Abstract
This study aims to assess the thermal homogeneity of the intraperitoneal (IP) cavity and the relevance of using a fixed telemetric temperature sensor at a given location in studying rodents. Ten rats were intraperitoneally implanted with three Jonah® capsules each; after assessing the accuracy and reliability of the sensors. Two capsules were attached, one to the right iliac fossa (RIF) and the other to the left hypochondrium (LH), and another was placed between the intestines but not attached (Free). In the ex vivo condition, the differences between sensors and reference values remained in the range of ±0.1℃. In the in vivo condition, each sensor enabled the observation of temperature patterns. However, sensor location affected mean and median temperature values while the rats were moving freely. Indeed, temperature data collected in the LH were 0.1℃ significantly higher than those collected in the RIF and temperature data collected in the LH were 0.11℃ significantly higher than those collected with the Free capsules. In in vivo conditions, intra-sensor variability of temperature data was not affected by sensor location. Taking into account sensor accuracy, similar intra-sensor variability, and mean differences observed between the three locations, the impact of sensor location within the IP cavity could be considered negligible. In in vivo conditions, temperature differences between locations regularly exceeded ±0.2℃ and reached up to 2.5℃. These extreme values could be explained by behavioral factors such as food or water intake. Finally, considering the good thermal homogeneity of the IP cavity and possible adverse consequences of sensor attachment, it seems better to let sensors range free within the cavity.
Body temperature is a basic physiological marker, frequently measured during animal experiments, especially with rodents. The measurement site selected for data collection varies across studies according to their objectives, and the accuracy or reliability level needed. In fact, temperature could be over- or under-measured given the specificity of the effects of some pharmacological drugs, 1 environment temperature influences 2 or body temperature dissipation. 3 Thus, invasive measurement techniques are used for accurate brain temperature measurement, 4 while tail skin 5 or intra-abdominal 6 measurements are most often performed. Because of ease of use, temperature is generally performed in active rodents using rectal probe thermometers 7 which have to be inserted at least 6.5 cm deep into the rectum7,8 to ensure reliability. However, it has been reported that body temperature is seriously affected by this method of measurement; so much so that the stress caused may lead to a temperature increase of 0.6℃ to 0.7℃. 9 Furthermore, it is well established that monitoring unstressed animals is a necessary condition of guaranteeing physiological measurement quality. 10 In this context, telemetric devices, implanted subcutaneously or intraperitoneally, constitute an effective alternative solution, enabling (i) the improvement of animal welfare, (ii) the enhancement of data reliability by avoiding handling-induced factors, 11 and (iii) continuous temperature monitoring without additional care for the animals 9 which does not interfere with normal circadian rhythms. 12 All these factors contribute to improving the quality of data collected and thus allow a reduction of 60 to 70% in numbers of animals used. 13
Regarding the use of telemetric devices in animals, the influence of intraperitoneal (IP) versus subcutaneous telemetry implantation in young Mongolian gerbils has already been investigated by Moons et al. 14 The authors have reported that the IP technique is more invasive but less troublesome than the subcutaneous technique but they were not interested in the possible impact of device location in the IP cavity on the data collected. Although temperature is roughly the same throughout the body, some previous work has already demonstrated that sensor location can influence values measured. 15 Thus, factors such as the size of species, methods of thermoregulation, housing condition or ambient temperature are main determinants of consistency between measurement sites. The IP and subcutaneous temperatures in free-moving rhesus macaques have been compared; 15 and a good correlation was observed between sites, with consistent subcutaneous values about 0.5–0.8℃ lower than the IP measurements. Thus, it seems that IP temperature measurement is closer to the core temperature. 16 The thermal core consists of ‘…those inner tissues of the body whose temperatures are not changed in their relationship to each other by circulatory adjustments and changes in heat dissipation to the environment …’; 17 such as the deep body sites or the hypothalamus. 18 Its measurement is commonly required invasive procedures such as intra-abdominal radiotelemetry, 19 pulmonary artery catheterization, oesophageal probes, and urinary bladder catheterization.18,20 Although the rodent model is currently used in laboratory experiments, no studies have yet focused on thermal homogeneity in the species.
However, the recommendations related to rectal temperature measurements (6.5 cm deep) suggest the importance of thermal heterogeneity in rodents. Thus, even in IP measurement, the location of the sensor within the cavity could also affect the values collected. Although displacements of the sensor within the IP cavity are very limited; due to adhesions occurring with the device, it is usually fixed within the abdomen.21,22 Furthermore, a mean absolute difference of ±0.17℃ has previously been reported between non-attached sensors implanted intraperitoneally in rats. 23 The authors hypothesized that the rodents’ activities such as food/water intake and locomotion/sleep could affect each sensor differently depending on its location. 23
To our knowledge, no study has focused on the possible impact of sensor location within rats’ IP cavities on temperature values collected. In their studies, Green et al. 21 and Deveney et al. 22 have proposed IP device implantation without precisely indicating the fixation site used. Given the possible influence of the measurement site on collected data and the lack of obvious recommendations related to temperature measurement sites in the IP cavity, the present study questions the thermal homogeneity of the IP cavity and the importance of attaching the sensor at a given location. In order to achieve this objective we arbitrarily chose to fix the sensors to ‘two extremities’ of the IP cavity, with the third sensor remaining free in the middle of the cavity.
Materials and methods
This study was carried out in strict accordance with the European Communities Council Directive 86/6609/EEC as well as with French legislation. The study was approved by the regional ethical committee of Basse-Normandie (assigned number: N/0260412/10/04-15). As shown in Figure 1, the experimental procedure was divided into two main parts. First, an ex vivo assessment of the sensor’s accuracy and reliability was made. Second, in vivo measurements were performed after the sensor was implanted surgically.
Chronology of the experimental procedure.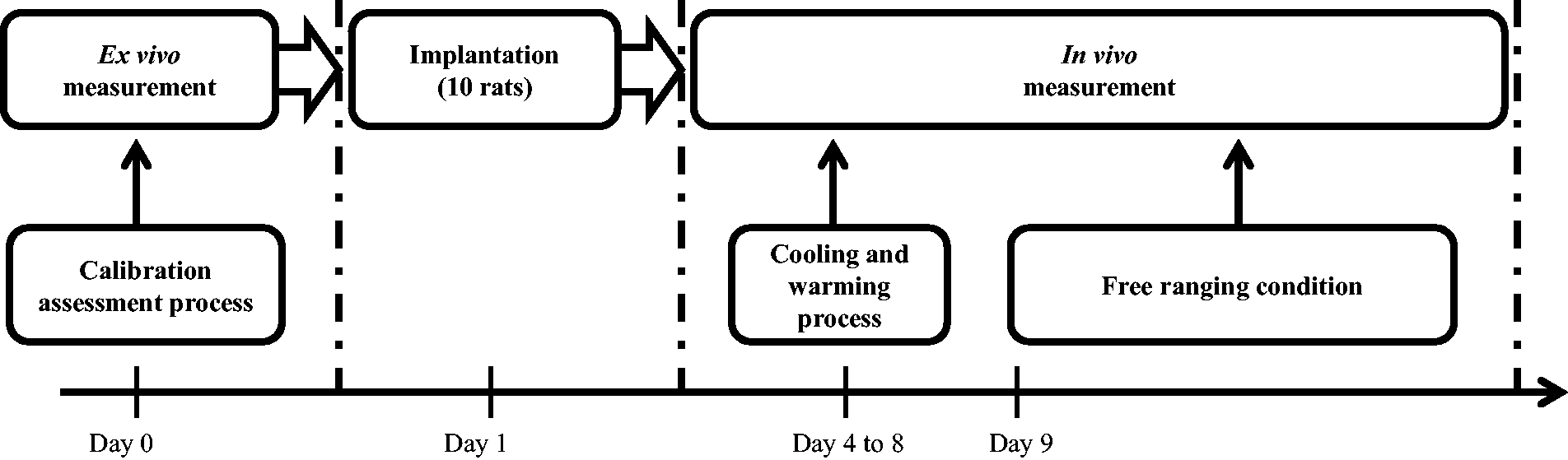
For the first part of the experiment, 30 Jonah® (Respironics, Herrsching, Germany) capsules were activated and immersed in a water bath successively heated to 35, 37 and 39℃. A thermostat bridge (JULABO®, model SE; Julabo, Seelbach, Germany) was placed on the bath along with a heating and stirring system. This thermostat bridge was equipped with an internal temperature sensor. A thermal sensor PT-100® (Julabo) was connected to the thermostat bridge; this traceable thermometer was used as the standard measure of the water bath temperature (external sensor). The whole (thermostat bridge and PT-100) having been calibrated as a system (COFRAC no. 2.1336; Certificate no. 83GG4576); the reported resolution of the thermostat bridge and PT-100 was 0.01℃ and the uncertainty budget of the system was 0.07℃. For each preset temperature, the data collection was performed for 20 min with a sample frequency of one minute.
For the second part of the experiment, 10 male Sprague Dawley rats (weight 636.7 ± 54.8 g) underwent an IP implantation of three Jonah® capsules under general anesthetic. An incision of 15 mm in length was performed and one of the three capsules was placed in between the intestines without any attachment (Free capsule). The other two were attached in the IP cavity with non-resorbable sutures, respectively in the left hypochondrium (LH capsule) and the right iliac fossa (RIF capsule). As previously described by Chapon et al., 23 isoflurane (3%, O2 flow rate =2 L/min; maintained at 2%, O2 flow rate =3 L/min) was used for the surgical procedure. For two days, each rat received a dose of proparacetamol (Perfalgan; Bristol-Myers Squibb Rueil-Malmaison, France) intraperitoneally (25 mg in 1 mL of saline solution) in order to decrease nociception caused by the laparotomy. They also received an IP dose of 50 mg/kg of amoxicillin and 6.25 mg/kg of clavulanic acid (Augmentin®; Sandoz, Princeton, NJ, USA) for three days.
After surgical implantation the animals were individually housed in transparent polycarbonate cages (Techniplast, Buguggiate, Italy) fitted with wood chip beddings (Special Diet Services, Saint-Gratien, France). Rodent laboratory food and water were provided ad libitum under standard laboratory conditions (21℃, 12–12 h light–dark reversed cycle).
Three days after implantation of the three capsules in each rat, continuous and simultaneous data recording started, with a sampling frequency of one minute. Between days 4 and 8 after the implantation procedure, each rat was injected in a random order with a 3 mg/kg dose of apomorphine hydrochloride to induce a temperature decrease or with a 25 µg/kg dose of lipopolysaccharide (LPS) of Escherichia coli (serotype 0111:B4) to induce a temperature rise. The objective of these drug injections was to explore a possible influence of site measurement to the pharmacological temperature variation induced. Drugs were prepared before the injections as previously described by Harkin et al. 24 Apomorphine hydrochloride was dissolved in a vehicle containing 0.89% NaCl with 1 mg/mL ascorbic acid added as an antioxidant. Escherichia coli LPS (serotype 0111:B4) was dissolved in 0.89% NaCl. Based on the results of Harkin et al., 24 the LPS was injected around 17:00 h and the apomorphine around 13:30 h in order to obtain a variation opposed to the normal kinetics associated with the circadian temperature rhythms. All injections were made in a volume of 1 mL/kg body weight and given subcutaneously. Later data was collected in the free ranging condition for a minimum period of 24 h during which no disturbing procedures were performed (Figure 1).
Data analysis and statistics
Soon after sensor implantation, the data collection was interrupted for rat 7, which might have been due to the low battery level of the Jonah® capsule resulting from too long a storage time before its activation. Therefore, the data analysis for the calibration and free ranging conditions was carried out with the data temperature corresponding to the nine remaining animals. Also, during the pharmacological apomorphine effect phase, the data collection for rats 5 and 6 was disturbed; therefore data analysis was carried out with the seven remaining animals for this phase.
All statistical analyses were carried out using R 2.15.0 software (www.r-project.org). In the calibration part, because of the large number of observations, Student’s t-test was utilized since the normality hypothesis was not rejected for any sample. For all other analyses, preference was given to non-parametric approaches (Friedman’s test, asymptotic general independence test) due to the robustness of these methods in abnormal circumstances and the limited number of observations. The level of statistical significance was set at P < 0.05. The method of non-linear least squares was utilized for determining parameter estimates of the cosinor function in the free ranging condition. Confidence intervals for amplitude, acrophase, batyphase, and mesor values, respectively, result from a non-parametric panel bootstrap experiment with 1000 repetitions. Comparison of temperatures recorded during the free range and apomorphine/LPS periods was carried out by a linear mixed model. In order to identify serial within-subject correlation, we investigated different correlation structures. Taking the Bayesian information criterion (BIC) as the selection criterion, the AR(1) error structure typical for time series was selected.
Results
Calibration assessment
For the three steps of the accuracy assessment at 35, 37 and 39℃, respectively, data collected using each sensor were compared with the reference temperature of the water bath. For each sensor, the t-tests indicated a significant difference to the reference temperature (P < 2.2 e–16), for all target values. It seems that e-Celsius® slightly underestimated the temperature and VitalSense® slightly but systematically overestimated the temperature in comparison with the reference temperature; however, the differences observed were margely lower than the accuracy degree of the devices. In fact, all differences remained in the target range of ±0.1℃.
Free ranging condition
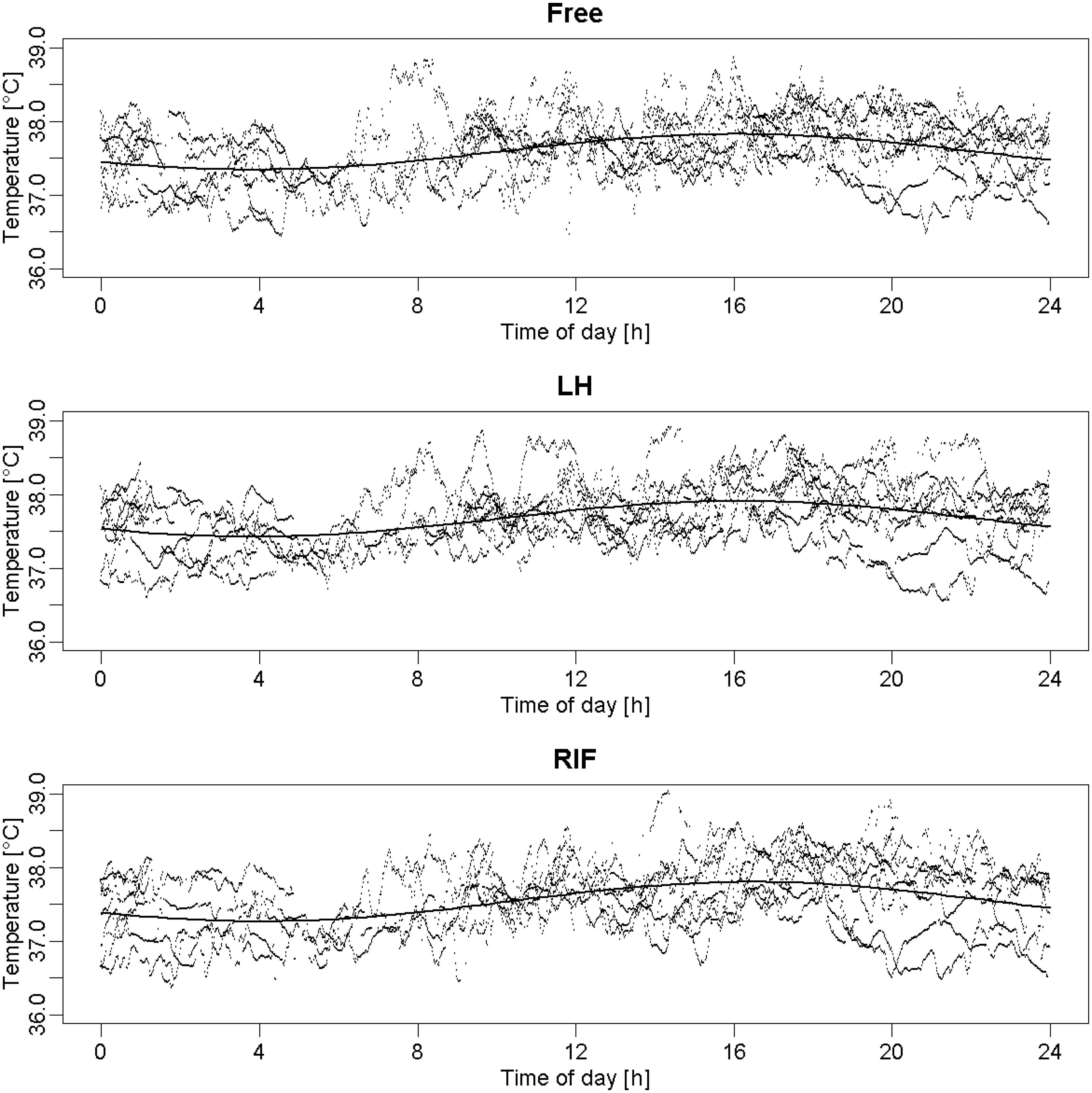
As an example, Figure 2 illustrates the circadian evolution of temperatures in rats 2 and 4. Raw data collected from each of the three Jonah® capsules enabled the modeling of a classic temperature pattern (Figure 3) with a function of the form: M + A * cos(omega * t + O).25,26 This model did not appear to be affected by the location of the capsules (Table 1), as the 95% intervals (below each point estimate) of the amplitude, acrophase, batyphase, and mesor, respectively, were all overlapping. Due to the heterogeneity of the rats and the low sample size, these intervals were rather large. However, one may note that the borders for each of the respective statistics lay relatively close together.
Circadian rhythmicity for rats 2 and 4 during the 24 h free ranging period. Raw temperature data issued from each of the three locations for each rat (Free, LH and RIF capsules) during the 24 h free ranging condition and their corresponding cosinor model (y-axis: temperature collected data [℃]; x-axis: daytime [h]). LH: left hypochondrium, RIF: right iliac fossa. Characteristics of the mean circadian model issued from each of the three locations. Amplitude: difference between the maximal and minimal values; acrophase: daytime of maximal value; batyphase: daytime of minimal value; and mesor: mean value during the day. LH: left hypochondrium, RIF: right iliac fossa.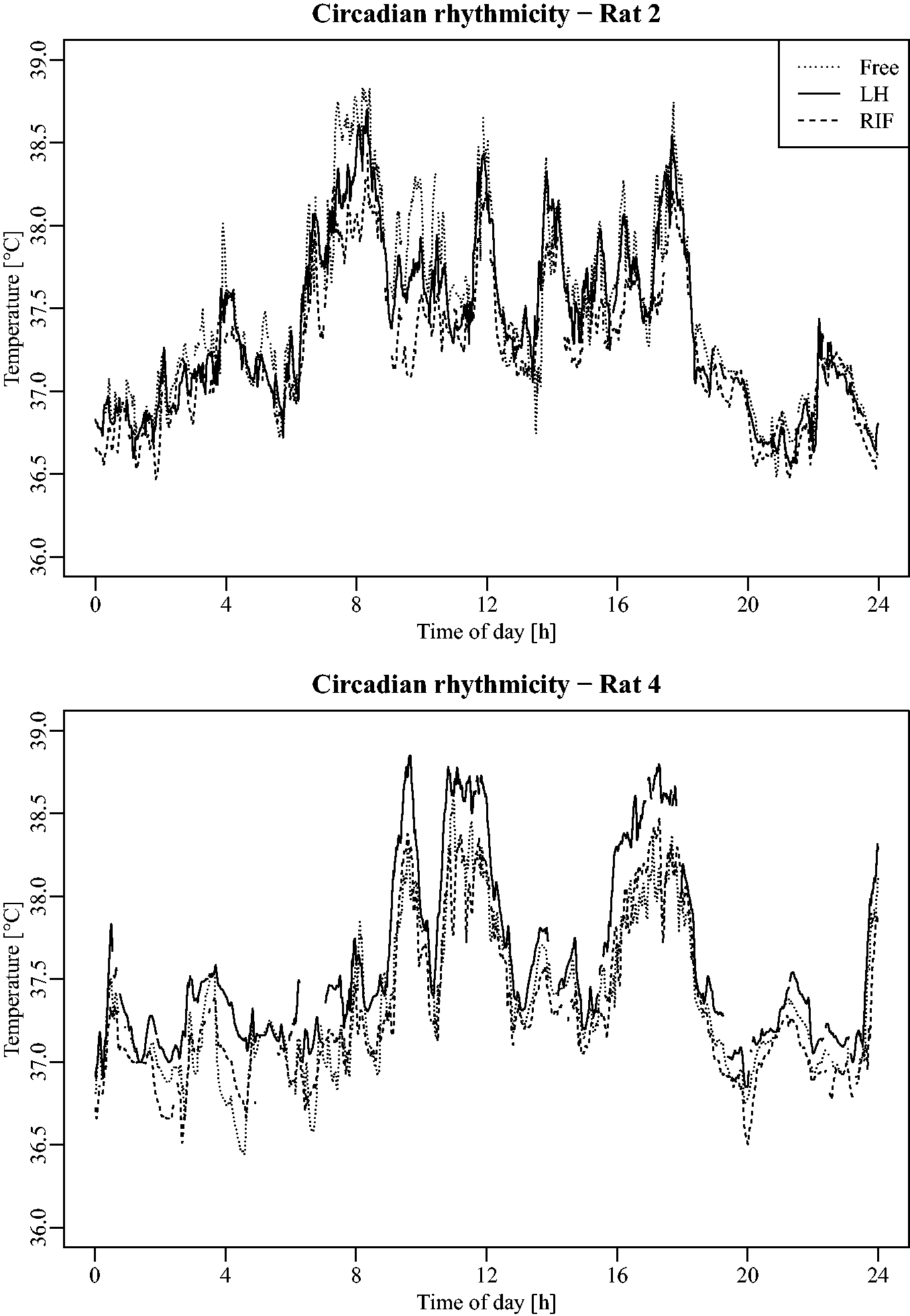

Regarding the reliability of temperature measurement, Figure 4 displays the temperature differences issued from the inter-sensor comparison calculated during the 24 h period of the free ranging condition. The results indicate a mean difference of 0.01 ± 0.14℃ between the Free and RIF capsules, 0.1 ± 0.12℃ between the LH and RIF capsules, and 0.11 ± 0.13℃ between the LH and Free capsules. The 90% interquartile range (IQR90) of these differences presents average values of 0.67 ± 0.16℃, 0.58 ± 0.18℃ and 0.55 ± 0.08℃, respectively, for the Free/RIF, LH/RIF and LH/Free capsule comparisons (Table 2).
Box plot displaying temperature differences issued from inter-sensor comparison for each rat during the 24 h free ranging condition (the three extreme values were deleted). The grey horizontal lines are located at ±0.1℃. Small circles represent observations outside the end of the whiskers. LH: left hypochondrium, RIF: right iliac fossa. Mean statistic for the temperature differences (℃) issued from location comparisons during 24 h of free ranging condition. NS: non-significant difference, LH: left hypochondrium, RIF: right iliac fossa, SD: standard deviation, IQR90: 90% interquartile range. *Indicates significant difference; P < 0.05.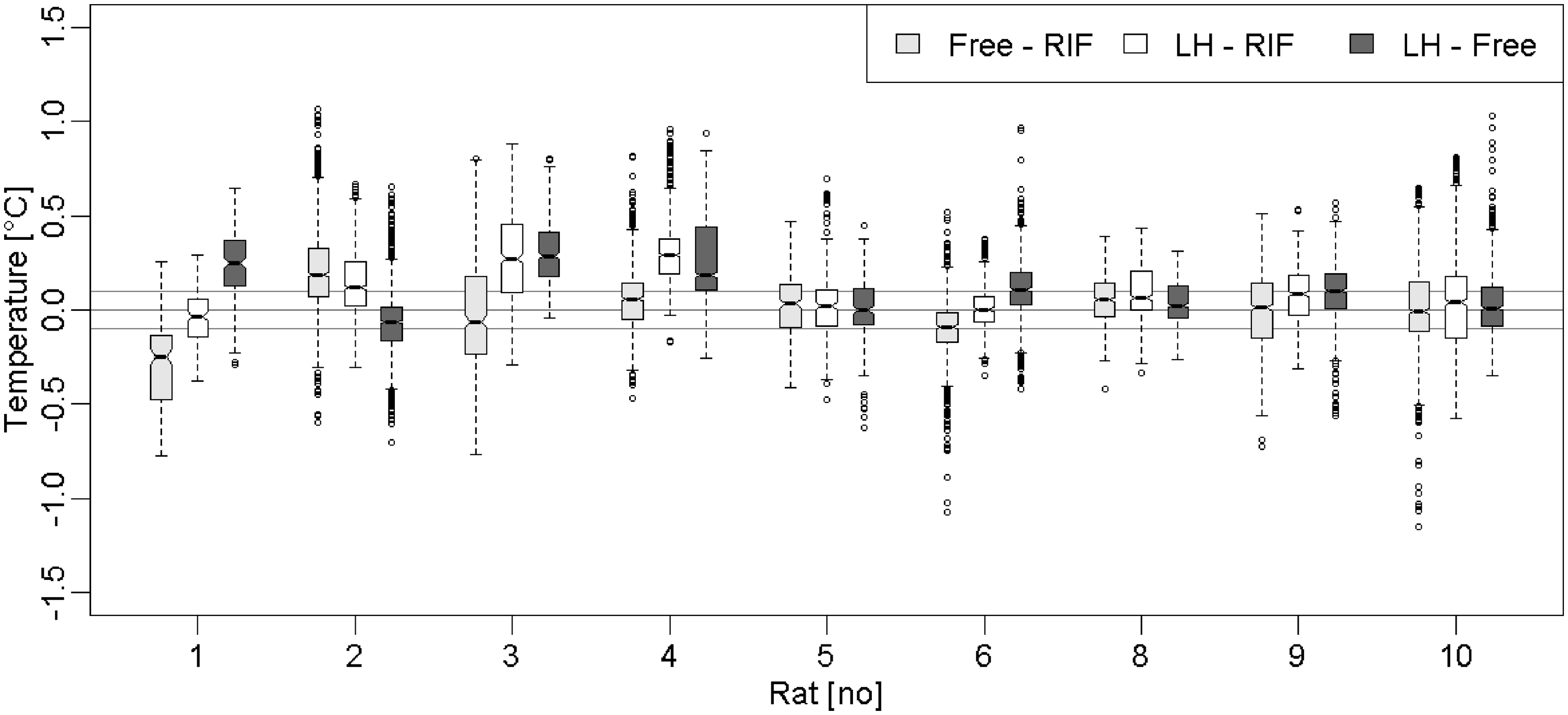

In contrast with the calibration assessment condition, most of the calculated differences exceed the range of ± 0.2℃; most of the values are included in the range of ±1℃ with the exception of three extreme values of up to ±2.5℃.
Further to this, we analyzed four statistics (mean, median, standard deviation, and IQR90) of the raw temperature data for each rat and location. Comparisons of these statistics using a Friedman’s test did not indicate any significant effects of capsule location on the standard deviation and IQR90 values (P = 0.72) during the course of the 24 h of free ranging. Conversely, a significant effect was observed on the mean (P = 0.013) and the median (P = 0.016) values. Post hoc analysis applied to the mean values using an asymptotic general independence test revealed that LH data were significantly higher than the RIF (+0.1 ± 0.12℃; P = 0.021) and Free (0.11 ± 0.13℃; P = 0.040) values; Free and RIF data were not significantly different (P = 0.97). As for the median, post hoc analysis revealed similar results: LH data were significantly higher than the RIF (0.1 ± 0.11℃; P = 0.026) and Free (0.10 ± 0.12℃; P = 0.048) values. Then again, Free and RIF data were not significantly different (P = 0.97).
Apomorphine and LPS injection
Figure 5 displays rats’ temperature values during the LPS and apomorphine phases. Graphical analysis indicates that the sensors enabled the observation of the pharmacological effects of the LPS and apomorphine injections. Regardless of the sensor and considering the level of pre-injection temperature, the apomorphine injection showed an average decrease of 1.5 ± 0.4℃ in animal temperature, while LPS a rise of 1 ± 0.6℃. For apomorphine, the involved temperature decrease was observed a few times after injection, and the mean minimal values reached were 36.32 ± 0.6, 36.55 ± 0.6 and 36.3 ± 0.6℃ for RIF, LH and Free capsules, respectively. Rats regained their initial temperature around 4 h after injection. As for the LPS effect, the temperature rise started 2 h after injection and the effect persisted for around 7–8 h. Implanted sensors indicated mean temperature peak values of 38.8 ± 0.7, 38.9 ± 0.6 and 38.9 ± 0.7℃, for RIF, LH and Free capsules, respectively, observed around 6 h after injection. In order to verify the pharmacological effects on body temperature, we compare the temperature values collected during the free ranging condition and the LPS/apomorphine effect phases. For this comparison, the temperature data corresponding to the most important pharmacological effects were considered: from 14:30 to 16:00 h for the apomorphine phase and from 22:00–00:00 h for the LPS phase. A linear mixed model revealed a significant difference between the temperatures collected at all three locations during the free ranging condition and the apomorphine and LPS phase, respectively (apomorphine – Free: 0.70℃, P < 0.05; LH: 0.63℃, P < 0.01; RIF: 0.64℃, P < 0.05. LPS – Free: 0.80℃, P < 0.001; LH: 0.66℃, P < 0.001; RIF: 0.69℃, P < 0.001).
Raw temperature data issued from the three locations (RIF, LH and Free capsules) during the LPS and apomorphine phase (y-axis: temperature collected data [℃]; x-axis: daytime [h]). LH: left hypochondrium, RIF: right iliac fossa, LPS: lipopolysaccharide of Escherichia coli (serotype 0111:B4).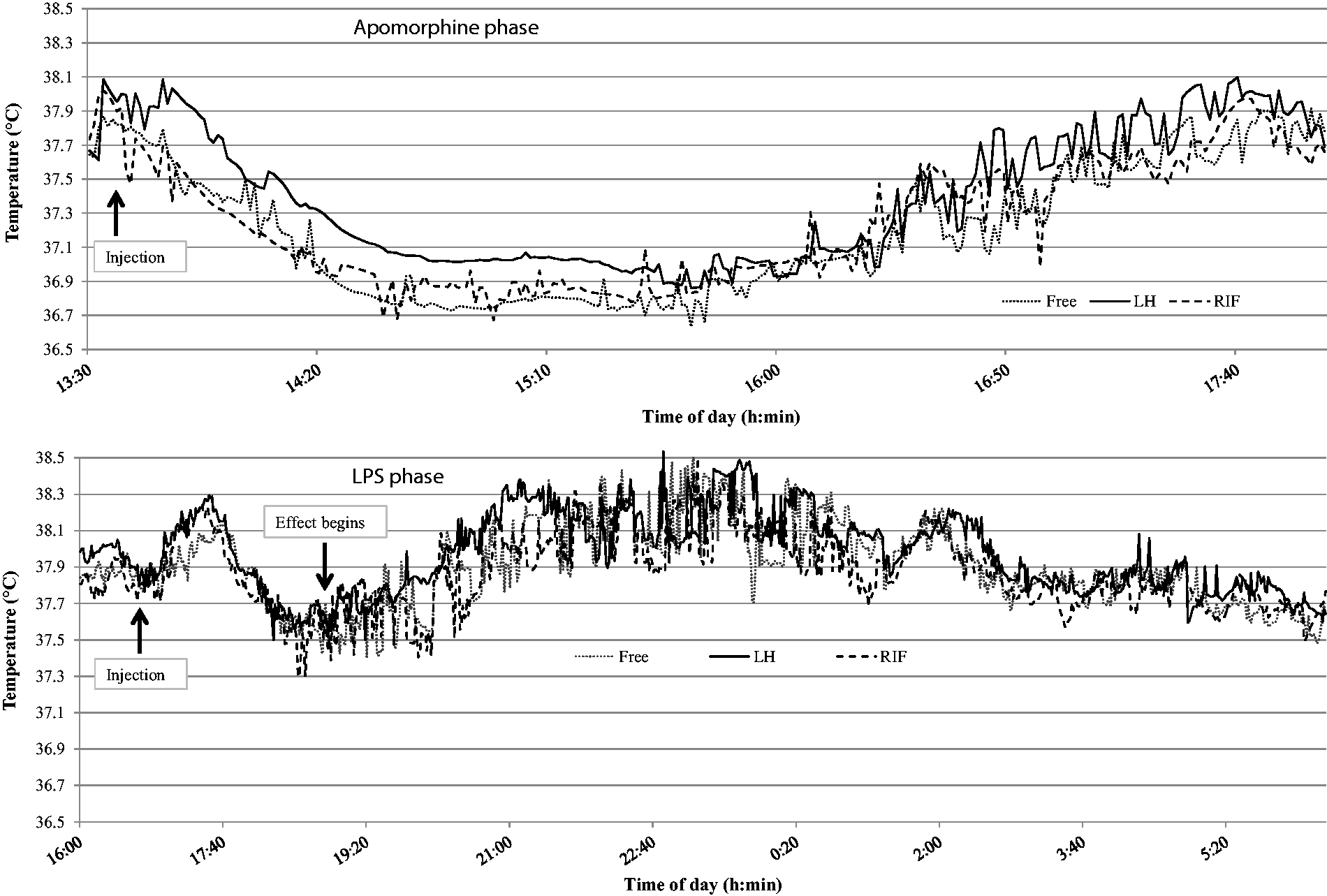
Descriptive statistics for apomorphine and LPS pharmacological effect periods (℃).
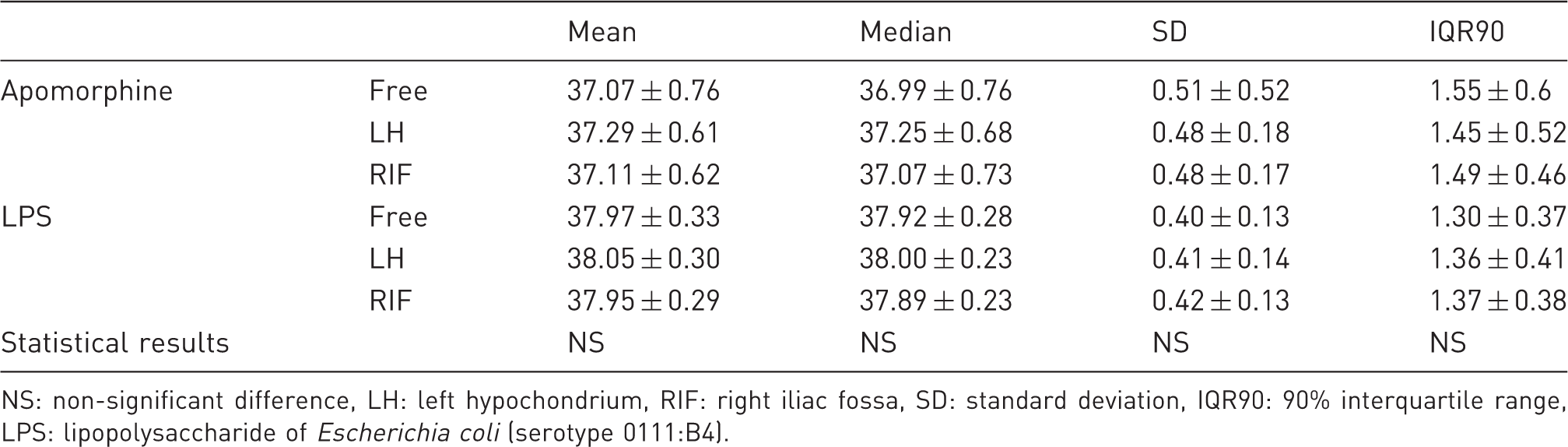
NS: non-significant difference, LH: left hypochondrium, RIF: right iliac fossa, SD: standard deviation, IQR90: 90% interquartile range, LPS: lipopolysaccharide of Escherichia coli (serotype 0111:B4).
Discussion
Considering the advantages inherent in the use of IP sensors for continuous temperature monitoring such as improvement of reliability of data collected and animal welfare, this study was designed to assess the thermal homogeneity of the IP cavity and the importance of attaching the telemetric temperature sensor at a given location. The aim of the experiment was to check the reliability of temperature data issued from three sensors implanted at three locations of the IP cavity in free ranging conditions or while temperature variation was pharmacologically induced. Thus, analysis of (i) raw data collected issued from each sensor and (ii) pairwise inter-sensor comparison were performed.
The first part of the experimental procedure consisted of an ex vivo assessment of sensor reliability and accuracy. Thirty sensors were simultaneously tested in a water bath at 35, 37 and 39℃. The results indicated that the maximal temperature difference observed between sensors and reference values was 0.1℃, which is close to the accuracy of the calibration system. This level of accuracy is classically observed for this kind of telemetric system.27,28
A first examination of the temperature data collected when rodents were in free range conditions enabled the modeling of a classic temperature pattern. Parameters of the temperature patterns were not affected by the sensor locations; both mean amplitude, acrophase and batyphase values issued from the different locations were close together. However, visual analysis indicated that calculated temperature differences among locations regularly exceed the range of ±0.2℃ and high differences of up to 2.5℃ are occasionally observed. This level of difference cannot be explained by technical reasons; thus extreme values might rather be explained by behavioral factors such as food or water intake, as shown previously. 23 The sensor location significantly affects the mean and median temperature values but the intra-sensor variability level was similar (standard deviation and IQR90). Temperature data collected in LH are 0.1℃ significantly higher than those collected in RIF and temperature data collected in LH are 0.11℃ significantly higher than those collected with the Free capsule. With a mean gap of 0.01℃, temperature data collected in the Free and RIF locations were not significantly different. Considering the level of accuracy of the sensor commonly used in research protocol, differences observed between locations are clearly negligible; studied phenomenon could be observed whatever the sensor location.
These observations are confirmed while temperature variations were induced by apomorphine or LPS injections. Indeed, whatever the location, the implanted sensors enabled the detection of a characteristic decrease of temperature or fever, comparable with those previously reported. 24 Thus, temperature kinetics, and both maximal and minimal values reached were similar for the LH, RIF and Free sensors. Furthermore, a Friedman variance analysis performed on raw data did not indicate any significant effects of sensor location on mean, median and intra-sensor variability of temperature data collected during the apomorphine and LPS pharmacological effect periods. By contrast with the 24 h of free ranging conditions, the lack of significant differences between locations for mean and median parameters may be explained by the shorter periods of data collection in the apomorphine and LPS effect conditions.
Considering (i) the sensor accuracy (±0.1℃) indicated by the manufacturer and confirmed by the calibration assessment process, (ii) the similar intra-sensor variability and (iii) the level of mean differences observed between the three locations, the impact of sensor location within the IP cavity may be considered to be negligible.
The results of the present study cannot reasonably be compared with previous research, and to the extent of our knowledge no experimentation has previously assessed thermal homogeneity in rodent IP cavities. Although it has been recognized that a deeper probe insertion in the colon (>6.5 cm) may allow a better correlation with body core temperature,11,29 this procedure is presumably more stressful and harmful for the rodents than a slight insertion.
Conclusion
Our study presents the assessment of thermal homogeneity of the IP cavity and the relevance of attaching the telemetric temperature sensor at a specific location. In rats, the location of the sensor in the IP cavity has a minor impact on the temperature data collected. This result is confirmed in both conditions; during 24 h of free ranging conditions and during temperature variations induced by apomorphine or LPS injection. Thus, the attaching of a temperature sensor in the IP cavity may be considered irrelevant. Furthermore, leaving the sensor unattached within the IP cavity may avoid the irritation and strangulation of organs which could lead to the onset of peritonitis. 14
Footnotes
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
