Abstract
General anaesthesia disrupts thermoregulation in mammals, which can cause hypothermia. Decreases in core body temperature of 1℃ cause significant postoperative complications in humans, and peri-anaesthetic hypothermia in mice increases data variability, which can potentially increase animal use. In rats, the impact of different temperature management strategies on the incidence and severity of hypothermia, and the accuracy of different temperature measurement methods, is unknown. Eighteen adult male and female SD rats were block-randomized to one of three treatment groups: no-warming (NW), limited-warming (LW, heat pad during anaesthesia), and pre-warming (PW, warm air exposure before anaesthesia, followed by heat pad). Anaesthesia (isoflurane) duration was for 40 min. Core body temperature (intra-abdominal telemetric temperature capsule) was recorded during anaesthesia and recovery. During anaesthesia, rectal, skin, and tail temperatures were also recorded. In the PW group, core temperature was maintained during anaesthesia and recovery. By contrast, the NW group was hypothermic (11% temperature decrease) during anaesthesia. The LW group showed a decrease in temperature during recovery. Recovery to sternal recumbency was significantly faster in the PW (125 [70–186] s, P = 0.0003) and the LW (188 [169–420] s, P = 0.04) groups than in the NW group (525 [229–652] s). Rectal temperature underestimated core temperature (bias −0.90℃, 95% limits of agreement −0.1 to 1.9℃). Skin and tail temperatures showed wide 95% limits of agreement, spanning 6 to 15℃, respectively. The novel strategy of PW was effective at maintaining core temperature during and after anaesthesia. Rectal temperature provided an acceptable proxy for core body temperature.
General anaesthesia disrupts thermoregulatory control mechanisms in mammals, and peri-anaesthetic hypothermia commonly occurs in humans, companion animals, and laboratory species.1–6 In humans, small decreases (1℃) in core body temperature are associated with a host of side-effects, including surgical site infection, impaired clotting, morbid cardiac events, impaired drug metabolism, and increased anaesthesia recovery time.7–9 Despite this strong link between hypothermia and negative clinical outcomes in humans, the incidence of peri-anaesthetic hypothermia remains high, and is estimated to be between 53 and 85%.10–12
In contrast to the large body of literature on peri-anaesthetic hypothermia in humans, there are relatively few studies in laboratory rodents5,6,13 or companion animals.1,2,14–16 In the latter, the incidence of postoperative hypothermia (oesophageal temperature of less than 38.5℃) is high, and is present in approximately 80–97% of dogs and 97% of cats.1–3
Though the evidence base is currently sparse, there are indications that complications associated with hypothermia in humans are relevant to companion animals 14 and laboratory rodents. 17 Peri-anaesthetic hypothermia may negatively affect experimental outcomes and data quality, unnecessarily increasing animal use.18–20 Furthermore, hypothermia may be a welfare concern; hypothermic human patients require more postoperative analgesia,21,22 and it has been suggested that shivering during the recovery period may cause surgical sites to jar and contribute to pain. 23
Hypothermia during general anaesthesia is caused by impaired thermoregulation. In healthy conscious mammals, core body temperature is maintained within a narrow interval, known as the inter-threshold range, with variations in core temperature limited to ±0.3℃ through a combination of autonomic and behavioural responses. General anaesthesia causes depression of the hypothalamus, which widens the inter-threshold range 10–20 fold, allowing core temperature to fluctuate by ±2–4℃.24,25 Therefore, hypothermia occurs before any heat-conserving mechanisms (e.g. vasoconstriction) are triggered. Loss of control of the arteriovenous shunting, which is critical to maintaining a normal core body temperature, results in a rapid redistribution of heat from the core to peripheral tissues following induction of general anaesthesia, and a subsequent decrease in core temperature occurs. During the first hour of anaesthesia, heat redistribution is responsible for 80% of the observed reduction in core temperature. 26
Hypothermia begins with the induction of general anaesthesia and is difficult to reverse once established. 27 Initiating surface warming before induction of general anaesthesia, ‘pre-warming’, is successful at delaying the onset of hypothermia during anaesthesia by reducing the core-periphery temperature gradient.9,27,28 This technique delays the consequences of the redistribution of blood from the core to the periphery; however, the effectiveness of this temperature management strategy has not been previously assessed in rodents.
The primary objective of this study was to compare the effect of different temperature management strategies on peri-anaesthetic core body temperature in rats. We hypothesized that warming rats prior to anaesthesia (pre-warming) with forced-air warming would prevent peri-anaesthetic hypothermia and would speed recovery.
A secondary objective was to compare rectal, skin, and tail temperature measurements as proxies for core temperature.
Animals
Female (n = 12) and male (n = 12) Sprague–Dawley rats, aged 5–6 weeks, were obtained from Charles River, Canada, or were surplus stock from the University of Calgary Animal Resource Centre.
Materials and methods
This study was approved by the University of Calgary Health Sciences Animal Care Committee, which operates in accordance with the guidelines of the Canadian Council on Animal Care (protocol ID: AC15-0062).
The rats were allowed to acclimatize to the housing facility for a minimum of seven days before surgery. Housing consisted of a plastic cage (47 × 25 × 21 cm) with wood chips, shredded paper, and paper rolls as enrichment. The rats were housed in a controlled temperature (23℃) and humidity (22%) environment, with a 12 h/12 h light/dark cycle with lights on at 07:00 h. All animals were housed together with a cagemate that received surgery on the same day. Food (Prolab 2500 Rodent 5P14, LabDiet; PMI Nutrition International, St Louis, MO, USA) and tap water were provided ad libitum. The rats were habituated to the surgeon (CS) for 3–5 days before surgery, and were considered to be habituated when they readily accepted a cereal treat offered by hand. The rats were placed into the anaesthesia induction chamber for 5–10 min with a cereal treat for 3–5 days before the experiment without exposure to anaesthetic gas or warmed air.
The experiment consisted of two phases: surgical implantation of the telemetric temperature capsule followed by the hypothermia experiment. The animals were block-randomized by sex (random.org), with eight animals per treatment group, before entering the first phase. A sample size of approximately nine animals per treatment group was calculated for an alpha of 0.05 and a power of 80% to identify a mean difference of up to 2℃ between groups with a standard deviation of 1℃ (G*Power 3.1.9.2). 29
Telemetric temperature capsule implantation
On the morning of the surgery, capsules (VitalSense; Philips Respironics, Koninklijke Philips NV, Amsterdam, The Netherlands) were activated and placed in a 37℃ water bath for 5 min to confirm their accuracy. Bath temperature was set with a calibrated thermometer (calibrated at 37℃ with a reading resolution of ±0.1℃) equipped with an immersion probe (Fluke infrared thermometer model 561 and Fluke 80PK-22 SureGrip Immersion Temperature Probe; Fluke Corporation, Everett, WA, USA). The capsules were subsequently submersed in 70% alcohol for a minimum of 30 min, then rinsed with sterile saline (0.9% NaCl) before implantation. The telemetry monitoring unit (VitalSense) was kept within one metre of the activated capsules throughout the study, according to the manufacturer’s instructions.
One researcher (CS) completed all the surgeries and conducted the experiments. Each rat was given buprenorphine (0.03 mg/kg SC, Vetergesic, 0.3 mg/mL; Champion Alstoe, Whitby, ON, Canada) and meloxicam (2 mg/kg SC, Metacam solution for injection, 5 mg/mL; Boehringer Ingelheim Vetmedica, Inc, St Joseph, MO, USA) 30 min before the induction of general anaesthesia. The rats were individually placed into a plexiglass induction chamber (25.5 [l] × 10 [w] × 12 [h] cm) and anaesthesia was induced with 5% isoflurane in oxygen (1 L/min) until loss of righting reflex occurred. The rats were then placed in dorsal recumbency on an absorbent pad on top of a heating pad (HotDog patient warming system; Augustine Biomedical and Design, Eden Prairie, MN, USA) set at 40℃, and anaesthesia was maintained with isoflurane (vaporizer setting of approximately 1.75%) delivered through a nose cone. The surgical site was prepared for surgery by clipping fur from the abdomen (pubis to xiphoid) and cleaning the skin with chlorhexidine and alcohol. A sterile drape was placed on the abdomen and a negative pedal withdrawal reflex was confirmed before starting surgery. A laparotomy was performed through a midline incision (approximately 15 mm long) and the capsule was inserted into the abdomen. The incision was closed in two layers with simple continuous (muscle) and intracuticular (skin) patterns. Following closure, isoflurane was stopped and each rat recovered with oxygen (1 L/min) on the heating pad until sternal recumbency was achieved. The rats were returned to their home cage and closely monitored for one hour postoperatively. All surgeries were performed between 14:00 and 16:00 h. The amount of food consumed per cage and the weight of each animal were measured every 24 h. Meloxicam (2 mg/kg) was administered subcutaneously at 24 and 48 h following surgery. After surgery, the rats were supplemented with an edible gel diet (DietGel Recovery; Clear H2O, Portland, ME, USA), which was provided to facilitate recovery and to encourage postoperative food consumption. Only rats showing an increase in body weight over the five days of recovery were entered into the hypothermia experiment.
Hypothermia experiment
The three treatment groups were: no-warming (NW), limited-warming (LW), and pre-warming (PW). All experiments were performed on the sixth day after capsule implantation between 09:00 and 11:00 h.
The animals were individually anaesthetised with isoflurane in oxygen as described above. After the loss of their righting reflex, the rats were kept in dorsal recumbency and anaesthesia was maintained for 40 min, after which isoflurane was stopped and they were allowed to recover as described above. Core (capsule) temperature was recorded every 5 min from the loss of the righting reflex until 120 min after recovery (return of sternal recumbency). Additionally, skin temperature at the xiphoid process and the lateral aspect of the tail base (infrared thermometer), and rectal temperature (thermometer inserted 6 cm into the rectum, Physio Logic Accuflex Pro, Model 16-639; AMG Medical, Montreal, QC, Canada) 30 were measured every 5 min during anaesthesia from the loss of the righting reflex until recovery. Rectal thermometer accuracy (reading resolution ± 0.1℃) was assessed using the method described for telemetry capsules (water bath temperatures of 32 and 37℃), and a correction factor was applied.
NW group
The rats were placed on an absorbent pad on the surgical table for the 40 min anaesthetic period, with no external heat source provided.
LW group
During the 40 min anaesthetic period and until return of sternal recumbency, each rat was placed on the same heat pad as that used for surgery, set at 40℃ (measured output of 36.9℃).
PW group
The anaesthesia induction chamber was pre-heated for 20 min with warm air delivered from a forced air warming system (Bair Hugger warming unit, Model 505; 3M Health Care, St Paul, MN, USA) at the highest setting (43℃). The induction chamber was empty during the first 10 min, then a rat was added for the remaining 10 min. The temperature of the inner surface of the chamber was measured with the infrared thermometer immediately after adding the rat and again 5 and 10 min later. At the end of this period, the core and skin temperatures (lateral tail base and xiphoid process) were recorded, the warm air was stopped, and anaesthesia was induced as described above. Following loss of the righting reflex, each animal was placed on the heat pad as described for the LW group.
Baseline temperatures for all animals were calculated by averaging the capsule temperatures between 08:00 and 18:00 h on the day before surgery. This temperature was used as the comparator for calculating the percentage temperature change over the course of the hypothermia experiment. The study design did not allow for blinding to treatment.
Statistical analysis
Temperature data were exported from the monitoring unit to an electronic spreadsheet, and then imported into a statistical software program (GraphPad Prism 6.0f; GraphPad Software Inc, San Diego, CA, USA) for analysis. Within-group comparisons were analysed with a one-way analysis of variance (ANOVA) for repeated measures and a Dunnett’s post hoc test to compare each time point with baseline. Comparisons between warming strategies were performed with a two-way ANOVA for repeated measures and a Tukey’s post hoc test. Between-group comparisons for time to achieve sternal recumbency following anaesthesia and time from start of anaesthesia to loss of the righting reflex were carried out using a Kruskal–Wallis test and a Dunn’s multiple comparison test. Agreement between rectal/skin/tail and core (capsule) temperatures was assessed using the Bland and Altman method, 31 with the criterion method (capsule) subtracted from the new method (rectal/skin/tail), so that a negative bias represents an underestimation of capsule temperature by the new method and vice versa. A P value of < 0.05 was considered to be significant.
Results
Due to unforeseen technical failures, nine rats were excluded as the capsules stopped transmitting data at unpredictable intervals during the recovery period following surgery. One rat was euthanized following dehiscence of the abdominal skin incision, and another was excluded as it failed to gain weight in the five days following the implantation surgery. To allow study completion with equal group sizes, five additional rats were allocated (block randomization) to the treatment groups so that 18 rats (182–323 g) completed the experiment, with six rats per treatment group. The PW and LW groups consisted of three males and three females each, while the NW group consisted of two males and four females. All the excluded animals were euthanized in accordance with CCAC guidelines (anaesthetized with isoflurane, followed by carbon dioxide after loss of the righting reflex). Data used for analysis are available from a repository (Pang, Daniel, 2017, ‘rat hypothermia’, doi:10.7910/DVN/HJ8LIF, Harvard Dataverse, V1, UNF:6:L//oHDbw9nYXf8UARLzxQw==).
The mean core temperature of all rats the day before the experimental day was 37.5 ± 0.53℃.
In the PW group, the mean chamber temperature when the rat was first added (after 10 min of warming) was 35.7 ± 3.5℃. This increased after a further 5 and 10 min to 36.3 ± 3.8℃ and 37.5 ± 2.6℃, respectively.
Pre-warming was successful at raising core body temperature before general anaesthesia (P = 0.04, Figure 1, Table 1). The PW group maintained significantly higher core body temperatures than both the NW and the LW treatment groups (Figure 1). The heating pad was effective at maintaining core temperatures close to baseline throughout anaesthesia in the LW group (with mean capsule temperatures lying within approximately 1% [0.38℃] of baseline). In contrast to the PW and LW groups, the NW group exhibited a rapid, linear drop in temperature from loss of the righting reflex until recovery, with a mean temperature reduction of 11.4% (approximately 4.1℃) at the nadir (Figure 1).
The percentage change relative to baseline (37.5℃) in the no-warming (NW), limited-warming (LW), and pre-warming (PW) treatment groups. The grey box indicates the period of general anaesthesia, delimited by dashed vertical lines indicating loss of the righting reflex and turning off of the vaporizer. Matching letters denote significant differences between groups: d (P < 0.0001); c (P < 0.001); b (P < 0.01); a (P < 0.05). A 1% change is approximately 0.38℃. Comparisons within each treatment group between each time point and baseline. Significant differences are set in italics. CI: confidence interval.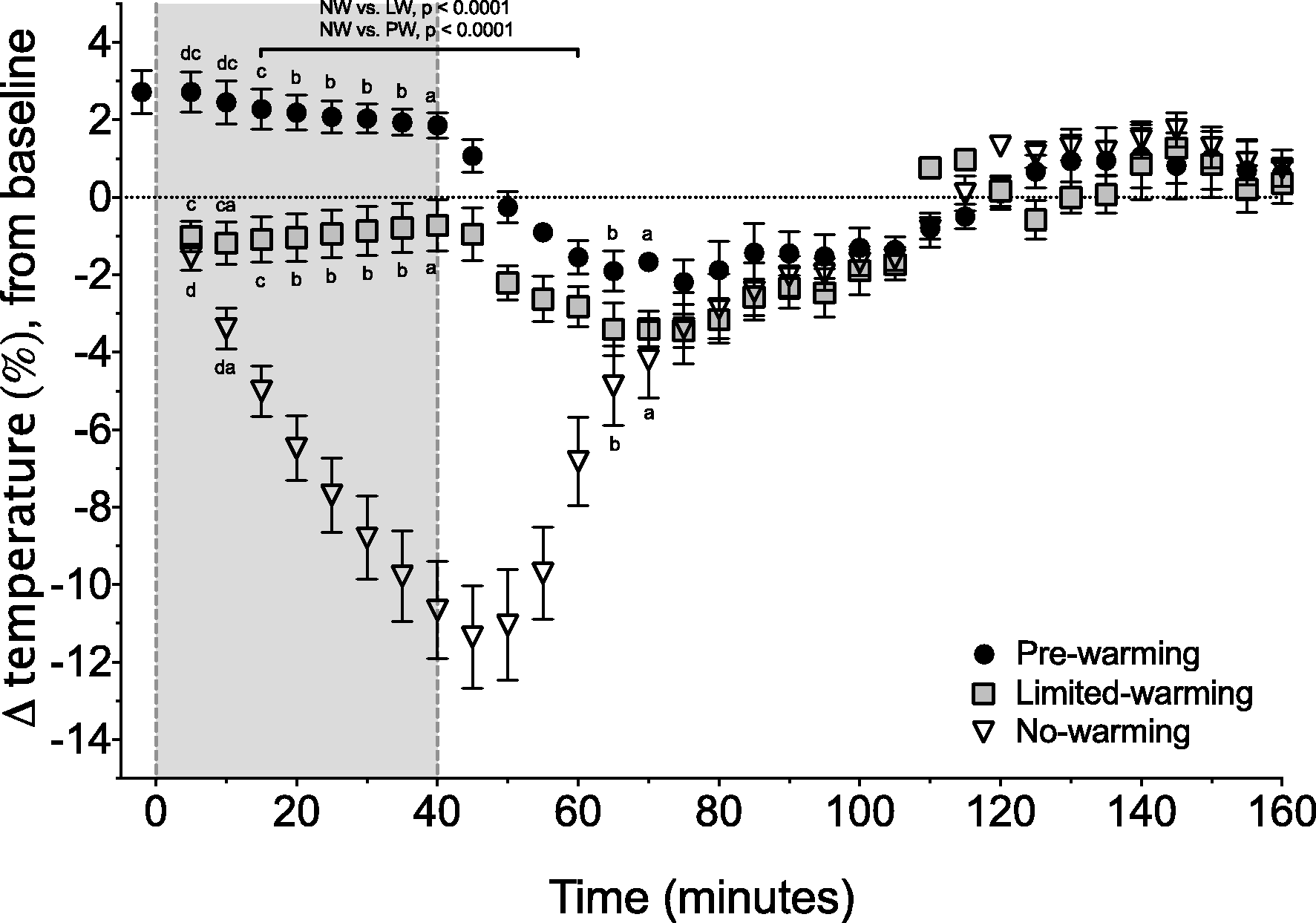
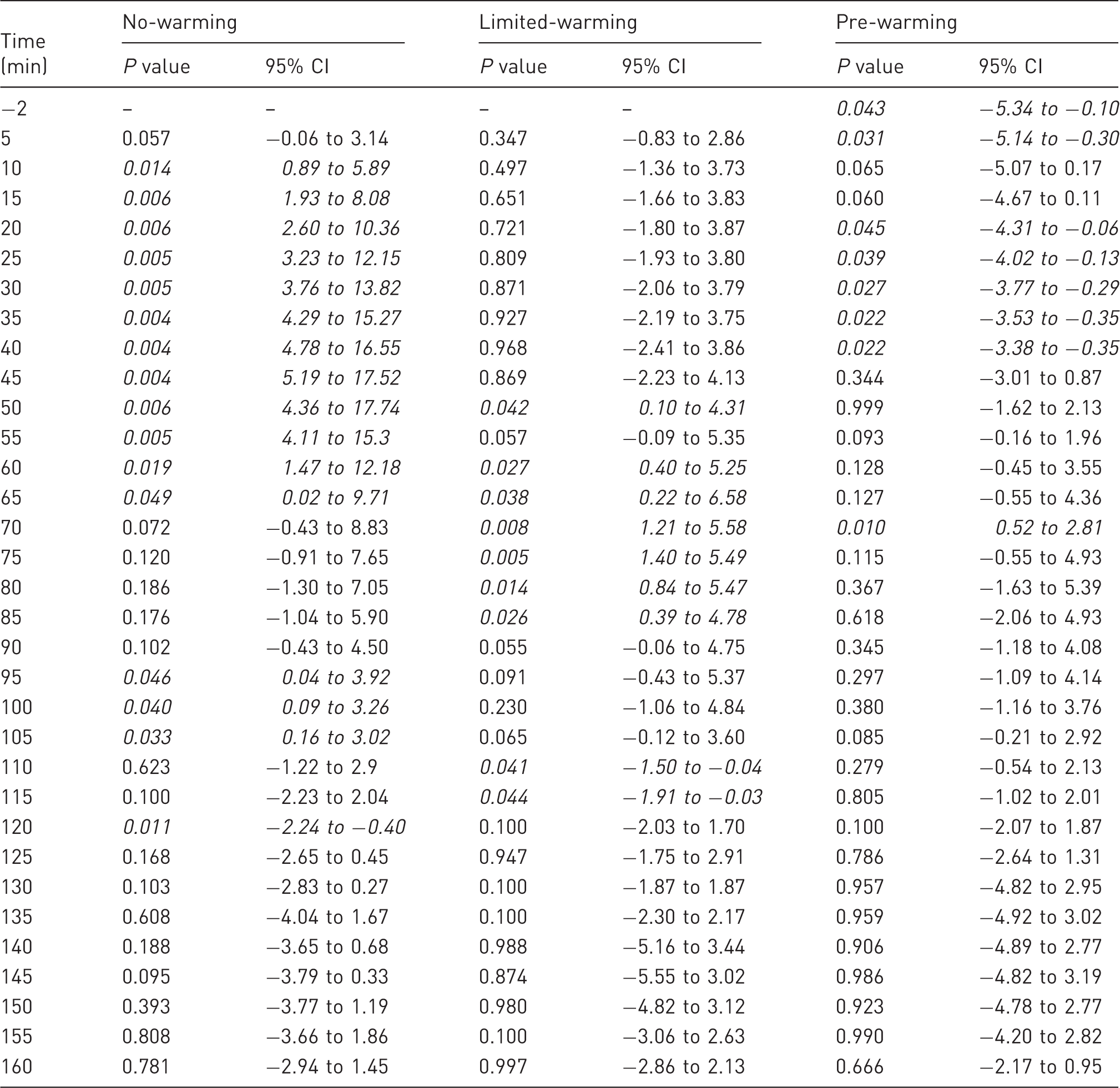
Following anaesthesia, temperatures from all groups converged over a 25 min period, with significant reductions compared with baseline in the NW and LW groups (Table 1). The NW group maintained core temperatures that were significantly lower than the other groups for 20 min after the end of anaesthesia.
Recovery to sternal recumbency was fastest in the PW group (125 [70–186] s), approximately five times faster that the NW group (525 [229–652] s, P = 0.0003, Figure 2). Return to sternal recumbency was also faster in the LW group (188 [169–420] s, P = 0.04) than the NW group by approximately 2.5 times. Time to sternal in the PW group was not statistically different from the LW group (P = 0.117). Time from start of anaesthesia to loss of the righting reflex did not differ between groups: pooled median time of 105 (85–141) s, P > 0.99 for all comparisons.
Time to achieve sternal recumbency was four times faster in the pre-warming group (PW) than the no-warming group (NW). The difference between PW and the limited-warming group (LW) was approximately one minute. Data are presented as individual data points with median and interquartile range.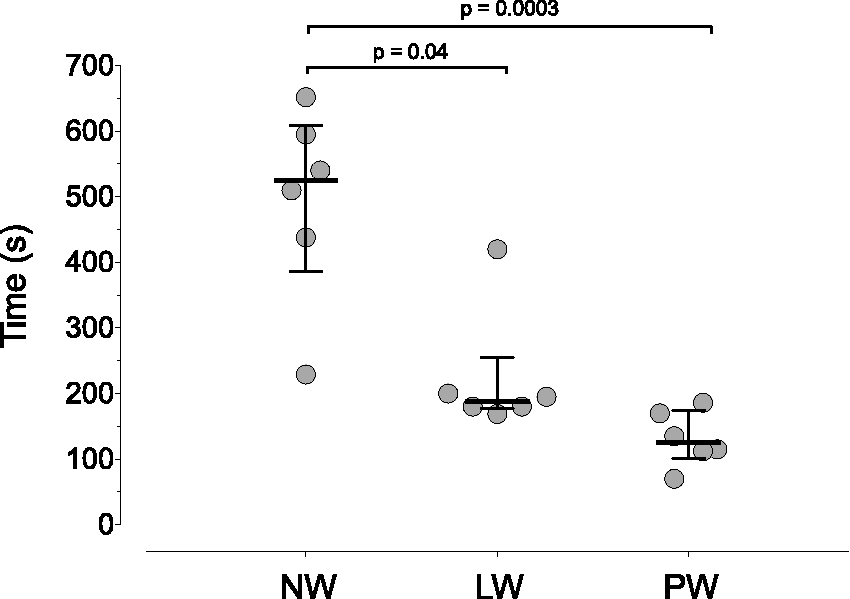
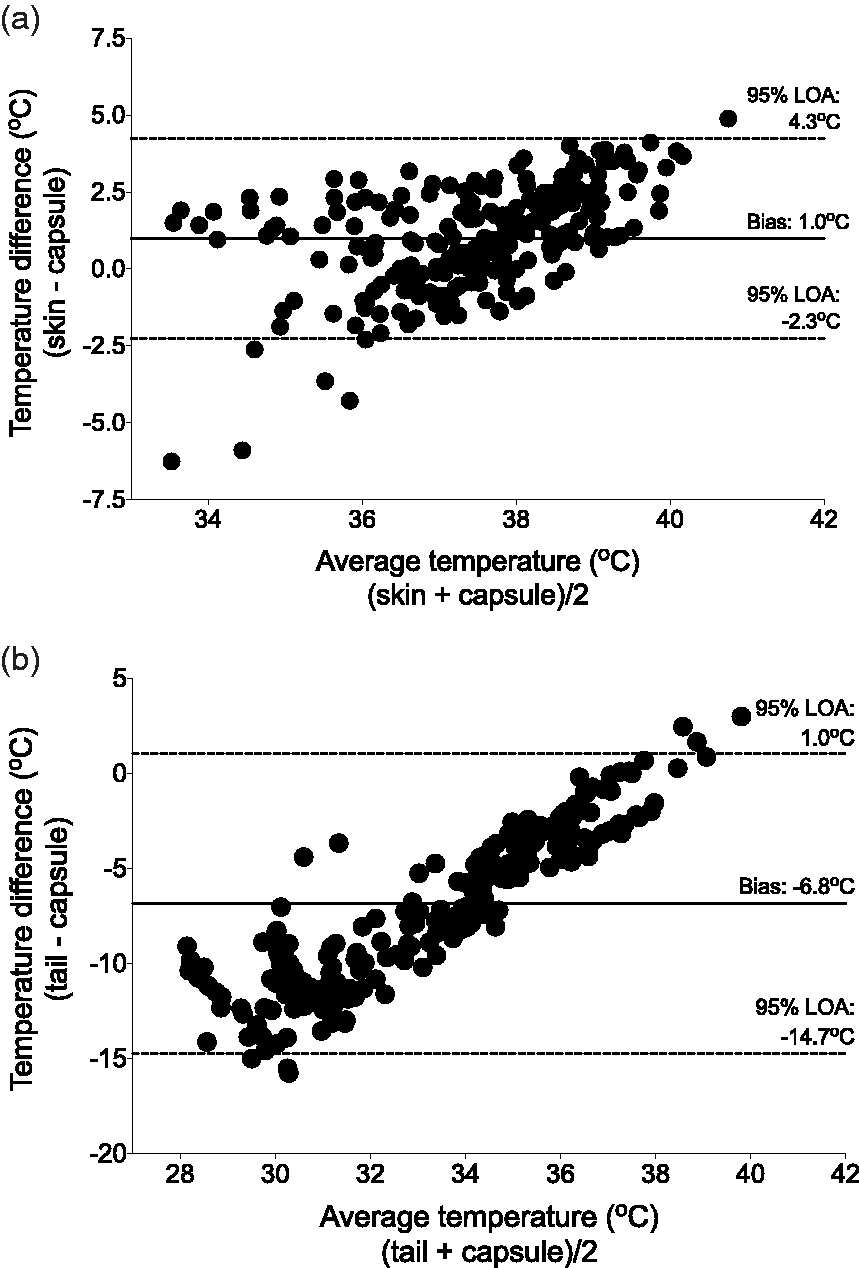
Rectal temperatures underestimated capsule temperatures (bias –0.9℃, 95% limits of agreement 0.1 to −1.9℃, Figure 3), and this pattern was maintained when the different treatment groups were analysed individually (Supplemental Figure S1, see http://journals.sagepub.com/doi/full/http://journals.sagepub.com/doi/full/10.1177/0023677217712539 for all supplementary data). Skin temperatures underestimated capsule temperatures with a similar bias (−1.0℃), but the 95% limits of agreement were wider and the presence of a positive trend indicates the presence of a proportional difference between the measurement methods (4.3 to −2.3℃, Figure 4). Tail temperatures showed the largest bias (−6.8℃) and the widest 95% limits of agreement (1.0 to −14.7℃, Figure 4).
Bland and Altman plot of rectal temperature versus telemetric capsule temperature. Rectal temperature underestimates core (capsule) temperature by 0.9℃, with 95% limits of agreement (LOA) ranging from 0.1 to −1.9℃. Bland and Altman plots of skin (a) and tail (b) temperatures versus telemetric capsule temperature. Skin temperature overestimates core (capsule) temperature by 1.0℃, with 95% limits of agreement (LOA) ranging from 4.3 to −2.3℃. Tail temperature underestimates core (capsule) temperature by 6.8℃, with 95% LOA ranging from 1.0 to −14.7℃.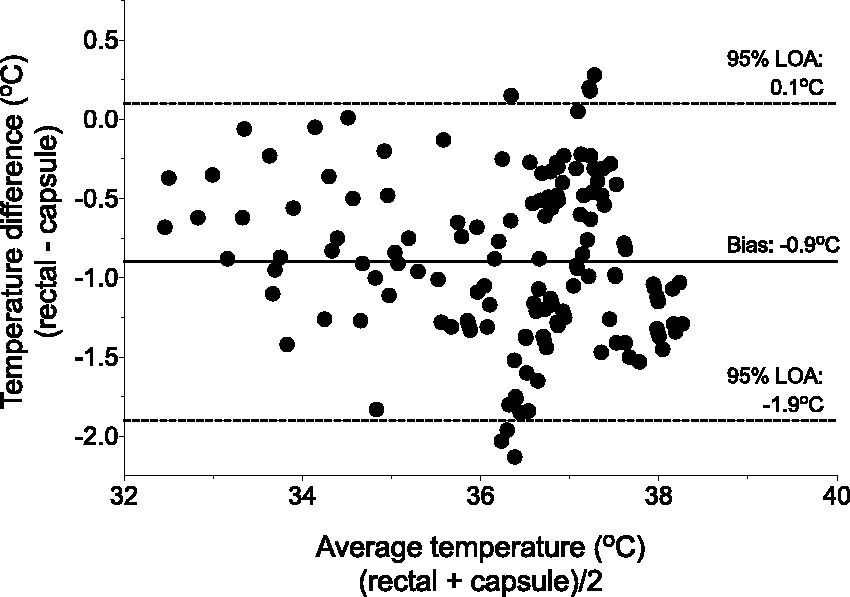
Discussion
Hypothermia is a common side-effect of general anaesthesia in mammalian species and prolongs anaesthetic recovery.14,16,17 In laboratory rodents, poor control of peri-anaesthetic temperature has been shown to have negative effects on data quality.18,20 Efforts to maintain normothermia in these species have had mixed successes, with difficulty maintaining normothermia while mitigating the risk of thermal burns.5,6,32 The results presented here show that: (1) PW raises core temperature, and prevents hypothermia during and after anaesthesia; (2) LW is effective at maintaining core temperature during anaesthesia, but with a risk of hypothermia during recovery; (3) warming before or during anaesthesia, or both, results in a shorter recovery period; and (4) proxy measures of core temperature vary considerably in accuracy and agreement.
In the present study, a PW period of 10 min was successful at preventing hypothermia in rats during the subsequent 40 min of general anaesthesia by maintaining core body temperature above baseline. In humans, PW that delays hypothermia successfully during anaesthesia does not require significant increases in core body temperature.27,33 Raising skin temperature by 4–5℃ (forced air warmers set at 40 or 43℃) only raised core temperatures by 0.3–0.5℃, yet was effective at maintaining core temperature for up to 3 h of anaesthesia.27,33 Our observation, that core temperature increased by approximately 1℃, indicates that a shorter or less aggressive PW protocol may be equally effective at preventing hypothermia. As the anaesthetic period was limited to 40 min, the effective duration of the PW strategy is unknown. Behavioural observations were not recorded during PW; however, rats typically settled quietly in the chamber after briefly investigating the warm air inlet and no escape behaviours were observed.
There have been mixed reports of the efficacy of external heat sources in preventing hypothermia in rats.5,6 Taylor 6 showed a heat pad (surface temperature of 37.2–38.9℃) to be effective at raising rectal temperature by up to 0.96 ± 0.1℃. If a similar underestimation of core body temperature by rectal temperature existed, as identified in this study, this indicates that a heat pad may be effective at maintaining core temperature for the studied anaesthetic period of 60 min. However, as rectal thermometer insertion depth was not reported, the relationship between rectal and core temperature is unclear. Thermometer insertion depth has a considerable effect on the accuracy of measured temperatures, with a minimal insertion depth of 5–6 cm required to reflect core temperature in rats weighing 200–300 g. 30 Recent work that evaluated a heated (40℃) surface for maintenance of normothermia found this strategy to be largely ineffective, with rectal temperatures decreasing from a mean of 37℃ to 31℃ over a 60 min anaesthetic period. 13 However, the short rectal thermometer insertion depth of approximately one centimetre limits quantitative interpretation of the results.
In contrast, Albrecht et al. have shown that a warm water heating pad maintained core body temperature (measured with intra-abdominal telemetry) close to, but slightly below (approximately 0.6℃), baseline temperatures during anaesthesia with isoflurane, ketamine–xylazine, or medetomidine–midazolam–fentanyl. 5 This difference is similar to the 2% decrease observed in the present study in the LW group.
The linear decrease in temperature observed in the NW group is similar to that reported in other studies, emphasizing the rapid onset and potential for substantial hypothermia when no external heating sources are applied.6,13 The consequences of hypothermia remain relatively unexplored in laboratory rodents compared with humans, though detrimental effects on data quality have been identified in mice.18,20 Marschner et al. have shown that placing mice in a warm ventilated chamber between injection of anaesthetic drugs and induction of anaesthesia successfully maintained rectal temperature within the target range of 99.3% of temperature recordings. 20 This improvement in temperature resulted in a reduction in data variability.
Overall, the varied reports in the literature indicate that maintenance of normothermia cannot be assumed based on provision of an external heat source, with wide variation in the efficiency and safety of the heat sources. 32 Reporting of results could be improved with clear descriptions of rectal thermometer insertion depth, heat source temperature output, and equipment calibration.
Unexpectedly, we observed hypothermia in the LW group during recovery from anaesthesia, persisting for approximately 30 min. This emphasizes the importance of continuing the warming procedures during the recovery phase. As studies typically focus on temperature management during the intra-anaesthetic period, the incidence of post-anaesthetic hypothermia in rodents is unknown.
Recovery to sternal recumbency was significantly faster with either of the warming strategy than with NW, which was consistent with previous studies in mice and dogs.14,17 In humans, the documented consequences of perioperative hypothermia (as little as a 1℃ decrease) are widespread, including prolonged recovery and hospitalization, increased surgical site infection, haemorrhage, and thermal discomfort.7–9,23,34 To what extent these are relevant to veterinary medicine is unclear as the evidence base is limited, although it is reasonable to assume that effective temperature monitoring and management is an important part of peri-anaesthetic care.
Free-moving telemetry capsules in the abdomen provide an accurate measurement of core temperature without the confounding effect of handling causing an increase in temperature.19,35 However, using intra-abdominal telemetric monitoring increases study cost and complexity. Identifying a non-invasive proxy for core body temperature is appealing for routine peri-anaesthetic temperature monitoring. These results suggest that rectal temperatures are a suitable substitute for core body temperature over the temperature range studied (approximately 32.5–38.0℃). The systematic underestimation of core temperature by 0.9℃ and limited excursion of the upper limit of agreement (0.1℃) means rectal temperature is unlikely to overestimate core temperature and miss the presence of hypothermia. As mentioned previously, thermometer insertion depth should not be less than 5–6 cm to ensure accuracy and consistency of readings. 30
Skin temperature showed a similar bias as rectal temperature but with 95% limits of agreement approximately three times wider. The possibility of overestimating core body temperature by over 4℃ raises the risk of failing to identify hypothermic animals. Similarly, tail temperatures inconsistently reflected core temperatures, with large underestimations at low temperatures.
An unexpected and disappointing complication encountered was the frequency of capsule failure, resulting in the exclusion of nine rats at variable intervals after implantation surgery. This problem was not known before starting the study. Fortunately, the low variability in collected data and the large effect size between the NW and other treatment groups allowed identification of statistically significant and meaningful differences.
Conclusion
Peri-anaesthetic warming through PW and LW strategies was effective in preventing hypothermia during the anaesthesia period, though hypothermia occurred during recovery in the LW group. Pre-warming increased core body temperatures throughout the 40 min anaesthesia period and the use of a shorter PW protocol may be equally effective. Rectal temperature recordings underestimated core temperatures by 0.9℃ with narrow limits of agreement. In contrast, neither skin nor tail temperatures provided acceptable approximations of core body temperature.
Footnotes
Acknowledgements
We wish to thank Dr Julie Reimer and Cassandra Klune for providing assistance during surgery, Dr Amy Larkin for guidance on surgical technique, and Guy Jennings (Bio-Lynx Scientific Equipment Inc) for technical service and support.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by a Natural Sciences and Engineering Research Council of Canada Discovery Grant awarded to DP (424022-2013). CS received a travel fellowship from the Canadian Association for Laboratory Animal Medicine to present preliminary findings at the Canadian Association for Laboratory Animal Science 55th Annual Symposium.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
