Abstract
Egyptian fruit bats are increasingly used as model animals in neuroscience research. Our aim was to characterize suitable injectable anaesthesia for this species, possibly replacing inhalant anaesthesia, thus minimizing occupational health hazards. Eight bats were randomly assigned by a crossover design for subcutaneously administered combinations of medetomidine-midazolam with: saline (MM-Sal), ketamine (MM-Ket), fentanyl (MM-Fen), morphine (MM-Mor), or butorphanol (MM-But). The anaesthetic depth and vital signs were monitored at baseline and every 10 min until bats recovered. If after 180 min the bats did not recover, atipamezole was administered. Mean induction times were 7–11.5 min with all combinations. Twitching during induction was common. All combinations produced anaesthesia, with significantly decreased heart rate (from 400 to 200 bpm) and respiratory rate (from 120–140 to 36–65 rpm). Arrhythmia and irregular breathing patterns occurred. MM-Fen, MM-Mor, and MM-But depressed respiration significantly more than MM-Sal. Time to first movement with MM-Ket and MM-But lasted significantly longer than with MM-Sal. Recovery time was significantly shorter in the MM-Sal (88 min) in comparison to all other treatments, and it was significantly longer in the MM-But (159 min), with atipamezole administered to four of the eight bats. In conclusion, all five anaesthetic protocols are suitable for Egyptian fruit bats; MM-Ket produces long anaesthesia and minimal respiratory depression, but cannot be antagonized completely. MM-Fen, MM-Mor, and MM-But depress respiration, but are known to produce good analgesia, and can be fully antagonized. Administration of atipamezole following the use of MM-But in Egyptian fruit bats is recommended.
Introduction
Egyptian fruit bats (Rousettus aegyptiacus) have been studied for decades in various research fields, 1 including investigation of their wing anatomy, 2 metabolism and adaptability to different climates,3,4 and recently their suspected involvement in the transmission of several zoonotic diseases.5,6 In 2011, Egyptian fruit bats were introduced as an animal model for research of spatial cognition, navigation, and spatial memory in the mammalian hippocampus, 7 and ever since appeared in an increasing number of publications in this field.7–12
Research using Egyptian fruit bats involves procedures such as imaging or surgery that require anaesthesia, for both long and short durations. Isoflurane inhalation anaesthesia is often the method of choice, having the advantages of a wide safety margin, very little metabolization, and quick induction and recovery. 13 However, it has some limitations: first, although portable inhalation anaesthetic systems are available, 14 and have been used to anaesthetize bats, 15 these are not commonly available under field conditions. Second, isoflurane is usually administered to Egyptian fruit bats via facemask, as is the case with other small animals, posing occupational health concerns due to the risk of anaesthetic gas leakage,16,17 especially when prolonged procedures are routinely performed in experimental settings. These concerns have been our primary motivation in developing reliable injectable protocols for anaesthesia of Egyptian fruit bats, which could potentially replace isoflurane anaesthesia at our bat facility.
Several injectable protocols have been reported in various bat species. Single agents such as ketamine, xylazine, and medetomidine have been used, 18 but combining several anaesthetic agents is usually advantageous, as it often results in lower individual dose requirements, reduces undesirable side effects and improves effectiveness.13,19 The most common combinations used for bat anaesthesia are alpha-2 adrenergic agonists, such as xylazine or medetomidine, with the dissociative anaesthetic ketamine.18–22 At our facility, a combination of ketamine 15 mg/kg and medetomidine 0.06 mg/kg was used for induction of anaesthesia in Egyptian fruit bats, followed by isoflurane administration via facemask. 7 These combinations are usually effective, as alpha-2 agonists potentiate ketamine and provide muscle relaxation, and ketamine stabilizes heart rate and blood pressure and provides some analgesia. 7 However, alpha-2 agonist and ketamine combinations may be associated with prolonged recoveries, increasing the risk of hypothermia. 23 Administration of alpha-2 antagonists in order to shorten recovery may not be recommended, as the bats will remain under the influence of ketamine, increasing the risk for catatonic reactions such as tremors or muscle rigidity. 24
A combination of medetomidine, midazolam, and fentanyl (MM-Fen) has also been reported in bats. 25 This combination was described in several animal species and is often used in research settings.23,24,26,27 Although none of the MM-Fen components are anaesthetics per se, this combination has been shown to induce anaesthesia with surgical tolerance,17,28 and has been used in various surgical procedures in several species,29,30 including bats.25,31 MM-Fen offers several advantages: (a) significant analgesia provided by fentanyl, a rapid acting synthetic opioid, 32 and medetomidine; 33 (b) good muscle relaxation, provided by midazolam and medetomidine;33,34 (c) amnesia, an important feature of surgical anaesthesia, 35 provided by midazolam;36,37 and (d) rapid and complete antagonization with atipamezole, flumazenil, and naloxone, 23 reducing the risk of anaesthetic related morbidity or mortality. 38 These important benefits could potentially make MM-Fen a suitable replacement for inhalation anaesthesia.
Our aim in this study was to evaluate the efficacy of MM-Fen combination in Egyptian fruit bats, and compare it to other medetomidine-midazolam based protocols with morphine, the prototypical opioid analgesic, butorphanol, a synthetic agonist-antagonist opioid, 39 ketamine, or saline. We aimed to compare onset, duration, and quality of anaesthesia induced by the combinations, duration and quality of recovery from anaesthesia, and the side effects of each combination.
Materials and methods
Ethics statement
This study was approved by the Institutional Animal Care and Use Committee of the Weizmann Institute of Science (permit 15061214-1), in compliance with Israeli animal protection law and The Guide for the Care and Use of Laboratories Animals, 8th edition. Housing facilities and animal care and use programs at the Weizmann Institute are fully accredited by the Association for the Assessment and Accreditation of Laboratory Animal Care International (AAALAC).
Animals and housing conditions
Eight healthy adult male Egyptian fruit bats (age unknown, average weight (mean ± SD) 177 ± 14 g) were randomly selected from the institute's bat colony. No females were selected for this study, since almost all healthy females in the colony are either pregnant or nursing throughout the year, and we did not want to risk the health and development of the pups with frequent anaesthetic events. All eight bats were kept together in a large mesh cage (length 3 m, Width 2 m, height 2.2 m) in a controlled-environment room (temperature 23℃, humidity 50%, 12:12 light/dark reverse photoperiod cycle as Egyptian fruit bats are nocturnal) .The bats were fed ad libitum with varied seasonal fruit, supplemented with Lori powder (Versele-Laga, Orlux, Belgium) on weekends. Clinical examination and weight monitoring were performed once a week.
Study design
Five anaesthetic protocols were tested. All protocols included medetomidine (Domitor, Orion Pharma, Espoo, Finland; 1 mg/mL) 0.15 mg/kg and midazolam (Midolam, Rafa, Israel; 5 mg/ml) 1.5 mg/kg (MM) combined with a third drug: saline (MM-Sal), ketamine (Clorketam, Vetoquinol, Lure, France; 1 g/10 mL) 10 mg/kg (MM-Ket), fentanyl citrate (Fentanyl, Janssen Pharmaceutica, Beerse, Belgium; 0.0785 mg/mL) 0.015 mg/kg (MM-Fen), morphine (Morphine HCl, Teva, Israel; 20 mg/mL) 1.5 mg/kg (MM-Mor), or butorphanol (Morphasol, aniMedica GmbH, Germany; 10 mg/mL) 1 mg/kg (MM-But). Doses of the drugs were selected after a pilot study in which increasing doses of medetomidine and midazolam were injected until anaesthesia induction was achieved within 10 min (unpublished results). All bats were subjected to all five anaesthetic protocols, using a randomized crossover design (with the help of an online randomization software; www.random.org/lists/) with at least a ten-day washout period between anaesthetic events. All assessments were performed by the same investigator, who was not aware of the administered drug protocol (blind design).
On the morning of each experiment (at the beginning of the dark photoperiod, and before feeding) each bat was captured using protective leather gloves (Sekuroka@-nappa leather gloves, Carl Roth GmbH and Co.), weighed (Citizen CT 1200 scale, Citizen, India, 1200 ± 0.1 g) and placed in a metal mesh cage (35.5 × 25 × 24 cm3) that enabled observation. An acclimatization period of 30 min was applied in order to enable the bats to recover from the stress of capture. At the end of the acclimatization period, a physical examination was performed. The following parameters were examined to serve as baseline: (a) reflexes, including withdrawal (toe pinch), tested by pinching a finger with plastic forceps and palpebral reflex (gently touching the medial canthus of the eye with plastic forceps); (b) response to noise by scrunching up bubble wrap while observing ear movement and eye blinking; (c) heart rate and pattern by stethoscope auscultation (heart rate up to 300 was counted, and when it was uncountable it was termed 400); (d) respiratory rate and pattern by observation of thoracic excursions; and (e) rectal temperature using a digital thermometer.
Anaesthetic mixtures of 0.75 mL were prepared per 1 kg body weight, with addition of saline to compensate for volume difference when necessary (Appendix A), in order to shield the investigator from the protocol used. Injection volume was calculated per body weight at the beginning of each trial, then drawn into an insulin syringe (BD Micro-Fine Plus Demi 0.3 mL, Becton, Dickinson and Company, USA), and injected subcutaneously between the bat's scapulae. The bat was returned to its cage and observed continuously. As soon as the bat stopped moving and did not show resistance to handling, it was placed on an electric heating pad, which was covered with an absorbent pet sheet. Oxygen at 1 L/min was administered via a facemask, and the bat was covered with bubble wrap. Core body temperature was kept at 37.0–38.5℃ throughout the anaesthesia period by manipulating the heating pad and bubble wrap.
Several time periods were recorded: (a) time to first effect – time from injection of anaesthetics to first observed ataxia or disorientation; (b) induction time – time from injection of anaesthetics until the bat stopped moving and accepted the face mask; (c) time to first movement – time from injection of anaesthetics until the first voluntary movement, and (d) recovery time – time from injection of anaesthetics to flying. If after 180 min the bat did not recover spontaneously, 0.05 mg of atipamezole hydrochloride (Antisedan, Orion Pharma, Espoo, Finland; 5 mg/mL; 0.01 mL diluted with 0.09 mL saline) was administered subcutaneously in order to antagonize medetomidine and shorten recovery, and ‘recovery time’ was recorded as 180 min.
The following parameters were assessed and recorded every 10 min until recovery, measured as described above: (a) the palpebral reflex; (b) withdrawal reflex (toe pinch); (c) response to noise; (d) heart rate; (e) respiratory rate; and (f) rectal temperature. We did not succeed in eliciting a clinch response (placing an instrument against the feet pad resulting in toe flexion), which was reported in other bat species as reflecting anaesthetic depth. 19 Since the withdrawal reflex (toe pinch) is a noxious stimulus, this reflex was also used to assess analgesia. Reflexes and response to noise were measured on a scale of 0–2, where 2 indicates normal response compared to baseline, 1 indicates a weak response, and 0 indicates absence of response. Side effects including arrhythmia, irregular breathing pattern and twitching were recorded as existing (1) or not (0), regardless of severity or duration. Ten minutes after recovery a final evaluation of all parameters was performed.
Statistical analysis
Sample size estimation was based on the expected difference in duration of anaesthesia within the bat group, between the MM-Sal treatment and all other treatments that included a third drug. Assuming the average difference in time of anaesthesia between the two regimens of treatment will be 10 min, with a standard deviation of no more than 10 min, a significance level of 5% (one-tailed) and a power of 80%, a sample size of eight bats was found to be sufficient to prove that the difference is statistically significant.
Repeated measures analysis of variance (ANOVA) was used to test the effect of the anaesthesia protocol, the effect of time, and the interaction between them. Additionally, the Friedman non-parametric test was used to analyse the effect of protocol on the duration variables and to assess if there was a change in bats' body weight over the study period. The Wilcoxon paired signed rank test was used to test the differences between pairs of measurements. The McNemar test was used to assess the change in the dichotomous variables between pairs of anaesthesia protocols. Significance was set at p ≤ 0.05.
Results
Mean ± standard deviation (SD) of the time periods in minutes (see definitions in Methods) following subcutaneous injection of five medetomidine-midazolam based combinations with: saline (MM-Sal), ketamine (MM-Ket), fentanyl (MM-Fen), morphine (MM-Mor), or butorphanol (MM-But) in eight Egyptian fruit bats.
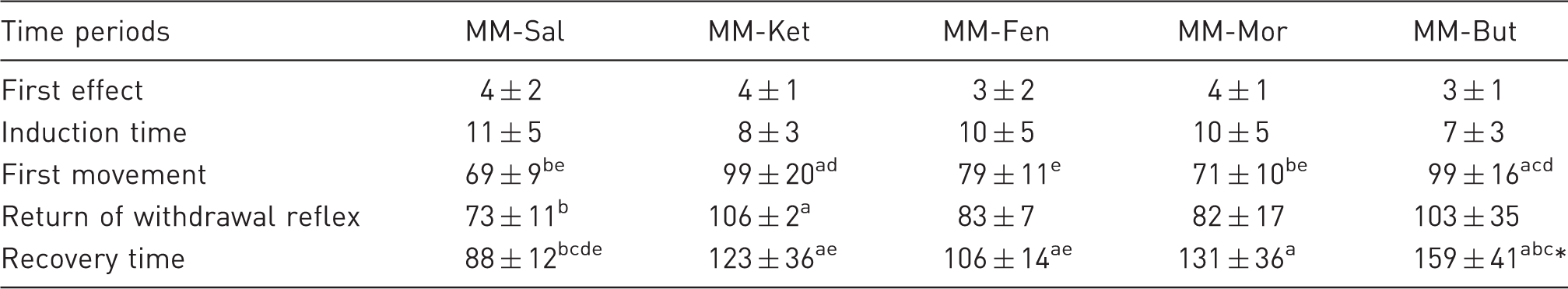
Significant difference between the indicated treatment and MM-Sal (p ≤ 0.05).
Significant difference between the indicated treatment and MM-Ket (p ≤ 0.05).
Significant difference between the indicated treatment and MM-Fen (p ≤ 0.05).
Significant difference between the indicated treatment and MM-Mor (p ≤ 0.05).
Significant difference between the indicated treatment and MM-But (p ≤ 0.05).
In four out of eight bats, atipamezole was administered at 180 min, and recovery time was recorded as 180 min.
The number of bats from each treatment (percentage) that showed side effects during anaesthesia following subcutaneous injection of five medetomidine-midazolam based combinations with: saline (MM-Sal), ketamine (MM-Ket), fentanyl (MM-Fen), morphine (MM-Mor), or butorphanol (MM-But) in eight Egyptian fruit bats.
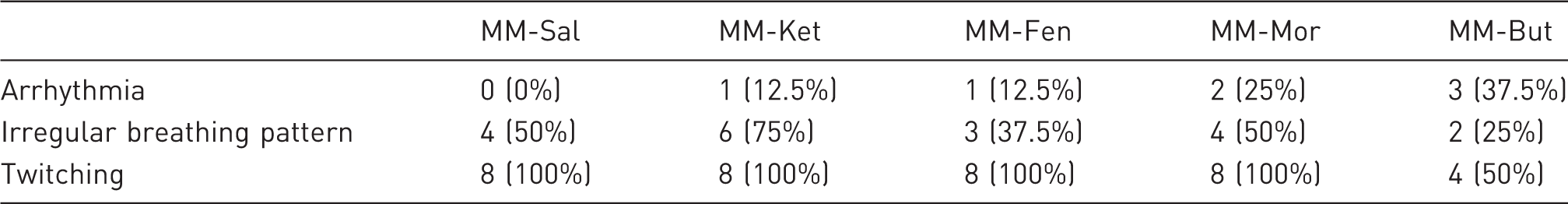
Mean heart rate (Figure 1) and respiratory rate (Figure 2) were compared for the first 60 min following injection (on further time-points some of the bats had already recovered). Following all treatments the heart rate dropped significantly from an estimation of 400 beats per minute (bpm) to approximately 200 bpm within 20 min from injection (p < 0.001; Figure 1), with no significant difference between treatments. Arrhythmia was detected in some bats in all treatments except in the MM-Sal (Table 2). Heart rate increased concurrently with regaining of withdrawal reflex and response to noise, reaching pre-anaesthetic values after recovery.
Mean ± SD (error bars) of heart rate (beats per minute; bpm) during the first 60 min following subcutaneous injection of five medetomidine-midazolam based combinations with: saline (MM-Sal), ketamine (MM-Ket), fentanyl (MM-Fen), morphine (MM-Mor), or butorphanol (MM-But) in eight Egyptian fruit bats. Mean ± SD (error bars) of respiratory rate (respirations per minute; rpm) during the first 60 min following subcutaneous injection of five medetomidine-midazolam based combinations with: saline (MM-Sal), ketamine (MM-Ket), fentanyl (MM-Fen), morphine (MM-Mor), or butorphanol (MM-But) in eight Egyptian fruit bats.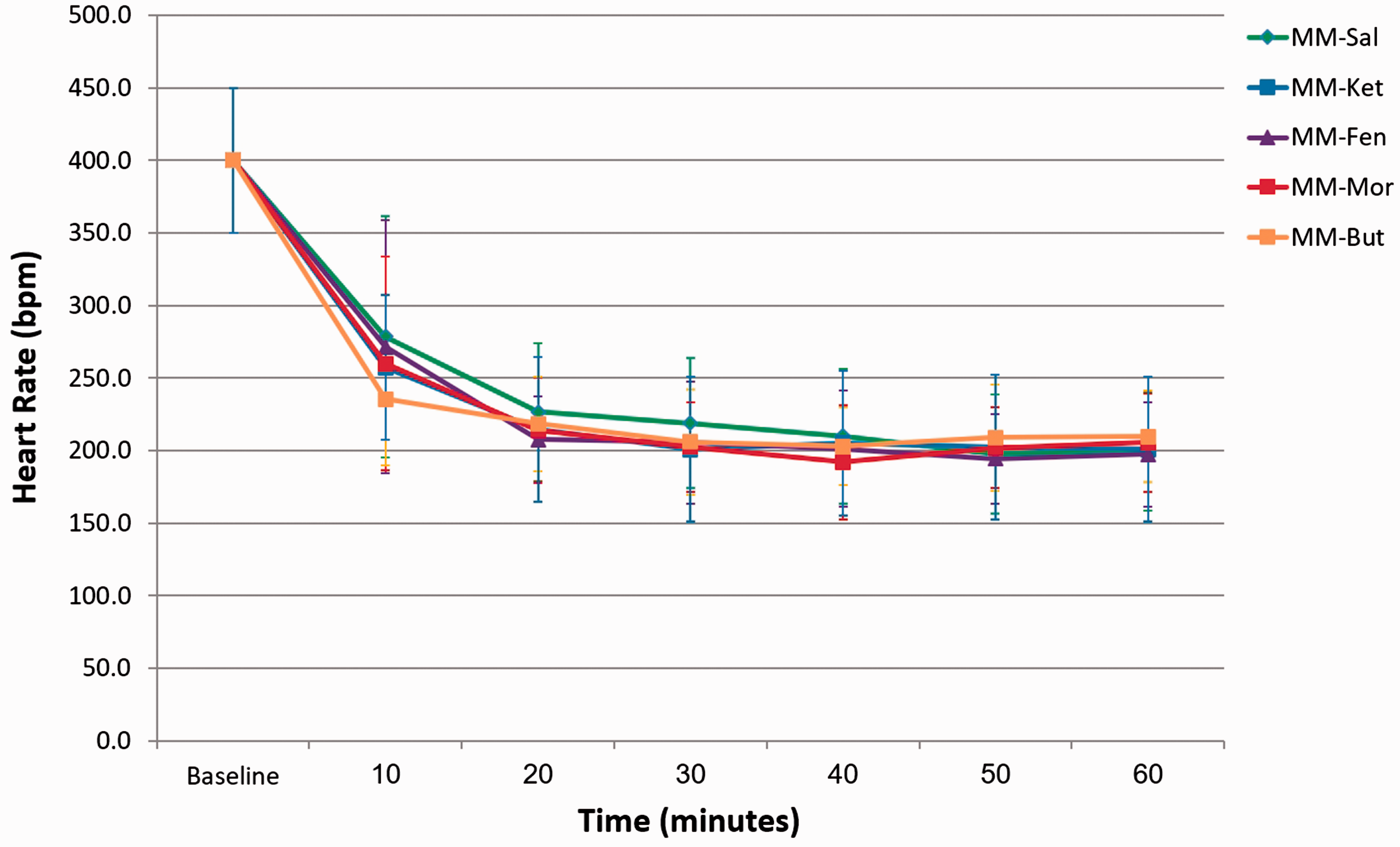
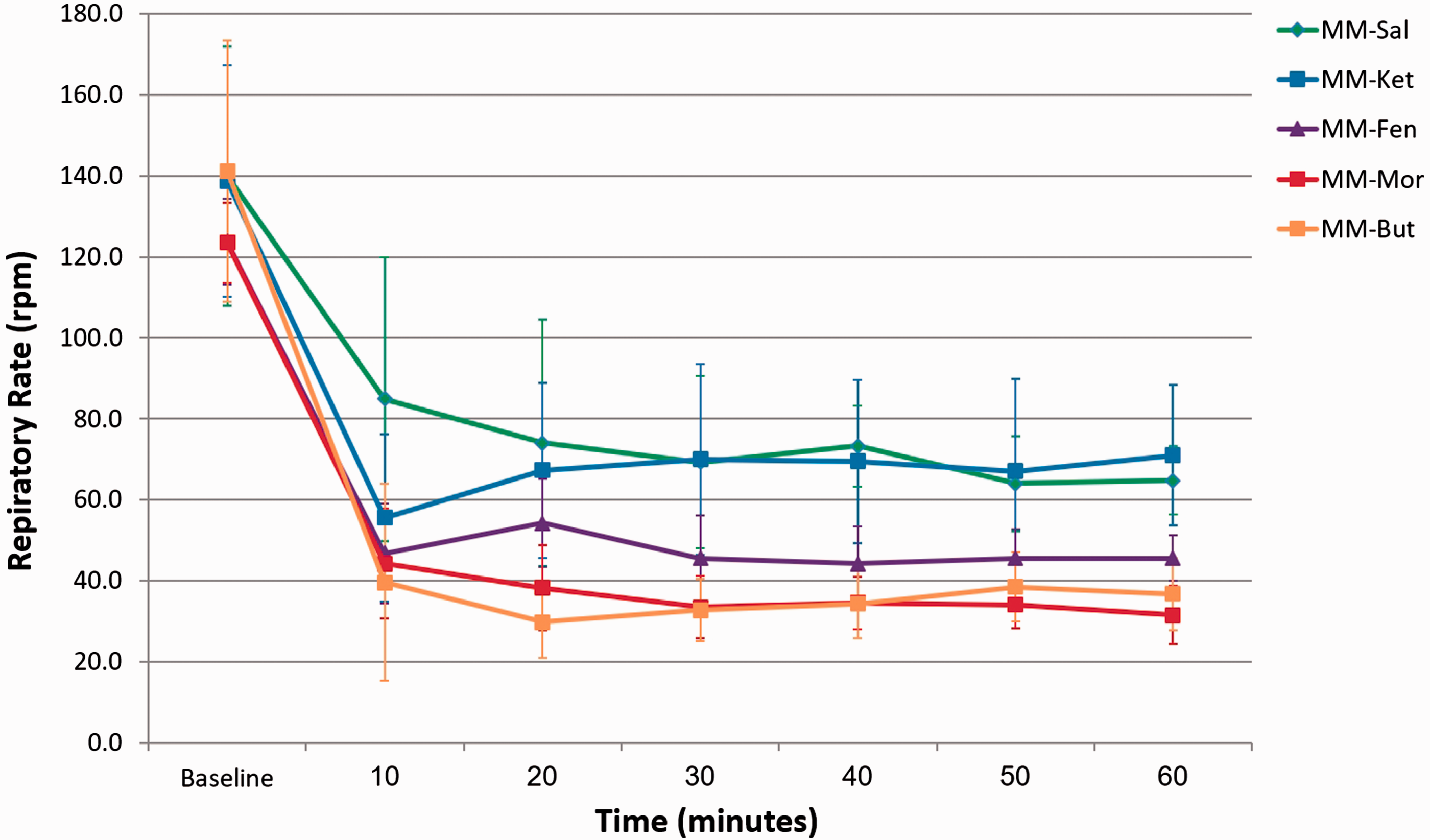
Respiratory rate decreased significantly from approximately 120–140 respirations per minute (rpm) to 36–65 rpm within 20 min following injection (p < 0.001; Figure 2). Significant differences in pooled respiratory rates (10–60 min) were observed between protocols: MM-Sal had significantly higher respiratory rate than MM-Fen (p = 0.03), MM-Mor (p = 0.002), and MM-But (p = 0.02); and MM-Ket had significantly higher respiratory rate than MM-Mor (p = 0.019). Irregular breathing pattern was observed for short periods during anaesthesia in some of the bats with all treatments (Table 2). Respiratory rate increased concurrently with regaining of withdrawal reflex and response to noise, reaching baseline values after recovery.
Rectal temperatures decreased significantly in all treatments at 10 min following injections (from 39.0 ± 0.2 to 37.3 ± 0.1℃, p < 0.001; no difference between treatments, p = 0.88). Because the bats were subsequently warmed actively, comparisons during the anaesthesia period were not performed.
Spontaneous movement, mainly manifested as ear movements, appeared in all treatments prior to regaining withdrawal reflex (Table 1). Mean time to regain spontaneous movement was significantly longer in the MM-Ket and in the MM-But in comparison to the MM-Sal treatment (p = 0.015 and p = 0.004, respectively), and in comparison to the MM-Mor (p = 0.019 and p = 0.008, respectively). Time to regain movement with MM-But was also significantly longer in comparison to the MM-Fen (p = 0.029). Mean time to regain withdrawal reflex was significantly longer in the MM-Ket in comparison to MM-Sal treatment. The MM-But had a similar mean time for regaining withdrawal reflex as the MM-Ket, however, it was not significantly different from the MM-Sal (p = 0.054).
Mean recovery time was significantly shorter in the MM-Sal in comparison to all other treatments, and it was significantly longer in the MM-But in comparison to all other treatments, except for the MM-Mor (p = 0.051) (Table 1). Notably, in four out of eight bats from the MM-But treatment, recovery time was recorded as 180 min following atipamezole administration. Without this intervention, recovery time would have been even longer. These bats were administered a mean of 0.3 mg/kg atipamezole (range 0.27–0.35), and subsequently started flying within 1–8 min.
During the evaluation at 10 min following recovery, all parameters returned to baseline values, except for the temperatures, which were significantly lower than baseline (37.6 ± 0.6℃, p < 0.001; no difference between treatments, p = 0.30).
Mean body weight of the eight bats showed no significant changes throughout the study period (p = 0.094). The bats were under continuous medical observation throughout the study period and at least six months following the end of the study, with no perianaesthetic morbidity or mortality observed.
Discussion
All five medetomidine-midazolam based combinations in the present study induced anaesthesia for varying durations. The subcutaneous route of anaesthetic administration was selected in this study because intravenous access is more difficult to obtain in conscious bats and may result in severe handling stress or injury. Likewise, the intramuscular route was not used because bats have small muscle mass, and this route of administration is considered to be more painful and stressful than the subcutaneous injection. Another reason to avoid intramuscular injections was the concern that muscle soreness post-injection may impair flight, thus prolonging recovery from anaesthesia. Drug absorption rates are considered to be lower with subcutaneous administration, and this may have affected the induction time or anaesthetic depth in our study. However, no such differences were reported in a recent study comparing administration routes of MM-But in mice. 40
Twitching of wings, legs, or generalized twitching that included ears and eyelids was common with all protocols. Twitching usually started shortly after the first signs of ataxia, and generally lasted for approximately 20 min, until the bats reached a deeper anaesthetic plane. This side-effect may be specific to Egyptian fruit bats, as it was not reported in other species under MM-Fen or MM-But anaesthesia,24,41,42 although muscle twitching following sedation with medetomidine was described in dogs and cats. 43 Wing flapping was reported in bats in response to ketamine administration, 13 and may be similar to the phenomenon that we described as twitching.
Baseline vital signs (heart rate estimation of 400 bpm, respiratory rate above 120 rpm and temperature above 39℃) may reflect high stress levels during physical examination, as expected in wild animals. In studies that monitored heart rate and temperature telemetrically in resting Egyptian fruit bats, heart rates of 226 ± 28 bpm and temperatures of 36.5 ± 1.17℃ were reported.44,45 We found no published resting respiratory rates for Egyptian fruit bats, but estimate that observed baseline respiratory rates were also higher than normal due to handling stress. Telemetric methods should be considered whenever possible to remove the likelihood of biased baseline results.
Under all five treatments, heart rate decreased significantly from baseline to values of approximately 200 bpm, which is lower than the published resting heart rate. Medetomidine, an alpha2-adrenoreceptor agonist, which was included in all protocols, tends to produce initial peripheral vasoconstriction, resulting in reflex bradycardia. Later, centrally decreased sympathetic tone preserves this effect. 43 Since opioids may potentiate bradycardia caused by alpha2-agonists by increasing parasympathetic tone, 32 we expected to see lower heart rates in the three protocols that included opioids, but no such differences were observed. Similar findings were shown in dogs and sheep,46,47 where heart rates decreased following administration of dexmedetomidine alone, or dexmedetomidine in combination with different opioids, with no difference between treatments.
Respiratory depression occurred under all protocols, 48 with the three opioid combinations producing lower respiratory rates than MM-Ket and MM-Sal. This finding was consistent with the literature, as opioids are known to produce dose-dependent depression of ventilation, 32 while ketamine, when administered as a single agent, usually has minimal effects on the respiratory system. 32 Respiratory depression under opioids is known to be greater with pure mu-agonist opioids,32,49 than with butorphanol, an agonist-antagonist. 32 Surprisingly, in the present study there was no difference in respiratory rate between the pure mu-agonists combinations, with morphine or fentanyl, and the butorphanol combination. Respiratory depression is a common effect of sedative and anaesthetic drugs, and may induce hypoventilation, affecting partial pressure of carbon dioxide (PaCO2), partial pressure of oxygen (PaO2), and acid–base balance. Because blood gas analysis was not available during this study, and these parameters were not monitored, oxygen was administered throughout the anaesthesia period, as oxygen administration was reported to improve arterial oxygenation during anaesthesia in other species.50,51
Temperature dropped significantly 10 min after injection of the anaesthetics. The high baseline temperatures can probably be attributed to stress, and some decrease following induction of anaesthesia was expected. However, it is known that homeostatic mechanisms that regulate body temperature are depressed during anaesthesia, and severe hypothermia may develop. 39 This is of special concern in bats, as they have a high ratio of surface area to body mass due to their small size, which is further increased by their large wings. 18 To prevent hypothermia, bats were actively warmed during this study.
Recovery time was shortest with MM-Sal, while the longest duration to recovery was with MM-But, and would have been longer had we not administered atipamezole at 180 min in four out of eight bats. However, the withdrawal reflex and voluntary movements recovered much earlier with the MM-But, at similar times as the MM-Ket. These findings suggest that the MM-But produces anaesthesia of similar duration to MM-Ket, but recovery is prolonged. Therefore, administration of atipamezole following the use of MM-But in Egyptian fruit bats is recommended.
The primary motivation to perform this study was to develop injectable anaesthesia protocols suitable for surgeries. General anaesthesia is defined by the American Society of Anesthesiologists (ASA) as a drug induced loss of consciousness during which patients are not arousable, even by painful stimulation. 52 Unconsciousness is difficult to assess in immobilized animals, thus the loss of withdrawal reflex (toe pinch), a noxious stimulus, is commonly used to indicate a state of surgical anaesthesia.17,53 Although medetomidine, midazolam, and opioids are classified as sedatives and not anaesthetics, combinations of medetomidine-midazolam-opioid have been reported to produce surgical anaesthesia in several animal species.17,28,54,55 Moreover, these combinations were used for conducting surgical procedures in experimental settings.25,29–31 In the present study, all five combinations induced loss of withdrawal reflex for periods of over an hour, indicating their effectiveness in inducing anaesthesia. MM-Sal also induced loss of withdrawal reflex, although for a shorter duration than all other combinations. It was reported previously that high doses of medetomidine alone can induce surgical anaesthesia in dogs. 56 Despite this finding, we do not recommend the use of medetomidine-midazolam for surgical procedures. In our Egyptian fruit bats facility, surgical procedures lasting 4–6 h are conducted under MM-Fen anaesthesia, with repeated injections administered every 45–60 min, providing an adequate surgical plane of anaesthesia and analgesia. 11
The present study has several limitations: baseline measurements of vital signs should be performed telemetrically, and additional measurements, such as electrocardiogram, blood pressure, and blood gas analysis (especially PaO2, PaCO2, and pH), should be performed for better assessment of perfusion, oxygenation, ventilation, and acid–base status. An assessment of muscle tone should also ideally be performed. In the course of the study we noticed a marked decrease in penile tone as the bats reached deeper planes of anaesthesia. Correlation of this relaxation with other indicators should be examined further. Penile tone may prove to be a valuable indicator of anaesthetic depth in male fruit bats. Female bats should be studied too, because they could potentially respond differently to the anaesthetic protocols, as was reported in other species. 42
In conclusion, all five examined protocols in this study induced anaesthesia in adult male bats with no apparent morbidity or mortality, and could potentially replace inhalation anaesthesia. Choice of optimal combination would depend on the exact procedure and setting. For non-painful procedures, the MM-Ket is a good choice, particularly when oxygen is not available, as it caused minimal respiratory depression. For painful surgical procedures, the opioid combinations are more suitable, as they offer good analgesia and can be fully antagonized in emergency cases.
Footnotes
Acknowledgements
The results of this investigation were presented at FELASA Congress 2016, and published in the proceedings of the conference. We thank Dr. Annette Denziger and Prof. Dr. Lutz Wiegrebe for originally introducing the authors to MM-Fen anaesthesia in bats. The authors would like to thank, Tali Bdolah-Abram for her help with statistical analysis, Prof. Alon Harmelin for critical reading of the manuscript, Ruth Brandt for editing recommendations, Shaul Yadai for bat caretaking and Prof. Nachum Ulanovsky for providing the facilities and bats for this study.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
