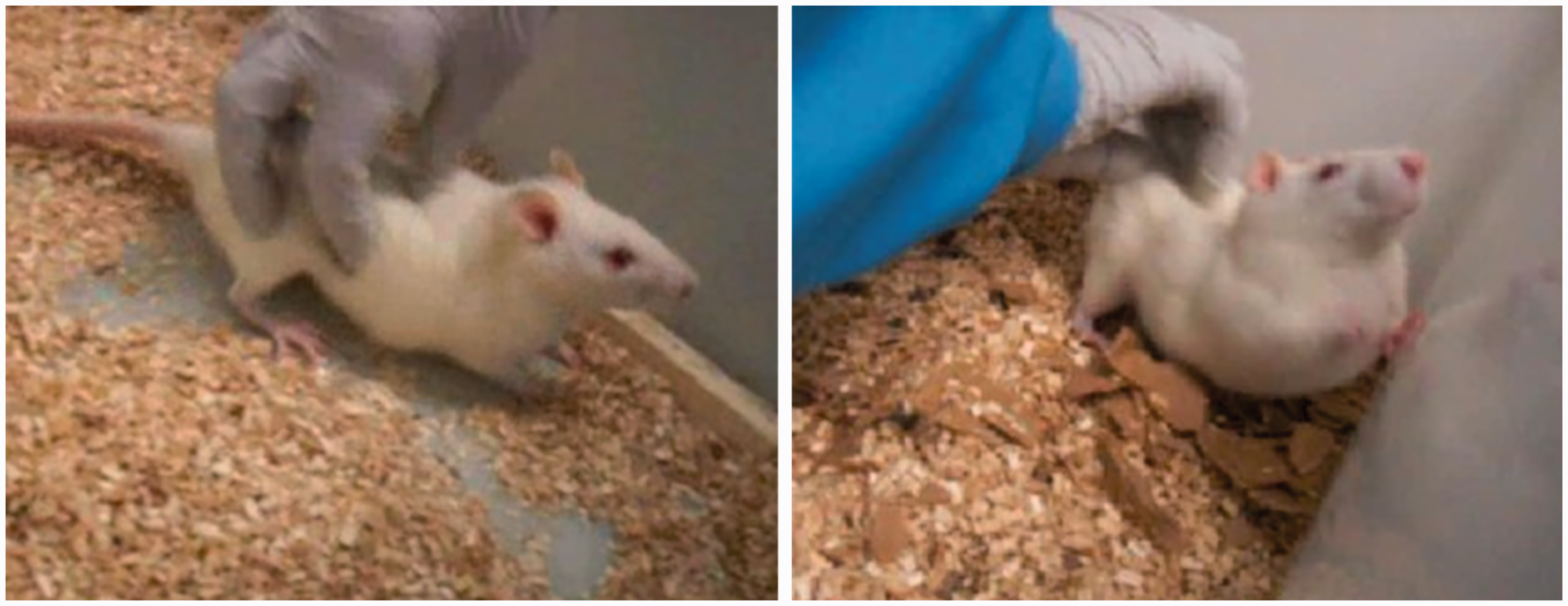
Oestrous cycle monitoring
Rats have an oestrous cycle of approximately 5 days. As guided by the literature,3,13,14 proestrus (Figure 1(a)) was identified by the presence of a large number of spherical nucleated cells, which resulted in a very cloudy cell suspension. Leucocytes were often present but in very small numbers, with cornified (keratinised, non-nucleated) cells being rare. A predominance of cornified cells confirmed oestrus (Figure 1(b)), with a number of these cells becoming spindle-shaped in appearance. Metoestrus (Figure 1(c)) was characterised by an abundance of leucocytes with a small number of cornified, particularly spindle-shaped, cells present. During dioestrus (Figure 1(d)), few cells of any cell type were present, which was characteristic of the stage in itself. A dioestrus lavage would typically contain a lot of mucus, sticking leucocytes together in lines. No vaginal cytology was undertaken in the Lister-Hooded rats.
Additionally, in 57 Wistar rats that were culled for a separate experimental purpose, the opportunity to remove their uteruses was taken and their post-mortem uterine mass increases confirmed proestrus and the other ovarian stages identified by the vaginal lavage method used (Figure 3). These Wistar females were fully anaesthetised (isoflurane; IsoFlo, Abbott) and decapitated to collect trunk blood and tissue samples for other studies.15,16
Wistar rats have a gestation period of approximately 21 days, which is slightly shorter than that of Lister-Hooded rats (23 days). Wistar rats were checked regularly close to their expected delivery date (d21 post mating), and cages were carefully checked for pups and counted, without disturbing the dams. Signs of blood were also checked for, in case of postnatal death. Pups were sexed at weaning at approximately 25 days old. Lister-Hooded rats that showed the oestrus dance and were paired with a male but failed to produce a litter (or any signs of giving birth, e.g. blood) were classed as not pregnant and used as a non-pregnant body mass comparison group. Three Lister-Hooded rats were suspected of pseudopregnancy and placed in another group for data analysis purposes.
Statistics
Values represent mean ± standard error of the mean. Statistical analysis was carried out using a one-way analysis of variance (ANOVA; uterine masses, uterine masses/body mass) and repeated measures ANOVA (body mass percentage increases following mating for nulliparous, primi/multiparous and not pregnant/pseudopregnant rat groups, with age as covariate) with Tukey post hoc tests on normally distributed data. The ANOVA F test results are reported in the format, e.g. F(2,27) = 18.06; P < 0.01; where the bracketed numbers are the degrees of freedom between groups and within groups. Pearson correlations were used to determine any relationships of body mass and body mass percentage increases following mating with age at mating. All statistical tests were performed using IBM SPSS Statistics 21. P < 0.05 was considered statistically significant.
Results
Observations of reproductive behaviour and body mass data were obtained from three different strains of rat: Wistar, Lister-Hooded and Sprague-Dawley. The initial work was carried out in Wistar rats, validated with cycle monitoring, which progressed to checking only for the oestrus dance, once the method was refined. This simpler method was then used in Wistars for pregnancy and lactation time points and in two more rat strains: Lister-Hooded and Sprague-Dawley. All rats were adjusted to reverse lighting conditions, then only checked to confirm the presence of an oestrus dance before being paired with a male if they did exhibit this proestrus behaviour, by taking body mass measurements at the designated time points. Results are not presented for Sprague-Dawleys as this method was not successful; females only displayed the oestrus dance once they were placed with a male in his home cage.
Behavioural observations: ‘oestrus dance’
The majority of females presented an obvious oestrus dance during handling in proestrus (except for Sprague-Dawleys, as mentioned above), every 4–5 days, so this distinctive behaviour was used as confirmation of readiness to mate/conceive. Additionally, animals were observed to have an agitated demeanour and be more physically active during proestrus. For the breeding colony of Wistars used in this study, the females demonstrated this sexual behaviour shortly after lights out and the Lister-Hoodeds, within the first 6 hours following lights out.
Confirmation of proestrus cycle stage in Wistars
The Wistar rats in the initial study were monitored for two (and up to four) complete ovarian cycles to ensure they were cycling normally. After they were culled at the different cycle stages, increased fluid in the uterus was used as an additional confirmation of successful proestrus identification by vaginal lavage. Intact uterus mass was found to be significantly highest at proestrus (F(3, 39) = 7.998, P < 0.001), when the uterine cavity became fluid filled (Figure 3(a)). When corrected for individual rat body mass, uteri remained significantly heavier at proestrus (F(3, 39) = 8.035, P < 0.001) than at all other cycle stages.
Refinement of time-mating procedure
Using oestrus dance observation alone to identify oestrus females that were then time-mated with proven males for 24 hours, 48/54 (89%) Wistar and 80/91 (88%) Lister-Hooded rats successfully conceived and littered down.
Wistars
Body masses were recorded pre-mating and during early pregnancy: d3P and d4P (but these d4 measurements were taken in rats fasted overnight). For seven Wistar rats that were 212 +/– 2.63 days old (range 205–218), their mean pre-mating body mass was 254 +/− 6.4 g (range 223–276) and by d3P was 267 +/− 7.6 g (232–294), with no lordosis behaviour observed at this latter time point.Their body masses had increased by 5.1% +/− 0.54 by d3P (3–7%); the d4P body masses are not shown as the rats were fasted as a requirement for a different study.
Lister-Hooded
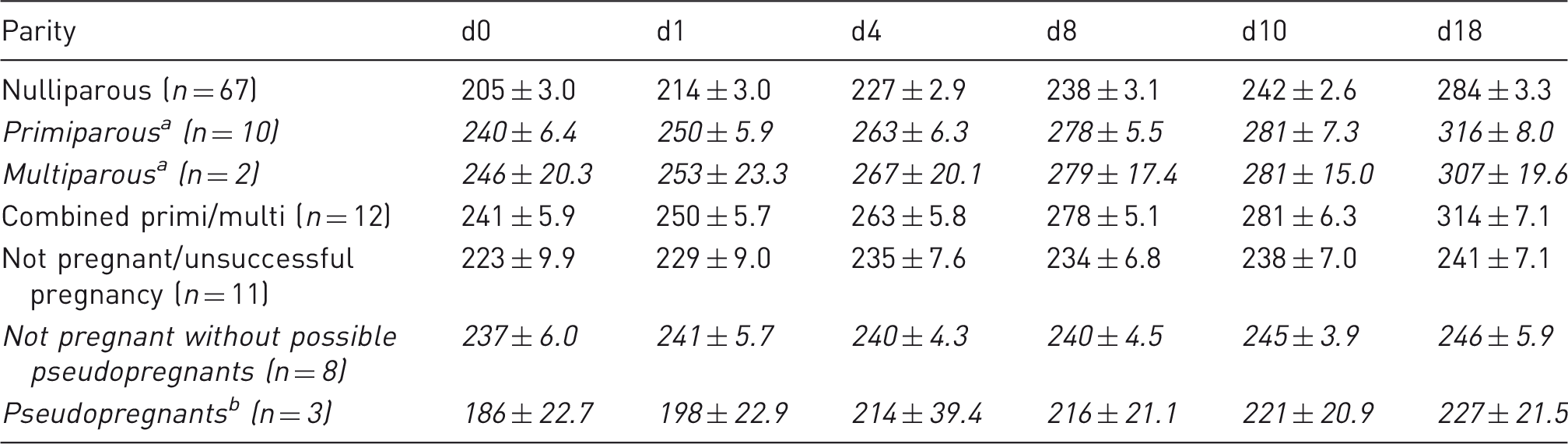
Changes in body mass after mating in Lister-Hooded rats. Mean body masses (g) before (d0) and after mating (d1–18) in nulli/primi/multiparous Lister-Hooded rats and those that did not conceive/have a successful pregnancy.
Primi- and multiparous are shown separately (in italics), then as combined as there were only 2 multiparous and their values were similar.
Possible pseudopregnants were separated from the not pregnant group.
Percentage changes in body mass after mating in Lister-Hooded rats. Mean body mass differences (%) after mating in nulli/primi/multiparous Lister-Hooded rats and those that did not conceive (within columns, a>b, P<0.001).
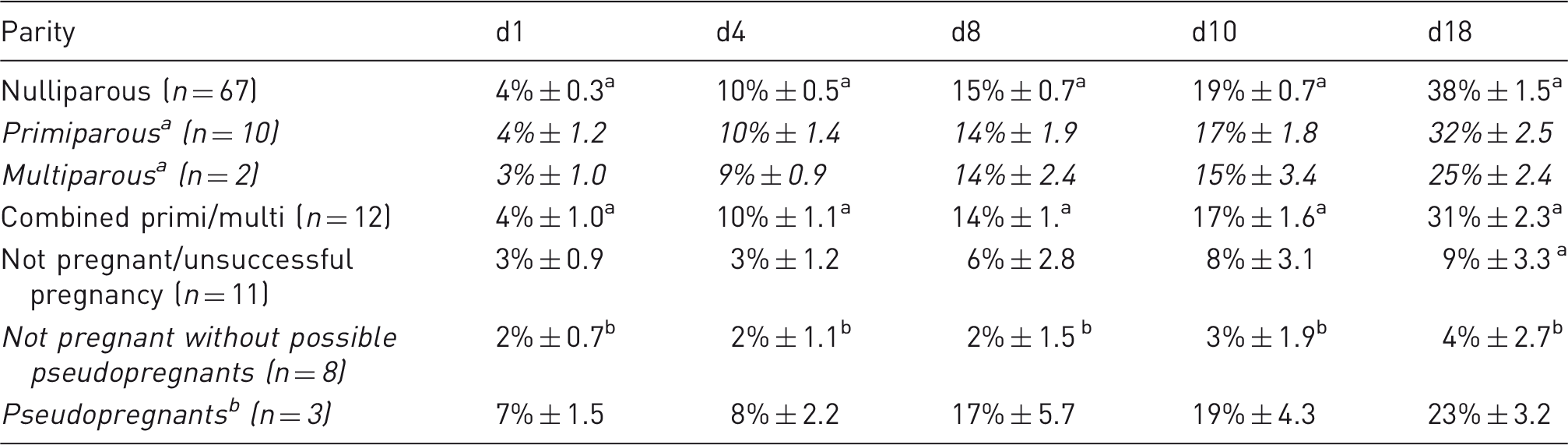
a Primi and multiparous are shown separately (in italics), then as combined as there were only 2 multiparous and their values were similar.
b Possible pseudopregnants were separated from the not pregnant group.
The body mass of all pregnant rats increased between d1P and d4P (with no further lordosis behaviour observed) and between all later time points (Tables 1 and 2; also see Figure 4). The day following mating, all rats showed modest increases of ∼ 4%, then by day 4 of pregnancy (when no physical signs are visible), consistent body mass percentage increases of 10% were recorded, followed by ∼ 15–19% by days 8–10, then up to 38% by day 18 (Table 2).
Percentage body mass increases throughout pregnancy from mating, for nulliparous and combined primi/multiparous pregnant and not pregnant (excluding three pseudopregnant).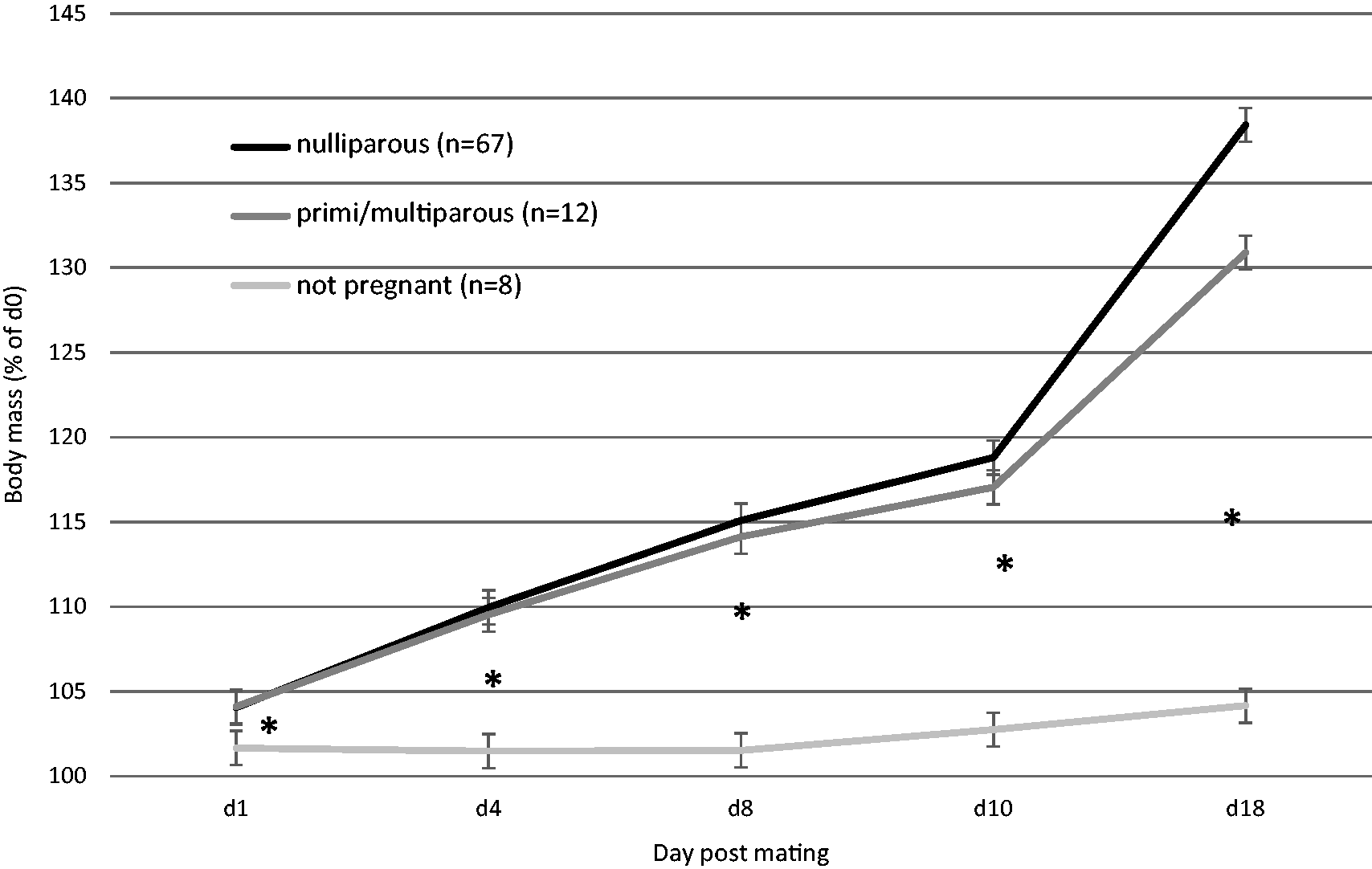
Primi- and multiparous rats were heavier prior to mating than nulliparous rats (d0, Table 1) as they were slightly older). However, the pattern of body mass increase (% increase of d0, Table 2) was similar for all pregnant rats until d18, when it was reduced for the rats that had already had prior litters (31% cf. 38%, Table 2). Nulliparous rats had the highest body mass percentage gains, especially by d18 of pregnancy, as they were younger and still physically growing (Table 2; re: age, see next section and Figure 5).
Body mass at mating (d0) by age (in days) in nulliparous Lister-Hooded rats. There was a positive correlation for nulliparous rats only (r = 0.739, n = 67, P < 0.001). Body masses at mating (d0) of nulliparous Lister-Hooded rats positively correlated with their age.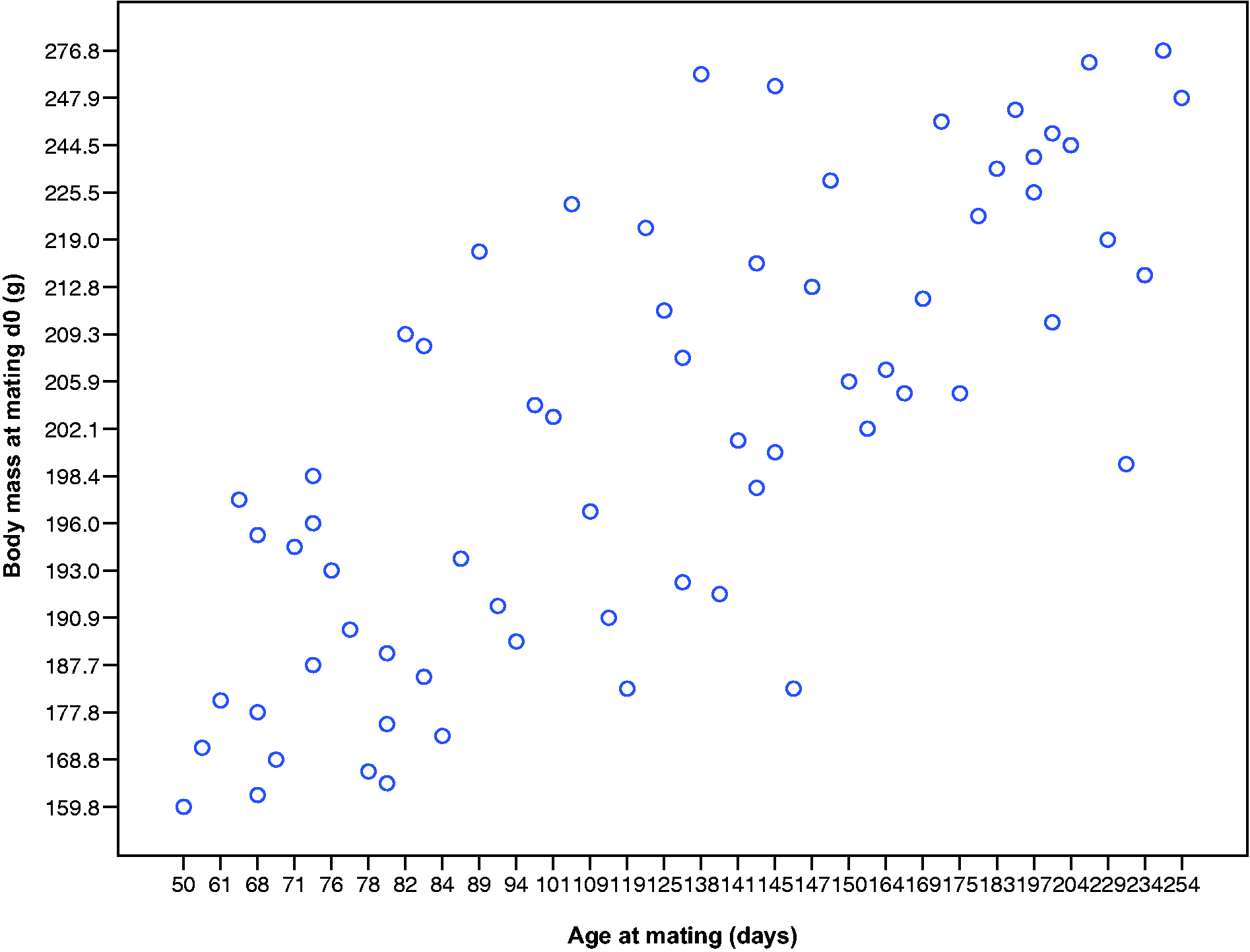
The not pregnant rat group, not including three suspected pseudopregnant animals, showed a very slight body mass gain (up to 4%) from d0 through to d18 post mating (Figure 5) but values were significantly lower than all the pregnant rat time points (F(2,67) = 22.563, P < 0.001; with age as covariate). The small pseudopregnant group had similar body mass gains to pregnant rats, although the increase by d18 was reduced compared to the younger rats with successful pregnancies (23% cf. 38% nulliparous cf. 32% primiparous, Table 2). Although the numbers are small (n = 3) and age range was wide (53–230 days), the pseudopregnant rats had a similar final body mass increase to multiparous rats (23% cf. 25%).
Pup totals and sex produced by nulliparous, primiparous and multiparous Lister-Hooded rats. Data shown ± standard error of the mean.
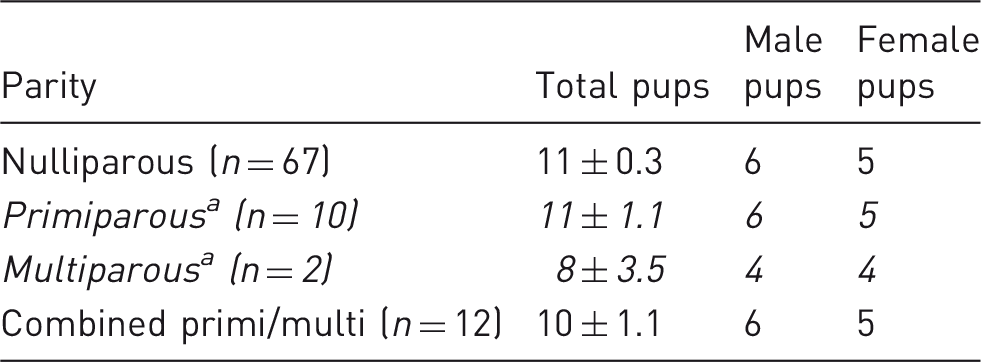
a Primi and multiparous are shown separately (in italics), then as combined as there were only 2 multiparous and their values were similar.
Larger body mass increases in nulliparous rats in late gestation
Body mass increases by the end of gestation were larger in younger nulliparous rats (Table 2) so we explored these data further. Figure 5 shows the range of ages of the nulliparous rats against their body masses at mating (d0). There was a positive correlation for nulliparous rats only (r = 0.739, n = 67, P < 0.001 cf. primi/multiparous r = 0.244, n = 12, P = 0.445, ns), suggesting that they were still growing physically/maturing to adult body sizes despite being reproductively competent.
Likewise, for nulliparous rats only, there was a negative correlation between age at mating and body mass increases throughout pregnancy: the youngest pregnant rats had the highest body mass percentage increases by d18, with the oldest having the smallest increases (d1%–d18%: r = −0.320 to −0.648, n = 57–67, P = 0.008 to < 0.001; Figure 6). There were no significant correlations for primi- or multiparous rats (d1%–d18%: r = −0.274 to –0.501, n = 11−12, P = 0.097–0.389, ns).
Relationship between age at mating (days) and percentage body mass increases from mating to day 18 of pregnancy in nulliparous Lister-Hooded rats (r = −0.608, n = 74, P < 0.001). Age of nulliparous Lister-Hooded rats negatively correlated with body mass increases during gestation.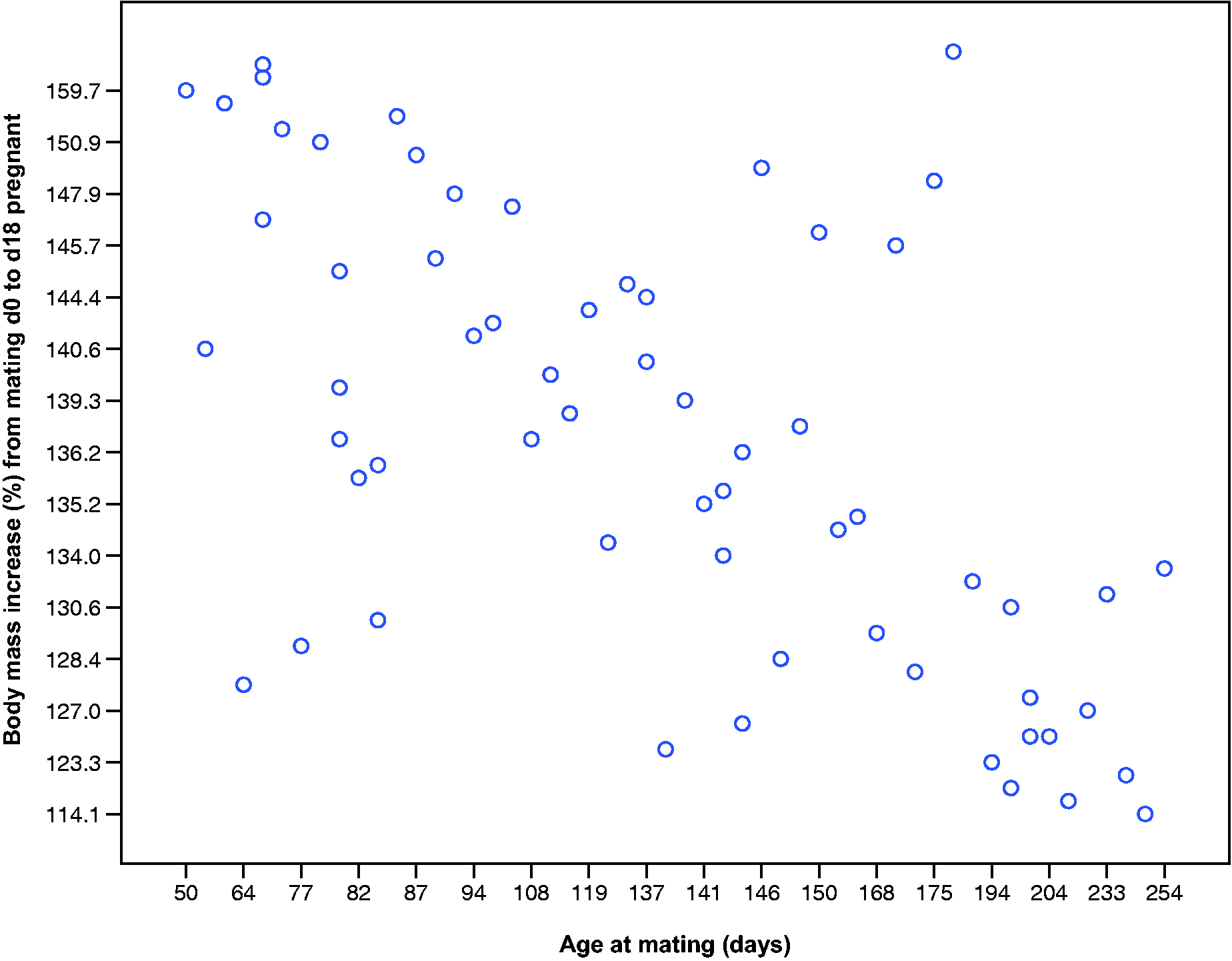
Discussion
We have described an alternative, non-invasive and consistent timed-mating method that uses gentle physical manipulation during handling to instigate lordosis behaviour in female Wistar and Lister-Hooded rats. Implementing the oestrus dance to time-mate the dams was very successful and a distinct refinement of the commonly used time-mating procedure (over 5 days with male using five females/time point required). This new and accurate method has important welfare implications as it greatly reduced the number of animals needed and produced a pregnancy rate approaching 90% with only a single, unproven female required per time point. This represents a further refinement as proven breeders are nearly always used with the existing method. Additionally, the disturbance of females with either oestrous monitoring and/or to check for the appearance of a vaginal plug to confirm mating was reduced, and then removed entirely. Females were subject to less stressful situations, as they were only paired with males when sexually receptive.
In the current study, pregnancy in Wistar and Lister-Hooded rats could be confirmed non-invasively, with confidence as early as d4 (when visualisation of any conceptus is not yet possible) using body mass increases of 10% from prior to mating body masses, compared to the not pregnant control group. This was much earlier and an improvement on previous studies that used body mass increases from mating to d14–d19 of pregnancy. 11 Furthermore, confirmation of pregnancy in unproven, nulliparous dams was very reliable as they had the highest body mass gains towards the end of pregnancy. This is a potential improvement in study design for future studies, which would not need to use proven breeders. Study animals could be selected with less physiological variation: those within a reduced age and body mass range, and that were also nulliparous.
This work has also revealed a further consideration for studies using unproven nulliparous rats. As they were still growing, this resulted in the youngest rats achieving the highest body mass gains from mating to d18 of pregnancy. Thus, we can further predict from this work that older nulliparous rats, over approximately 20 weeks (5 months) old, are likely to have smaller body mass increases to d18P, closer to the values achieved by primi- and multiparous dams. These slight body mass differences may also reflect reducing litter sizes, as the multiparous dams had smaller litter sizes than the nulli- and primiparous groups.
Non-pregnant animals showed small body mass increases, which might be expected to be greater in younger, growing animals, but the much larger increases in mass due to pregnancy in the other rat groups became apparent as early as the day following mating and had more than doubled by day 4 of pregnancy, continuing to increase further thereafter. Based on our findings for Lister-Hooded and Wistar rats that displayed the oestrus dance, we would predict that increases of greater than 3% by day 1 following mating indicate conception and over 8% by day 4 confirm a successful pregnancy.
However, we offer one potential caveat with this body mass increase method, relevant to studies using or requiring multiparous dams. Based on our findings, albeit with very small numbers, there could be the possibility of a false positive pregnancy diagnosis in a multiparous dam when a pseudopregnancy had actually occurred (n = 3), as their body mass increases mirrored the values of the multiparous dams (n = 2) for the whole study period. This was surprising as pseudopregnancy is expected to resolve by day 10, so it is possible that these pseudopregnant animals did conceive but their pregnancies were resorbed at a late stage, which could explain the late pregnancy body mass gains. Further studies are needed using larger sample sizes of pseudopregnant animals compared with multiparous dams to confirm this observation.
In our study, the rats were housed under reverse lighting conditions, so they were observed and weighed during their active phase. This was simple to instigate in separate rooms within the facility, as they each had individual light cycle monitors. After being housed in rooms with lighting schemes that suit human workers, brought-in rats easily adapt to reverse lighting within about 2 weeks. Rats born under reverse lighting did not require any adjustment time. Reverse lighting rooms were fitted with red overhead lights, and workers also used red headtorches to allow cleaning, feeding and other day-to-day tasks without disrupting the lighting conditions.
We were unable to observe the behavioural signs of oestrus in Sprague-Dawley rats, which were also studied under reverse lighting, before they were placed in the home cages of male rats, so our new methods were not replicated in all strains tested. Thus, more animal and labour-intensive studies are required to achieve the same outcomes for timed-mating in this rat strain when housed under similar conditions, and this may also apply to other rat strains not studied here. It remains unknown why some laboratory rat strains show overt behavioural signs of oestrus on handling whilst others do not unless they are in close proximity to a sexually mature male rat, which may indicate that the role of pheromonal or other intersexual signals varies depending on genetic background. It was noted that cytological changes in Sprague-Dawleys were much harder to follow, thus their ovarian cycles may have been irregular and less predictable. Sprague-Dawleys have demonstrated more anxiety-like behaviours and reduced motor function compared with other rat strains,17,18 so their natural behaviours may be more inhibited during handling. Thus, testing of other rat strains to find out which ones can be gently stimulated to show lordosis behaviour will also be required before attempting to fully adopt this new method.
We have established a new robust method that combines a behavioural method to increase mating success rates and additionally validated the use of body mass increases (as % body mass since mating) across a range of ages and parities to confidently predict whether a nulli-, primi- or multiparous dam is, and/or remains, pregnant from day 4 of pregnancy. Based on our findings it should also be expected that the youngest nulliparous females will gain more body mass in the later stages of pregnancy, which highlights the importance of them having access to enough additional food at this stage of pregnancy, compared with primi- and multiparous dams.
Setting up a study that relies on early embryonic stages requires practice of cytological methods to confirm reproductive cycle staging, but once reproductive cyclicity has been established in the breeding stock, the use of behavioural signs of oestrus, induced by gentle handling, can replace the need to undertake the previously labour-intensive and potentially disruptive procedures in the females. This refined breeding management procedure has been adopted as a new efficient practice in the Biomedical Research Unit (BRU) at The Open University for two strains of rat, Wistars and Lister-Hooded, housed under reverse lighting, which is effective for nulliparous (unproven) as well as proven females. This has resulted in a reduction in the number of rats required to provide new rat litters or females at specified time points of pregnancy or lactation.
Supplemental Material
sj-vid-1-lan-10.1177 0023677218774076 - Supplemental material for Improved timed-mating, non-invasive method using fewer unproven female rats with pregnancy validation via early body mass increases
Supplemental material, sj-vid-1-lan-10.1177 0023677218774076 for Improved timed-mating, non-invasive method using fewer unproven female rats with pregnancy validation via early body mass increases by Agata K Stramek, Michelle L Johnson and Victoria J Taylor in Laboratory Animals
Footnotes
Acknowledgements
The authors thank Steve Walters, Karen Evans, Sophie Brooks and Gareth Williams for outstanding technical support and other life, health and chemical sciences (LHCS) researchers for the opportunity to monitor their breeding stock. We are also grateful to anonymous reviewers whose comments on an earlier version have helped us improve the current manuscript. The School of Life, Health and Chemical Sciences (LHCS), The Open University, provided the funding for this work.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
