Abstract
This study was conducted to investigate the effects of blood sampling on animal welfare in a total of 60 NTac:SD rats and 72 C57BL/6NTac mice of both sexes. Blood was sampled either by sublingual vein puncture, tail vein puncture or by retrobulbar plexus/sinus puncture under light isoflurane anaesthesia and, additionally, by facial vein puncture in mice. Non-punctured animals as well as isoflurane-anaesthetised animals were used as controls. Pre- and post-puncture sucrose intake (1.5% w/w) was measured in rats, and nest building scores were studied in mice for 24 h post-puncture. Post-puncture activity and anxiety levels of rats and mice were measured using an elevated plus maze test and an open field test. Stress levels 24 h post-puncture were assessed by analysing faecal corticosteroid metabolites. Sucrose intake and faecal corticosteroid levels were not affected by the blood sampling procedures. Rats showed reduced activity in the open field test and an increased level of anxiety in the elevated plus maze test following retrobulbar plexus puncture and isoflurane anaesthesia. In mice, nest building activity was affected in all the groups compared with the control group, except for animals subjected to facial vein puncture. Retrobulbar sinus puncture, tail vein puncture and sublingual puncture in mice resulted in reduced activity and increased anxiety. We conclude that, of the tested methods, puncture of the tail vein and the sublingual vein have the least adverse effects in rats, whereas facial vein puncture had the least adverse effects on the welfare parameters in mice.
Introduction
Laboratory rodents are often subjected to blood sampling in connection with experimental studies or health monitoring. 1 Several studies have been made to assess the effect of sampling methods on, for example, stress levels, 2 tissue damage 3 and food consumption 3 in the animals. However, in these studies, only a few techniques are compared using only one species,2–4 and extrapolating results from one species to another is problematic. Moreover, when comparing methods, it is of crucial importance that all tests are carried out under identical circumstances. Retrobulbar plexus/sinus puncture has been shown to have adverse effects such as tissue damage5–8 and contamination from the Harderian glands. 9 Therefore, alternative approaches such as facial vein puncture and sublingual puncture offering similar volumes of blood have been suggested for use in mice, though contamination may be difficult to entirely avoid using facial vein puncture. However, it has also been demonstrated that tissue damage levels are significantly lower in mice sampled by sublingual puncture compared with mice sampled by facial vein puncture. 3 The objective of the present study was to analyse and compare stress levels, behavioural consequences and the physiological effects of three commonly used techniques for blood sampling in rats (retrobulbar plexus puncture, tail vein puncture and sublingual puncture) and four techniques in mice (adding the facial vein puncture) (Table S1 in Supplementary Material online). Retrospectively, vena saphena puncture in mice should also have been included in this study as a less harmful alternative to both sublingual puncture and the retrobulbar sinus puncture. 6 Sublingual blood sampling was performed without anaesthesia in both mice and rats. Isoflurane anaesthesia has been shown to be aversive to rats,10,11 and blood sampling without anaesthesia in rats is an accepted standard method in Denmark. Since isoflurane re-exposure is also aversive to mice, 12 sublingual puncture was performed in the same way as for rats in this study. To assess the effect of blood sampling on animal welfare, a multitude of standard behavioural tests were applied to evaluate the combined changes in stress level, level of anxiety, activity and reduced welfare. For the assessment of anxiety level and overall activity, the elevated plus maze and the open field tests were used. 13 For the assessment of stress level, the level of faecal corticosteroid metabolites was measured. 2 Reduced welfare was evaluated in mice by assessing nesting behaviour,14,15 and in rats by a sucrose preference test allowing for detection of stress and anhedonia (a depression-like emotional state with a lack of feeling pleasure in normally pleasurable activities). 16
Materials and methods
Animals and husbandry
A total of 30 male and 30 female rats (NTac:SD) and 36 male and 36 female mice (C57BL/6NTac), six weeks of age, were included in the study. All animals were health monitored according to FELASA guidelines. 17 Detailed information on the animals and animal housing is presented in the Supplementary Material.
All experimental protocols involving animals were ethically reviewed and approved by the Animal Experiments Inspectorate (the national authority under the Ministry of Environment and Food of Denmark (licence number: 2012-15-2934-00256 C1)).
Timeline for the studies
Overall, two punctures were performed on each animal. The effects of the first puncture were investigated using faecal corticosterone excretion and home cage behavioural observations (nesting behaviour in mice and sucrose preference in rats). The effects of the second puncture were evaluated using open field and elevated plus maze tests in both species. Two consecutive punctures were performed to avoid confounding of the behavioural home cage observations (nesting and sucrose preference) due to stress resulting from the open field test and the elevated plus maze test. For details of the timeline of the study, please refer to the Supplementary Material (Figure S1).
Blood collection techniques and controls
For all blood sampling procedures, 1–2 drops of blood were drawn on each sampling occasion. Moreover, the second blood sample was always drawn from the opposite side of the animal.
Retrobulbar plexus/sinus puncture in rats and mice (eye)
Light isoflurane (Isobavet®, Schering Plough Animal Health, Denmark) anaesthesia was induced using a face mask, a precision vaporiser (Penlon PPV2) and a rodent ventilator (Hallowell EMC). Induction time (time until loss of righting reflex) was 1–3 min in rats and 1–1.5 min in mice. A concentration of 3–4% isoflurane was used. The animals were scruffed and a micro haematocrit tube (length 75 mm in both species) with an outer/inner diameter of 1.55/1.15 ± 0.05 mm for rats and 1.35/0.95 ± 0.05 mm for mice (Vitrex Medical A/S, Denmark) was inserted at the median canthus of the eye with a firm twist. As soon as blood appeared in the tube, it was removed, and a saline gauze sponge was gently placed over the eye to stop any residual bleeding. The animal was then returned to its home cage and continuously monitored until fully recovered (approximately 2–3 min).
Tail vein puncture in rats and mice (tail)
Rats were restrained in a plastic tube rat restrainer. The right lateral tail vein was raised using thumb and index finger, and was punctured with a 21G, 40 mm sterile needle (Terumo, Belgium) 3 cm caudally from the tail base.
Mice were restrained in a box restrainer, and the tail was grasped between the thumb and index finger. The right lateral tail vein was punctured with a nick incision approximately one-third from the tail tip using a number 11 BP blade (Kruuse, Denmark). Any residual bleeding was stopped by applying gentle pressure with a gauze sponge on the puncture site. The mice were then returned to their home cages.
Sublingual vein puncture in rats and mice (tongue)
The rats were scruffed and the sublingual vein was punctured at the base of the tongue with a 21 G, 40 mm sterile needle (Terumo, Belgium). Each rat was immediately inverted to allow the collection of 1–2 drops of blood from the punctured site and was then returned to its home cage. In mice, exposure of the vein was not possible, which is why the puncture was made towards the lateral side at the base of the tongue using a 23G, 30 mm sterile needle (Terumo, Belgium). After sampling, the mouse was returned to its home cage. No residual bleeding was noted in any of the animals.
Facial vein puncture in mice (cheek)
The mice were scruffed and a puncture was made approximately 3 mm caudally and 1 mm dorsally from the lateral whisker and sebaceous gland in the mandibular region with a 21G, 40 mm sterile hypodermic needle. The needle was introduced only to the depth of the bevel. No residual bleeding was observed.
Isoflurane anaesthesia control in rats and mice (iso)
The rats and mice were anaesthetised and handled in exactly the same way as the retrobulbar plexus/sinus puncture groups, excluding only the retrobulbar plexus/sinus puncture.
Control group for behavioural testing in rats and mice
Naïve control animals were subjected to the same behavioural tests at the same time as the punctured animals, as shown in the timeline (Supplementary Material Figure S1). No additional handling was carried out.
Behavioural tests and stress hormone analysis
Sucrose preference in rats
The animals were supplied with one bottle of 1.5% sucrose (BDH Prolabo GPR Rectapur, Belgium) and one bottle of tap water for four days. After 24 h of habituation, the bottles were weighed, followed by two additional readings at 48 h and 72 h. Puncture was performed after 72–75 h, whereafter bottles were weighed 24 h later for measuring post-puncture intake of sucrose and tap water.
Nest building scoring test in mice
The test was based on a modification of the nest building scoring system documented by RM Deacon, 18 as described by Pedersen et al. 19 The mice were deprived of nesting material two weeks prior to testing and were only provided with a red mouse igloo with three entrance holes (Bio-Serv Mouse Igloo). At test start, the igloo was placed in the left rear corner of the cage. When the animals were returned to their home cage after puncture, one nesting pad (NestLets® Ancare, USA) was supplied in the right front corner of each cage (t = 0). Nest building scoring was done at t = 1, 2, 3, 4, 5, 6, 7, 8 and 24 h. Scoring from 0 to 5 was carried out based on the percentage of nesting pad shredded (0%, 1–24%, 25–49%, 50–74%, 75–99% and 100%). This score was added to the number of holes covered with the shredded material (0–3), resulting in a maximum score of eight (for details and pictures of scorings, please refer to Pedersen et al. 19 ).
Elevated plus maze (EPM) in rats and mice
Both rats and mice were subjected to the EPM test after the second vein puncture. In the EPM for rats, all arms (closed and open) were 50 cm long and 10 cm wide. In the EPM for mice, all arms were 8 cm wide and 21 cm long. The animal was placed randomly in the central area with its head facing to one of the two open arms and away from the handler. The test time was 5 min and no handler was present. The surface of the EPM was cleaned between tests using wipes moistened with 70% ethanol. The test was recorded on video and manually analysed. The number of entries (both front and hind paws entering) and duration of time spent in the open arms, in the central area and in the closed arms were measured. Risk assessment was noted when the animal had its front paws in an open arm and the remaining part of the body in the central area and/or in a closed arm. Overall activity was calculated as the sum of the total number of entries into the open arms, into the closed arms and number of risk assessments made. The animals were passed on to the open field test immediately after the EPM test.
Open field test in rats and mice
Each animal was placed with its face away from the handler in the centre of a circular open field (97 cm diameter) and video recorded for a period of 5 min with no operator in the room. Between tests, the entire open field surface was cleaned with 70% ethanol and between species the open field was cleaned and washed thoroughly. For manual analysis of the video, the entire arena was divided into four quadrants as well as into an outer (0–30 cm from the wall) and a central area. Entries to the various quadrants were analysed as a measure of overall activity. Additionally, numbers of entries and the subsequent duration of time spent in the central area were recorded. After the behavioural tests, animals were returned to their home cages.
Faecal corticosteroid metabolites (FCMs) assay in rats and mice
Twenty-four hours after puncture, all bedding material from each cage was collected and frozen in labelled plastic bags at −20℃ until segregation and analysis.20,21 An electric sieve shaker was used for complete segregation of the smaller mouse faecal pellets from the bedding material, while faecal pellets from rats were manually picked from the bedding with a pair of clean forceps.
Statistical procedures
Statistical tests were performed with Graphpad Prism 5.02. Each cage was the experimental unit of the nest building test, sucrose intake and FCM assay, while the individual animal was the experimental unit of the EPM and open field tests. Data distribution normality was tested with the Shapiro–Wilk test. Normally distributed data were analysed with analysis of variance with Tukey’s multiple post hoc comparison tests, while other data were analysed with the Kruskal–Wallis test with Dunn’s multiple post hoc comparison tests. p < 0.05 was considered statistically significant. The effect sizes for total sucrose and total fluid intake in rats and FCM assay in rats and mice were estimated with a post hoc power analysis in G Power software 3.0.10.
Results
None of the vein punctures needed repetition due to failure of the first attempts, as these were all successful. None of the daily inspections of the animals throughout the study revealed any animals with clinical signs of illness, injuries or pathologies related to the blood sampling procedures. No significant sex effect was revealed for any parameter recorded.
Sucrose preference in rats
When offered both 1.5% sucrose water and tap water, the daily sucrose intake per cage was 107.52 ± 14.1 g of sucrose compared with 5.64 ± 2.5 g of tap water (mean ± SD). None of the treatments had any effect on the intake of either 1.5% sucrose (p = 0.2337; Kruskal–Wallis statistic = 11.65) or total water (sucrose and tap water combined) (p = 0.3421; Kruskal–Wallis statistic = 10.10). A medium effect size of 0.378 was calculated with post hoc power analysis.
Nest building scoring test in mice
The overall 24 h nest building score differed significantly between groups (p < 0.0001, Kruskal–Wallis statistic = 66.79), and this was reflected by significant differences between the various groups in the multiple comparisons (Figure 1). It is worth noting that, compared with the control group, only the mice subjected to facial vein puncture maintained their nest building activity.
Pairwise comparisons of the overall nest building activity in mice for 24 hours (group-wise total of all scores obtained) using Kruskal–Wallis test with Dunn’s multiple comparison. Data are presented as mean ± SEM.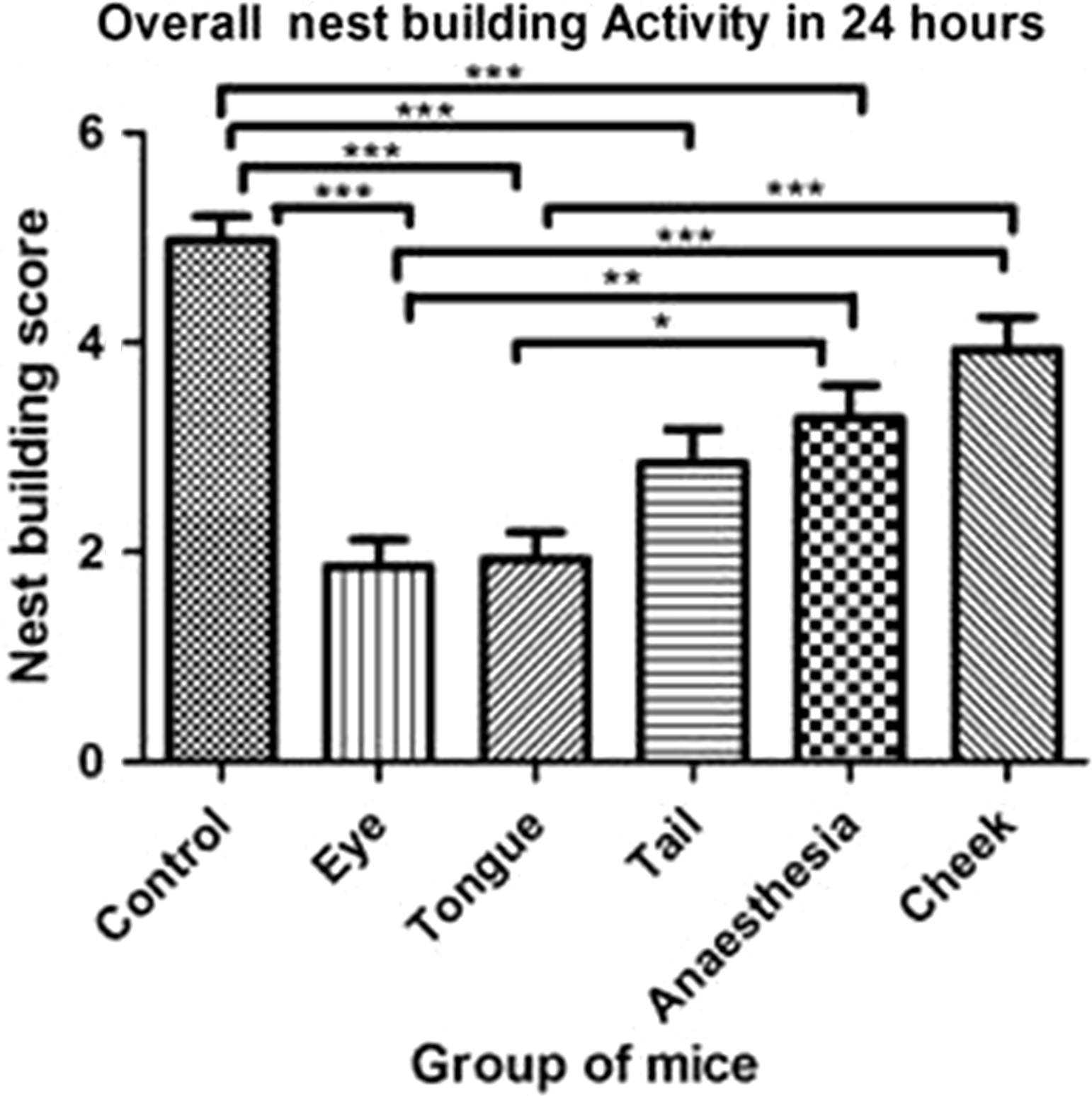
EPM in rats
The groups did not differ in their number of open-arm visits, while there was a significant difference in the number of closed-arm visits (p = 0.0008, Kruskal–Wallis statistic = 18.87), centre visits (p = 0.0004, Kruskal–Wallis statistic = 20.61), time spent in the central area (p < 0.0001, Kruskal–Wallis statistic =25.39), risk assessments (p < 0.0001, Kruskal–Wallis statistic = 34.66) and overall activity (p < 0.0001, Kruskal–Wallis statistic = 44.53). Pairwise comparisons are presented in detail in Figure 2(a)–(e). An important finding is that control rats had significantly higher activity compared with rats subjected to retrobulbar plexus puncture (risk assessment and overall activity) or anaesthetised with isoflurane (overall activity), whereas rats that were tail punctured showed significantly higher levels of activity compared with retrobulbar-plexus-punctured rats or rats anaesthetised with isoflurane (on all five parameters).
Activity of rats (n = 12 per group) in the EPM test. Number of visits to closed arms (a), visits to the central area (b), time spent in the central area (c), risk assessments in open arms (d) and overall activity of rats in the EPM (e) showed significant difference between groups in the Kruskal–Wallis test. Data are presented as mean ± SEM.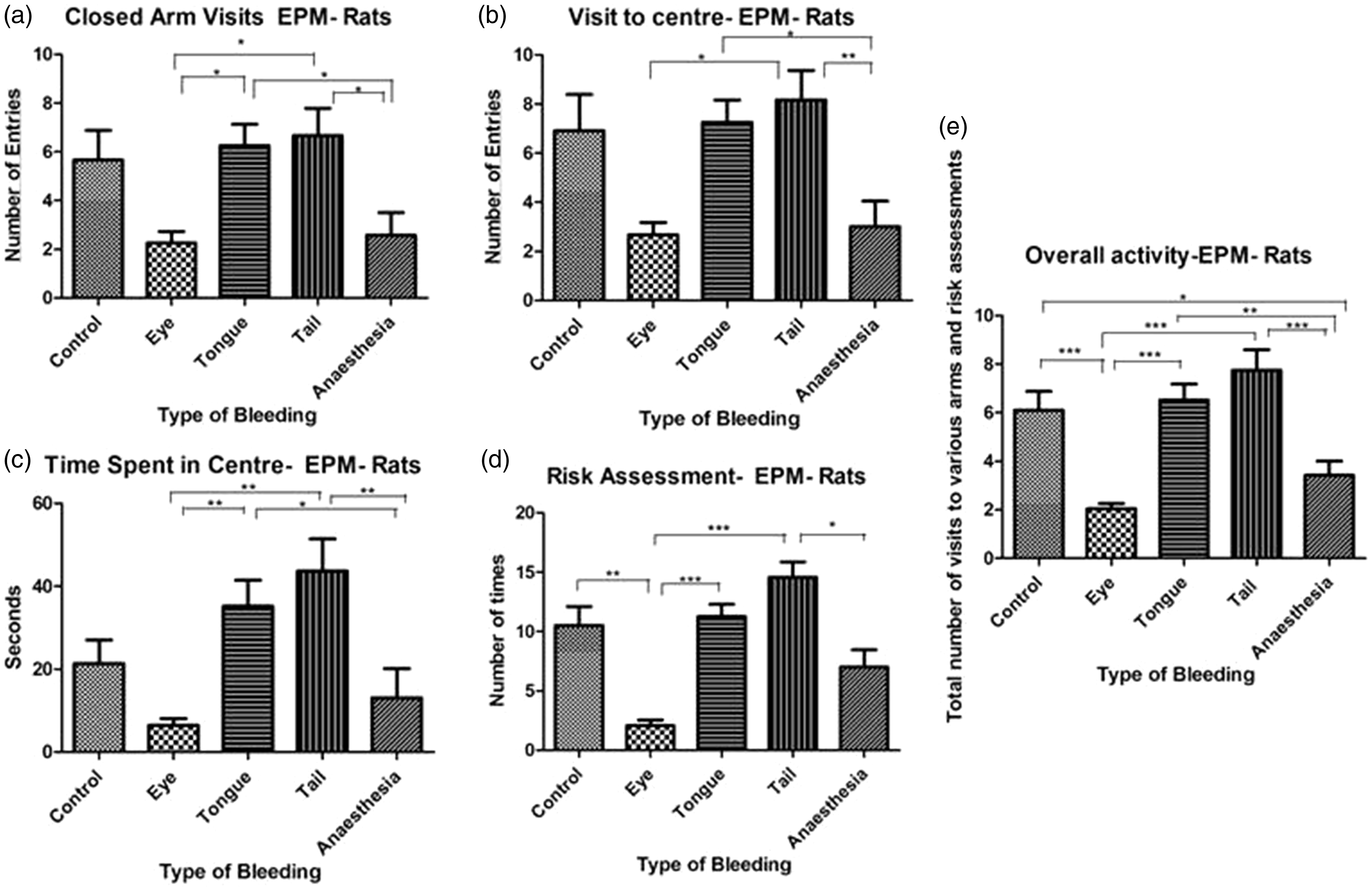
Open field test in rats
The groups spent significantly different amounts of time in the centre (p = 0.0162 and Kruskal–Wallis statistic = 12.17), had significantly different numbers of central area entries (p = 0.0068, Kruskal–Wallis statistic = 14.15) and differed significantly in their overall activity (p < 0.0001, Kruskal–Wallis statistic = 69.25). The significant differences between the individual groups are shown in Figure 3(a)–(c). The most prominent finding is the difference on all three parameters between rats being punctured either in the tail or in the retrobulbar plexus, indicating that the latter are less active and less prone to enter and spend time in the open field centre.
Activity of rats (n = 12 per group) in the OF test. Time spent at the centre (a), number of entries to central area (b) and the overall activity (c) in the OF showed significant differences between groups in the Kruskal–Wallis test. Data are presented as mean ± SEM.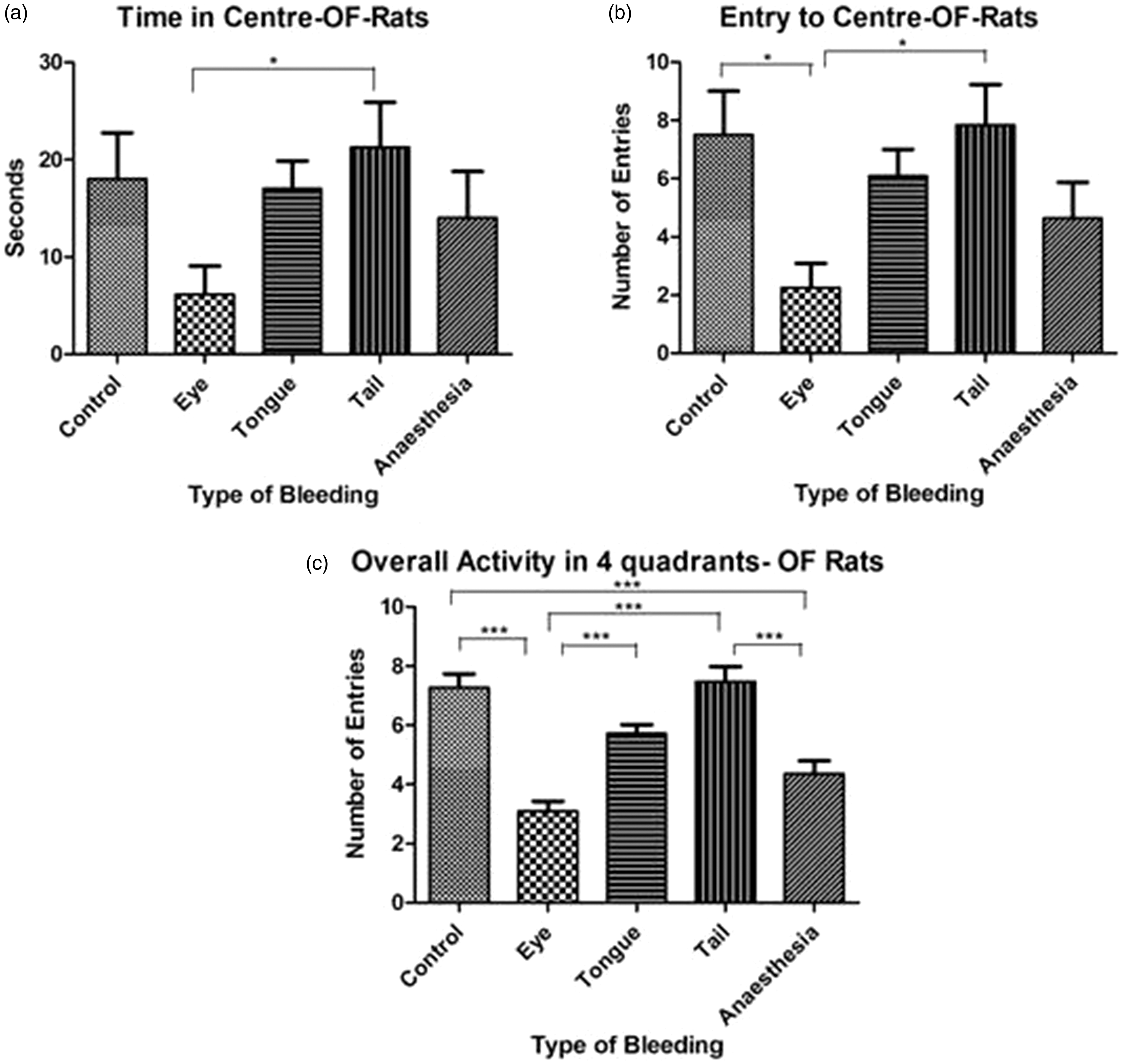
FCMs in rats and mice
FCM data did not follow a normal distribution and were analysed with Kruskal–Wallis test. None of the punctures had significant effects in rats and mice. Medium effect sizes of 0.377 in rats and 0.341 in mice were calculated with post hoc power analysis.
EPM test in mice
Overall, the groups spent significantly different amounts of time in the open arms (F(5, 48) = 2.662, p = 0.0334)), while there were no differences in their times spent in closed arms or their number of open-arm visits. The groups also differed significantly in their number of centre visits (F(5, 48) = 4.361, p = 0.0024)), in the amount of time they spent in the centre (F(5, 48) = 8.286, p < 0.0001)), in their open-arm risk assessments (F(5, 48) = 7.712, p < 0.0001)) and in their overall activity (p = 0.0008, Kruskal–Wallis statistic = 20.93). Pairwise comparisons (Figure 4(a)–(d)) showed that mice subjected to facial vein puncture did not differ from control animals in any of these parameters, whereas retrobulbar-sinus-punctured mice had fewer visits to the centre, spent significantly less time there and showed reduced overall activity compared with the control mice (Figure 4(a), (b) and (d).
Activity of mice (n = 12 per group) in the EPM test. Number of visits to the centre (a) and time spent in the centre (b) differed significantly in analysis of variance and risk assessments in open arms (c) and overall activity (d) differed between groups in the Kruskal–Wallis test. Data are presented as mean ± SEM.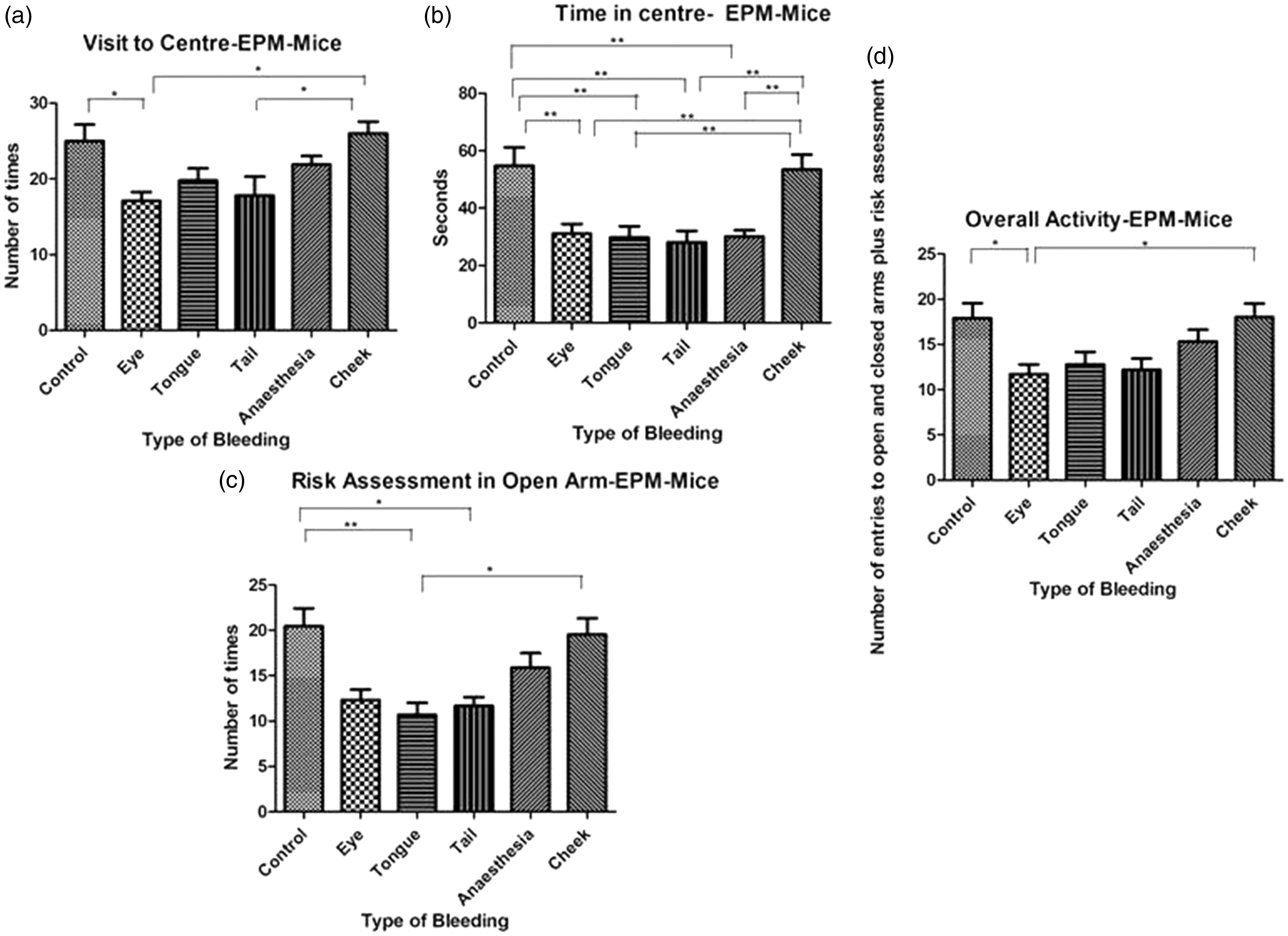
Open field test in mice
One mouse from the Iso group had to be excluded due to video recording problems. Overall, the groups differed significantly in their central area entries (F(5, 47) = 3.118, p = 0.0164)), the time they spent in the centre (F(5, 47) = 3.364, p = 0.0112)) and their overall activity (p < 0.0001, Kruskal–Wallis statistic =36.93). Significant differences between the individual groups were found on several parameters in the multiple comparisons (Figure 5(a)–(c)). For example, on all three parameters, mice subjected to retrobulbar sinus puncture were significantly less active than the control mice, while this was not the case for mice punctured in the tail or the facial vein. Moreover, it is important to notice that mice subjected only to isoflurane anaesthesia had a significantly higher number of centre entries and greater overall activity compared with mice subjected to retrobulbar puncture under isoflurane anaesthesia.
Activity of mice (n = 12 per group) in the OF test. Entry to the centre (a) showed significant difference between groups in ANOVA and time spent in the centre (b), and overall activity (c) in the OF test in mice showed significant differences between groups in the Kruskal–Wallis test. Data are presented as mean ± SEM.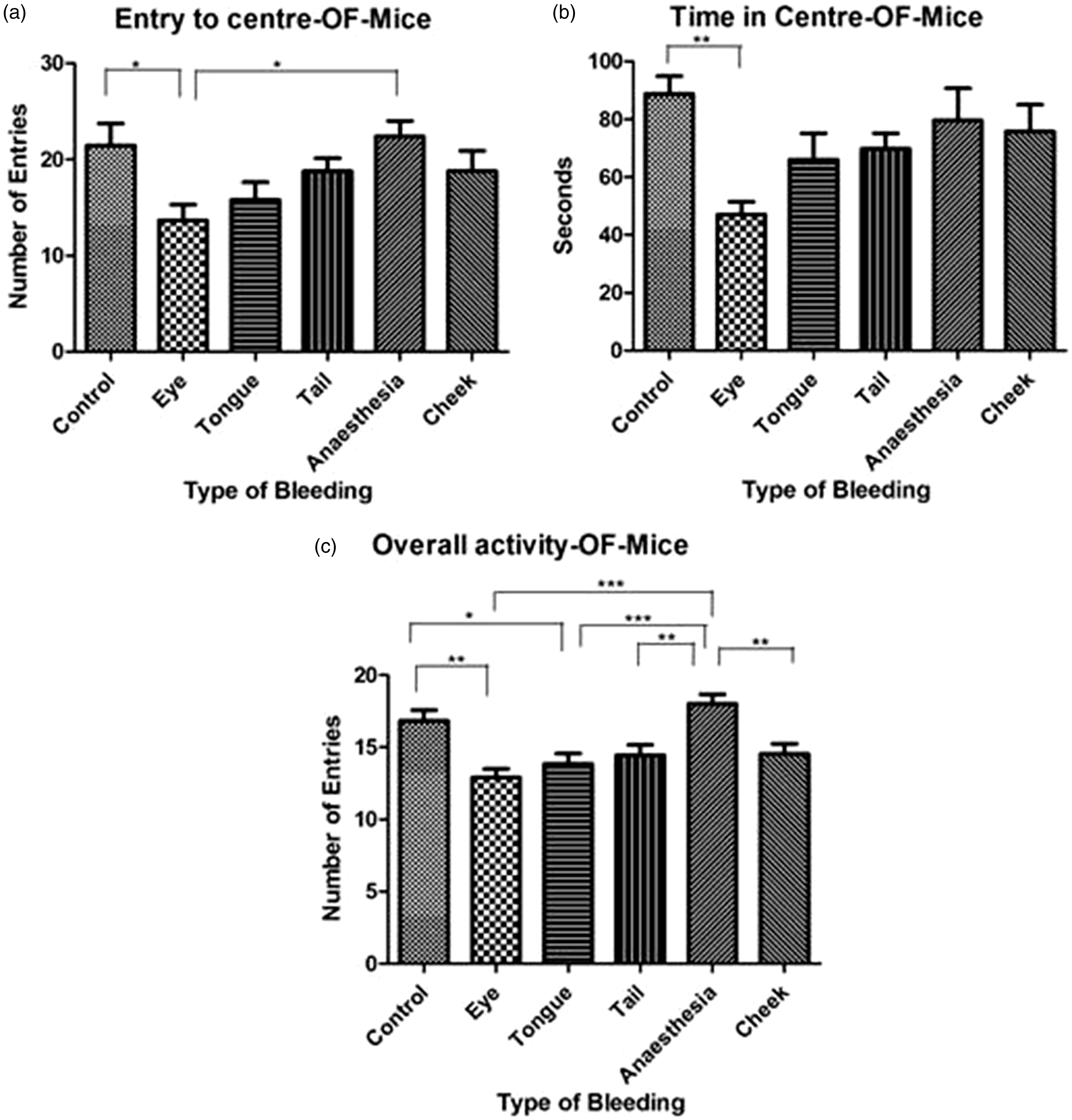
Discussion
In rats, only the retrobulbar plexus puncture resulted in a significantly lower risk assessment in the EPM and fewer centre area entries in the open field compared with control animals, whereas both retrobulbar plexus puncture and isoflurane anaesthesia (but none of the other sampling methods) resulted in a significantly lower overall activity in both EPM and open field compared with control animals. Since EPM and open field tests were carried out after the second puncture, the aversion towards repeated isoflurane anaesthesia, as demonstrated by Wong et al. 10 , may have contributed to this effect.
In mice, overall nest building after 24 h was significantly reduced by both anaesthesia and all the sampling methods except for facial vein puncture compared with control mice (Figure 1). Compared with the control mice, the retrobulbar-sinus-punctured mice showed an overall reduced activity in the EPM tests, whereas mice subjected to anaesthesia alone did not differ from control animals. Both mice punctured sublingually or in the retrobulbar sinus showed reduced activity in the open field test compared with control animals. Isoflurane anaesthesia did not significantly influence the overall activity in the open field test compared with control animals; however, all vein-punctured mice were significantly less active compared with mice subjected to isoflurane anaesthesia alone. Hence, in this study, facial vein puncture appears to be the most humane method of those that were investigated. Since nest building behaviour in tail-punctured mice was not significantly lower than for those punctured in the facial vein, tail vein puncture could be suggested as the second least harmful procedure. Even though sublingual puncture in conscious rats seems to be preferable as a method in this species, it must be heavily emphasised that this is indeed not the case for mice. Even under anaesthesia, it may be difficult for the operator to see the sublingual veins in mice, 5 and moreover, this method severely affected nest building behaviour in the mice in our study. Additionally, it is much easier to see other veins, such as the saphenous vein, and hence it could be argued that sublingual puncture should be avoided in mice. The findings in this study indicate that puncture of the retrobulbar plexus/sinus is the most stressful technique in both species, which is in agreement with previous observations.4,22 Retrobulbar sinus puncture impairs the normal nest building behaviour in mice, and from the nest building scores it is evident that even after 24 h the mice have not returned to their normal ethological activity levels. This effect could be due to tissue damage to the eye and the surrounding tissues, which is known to be caused by retrobulbar sampling.5–8
The most striking difference between mice and rats noted in this study is the behavioural differences after puncture of the sublingual vein. In mice, sublingual puncture severely affected both nest building behaviour and behaviour in EPM and open field tests, which indicates that this procedure is highly stressful. In rats, however, the other tested methods seemed to be more harmful. In rats, neither tail vein nor sublingual puncture affected any of the activities in open field and EPM compared with control animals, whereas retrobulbar plexus puncture significantly decreased several behavioural parameters.
Tail vein puncture in rats has previously been shown not to change the initial low level of corticosterone, 23 and the present work demonstrates that of the three techniques tested in rats, the tail vein puncture affects animal welfare the least. Most of the parameters assessed in the EPM and the open field tests suggested that isoflurane raised the anxiety levels and reduced the activity levels in the rats severely, whereas in mice, the overall activity in EPM and open field was not affected by prior isoflurane anaesthesia. However, nest building activity measured over 24 h was reduced significantly, indicating a long-term effect of prior anaesthesia on mouse behaviour. The present study differentiates clearly between the reduced activity and raised anxiety levels caused by retrobulbar sinus puncture from that of anaesthesia alone, since retrobulbar-sinus-punctured mice were still found to be significantly less active in the open field test compared with the isoflurane-treated control mice.
Since elevated FCM levels are considered to reflect substantial subacute or persistent elevation of hypothalamic–pituitary–adrenal activity, 21 it seems that none of the methods applied were stressful to an extent that caused a long-term effect on this system. However, since we have only measured the total amount of FCM excreted during 0–24 h after blood sampling, additional faecal sampling would be necessary to assess whether there is an effect of blood sampling on FCM in the hours right after the sampling. Moreover, additional sampling would be necessary to establish whether blood sampling may have an effect on the circadian rhythm of corticosterone excretion.
A study by Tsai et al. 24 on vein puncture and welfare in BALB/cO1aHsd mice indicates that the retrobulbar puncture presented a lesser impact on welfare in comparison with the facial vein puncture, which is in contrast to the present study. The present work did not, however, analyse and summarise the tissue damage as reported by Tsai et al. 24 and the differences in the end results can be due to the parameters studied and the differences in approaches to the performed tests. Teilmann et al. 25 demonstrated that facial vein phlebotomy induced a higher stress response and a greater body weight loss than retrobulbar blood sampling without anaesthesia in mice, which is not in accordance with the findings of the present study, which concludes that facial vein puncture is a better choice for blood sampling in mice in comparison with the retrobulbar sinus. Since isoflurane anaesthesia in itself may have a detrimental effect on animal welfare in both mice and rats, these contradictory results may be due to the use of anaesthesia for retrobulbar puncture in the present study. Henceforth, it could be of potential benefit to evaluate the use of a vasodilating sedative such as Hypnorm® (fluanisone and fentanyl) instead of isoflurane for retrobulbar puncture. Moreover, the sedating effect of the Hypnorm® may counteract the stress related to handling and immobilisation. Sublingual vein puncture has been recommended as a substitute for retrobulbar 5 and facial vein puncture 3 in mice; however, the present study concludes that sublingual and retrobulbar routes pose severe welfare issues and that the facial vein is the better choice among these three techniques in mice. It is important to be aware of other suitable alternatives for blood sampling in rodents, such as sampling from the lateral vena saphena in mice (saphenous venepuncture). A study by Aasland et al. 26 indicates that saphenous sampling is less stressful than tail sampling and hence may provide an even better blood sampling method in mice than the four methods assessed in our study.
In conclusion, the welfare parameters used in the study indicate that in this study and under the housing conditions offered in this study, tail vein puncture and sublingual puncture are the best choices of the bleeding techniques used in this study in young NTac:SD rats, and that facial vein puncture is the best method in young C57BL/6NTac mice. Following facial puncture, tail vein puncture may be considered a better choice than sublingual vein and retrobulbar sinus puncture in mice. However, other methods such as saphenous venepuncture may present an even better alternative. Further research should be carried out to evaluate the effect of isoflurane anaesthesia on animal welfare in combination with blood sampling. Moreover, alternative ways of alleviating the stress induced by handling, fixation and venepuncture should be explored. Lastly, the opposing results of the various studies on the effect of blood sampling on mouse and rat welfare may call for new, unconventional and much more subtle ways of assessing the welfare of our laboratory rodents during standard procedures such as blood sampling.
Footnotes
Acknowledgements
VSH and DBS contributed equally to this work. We thank Helene Farlov, Mette Nelander, Helle Porsdal and Trine Marie Ahlman Glahder, the technicians at University of Copenhagen, for taking care of the animals and assisting with the faecal corticosteroids assay.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by CALAR (Centre for Applied Laboratory Animal Research), Denmark.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
