Abstract
In accordance with the ‘refinement’ component of the 3Rs, the primary aim of this study was to investigate and compare ketamine + medetomidine (KM) and s-ketamine + medetomidine (SKM) anaesthetic protocols in C57BL/6J mice (both sexes). We sought to determine whether s-ketamine could provide adequate surgical tolerance at a 50% dose relative to that of ketamine racemate and whether antagonism of medetomidine could be initiated 15 min earlier. The second aim was to investigate the potential improvement in analgesia for both anaesthetic protocols by adding butorphanol or metamizole. Analgesia was tested via the pedal withdrawal reaction (PWR) to a painful stimulus. During anaesthesia, respiratory frequency, pulse oximetry, body temperature and PWR were monitored. Among the 16 mice in each group, the PWR was lost in all the KM + metamizole (35:56 ± 6:07 min), KM + butorphanol (43:45 ± 2:14 min) and SKM + butorphanol (24:03 ± 5:50 min) mice, 15 of the non-premedicated KM (37:00 ± 8:11 min) mice, and 9 of the pure SKM (20:00 ± 4:19 min) mice; the latter group increased to 11 mice (17:16 ± 5:10 min) with premedication of metamizole. In contrast to the racemic combination, s-ketamine at the dose used here did not lead to sufficient loss of the PWR. However, earlier partial antagonism of SKM resulted in a slightly shorter and qualitatively better recovery than later partial antagonism of SKM. The addition of metamizole or butorphanol to KM or SKM anaesthesia positively influences the analgesic quality. However, when butorphanol is added, controlled ventilation may be necessary, especially for male mice.
Introduction
To improve animal experiments in accordance with the 3R (reduction, refinement, replacement) guidelines, anaesthesia and analgesia are important factors that can be refined. The appropriate balanced anaesthetic regimen is determined based on the frequency and duration of the experiment and the painful stimuli. Mice are difficult animals to anaesthetize due to their high risk of hypothermia and hypoglycaemia1 and the large discrepancies in doses among different strains. Beside inhalant anaesthetics only few injectable combinations can be recommended for mice. Ketamine + α2-adrenoceptor agonist combinations are commonly used, but large variations in analgesic depth and duration among individuals have been reported,2–7 which makes standardization difficult and poses a risk of insufficient analgesia. Therefore, we sought to improve the recovery time and the quality of analgesia for surgical interventions. We compared ketamine + medetomidine (KM) and s-ketamine + medetomidine (SKM) in C57BL/6J mice. Each protocol was also evaluated with the addition of the analgesics butorphanol and metamizole. Butorphanol acts as an agonist mainly at κ-opioid receptors and as a partial agonist or antagonist at μ-opioid receptors. 8 Therefore, butorphanol has no addictive potency and is officially a non-restricted narcotic drug.7,9 The pyrazolone derivate metamizole (syn. dipyrone) is a nonsteroidal antipyretic analgesic. Although the complete mechanisms of action of metamizole have not been fully clarified, metamizole has been shown to inhibit NDMA receptors (similar to ketamine) and cyclooxygenase and act as an antagonist of TRPA1 channels.10,11,12 S-ketamine is used at a 50% lower dose than racemic ketamine, as previously described in hamsters, 13 cats 14 and humans. 15 Therefore, we investigated whether such s-ketamine doses would also achieve an adequate anaesthetic depth in mice. Furthermore, recovery was evaluated after antagonism of medetomidine at the earliest possible time point. For racemic KM, the earliest antagonism time point was after 40 min,16 without intense ketamine-induced catalepsy. When using s-ketamine, partial antagonism can be performed even earlier, as reported in hamsters. 17 We additionally investigated both sexes independently, as notable sex differences related to the metabolism of these drugs have been reported.4,6,18
Material and methods
Ethics statement
This study was performed in accordance with the German Animal Welfare Act (Deutsches Tierschutzgesetz) as well as Directive 2010/63/EU and approved by the local ethics committee of the approving authority, the district government of Upper Bavaria in Germany (Gz.:55.2-1-54-2532-186-2015). The acute and cumulative stress level was categorized as low for the current study.
Animals
In each group, eight male (weighing 30.4 ± 3.0 g) and eight female (weighing 23.4 ± 1.7 g) C57BL/6J mice (Charles River Laboratories, Sulzfeld, Germany) aged 15–20 weeks were included. The mice were housed under specific-pathogen-free (SPF) conditions in accordance with the ‘Federation of Laboratory Animal Science Associations' (FELASA) recommendations. 19 To reduce the number of animals used, each mouse was randomly allocated to two different anaesthetic protocols with a wash-out period of two weeks between experiments. In total, 48 mice (24 male, 24 female) were used in this study, which equals a reduction of experimental mice by half. Furthermore, each mouse was allowed an adaptation period of at least one week before the first intervention. After the end of the study and a convalescence period of at least three weeks, the mice were reused in a final low-stress experiment, with permission from our approving authority.
Mice were group housed (2–5 mice per cage, 435.7 cm2 floor area) in individually ventilated cages (cage type 9, Maxi-Miser Thoren-Caging-System, Thoren Caging Systems Inc., Pennsylvania, USA), in accordance with the European guidelines (2010/63/EU), on special wood bedding (Select Fine, Sniff, Germany). Autoclaved standard rodent diet (Altomin No. 1324SP, Lage, Germany) and water were given ad libitum. Autoclaved cellulose, aspen bricks, and nestlets (Bioscape, Castrop-Rauxel, Germany) and a red polycarbonate house (Bioscape, Castrop-Rauxel, Germany) were provided for enrichment. The room temperature was maintained at 22 ± 2℃ and a humidity of 45–60%. Animals were maintained under a 12/12 h light/dark cycle with 30 min of dawn and dusk.
Drugs and methods
Mice were not fasted before anaesthesia. After weighing and visual examination, either pre-emptive analgesia was given before intraperitoneal (IP) anaesthesia or (in the groups without additional analgesia) anaesthesia was directly administered by IP injection.
Sixteen mice (8 male, 8 female) each were assigned to the following groups:
KM: ketamine (100 mg/kg; Narketan 100 mg/ml, Vétoquinol GmbH, Ravensburg, Germany) and medetomidine (1 mg/kg) mixed in one syringe. SKM: S-ketamine (50 mg/kg; Ketanest-S 25 mg/ml, Pfizer Pharma PFE GmbH, Berlin, Germany) and medetomidine (1 mg/kg) mixed in one syringe. KM + butorphanol: butorphanol (2.5 mg/kg; Alvegesic® vet. 10 mg/ml, Selectavet, Weyarn-Holzolling, Germany) subcutaneously (SC) 30 min before ketamine (100 mg/kg) and medetomidine (1 mg/kg) administration. KM + metamizole: metamizole (150 mg/kg; metamizole 500 mg/ml, Hexal AG, Holzkirchen, Germany) orally (PO) by licking a drop containing the appropriate dose directly from a 1 ml syringe. The awake mouse was therefore briefly restrained 30 min before ketamine (100 mg/kg) and medetomidine (1 mg/kg) administration. SKM + butorphanol: butorphanol (2.5 mg/kg) SC 30 min before s-ketamine (50 mg/kg) and medetomidine (1 mg/kg) administration. SKM + metamizole: metamizole (150 mg/kg) PO 30 min before s-ketamine (50 mg/kg) and medetomidine (1 mg/kg) administration.
In the KM groups, medetomidine (Sedator 1 mg/ml Albrecht GmbH, Aulendorf, Germany) was antagonized after 45 min by atipamezole (5 mg/kg; Antisedan 5 mg/ml, Vétoquinol GmbH, Ravensburg, Germany) SC and in the SKM groups after 30 min.
The induction time was observed from the injection of anaesthetics until the loss of the righting reflex. The first measurement after loss of the righting reflex was defined as time point 0. Afterwards, data were recorded every 5 min until injection of the antagonist. Immediately after loss of the righting reflex, the cornea was protected with dexpanthenol eye cream (Bepanthen, Bayer Vital GmbH, Leverkusen, Germany). The recovery time was defined from the injection of the antagonist until the righting reflex was regained. Immediately before injecting the antagonist, each mouse was administered a pre-warmed infusion SC (0.2 ml Ringer's solution + 0.1 ml 20% glucose).
Monitoring of anaesthesia was performed by means of the respiratory frequency (RF; respiration counted by the investigator), heart rate (HR), peripheral oxygen saturation (SpO2) and rectal body temperature (BT) via a high-speed pulse oximeter including a temperature probe (‘PhysioSuite’ including ‘Thermostat’, Kent Scientific Corp., USA distributed by EMKA Technologies, Paris, France). Additionally, the pedal withdrawal reaction (PWR; pinching the toes with atraumatic forceps) was evaluated (positive reaction: mouse attempt to withdraw the limb; negative reaction: mouse did not react to toe pinch). Physiological BT was maintained at 38–39℃ by a heating pad (Horn GmbH, Gottmadingen, Germany), and 100% oxygen was supplied via a nose mask.
Surgical tolerance was defined as the period when the PWR to a painful stimulus was lost without any significant increase in HR or RF. In this study, the only painful stimulus was a toe pinch with atraumatic forceps to test the PWR.
Statistical analysis
The study was classified as an explorative study and the sample size was set at n = 8 in accordance to Arras et al. 2 The data were analysed using Excel and IBM SPSS Statistics, version 24 (IBM Corp., Armonk, NY, USA). All values are summarized as the mean (M) ± standard deviation (SD). ANOVA and pairwise t-tests were used to analyse the time differences in RF, HR and BT. For SpO2, the non-parametric Kruskal-Wallis and Wilcoxon tests were used. For unpaired comparisons, such as sex differences, two-sample t-tests or Mann-Whitney-U-tests were performed. All tests were two-sided with a 5% level of significance (p = 0.05).
Results
Comparison of anaesthetic stages and pedal withdrawal reaction (PWR) among groups (in mean ± standard deviation).
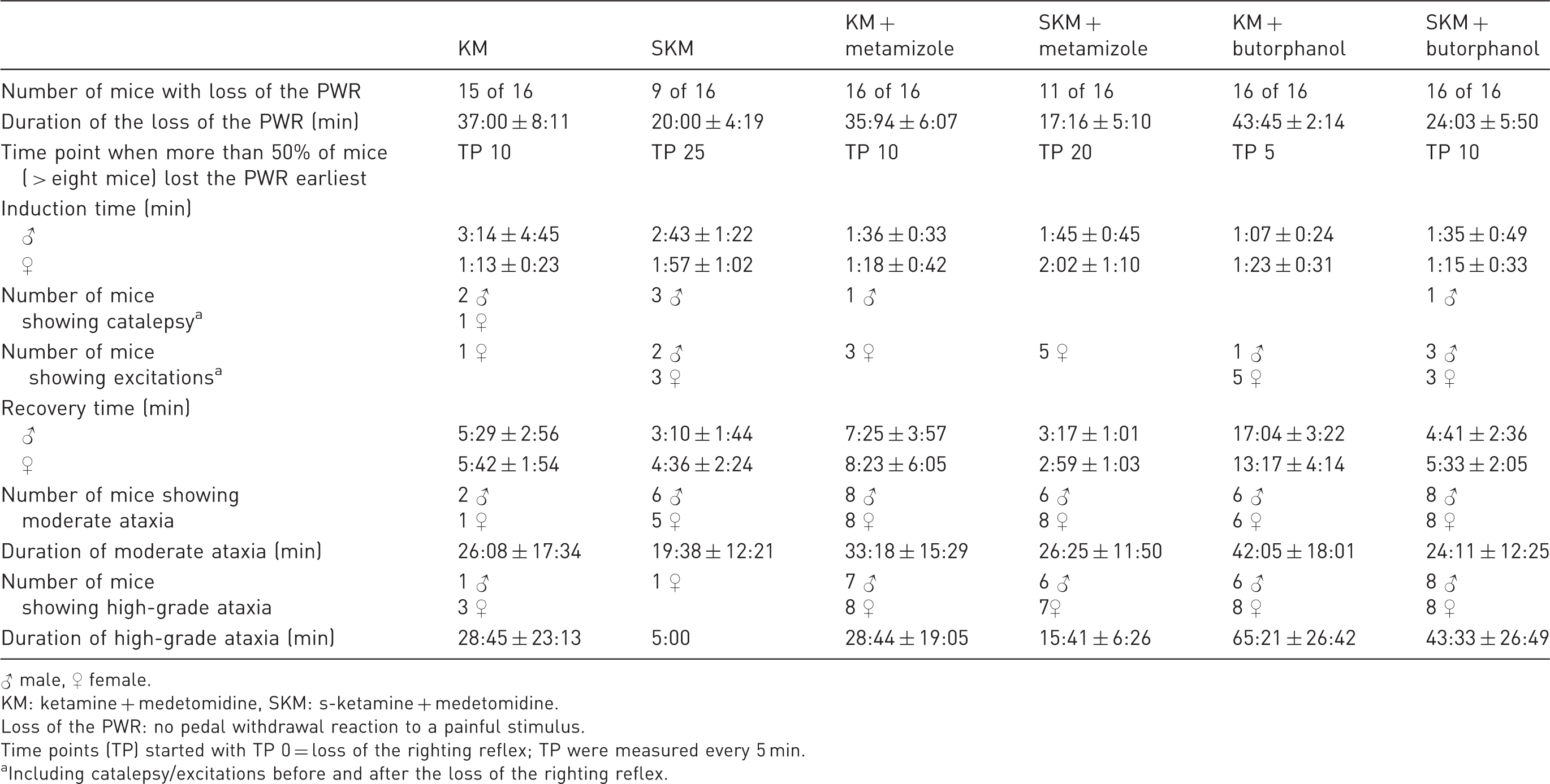
♂ male, ♀ female.
KM: ketamine + medetomidine, SKM: s-ketamine + medetomidine.
Loss of the PWR: no pedal withdrawal reaction to a painful stimulus.
Time points (TP) started with TP 0 = loss of the righting reflex; TP were measured every 5 min.
Including catalepsy/excitations before and after the loss of the righting reflex.
Comparison of heart rate during anaesthesia among groups and sex (in mean ± standard deviation).

KM: ketamine + medetomidine, SKM: s-ketamine + medetomidine; TP: time point.
Significant difference between current and former time points.
Significant difference between time point 0 and end time point.
Comparison of respiratory frequency during anaesthesia among groups and sex (in mean ± standard deviation).
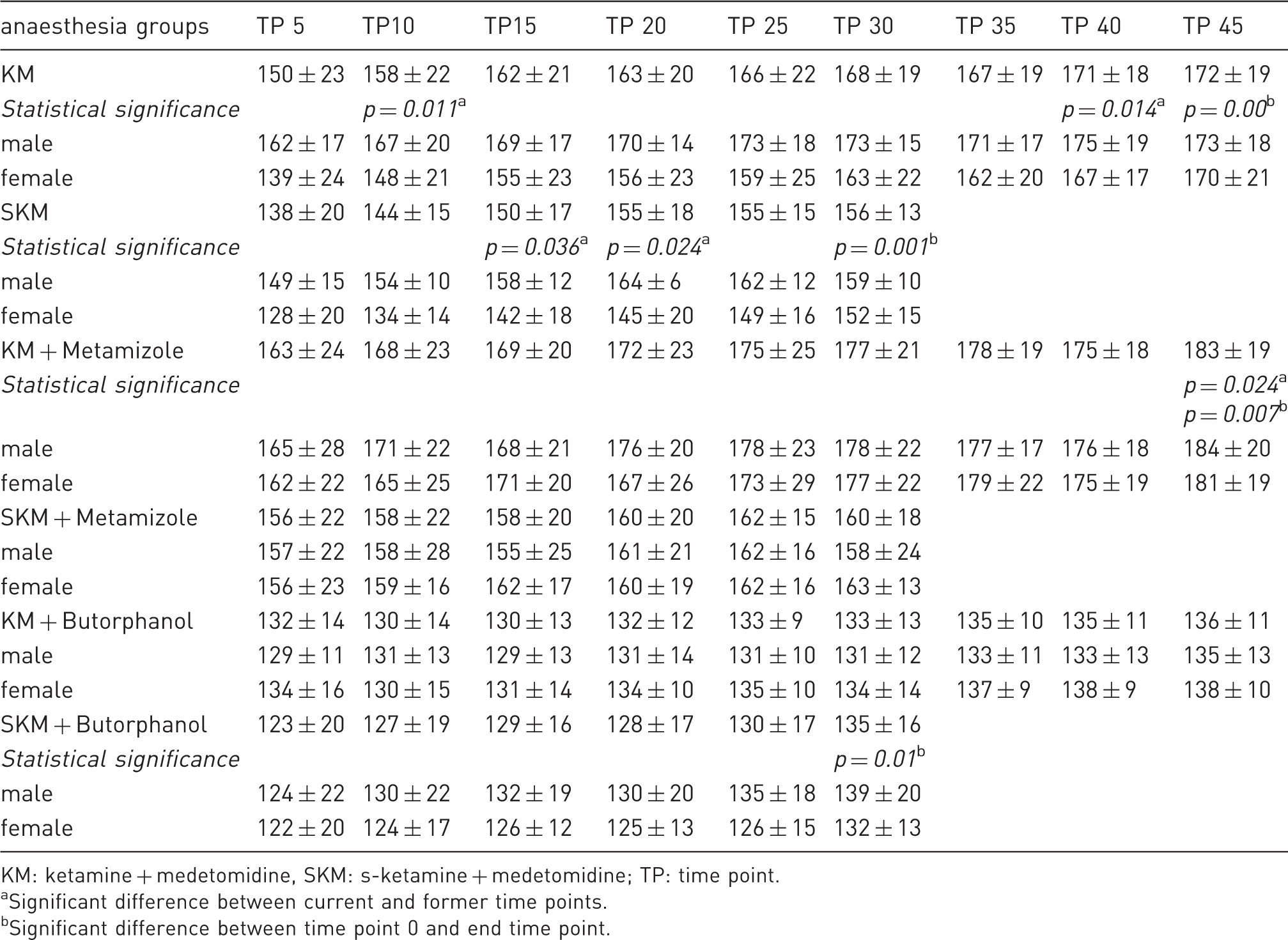
KM: ketamine + medetomidine, SKM: s-ketamine + medetomidine; TP: time point.
Significant difference between current and former time points.
Significant difference between time point 0 and end time point.
Data are not shown for SpO2 and BT, which were influenced by supplemental oxygen and heat. As expected, a massive decrease in both parameters was observed at only time point 0 (directly after induction) before external supplementation was initiated. However, the data quickly stabilized thereafter within a physiological range of 37.5–38.5℃. 7 SpO2 revealed sufficient spontaneous breathing ( > 95 vol%) in all groups. In only male mice administered KM +butorphanol, SpO2 was decreased to 93 ± 6 vol% at 40 min; additionally, rattling breathing indicative of increased bronchial secretion was observed in some of these mice.
Discussion
A refinement of anaesthetic protocols for experimental mouse studies according to the 3Rs was aimed in the current study by using s-ketamine at half of the dose of racemic ketamine in combination with medetomidine, while inducing an adequate surgical tolerance level as successfully reported in hamsters, 13 cats 14 and humans. 15 Furthermore, anaesthesia may be better controlled by earlier antagonism of medetomidine to avoid a potential ketamine hangover with intense catalepsy. 16 Additionally, analgesia for surgical procedures may be improved and prolonged by the addition of metamizole or butorphanol to basic KM or SKM anaesthesia. To test nociception during anaesthesia, we used the PWR as a reliable indicator of reaching surgical tolerance and sufficient analgesia in accordance with the studies of Arras et al. 2 and Buitrago et al. 20
The dose of ketamine (100 mg/kg) and medetomidine (1 mg/kg) was based on that used by Arras et al. 2 In contrast to that of SKM (50 mg/kg), the chosen dose of KM was sufficient to achieve a loss of PWR in nearly all of the C57BL/6J mice for a duration (37:00 ± 8:11 min) sufficient for surgical procedures, although results of KM anaesthesia were not sufficient in other studies.2,3,6,20 In contrast, the tested SKM dose was not satisfying since only nine out of 16 mice (three male, six female) lost the PWR for 20:00 ± 4:19 min. Sex differences due to drug dosages in another mouse strain have previously been reported by Cruz et al., 4 whom, in contrast to our findings, observed that female Swiss Webster mice needed 55% more ketamine than male mice. Catalepsy during induction occurred in only a few male mice and one female mouse, whereas excitations were observed more often in female mice. Ataxia was observed in all groups after the righting reflex was regained, as also reported by the literature,21,22 and lasted longer in the KM group than in the SKM group, which was already known in hamsters 13 and mice. 21 Ataxia lasted for at most 2 h, and after this time, the mice showed normal locomotion and behaviour. This finding concurs with those of a study by Hjálmarsdottir. 22 High-grade ataxia was more prevalent in female mice. This could have been caused by the ketamine which induces catalepsy by increasing the tonus of skeletal muscles, 7 and because of the persistent activity of the muscles due to increased action in some subcortical regions, 10 spontaneous limb movements can occur. 7 This finding is also in accordance with that from Cesarovic et al., 23 in which only s-ketamine was given as premedication, and all mice exhibited ataxia and tremor during induction. Furthermore, mice are a highly reflex-oriented species. 22 Thus, reflex activity can persist while the dose of narcotics cannot be further increased without risk of death. 24 Caused by the partial antagonization of medetomidine, ataxia may be attribute to the described ketamine effect. In the s-ketamine protocols, the lower dose of s-ketamine may be responsible for the shorter duration of ataxia.
Sex differences were also observed during induction with SKM and KM, in which unconsciousness and loss of the PWR were achieved earlier in females than in males. This result may be caused by the higher percentage of fat in male mice than in female mice, as more lipophilic ketamine 25 accumulates in fat, 26 which may lead to a delayed effect. Before additional heat supply, the male mice of the KM group had a higher BT than the female mice, as also reported by Cruz et al. 4 This difference may be due to the lower percentage of muscle in females than in males. 27 Within the SKM group, only three of the male mice, compared to six of the female mice, showed loss of the PWR over a certain time. As s-ketamine is the right-handed enantiomer of racemic ketamine, 15 this sex difference may be caused by possible differences in the enzymatic system of hepatic metabolism. 26 The slight, continuous increase in RF over the course of anaesthesia, especially in the KM, SKM, KM + metamizole and SKM + butorphanol groups, may be due to increased BT, pain, insufficient anaesthetic level, hypoxia or hypercapnia. Due to the application of an external heat supply, the BT slowly increased during the course of anaesthesia to a physiological level but not to hyperthermia. We can exclude pain and an insufficient anaesthesia level as reasons for the respiratory depression, as the mice simultaneously exhibited loss of the PWR. Hypoxia was not observed by pulse oximetry, except in some males of the KM + butorphanol group, in which light hypoxia (SpO2 < 95 vol%) was detected. Regarding hypercapnia, spontaneous respiration during anaesthesia may have led to sufficient but reduced gas exchange, as breathing became shallower. A compensatory reaction would have increased the RF.
The considerable decrease in HR in the SKM and KM groups followed by a continuous increase in HR after 15 min of anaesthesia may be caused by the phencyclidines and α2-agonists. Ketamine likely activates the central sympatho-adrenergic system and depresses the reuptake of noradrenaline at the synaptic junction. Due to these effects, stimulation of the cardiovascular system follows, as indicated by tachycardia and hypertension. 7 Medetomidine typically causes bradycardia, and within the first few minutes, peripheral vasoconstriction results in an increase in blood pressure due to increased peripheral vessel resistance. 7 As blood pressure was not monitored in the current study, we can only assume the simultaneous regulation of HR and blood pressure.
Overall, males showed higher RF and HR than females, except the female mice of the KM, SKM, KM + metamizole and KM + butorphanol groups, which initially had a higher HR at time point 0 than the male mice. This may be due to stress, which affects HR29 and RF. As conscious female mice of these groups showed a higher level of activity and defence reactions than male mice, the higher initial HR may be caused by their higher stress level during handling for drug administration. Hence, the induction time was very fast, and the adaptation of HR to anaesthesia in female mice may have taken longer than that in male mice. In accordance with the study by Xing et al., 30 the higher HR in male C57BL/6J mice after administration of anaesthesia may be influenced by their genetic background. Flandre et al. 31 previously reported a higher RF in male mice than in female mice, which may have been caused by differences in hormones. 32 When using SKM, inspiratory breathing was deeper and more effective in females than in males, as females regained physiological SpO2 immediately after receiving an oxygen supply, whereas males regained physiological SpO2 only after 15 min, despite their higher RF. Additionally, according to Voipio et al., 6 females take longer to regain the righting reflex during recovery than males, which may be caused by different hepatic metabolism of (s-)ketamine between males and females. 26
For ketamine + α2-agonist combinations, the depth and duration of surgical tolerance vary widely among individuals.2,3,5,6,20,21 Therefore, we investigated two additional analgesics that are commonly used in veterinary practice and are useful for treating moderate pain. Depending on the painful stimulus of the experiment, more potent analgesics could be necessary. Butorphanol, an opiate agonist-antagonist, 7 was recommended for use combined with dexmedetomidine + tiletamine + zolazepam in mice by Cagle et al. 33 and combined with medetomidine + midazolam by Kawai et al. 34 As shown in other species35,36 and in mice,37,38 metamizole is a potent analgesic and can reduce the required dose of other anaesthetics when administered in combination. The doses of butorphanol and metamizole were chosen in accordance with Erhardt et al. 7
With the exception of the SKM + metamizole group, the PWR was lost in all groups with additional administration of analgesia. However, compared to pure SKM treatment, metamizole premedication was able to increase the percentage of mice that lost the PWR from 56 to 69%. Anaesthesia was induced more quickly with premedication in male mice as well as in female mice of the SKM + butorphanol group than without premedication (see Table 1), which can be explained by the sedative effect of butorphanol 7 and by the antagonistic effect of metamizole on NMDA receptors of the central nervous system.11,39 The combination of KM + butorphanol induced the best analgesic effect. However, the addition of butorphanol to KM led to more side effects, especially short-term mild to moderate excitation. Opioids are able to reduce the threshold of excitation in the central nervous system as they are weak partial µ-agonists.7,40 In contrast, all four premedicated groups showed less catalepsy than the groups without premedication, as both butorphanol 41 and metamizole may reduce the high skeletal muscle tonus caused by (s-)ketamine. As metamizole is known to induce spasmolytic action in smooth muscles, supposedly by inhibiting the release of intracellular Ca2+,7,42 it may also influence skeletal muscle when administered in combination with α2-agonists, as not all mechanisms of action have been identified thus far. In the premedicated KM groups, both metamizole and especially butorphanol clearly prolonged the time to regaining the righting reflex after antagonism, which is caused by the slight sedative effect of butorphanol 33 that was unaffected by atipamezole antagonism 9 and by the dose-dependent sedative effect of metamizole. 43 The mean RF, HR and SpO2 were lower in the butorphanol groups directly after induction than in the metamizole groups. In contrast to basic KM and SKM, all groups that received premedication started with a markedly lower HR at time point 0, but the HR increased significantly during the anaesthetic period (p-values are shown in Table 3). The opioid analgesic butorphanol produces a sedative effect on the central nervous system and depresses breathing by affecting µ-opioid receptors25,40 and circulation (blood pressure and HR). Metamizole is known to decrease blood pressure after fast intravenous injection. 7 In this study, metamizole was given orally. As no primary decrease in HR was observed directly after induction in the metamizole groups, orally administered metamizole does not seem to notably influence the cardiovascular system. This observation may be due to the influence of stomach content on the onset of oral medication, 45 as rodents do not need to be fasted prior to anaesthetic induction. 44 Supplementation of 100% oxygen under spontaneous breathing was sufficient in all mice except male mice of the KM + butorphanol group beginning at time point 40, at which time these mice reached only 93 ± 6 vol%, and some exhibited the rattling breathing characteristic of increased bronchial secretion. Massive bronchial secretion during recovery was observed in 10 out of 16 male mice and in half of the female mice of both groups premedicated with butorphanol. Ketamine induces salivary and bronchial secretion,10,44 which appears to be intensified by the addition of butorphanol, especially in males. Whether premedication with atropine, as recommended by Flecknell, 44 is able to reduce this bronchial secretion needs to be examined in further studies. Additionally, the male mice of the KM + butorphanol group required the longest recovery period. As male mice have a higher percentage of fat, the extended recovery may be caused by the lipophilic property of butorphanol, 46 which may possibly allow butorphanol to accumulate in the body.
The noticeable difference in sex in all premedicated groups has been previously shown for basic anaesthesia, as the mean HR in male mice is consistently higher than that in female mice. Xing et al. 30 also reported a higher HR in male C57BL/6J mice than in female mice. A key reason for this sex difference, aside from an age difference, 30 is the contrasting hormone activity, which affects the cardiovascular system by modulating ventricular repolarization. 47
In summary, our aims were not completely achieved, as the PWR was not lost in all mice under the s-ketamine protocol, which has also been shown in dogs. 48 Therefore, more detailed dose-evaluation studies on the SKM combination are necessary. However, earlier antagonism of medetomidine in combination with s-ketamine resulted in a qualitatively better and shorter recovery. Improvements in analgesia were observed following the use of (s-)ketamine + medetomidine protocols with the addition of either butorphanol or metamizole. The addition of butorphanol to the basic anaesthetic protocols markedly increased the mean duration for which the PWR was absent. The addition of metamizole or butorphanol to SKM increased the number of mice that exhibited a loss of the PWR. Premedication, especially by pre-emptively administered butorphanol, allowed a notably shorter anaesthesia induction time than that without premedication, especially in males, and decreased ketamine-based catalepsy. However, the excitation rate during induction increased when using the KM-based protocols. A disadvantage of adding butorphanol to KM was the development of insufficient respiration and massive bronchial secretion in male mice after 40 min. Therefore, equipment for intubation and mechanical ventilation should be available when using this combination. Regarding recovery, butorphanol had a longer lasting sedative effect than metamizole, and ataxia was more pronounced. A dose reduction should be considered when using butorphanol in this mouse strain. No disadvantage was observed following the use of metamizole, and pre-operative application of this analgesic in combination with KM and SKM is therefore highly recommended.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
