Abstract
Pig experiments often require anaesthesia, and a rapid stress-free induction is desired. Induction drugs may alter the subsequent anaesthesia. Therefore, the aim of the present study was to compare, in pigs, the effects of two different injectable anaesthetic techniques on the induction and on the physiological variables in a subsequent eight hours of total intravenous anaesthesia (TIVA). Twelve domestic castrates (Swedish Landrace/Yorkshire) 27‒31 kg were used. The pigs were randomly assigned to different induction drug combinations of zolazepam–tiletamine and medetomidine intramuscularly (ZTMe) or midazolam, ketamine intramuscularly and fentanyl intravenously (MiKF). Time from injection to unconsciousness was recorded and the ease of endotracheal intubation assessed. The TIVA infusion rate was adjusted according to the response exhibited from the nociceptive stimulus delivered by mechanically clamping the dewclaw. The time from injection to unconsciousness was briefer and intubation was easier in the ZTMe group. Results from the recorded heart rate, cardiac index and arterial blood pressure variables were satisfactorily preserved and cardiovascular function was maintained in both groups. Shivering was not observed in the ZTMe group, but was observed in four of the pigs in the MiKF group. The requirement of TIVA was lower in the ZTMe group. In conclusion, ZTMe had better results than MiKF in areas such as shorter induction time, better intubation scoring results and less adjustment and amount of TIVA required up to six hours of anaesthesia. The results may have been due to a greater depth of anaesthesia achieved with the ZTMe combination at the dose used.
Introduction
The use of pigs as an experimental animal model when studying diseases and developing new surgical techniques for humans is increasing. In these studies, anaesthesia of the animal is usually required, and long-term anaesthesia is often needed. 1 The effects of anaesthetic and analgesic drugs differ between species 2 and may cause physiological changes, 3 thus possibly affecting the outcome and results of the research. 4
Pigs are easily stressed during their handling before anaesthesia is induced, which can interfere with the onset of action of the drugs targeting the central nervous system. 5 The ideal anaesthetic induction techniques in pigs should provide fast and reliable unconsciousness, minimal cardiovascular and respiratory depression, adequate analgesia and muscle relaxation. To reduce the discomfort for the animal, if an injection is used as induction technique, the injection of the drug should preferably be rapid. 6 In order to immediately provide an open airway after induction and easily perform endotracheal intubation, it is advantageous if salivation is reduced and the laryngeal reflexes are eliminated. 2
In pigs, it is desirable to administer anaesthetic drugs by a single intramuscular (IM) injection since venous cannulation can be stressful for the animal and difficult for the researcher if aggressive behaviour is exhibited by the pig. A butterfly needle can be placed in the muscular region located in the dorso-cranial aspect of the neck. 7 A known induction protocol for pigs used in our research laboratory is a combination of ketamine and midazolam given IM followed by an intravenous (IV) injection of an opioid before intubation. 8 It has been previously shown in a similar breed and age group of pigs that the induction of anaesthesia using a α2-agonist in combination with tiletamine–zolazepam administered IM can produce a reliable and rapid induction. 9 However, the effects of different drug combinations on the induction and the physiological variables during subsequent anaesthesia have not yet been compared.
The aim of the present study was to compare in pigs the effects of two different injectable anaesthetic techniques on the induction and on the physiological variables in a subsequent eight hours of total intravenous anaesthesia (TIVA). A specific objective was to also evaluate if the drugs used for induction affected the requirement of a predetermined drug combination during eight hours of anaesthesia.
Material and methods
Animals
The experimental protocol was approved by the Ethics Committee for Animal Experimentation, Uppsala, Sweden (Approval No. C250/09) and performed at the Department of Medical Science at Uppsala University, Sweden. The animals were treated according to the guidelines of the Swedish Board of Agriculture and the European Convention of Animal Care. Included in the study were 12 clinically healthy domestic crossbreed castrates (Swedish Landrace × Yorkshire), that were nine weeks old and weighed 27–31 kg. The pigs arrived from the breeder the same day as the study was performed. Food was withheld twelve hours before anaesthesia, but water was provided ad libitum. Upon arrival they were randomly assigned, using the R statistical program version 2.3.1, to one of two different induction drug combinations resulting in n = 6 in each group.
Anaesthesia, catheterisation and monitoring
The anaesthesia was induced while the pigs were still in the 95 cm × 37 cm × 62 cm transport crate. Using a butterfly needle (CHIRAFLEX Scalp vein set 21G × 3/4″, 0.8 × 20 mm Luer-Lock, Slovakia), the pigs received an IM (brachiocephalic muscle) injection with one of the following drug combinations.
ZTMe: zolazepam–tiletamine (Zoletil Forte® vet. 250 + 250 mg, Virbac, France) 5 mg/kg (Z 2.5 + T 2.5 mg/kg) in combination with medetomidine (Domitor® vet. 1 mg/ml, Orion Pharma AB Animal Health, Sweden) 0.05 mg/kg. MiKF: midazolam (Midazolam Actavis 5 mg/ml, Actavis AB, Sweden) 2 mg/kg, in combination with ketamine (Ketaminol® vet 100 mg/ml, Intervet AB, Sweden) 10 mg/kg followed by fentanyl (Fentanyl B. Braun 50 µg/ml B. Braun Medical AB, Sweden) 4 µg/kg IV before intubation.
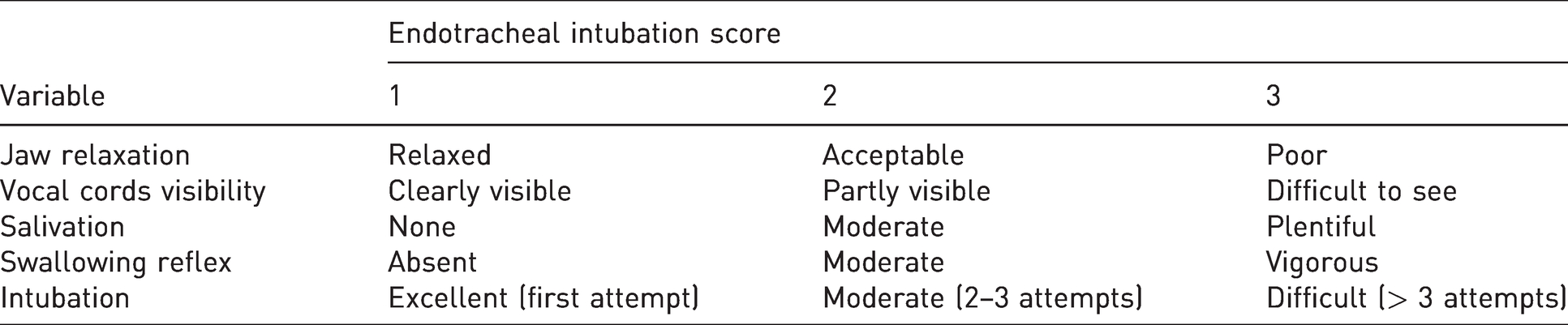
After the IM injection, the position of the pig was checked continuously, and the time to unconsciousness (sternal recumbency, head down, lack of reaction when manipulating the body, absence of palpebral reflex) was recorded. The anaesthetised pig was placed in dorsal recumbency on an operating table. A cannula (BD Venflon™ 20G × 32 mm, BD Medical, Sweden) was inserted in one of the auricular veins and the trachea was intubated with an endotracheal tube (6–8 mm ID. The endotracheal tube was connected to a ventilator (SERVOi® Anesthesia delivery system, MAQUET, USA). The pigs’ lungs were mechanically ventilated (volume control) with 40% oxygen in air, at a respiratory rate (RR) of 20 breaths/minute and a tidal volume of 7 ml/kg. The inspiratory to expiratory ratio was set to 1:2.5 and a positive-end-expiratory pressure to 0 cmH2O. The minute ventilation (MV) was adjusted to maintain arterial partial pressure of carbon dioxide (PaCO2) to 41–45 mmHg. The endotracheal intubation was performed and evaluated by a laboratory technician who was unaware of the drug combination used. Before the start of the study, an intubation scoring sheet based on a system used for humans 10 and pigs 11 was developed (Table 1).
The intubation procedure was performed according to a described textbook technique. 2 Scoring was rated from one to three, where a higher score indicated a more difficult procedure. All criteria needed to be met for each level, and if they were not, the procedure was classified one level higher.
Immediately after the intubation, TIVA was administered with a starting and base rate of 7 ml/kg/h (Baxter 6201 Volumetric Infusion Pump, Baxter, USA). The timer was set to zero and the anaesthesia infusion rate was recorded for 8 h. The TIVA mixture contained 0.015 mg/ml midazolam, 4 mg/ml ketamine and 0.5 µg/ml of fentanyl in 947 ml 0.9% Lactated Ringer’s solution (Ringer-acetate, Fresenius Kabi AB, Sweden). The starting infusion rate was midazolam 0.105 mg/kg/h, ketamine 28 mg/kg/h and fentanyl 3.5 µg/kg/h.
One catheter was placed percutaneously into the femoral artery and two catheters were inserted from an incision into the jugular vein (BD Careflow™ 3Fr 1.10 mm, 200 mm, BD Medical, Sweden). One Swan-Ganz catheter (Swan-Ganz CCO/VIP 7 Fr 110 cm Edwards Lifesciences LLC, USA) was placed using pressure monitoring and by observing the pulse pressure contour change when the catheter passed through the heart and into the pulmonary artery. One catheter (BD Careflow™ 4Fr OD 1.45 mm, 200 mm BD Medical, Sweden) was also placed in the right atrium under pressure monitoring. This allowed for the measurement of the central venous pressure (CVP), pulmonary artery blood pressure (PAP), pulmonary artery occlusion pressure (PAOP), cardiac output (CO), cardiac index (CI) and the sampling of mixed-venous blood. Thermodilution using three boluses of 10 ml ice-cold sodium chloride 0.9% injected via the Swan-Ganz catheter into the right atrium was performed to measure cardiac output. A minimum of three measurements were attained in accordance with the manufacturer's recommendations. After placement of the catheters, blood samples to measure arterial pH (pHa), PaCO2, arterial partial pressure of oxygen (PaO2), arterial oxygen saturation of haemoglobin (SaO2), mix venous oxygen saturation of haemoglobin (S
From the measurements obtained, the following calculations were made using standard equations:
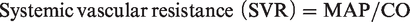
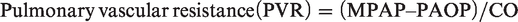
Noxious stimuli
The pigs were anaesthetised for 8 h without surgical provocation, except during the insertion of the catheters during the first 30 min. The amount of TIVA required was based on the response to a nociceptive stimulus. This was accomplished by mechanically clamping for 10 s and releasing the dewclaw three times per test interval just below the coronary band of the medial or lateral dewclaw on either the right or left pelvic limb. The site of the stimulation was changed slightly each test interval to prevent sensitisation to stimuli. 12 In total, two to three test intervals were performed on each dewclaw. The mechanical compression (10 kg), which was measured using a pinch gauge (Baseline® Lite Hydraulic Pinch Gauge, Fabrication Enterprises, USA), was created using forceps. Clamping was administered at 30-min intervals during the first 2 h and at 60-min intervals during the remaining hours of anaesthesia. When an obvious positive reaction, such as a reflex withdrawal of the leg occurred, the anaesthesia infusion rate was increased by 1 ml/kg/h. When no reaction was observed, the infusion rate was decreased by 1 ml/kg/h, but never below 7 ml/kg/h.
Statistical analysis
A repeated measures analysis using a mixed model method was used to analyse the physiological data. A mixed model approach that implemented the MIXED procedure of the SAS (SAS Institute Inc. (2017): Cary, NC, SAS Institute Inc.) system was used. The fixed part of the models included induction protocol, anaesthesia time and the interaction between them. The relationships between time points within an animal were modelled using a spatial power covariance structure. Pairwise comparisons between treatments were made at all time points and evaluated after the application of the Bonferroni correction. The assumptions were checked using diagnostic plots.
Time (minutes) data were analysed using a Mann–Whitney U test (NPAR1WAY procedure in SAS 2017). The intubation score data is ordinal with three values. We tested the effect of the treatments with the scores obtained during the intubation using a Mann–Whitney U test. The p-value was calculated in an exact way using a randomisation algorithm NPAR1WAY procedure of the SAS (ibid). Data are presented as median or mean ± SD. A p‒value of < 0.05 was considered significant in all tests.
Results
Anaesthesia
Both drug combinations provided satisfactory induction for the TIVA administered in this study. Time from injection to unconsciousness was briefer (4.0 ± 1.0 min and 6.8 ± 2.8 min) (p = 0.030) in the ZTMe group than in the MiKF group, and intubation was easier to perform in the ZTMe group (p = 0.041) (Figure 1). Shivering was not observed in the ZTMe group, but ten minutes after initiating TIVA, it was observed in four out of six pigs for up to 50 min in the MiKF group.
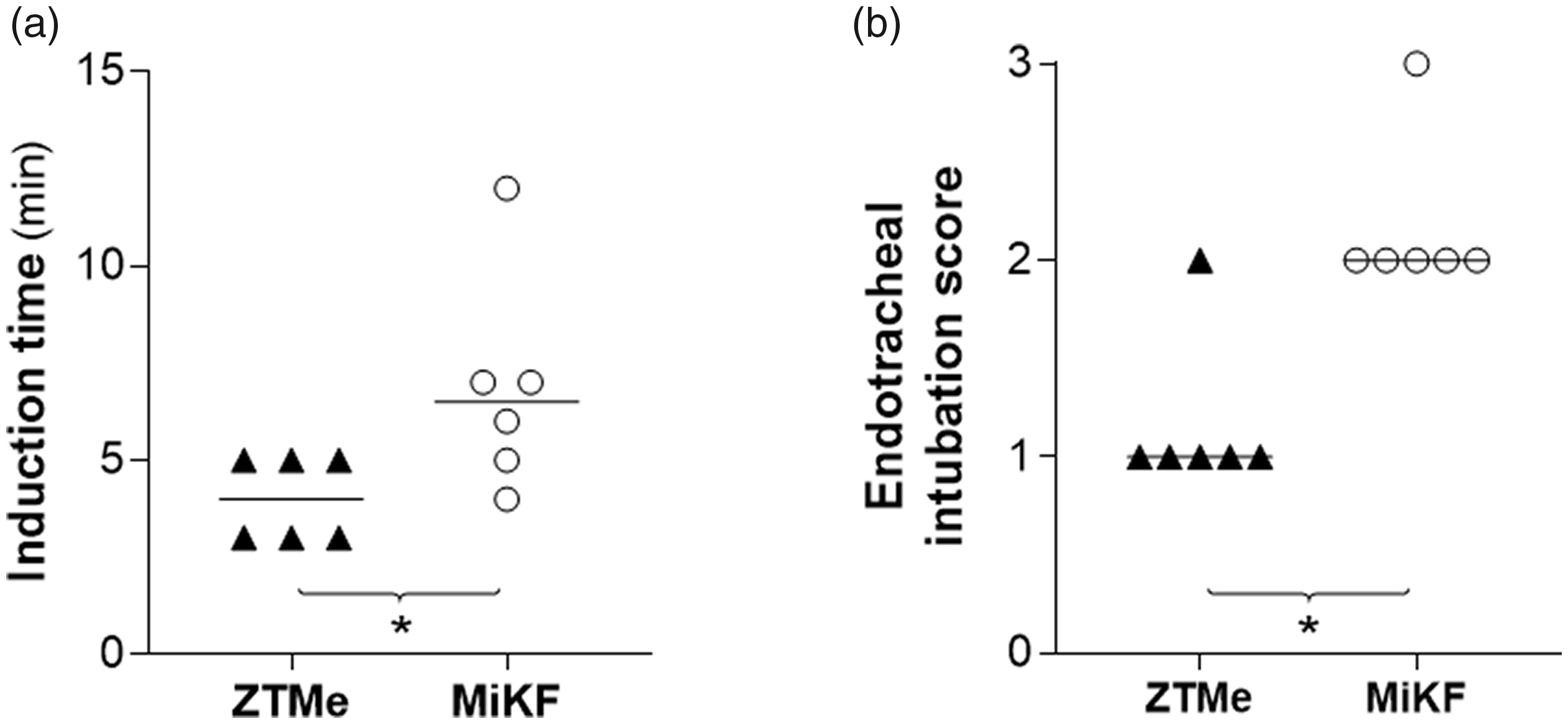
(a) Induction time (from injection to unconsciousness) (mean) and (b) scoring of endotracheal intubation (median) after intramuscular (IM) injection with zolazepam–tiletamine 5 mg/kg (Z 2.5 + T 2.5 mg/kg) in combination with medetomidine 0.05 mg/kg (ZTMe) n = 6 or midazolam 2 mg/kg, in combination with ketamine 10 mg/kg followed by fentanyl 4 μg/kg intravenously before intubation (MiKF) n = 6. Data considered significant (Mann–Whitney U test) at p < 0.05 (*p < 0.05).
The required TIVA infusion rate differed at different time intervals between the two groups (p = 0.040). Less adjustment of the rate, based on the response to the noxious stimuli, was required with ZTMe than with MiKF during the second to sixth hour of the anaesthesia (Figure 2).
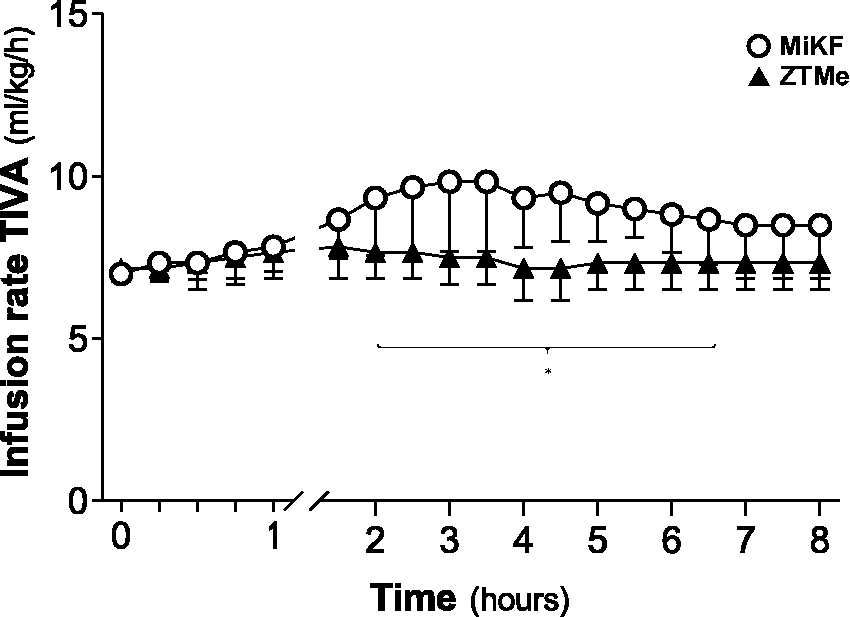
Infusion rate of total intravenous anaesthesia (TIVA) (mean ± SD) including 0.015 mg/ml midazolam, 4 mg/ml ketamine and 0.5 μg/ml of fentanyl. TIVA administration was based on a nociceptive stimulus response created by mechanically clamping the dewclaw. Data considered significant (mixed model) at p < 0.05 (*p < 0.05). ZTMe, zolazepam–tiletamine in combination with medetomidine; MiKF, midazolam in combination with ketamine followed by fentanyl.
Circulation
Significant differences were measured between the two drug combinations during the first hours of TIVA. The HR in the ZTMe group was initially higher (p = 0.004), but later decreased and was significantly lower (p = 0.012) 1.5‒3 h into TIVA (Figure 3) when compared to the MiKF group. Group differences in CI, MAP (Figure 3) and SVR (Table 2) were recorded over time. CI was significantly lower (p = 0.015) with ZTMe 1.5‒2 h into TIVA when compared to MiKF. MAP was significantly higher (p < 0.001) during the first 45 min of TIVA in the ZTMe group when compared to the MiKF group. SVR was higher (p = 0.009) during the first 3 h after induction with ZTMe when compared with MiKF. No significant differences between the induction drug combinations were found in PVR (Table 2) or MPAP values (Figure 3).
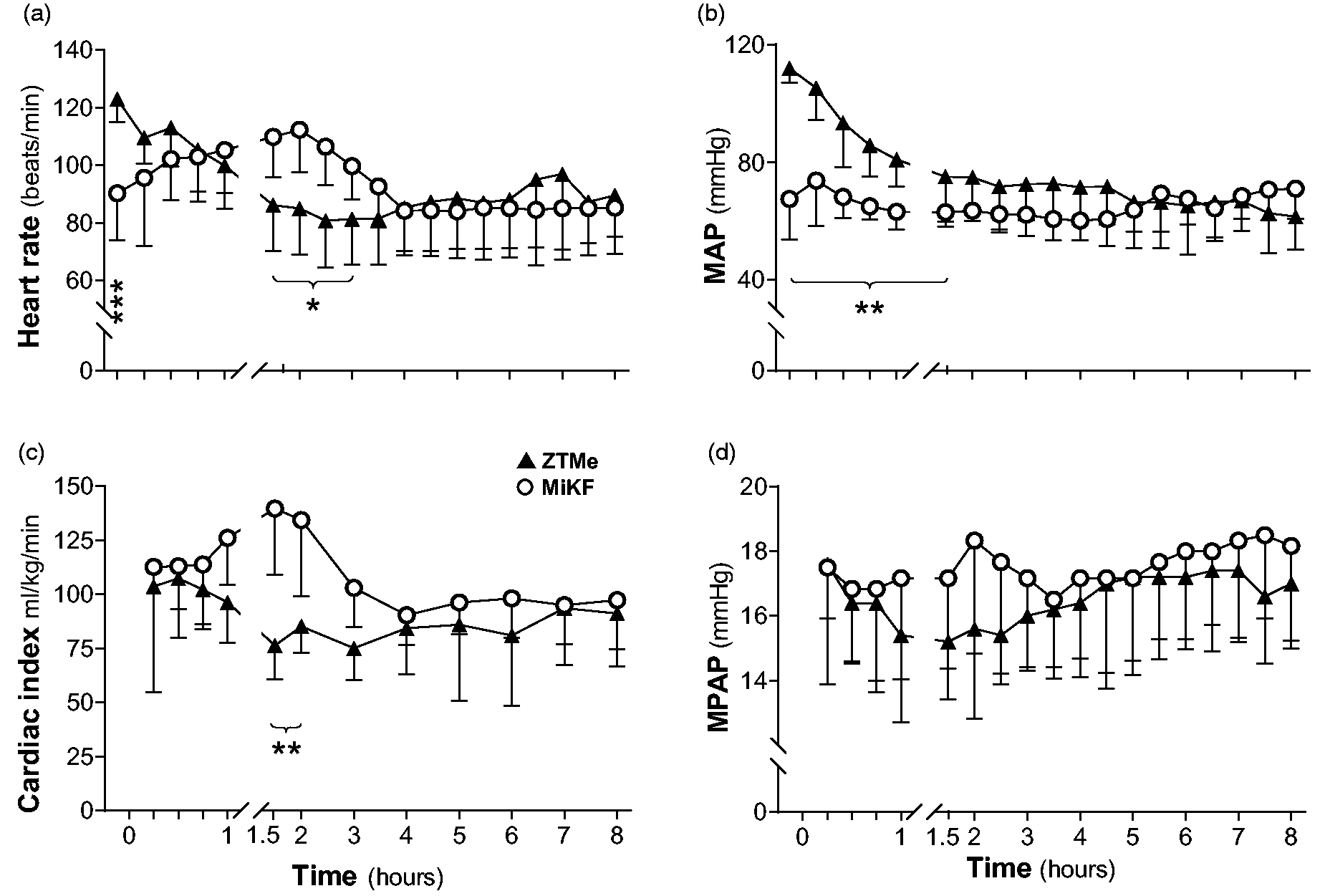
(a) Heart rate, (b) mean arterial blood pressure (MAP), (c) cardiac index and (d) mean pulmonary arterial pressure (MPAP) (mean ± SD) in pigs undergoing TIVA. Data considered significant (mixed model) at p < 0.05 (*p < 0.05, **p < 0.01, ***p < 0.001). TIVA, total intravenous anaesthesia; ZTMe, zolazepam–tiletamine in combination with medetomidine; MiKF, midazolam in combination with ketamine followed by fentanyl.
Physiological values (mean ± SD): blood temperature (Temp B), haemoglobin concentration (Hb), glucose, lactate, systemic vascular resistance (SVR) and pulmonary vascular resistance (PVR) obtained during total intravenous anaesthesia (TIVA) after intramuscular induction with zolazepam–tiletamine in combination with medetomidine (ZTMe) or midazolam in combination with ketamine followed by fentanyl (MiKF).
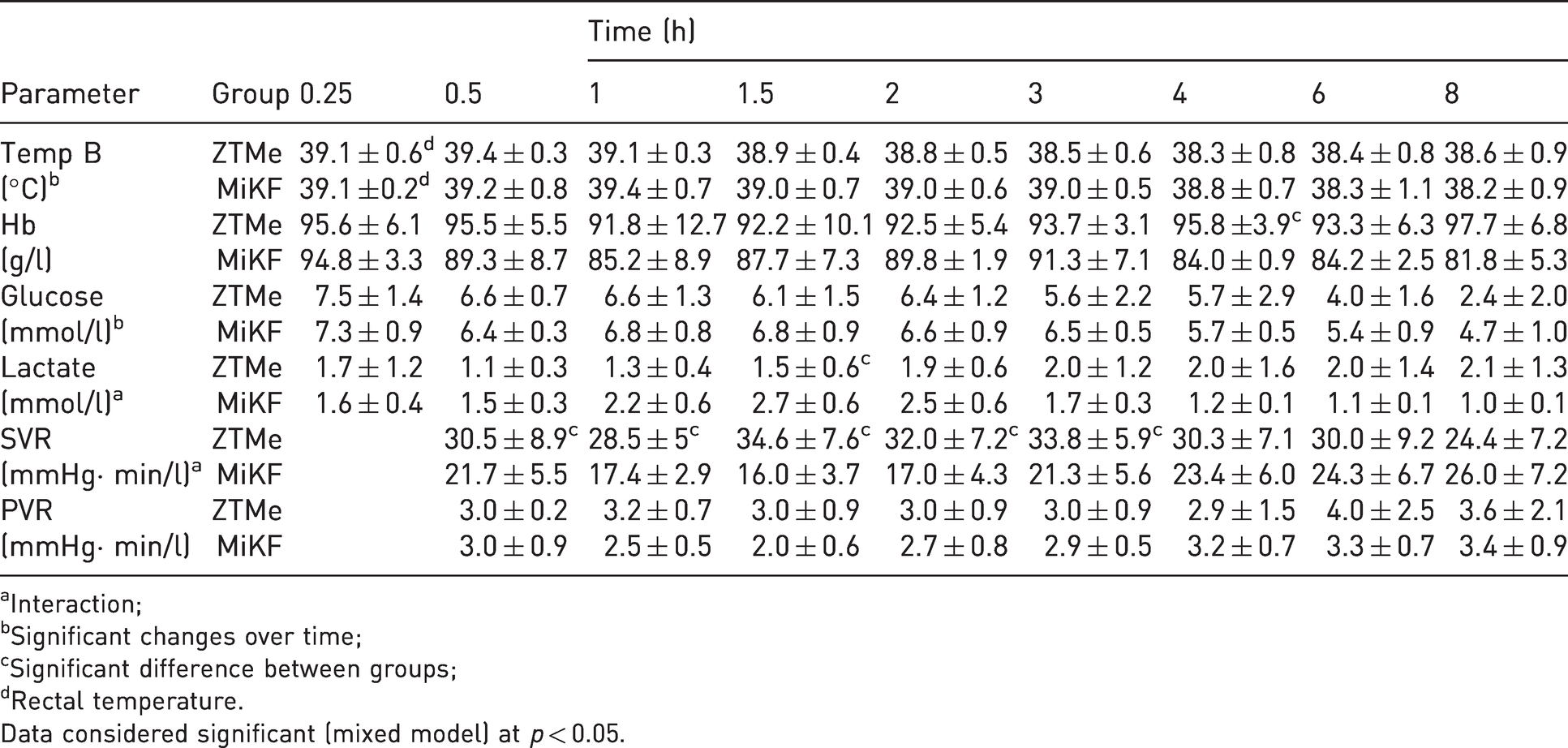
aInteraction;
bSignificant changes over time;
cSignificant difference between groups;
dRectal temperature.
Data considered significant (mixed model) at p < 0.05.
Ventilation
All pigs received mechanical ventilation from the start of TIVA and throughout the entire anaesthesia. The MV, adjusted to PaCO2 from the start of anaesthesia, was significantly lower (p = 0.047) in the early and late phase of TIVA in the ZTMe group when compared to the MiKF group. Acid base and blood gas data differed between the two groups. In the ZTMe group, pHa was significantly higher (p = 0.028) during the first two hours of TIVA compared to the MiKF group. The PaO2 and SaO2 was significantly higher (p < 0.001) and (p < 0.001), respectively, during the first 30 min after induction with ZTMe when compared to MiKF. S
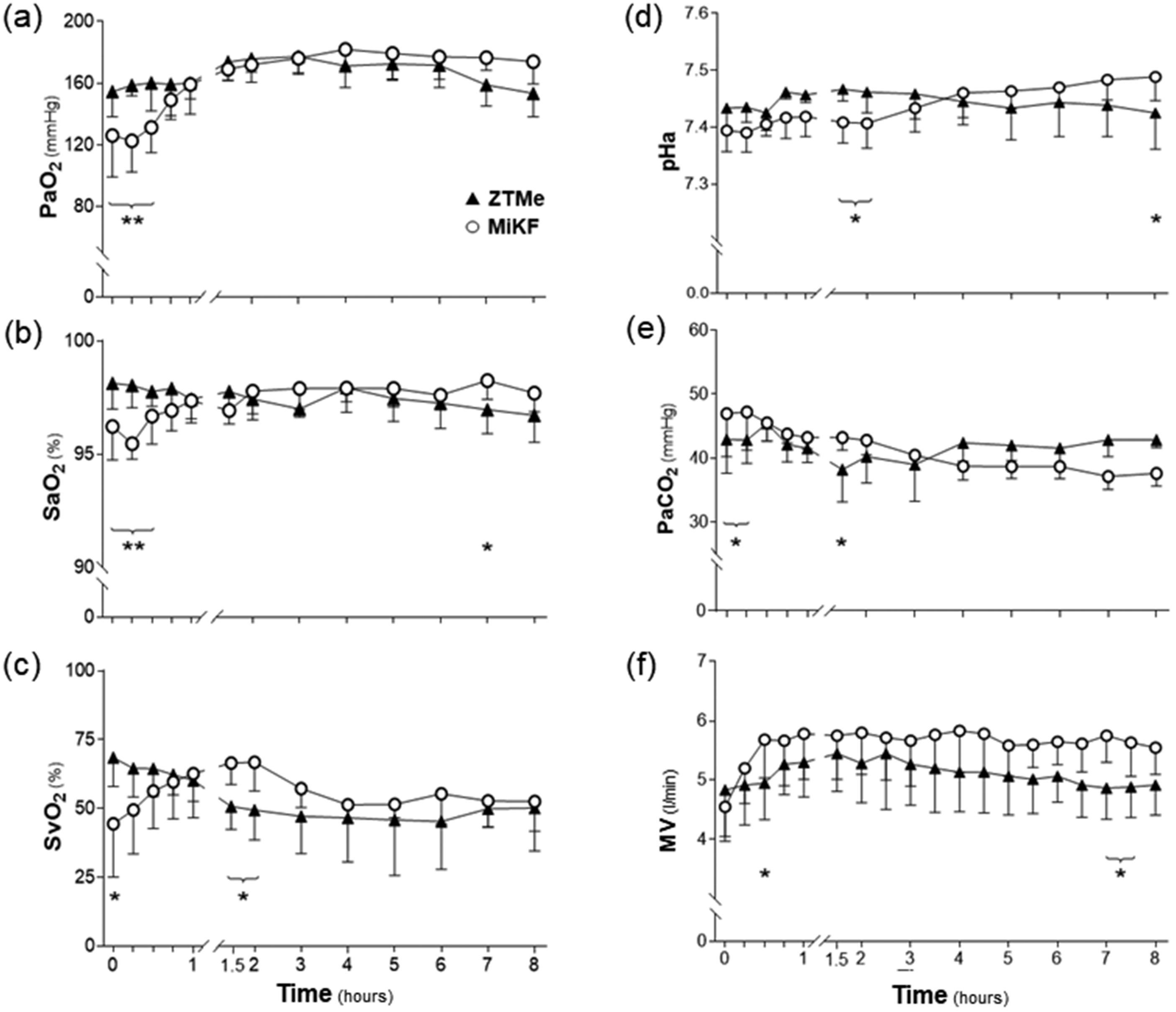
(a) Arterial oxygen partial pressure (PaO2), (b) arterial oxygen saturation of haemoglobin (SaO2), (c) mixed venous oxygen saturation of haemoglobin (SvO2), (d) arterial pH (pHa), (e) arterial partial pressure of carbon dioxide (PaCO2) and (f) minute ventilation (MV) (mean ± SD) in pigs undergoing TIVA. Data considered significant (mixed model) at p < 0.05 (*p < 0.05, **p < 0.01). ZTMe, zolazepam–tiletamine in combination with medetomidine; MiKF, midazolam in combination with ketamine followed by fentanyl.
Other physiological variables
Blood temperature and glucose concentration decreased over time in both groups. Lactate values differed between the induction techniques over time. Lactate was significantly lower (p = 0.028) 1.5 h into TIVA with ZTMe when compared to MiKF. There were minor differences in Hb. There were no differences in the amount of urine produced between the two induction drug combinations (see Table 2).
Discussion
The induction of anaesthesia in pigs with ZTMe was more rapid than with MiKF and endotracheal intubation was more easily achieved. The significant differences in the physiological variables, were for the most part, observed during the first 2 h of anaesthesia. Pigs induced with ZTMe needed less TIVA up to 6 h of anaesthesia to avoid a nociceptive response to the clamping of their dewclaw when compared to those induced with MiKF.
Induction of anaesthesia
The volume of the IM injection was not standardised and any differences in the reaction to the volume itself was not assessed. There is a recommended maximum volume for an IM injection at one site (0.25 ml/kg). 13 The volume of the ZTMe injection (0.05 ml/kg) was less than this amount. The volume of the MiKF injection (0.50 ml/kg) is twice the recommended amount. When injection volumes of 0.59–0.66 ml/kg were used in another study, pain was manifested in one third of the animals after half of the dose. 14 The large injection volume used in the MiKF group could have been divided into volumes less than 0.25 ml/kg to avoid possible pain and delayed time to unconsciousness. However, more than one injection could have also been an added discomfort for the pigs. Intubation was easier after induction with ZTMe than with MiKF. Intubation in pigs is difficult and it has been reported that laryngotracheal damage may occur due to the species-specific anatomical characteristics and the persistence of airway reflexes under anaesthesia. 3 Rapid onset of anaesthesia with good muscle relaxation has been described when medetomidine has been used in pigs, 15 and that could explain the ease of intubation experienced in the ZTMe group. Repeated doses of fentanyl in the MiKF group may have facilitated a smoother intubation, however, dose-related side effects are reported in pigs when fentanyl is given in high doses. 3
Shivering was observed in four out of six pigs in the MiKF group. Shivering has earlier been observed in pigs undergoing anaesthesia and the cause of the mechanisms involved varies.
16
The movements might interfere with planned interventions such as imaging and surgery, thus neuromuscular blocking (NMB) is recommended to treat this condition.
17
However, the masking of reflexes can be induced if NMB agents are used, which can make it difficult to assess the depth of anaesthesia. The NMB agents have no analgesic action, they do not produce loss of consciousness and the risk of accidental awareness due to inadequate supervision and poorly trained staff has been described.
16
In one study where shivering was observed in nine out of ten piglets after a fentanyl injection of 10 µg/kg IV,
17
the shivering subsided over time and was no longer obvious 43 min after the fentanyl injection. In the current study, shivering continued for up to 50 min in some pigs. The initial fentanyl injection of 4 µg/kg was administered before intubation and was followed by 3.5 µg/kg/h IV during TIVA. Post-operative shivering is a common complication of anaesthesia in many species including humans. This function of the body is associated with increased carbon dioxide production, increased oxygen consumption, decreased S
Required amount of total intravenous anaesthesia based on nociceptive stimulus
The infusion rate of TIVA based on the response to noxious stimuli differed between hour two and hour six. Mechanical nocistimulation has been shown to be applicable to pigs. 21 In some studies, a method to ensure an appropriate depth of anaesthesia has been an observed depression of the somatic reflex responses after mechanically clamping the dewclaw.21,22 That method has been compared with other methods such as electrical stimulations that are evaluated with the bispectral index and electroencephalography. 21 23 The conclusions in these studies suggest that clamping the dewclaw is acceptable for assessing nociception in anaesthetised pigs. The combination of zolazepam–tiletamine is reported to produce an anaesthetic effect up to 40 min,24,25 whereas the combination of midazolam and ketamine has been reported to provide safe and effective sedation in piglets for up to 2.5 h from induction. 8 The antinociceptive effect of an α2-agonist has recently been reported when the α2-agonist dexmedetomidine was infused for 80 min in pigs. 23 In that study, dexmedetomidine provided superior analgesia when compared to a fentanyl infusion based on the response to noxious stimuli. In dogs, the response threshold to an electrical stimuli increased significantly and lasted up to 6 h after an IM injection of medetomidine 0.03 mg/kg. 26 In another study involving conscious horses, an IM injection of dexmedetomidine 0.008 mg/kg was consistently associated with antinociception for up to 6.5 h when nociceptive electrical stimulation was performed. 27 Even though mechanical rather than electrical stimulation and pigs rather than horses and dogs were used in the present study, it is still possible that it is the α2-agonist medetomidine that enhanced the antinociceptive effect and prolonged the sedative effect for several hours in our pigs.
In the present study a positive response was defined as any reflexive withdrawal, whereas a negative response was defined as the absence of motion after stimulation. The TIVA rate was never set below 7 ml/kg/h, which was based on a known infusion rate used in research laboratories and according to textbook recommendations. 2
Physiological variations
The overall cardiovascular function and stability was satisfactorily preserved in all pigs. However, some of the differences that were measured were probably an effect of the medetomidine included in the ZTMe induction drug combination. The initial high MAP observed in ZTMe group was likely due to an increase in SVR through the activation of the postsynaptic receptors in the vascular smooth muscle, which is a well-known peripheral effect of α2-agonists. 28
Pigs in the ZTMe group exhibited a higher HR at the start of TIVA compared to the MiKF group. This was unexpected because α2-agonists commonly produce bradycardia. 14 In line with the findings in this study it has previously been reported that HR was higher in pigs treated with a dexmedetomidine infusion than pigs given a fentanyl infusion at a similar anaesthetic depth. 23 Further, tiletamine and ketamine which were included in the induction combination can affect HR by stimulating the sympathetic nervous system. 29
Pigs induced with the ZTMe combination required less MV to achieve a physiological PaCO2 and pH when compared to the MiKF combination while undergoing anaesthesia. As mentioned previously, it is possible that with MiKF induction the metabolism and carbon dioxide production can be higher, whereas the inclusion of an α2-agonist in the ZTMe induction reduced noradrenaline release and sympathetic nervous tone. The significantly lower S
Limitations of the study
The benefits of the ZTMe may have been due to the fact that the selected doses of the drug combination resulted in a deeper anaesthesia level than the MiKF. As different doses of the combination have not been evaluated in this study; it cannot be ruled out and therefore further investigation is needed. Instead of a separate infusion of the TIVA drugs, they were added to a Lactated Ringer’s solution. The volume of the Lactated Ringer’s solution infused (7.3 ± 0.8 and 8.7 ± 1.3 ml/kg/h in the ZTMe and MiKF group respectively) was based on the reflex withdrawal to a nociceptive stimulus and not according to any cardiovascular requirements. We could have used a separate infusion of Lactated Ringer’s solution for cardiovascular support. However, our volumes were within the range recommended for cardiovascular support (5–10 ml/kg/h). 2
Conclusion
The combination of ZTMe in the doses used provides a superior quality induction. With ZTMe, endotracheal intubation was more easily achieved. The overall cardiovascular stability was satisfactorily preserved with both drug combinations in all pigs. Some measured differences during the early phase of anaesthesia were probably an effect of the medetomidine included in the ZTMe combination. The required amount of TIVA, based on an assessment of nociception, was less for up to 6 h of anaesthesia with ZTMe. Similarly, there was less need for TIVA adjustments.
Footnotes
Acknowledgements
We are grateful to the staff at Hedenstierna Laboratory, Uppsala University for the precious technical support, to Eva-Maria Hedin for her invaluable help creating and editing the tables and figures, to Robin Quell for proofreading and English language editing and to Marianne Jensen-Waern for her critical reading of the manuscript.
Declaration of conflicting interests
The author(s) declare no potential conflicts of interests with respect to the research, authorship and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The research has received funding from The Swedish Research Council (VR) and the Swedish Research Council for the Environment, Agricultural Sciences and Spatial Planning (FORMAS).