Abstract
Endotracheal intubation of laboratory animals is a common procedure shared by several research fields for different purposes, such as mechanical ventilation of anaesthetized animals, instillation of cytotoxic nanoparticles, infectious agents or tumour cells for induction of disease models, and even for diagnostic and therapeutic purposes. These different research purposes, achieved in different animal models, require technical expertise and equipment that suits every research need from animal facilities. In this short report we propose a videoendoscopic intubation station that could be shared among the most common laboratory animals, namely the mouse, rat, guinea pig and rabbit, from neonates to adult animals. This report aims to contribute to the reduction of animals excluded from experiments due to false paths during direct and blind intubations and to the refinement of procedures by replacing surgical approaches such as tracheotomy.
Anaesthesia of laboratory animals is a generalized procedure in laboratory animal science; however, endotracheal intubation and mechanical ventilation is often impossible to perform due to a lack of specialized equipment and an absence of trained personnel in laboratory animal intubation.
Appropriate ventilation of anaesthetized laboratory animals may be imperative when fundamental physiological processes are being studied and normal blood pH, oxygen saturation (sO2) and partial pressure of carbon dioxide (PaCO2) are desirable. 1 Additionally, it allows resuscitation when respiratory arrest occurs, especially when drugs that induce marked respiratory depression are being used. 2
Endotracheal intubation also allows the delivery of inhalation anaesthesia, a method preferred in large laboratory animal anaesthesia since it combines the advantages of mechanical ventilation with the benefits of inhalation anaesthesia. Studies on endotracheal intubation with inhalation anaesthesia in mice and rats have also been published, suggesting their feasibility in small laboratory animals.3,4
The endotracheal route is also used for the induction of pulmonary diseases such as specific occupational diseases contracted by exposure to cytotoxic nanoparticles, 5 and instillation of tumour cells for lung cancer research, 6 and is even used as a route of infection in pulmonary infection models such as tuberculosis. 7 Furthermore, this route has recently been explored for new therapeutic strategies. 8
Due to the different body sizes of laboratory animals and their specific anatomic features, strategies described for videoendoscopic intubation require specie-specific tips and equipment.1,3,4,9,10 By combining and optimizing these specie-specific strategies, we present a videoendoscopic intubation station that can be shared among the most commonly used species housed in an animal facility, by applying the same intubation technique.
Animals used in this work were included in research projects underway in the institution, with each project being approved by the animal ethics committee of the Institution and by Direção Geral de Alimentação e Veterinária (DGAV), the competent national authority for animal protection. All personnel involved in the procedures were approved as competent for animal experimentation by DGAV.
Two animals of each species were used: Mus musculus, Rattus norvegicus, Cavia porcellus and Oryctolagus cuniculus. Animals were anaesthetized intraperitoneally with a combination of ketamine and medetomidine.
All species were intubated with the help of a videoendoscopic system with a 1.9 mm diameter, 10 cm length, 30° endoscope (HOPKINS II Telescope; Karl Storz GmbH, Tuttlingen, Germany) and a Telepack X System (Karl Storz GmbH) comprising a light source and an endoscopic camera (Figure 1).
Videoendoscopic intubation station consisting of a 1.9 mm, 10 cm, 30° endoscope and a Telepack X System with a light source and endoscopic camera.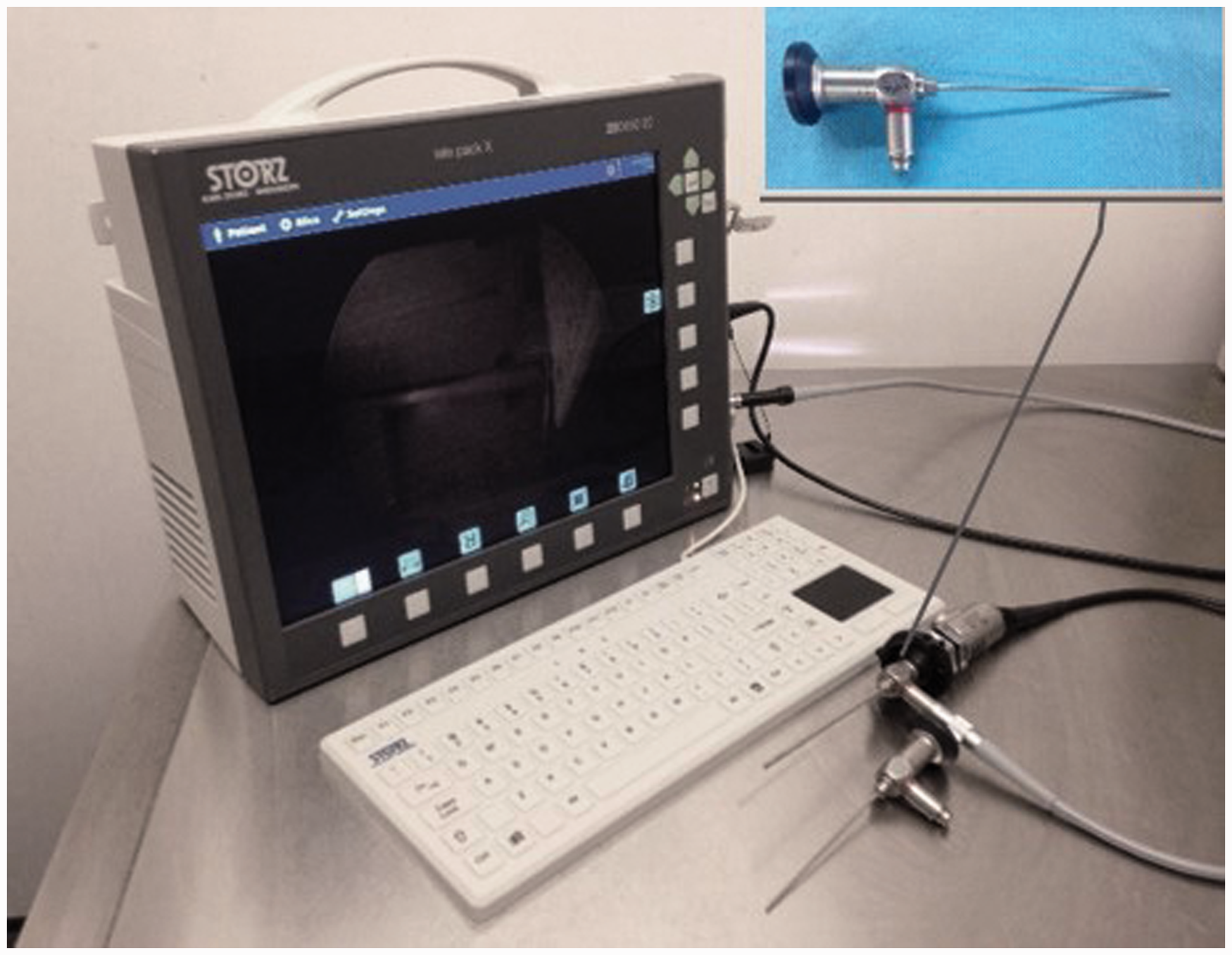
For the right-handed operator, animals were positioned in right lateral recumbency, the endoscope was handled with the left hand and the endotracheal tube with the right hand via the following steps (Figure 2):
The endoscope tip was introduced into the mouth above the tongue with the bevel pointed dorsally, allowing visualization of the palate. The endoscope was advanced until visualization of palatine veins was achieved (Figure 2, column 1). By following the palatine veins, the edge of the soft palate was visualized covering the epiglottis and closing the laryngeal opening. Gentle pressure was applied on the soft palate with the tip of the endoscope and the palate was displaced dorsally allowing the epiglottis to fall, thereby allowing visualization of the laryngeal opening (Figure 2, column 2). (If the epiglottis is already ventral to the soft palate, this step is no longer needed.) The endoscope was withdrawn slightly and vocal cords were visualized. An endotracheal tube was inserted into the mouth, parallel to the scope (Figure 2, column 3). The endotracheal tube was then aligned with the laryngeal opening and advanced through the vocal cords during inspiration, taking advantage of the opening of the glottis. Spinning movements of the endotracheal tube helped its progression (Figure 2, column 4). Videoendoscopic intubation of laboratory animals using a 1.9 mm and 30° Hopkins rigid endoscope. View of glottis of the mouse (row A); rat (row B); guinea pig (row C) and rabbit (row D). Visualization of the palatine veins and the soft palate (column 1); visualization of the epiglottis and vocal cords after dorsal displacement of the soft palate (column 2) – except for the guinea pig in which the epiglottis is behind the palatal ostium which makes it difficult to lower and exteriorize; introduction of the endotracheal tube side-by-side with the endoscope (column 3); Introduction of the endotracheal tube through the vocal cords, inside the trachea. (column 4).
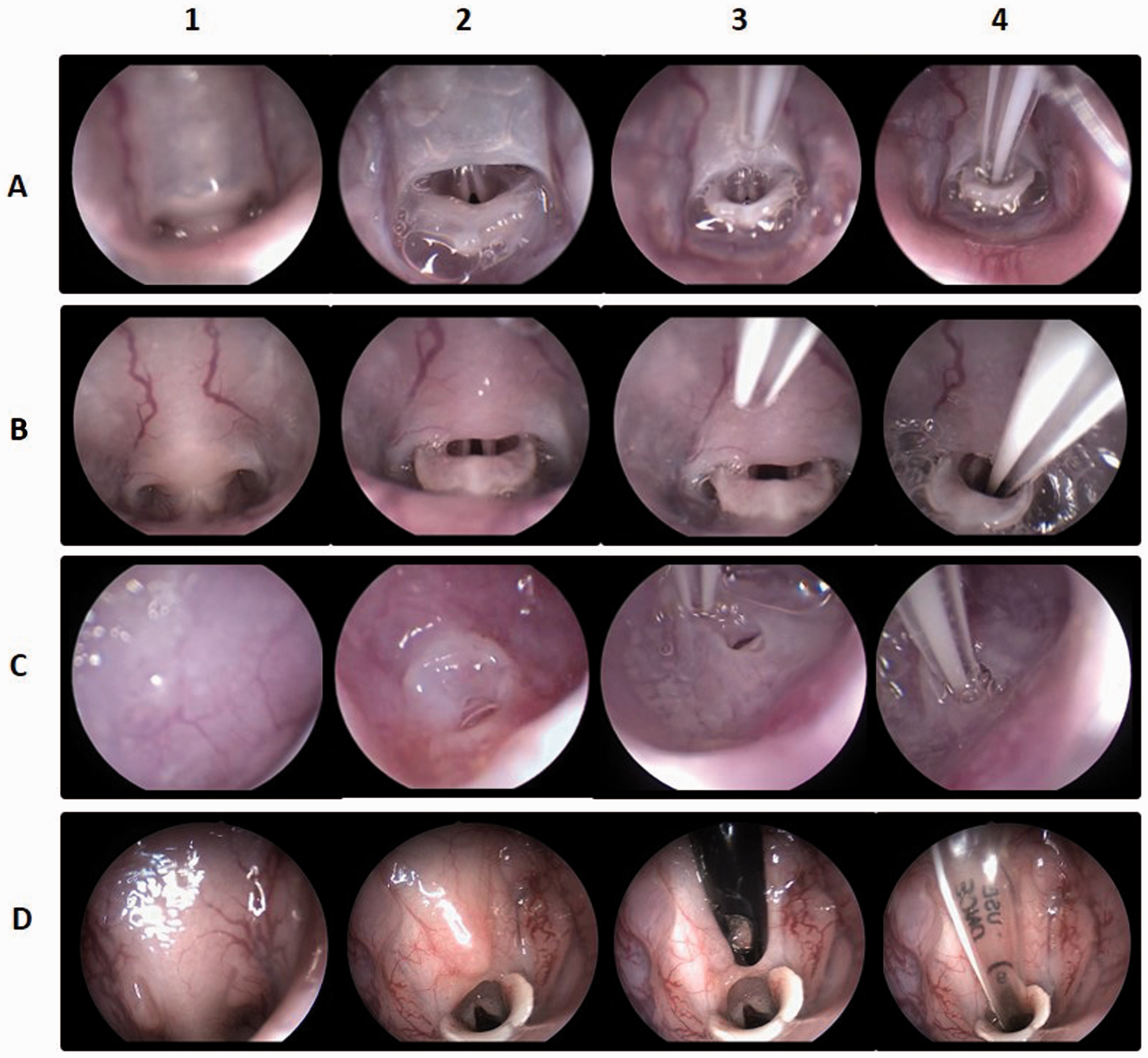
This side-by-side technique uses an endoscope with a very small diameter (1.9 mm) as a laryngoscope to lower the base of the tongue and the epiglottis. The 30° angled tip favours endoscope progression and allows a wide view over the soft palate. The endoscope enters the mouth laterally to the large incisors and the animal’s lateral position favours this approach. The camera head can be supported at the operating table while progressing through the mouth, allowing a more stable image. The animal’s lateral position has advantages over the dorsal position since the tongue does not fall over the endoscope, nor does it need to be held during endoscope progression and intubation. During intubation, the scope remains in the oral cavity and only the endotracheal tube progresses through the vocal cords. Therefore, this technique can be applied in animals, such as mice and neonatal rats, which require endotracheal tubes with smaller diameters than the scope, as long as their mouth opening allows the progression of the scope (1.9 mm). For these reasons, only one person is required, and no mouth gags are needed for mouth opening or platforms for specific body positions.
Guinea pigs present a unique anatomy with a soft palate continuous with the palatoglossal arches and the base of the tongue.
11
Only a small palatal ostium is visualized (Figure 2, column 3) and the soft palate must be displaced in order to visualize the epiglottis and the small laryngeal opening (Figure 2, row C). The choice of endotracheal tubes should be made according to the species, and adjustments must be made according to the size of the animals within the species:
Mus musculus (26 ± 1 g): 24 G intravenous catheter Rattus norvegicus (360 ± 15 g): 18 G intravenous catheter Cavia porcellus (380 ± 12 g): 18 G intravenous catheter Oryctolagus cuniculus (2300 ± 152 g): 2.5 mm internal diameter
Although the initial investment in a videoendoscopic station by the animal facility may be higher than the cost of the equipment used for direct intubation, this cost is not prohibitive and may be justified by the general usage by researchers in a greater variety of animal models, for the most diverse research fields and for procedures requiring endotracheal access.
Footnotes
Acknowledgment
The authors would like to thank Karl Storz GmbH.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was granted by Project ON.2 SR&TD Integrated Program (NORTE-07-0124-FEDER-000017), co-funded by the North Portugal Regional Operational Program (ON.2-O Novo Norte), under the National Strategic Reference Framework, through the European Regional Development Fund (ERDF). This work was also supported by FEDER funds through the Operational Programme Competitiveness Factors – COMPETE and National Funds through FCT – Foundation for Science and Technology under the project POCI-01-0145-FEDER-007038. A Miranda was supported by an FCT grant (SFRH/BD/52059/2012).
