Abstract
Laboratory rats are generally fed ad libitum, although this method is associated with obesity and an increased frequency of spontaneous tumours. It has been challenging looking for ways to limit feed consumption in group-housed rats without any setbacks to animal welfare and scientific results. The diet board, as a method of dietary restriction, was used in the present study. Diet board feeding allows group housing and should result in enhanced welfare compared with traditional methods of dietary restriction. With respect to animal model robustness and translatability of results it is important that the feeding regime does not affect diurnal rhythmicity of biological parameters. In the present study the effects of diet board feeding on diurnal rhythms of blood glucose, serum ghrelin, faecal immunoglobulin A (IgA) and faecal corticosterone were assessed. The diet board did not alter diurnal rhythms, and adds weight to the use of this method for dietary restriction which should benefit animal health and the validity of scientific results generated from the animals.
The world is not yet perfect, and certainly not when it comes to feeding laboratory rodents. The negative aspects of ad libitum feeding include a high variation in food intake, 1 and consequently a high variation in body weight and other variables. It can also result in high mortality and morbidity.2–5 Dietary restriction does solve these problems,6–8 but it also creates new ones. The most common method of implementing dietary restriction in rodents is to house them singly and give them a pre-calculated meal once a day, often during the daytime. 9 However, this practice compromises both animal welfare and scientific integrity. The lack of conspecifics is a serious threat to the wellbeing of rats.10,11 Feeding during the daytime disturbs the diurnal rhythms of physiology and behaviour, thus confounding research results.12–14 When comparing results obtained from animals fed ad libitum with those subjected to dietary restriction, it is not clear whether the possible differences reflect decreased caloric intake per se, or altered diurnal rhythms.
We have developed a novel method of dietary restriction. The diet board15,16 offers the possibility of combining dietary restriction with group housing and normal eating rhythms. The diet board is made up of two wooden boards onto which food pellets are embedded.15,16 The diet board is always in the cage, allowing the rats to eat as often as they choose. However, the rats need to gnaw wood before reaching the pellets. Their food intake is decreased by 15% compared with ad libitum fed controls. We have previously shown that diet board feeding causes a 15% decrease in weight gain, a 30% decrease in gonadal fat mass, a 3% decrease in skeletal growth, and elicits similar endocrinological changes as in traditional methods of dietary restriction.15–18
The aim of the present study was to investigate how diet board feeding affects diurnal rhythms of physiological variables. In order for the diet board to be generally accepted as a refinement alternative to the traditional methods of dietary restriction, it should meet two conditions: (i) Diet board feeding should result in enhanced health and welfare compared with traditional methods of dietary restriction. (ii) The quality of results obtained with the diet board should be as good as or better than in experiments using other methods of dietary restriction. 19 To ensure scientific quality and relevance, undisturbed diurnal rhythms would be a major achievement in the practice of restricting caloric intake in rodents. Our hypothesis was that diet board feeding does not alter diurnal rhythms of physiological variables in animals compared with ad libitum feeding.
Animals
The study was performed at the Laboratory Animal Centre, University of Eastern Finland, Finland. The study plan was reviewed and approved by the Finnish National Ethics Committee (licence number ESLH-2008-03945/Ym23).
A total of 48 barrier bred HsdBrlHan:Wist male rats (Laboratory Animal Centre, University of Eastern Finland, Kuopio, Finland) were used in this study. The barrier was free of the pathogens listed in the Federation of European Laboratory Animal Science Associations (FELASA) recommendations for health monitoring. 20 The animals were 12 weeks old at the commencement of this 10-week study. The rats were identified with ear notching.
Animal housing
All animals were kept in one animal room, housed in groups of three in solid bottom Makrolon® polycarbonate cages (55 × 35 × 20 cm; Tecniplast, Buguggiate, Italy) with a wire-grid hopper (Bayer AG, Leverkusen, Germany). The cage floor was covered with 300 g of aspen chip bedding (Tapvei Ltd, Kaavi, Finland). Tap water in polycarbonate bottles was available ad libitum. Each cage was provided with three aspen chewing blocks (Tapvei Ltd). The cages and chewing blocks were changed weekly, and the bottles twice a week. The room temperature was 21 ± 1℃ and relative humidity 55 ± 15%. The lights came on at 07:00 h and off at 19:00 h. The light intensity in the cages was approximately 130 lx. All animals were weighed twice a week (Sartorius 1B31; Sartorius-Werke GmbH, Göttingen, Germany).
Feeding
All animals were fed autoclaved (121℃, 20 min, 220 kPa, Finn-Aqua 121821 D; Steris Finn-Aqua, Tuusula, Finland) Lactamin R36 pellets (Lantmännen, Kimstad, Sweden). The control animals (n = 24) were fed ad libitum from the food hopper. The study group (n = 24) was fed solely from the diet board. 15 The diet board consisted of two crossed aspen boards (36.0 × 12.2 × 2.7 cm) with 20 vertically drilled holes (Ø 12.5 mm) filled with food pellets and fixed on site by autoclaving. Each hole had a 2 mm wide slot showing the pellets, thereby providing rats with an easier access to the food. Two corners of each board (6.0 × 6.0 cm) were removed. Control animals had a similar board of the same shape and size, but without the holes or food. Both boards were changed to new ones at the cage change stage.
Study design
The animals were randomly allocated into 16 cages with three rats in each. The cages were randomly divided into eight control cages (ad libitum feeding) and eight study cages (diet board feeding). The cages were placed on tables in the animal room, all at the same height. The study was arranged in two equal blocks with a two-week phase shift.
Sampling
Blood samples were collected six times at 4 h intervals beginning at 09:00 h on week 9. The samples were taken by piercing a hole in the vena saphena with a 25 G needle 21 and the resulting blood drops were collected into a test tube. The sample volumes did not exceed those recommended by Diehl et al. 22
Humane endpoints
Three different humane endpoints were established for the different phases of the experiment. During the first week of the experiment the limit was set to 15% weight loss. During experimental weeks 2 to 4, a failure to gain weight over a two-week period was added to the endpoint. From the fifth week onwards, weight loss of 5% or more during any one week was the endpoint. Dehydration, abnormal behaviour or appearance, or unexpected trauma or diseases were considered as general criteria for exclusion from the study at all times.
Materials and methods
Sample assays
Blood glucose concentrations were measured instantly after drawing the blood with Accu-Chek Compact Plus (Roche Diagnostics, Mannheim, Germany). The blood intended for serum ghrelin analysis was allowed to coagulate for 10 to 15 min, and was then centrifuged at 12 000 rpm for 15 min at room temperature (Eppendorf 5412; Eppendorf, Hamburg, Germany). The serum attained was placed into plastic tubes and immediately frozen at –20℃, and then shipped frozen to an assay laboratory where the samples were kept at –80℃ until analysis. The concentrations of serum ghrelin were measured using a commercial kit (total ghrelin RIA kit; Linco Research, St Charles, MO, USA).
Faecal samples were collected four times a day from each cage at 6 h intervals beginning at 14:00 h on week 6. On collection day, the cage floor was covered with only half the amount of normal bedding to make the sample collection easier. At each time point all faecal pellets among the bedding were carefully collected with forceps and stored at –20℃. The extraction of immunoglobulin A (IgA) and corticosterone from the faecal samples was carried out according to the method described by Pihl and Hau. 23 The concentrations of IgA (rat IgA quantitation kit; Bethyl Laboratories, Montgomery, TX, USA) and corticosterone (DRG Diagnostics, Marburg, Germany) were analysed with commercial kits using a Multiskan EX microtiter reader (Thermo Electron Corp, Waltham, MA, USA).
Statistics
The appropriate size of the experiment was estimated with the resource equation method, 24 and the degree of freedom for error was 13. The differences between the diet board and ad libitum groups were analysed with linear mixed models for repeated measures using all collected data. Time, treatment and their interaction were included as fixed effects into the model. A random cage effect was included in the models in order to accommodate for the possibility of correlated outcomes among cagemates. The linear mixed model’s assumption of normality of distribution was tested with Shapiro–Wilkinson’s test from the residual variance terms. Where necessary, the variables were log-transformed to normalize their distributions.
The results of normally distributed variables (corticosterone, glucose) are presented as model-based estimates of the arithmetic means and 95% confidence intervals (CI). For variables that were not normally distributed (IgA, ghrelin), the results are presented as model-based estimates of the geometric means and their 95% CI. In the graphic representation of the normally distributes, variable means and standard deviations from the raw data are used. For the variables not normally distributed the means and 95% CI from the raw data are used. Due to a technicality, the values in the graphic representation of the body weight constitute an exception: there the values are model-based estimates of the means and their 95% CI, even though the weights were normally distributed.
The statistical software package used to process and analyse the data was SPSS 14.0 for Windows (SPSS Inc, Chicago, IL, USA). The graphs were drawn with SigmaPlot 10.0 (Systat Software Inc, San Jose, CA, USA).
Results
Diet board fed animals resulted in significant differences (P = 0.016) in their body weights compared with ad libitum fed animals, with the diet board fed animals showing an approximately 10% lower body weight throughout the experiment (Figure 1). None of the animals reached the humane endpoints set for this experiment.
Body weights of diet board and ad libitum fed rats throughout the 10-week experiment. The error bars represent the 95% confidence intervals of the model-based estimates of the means. The figure is based on data from 48 rats.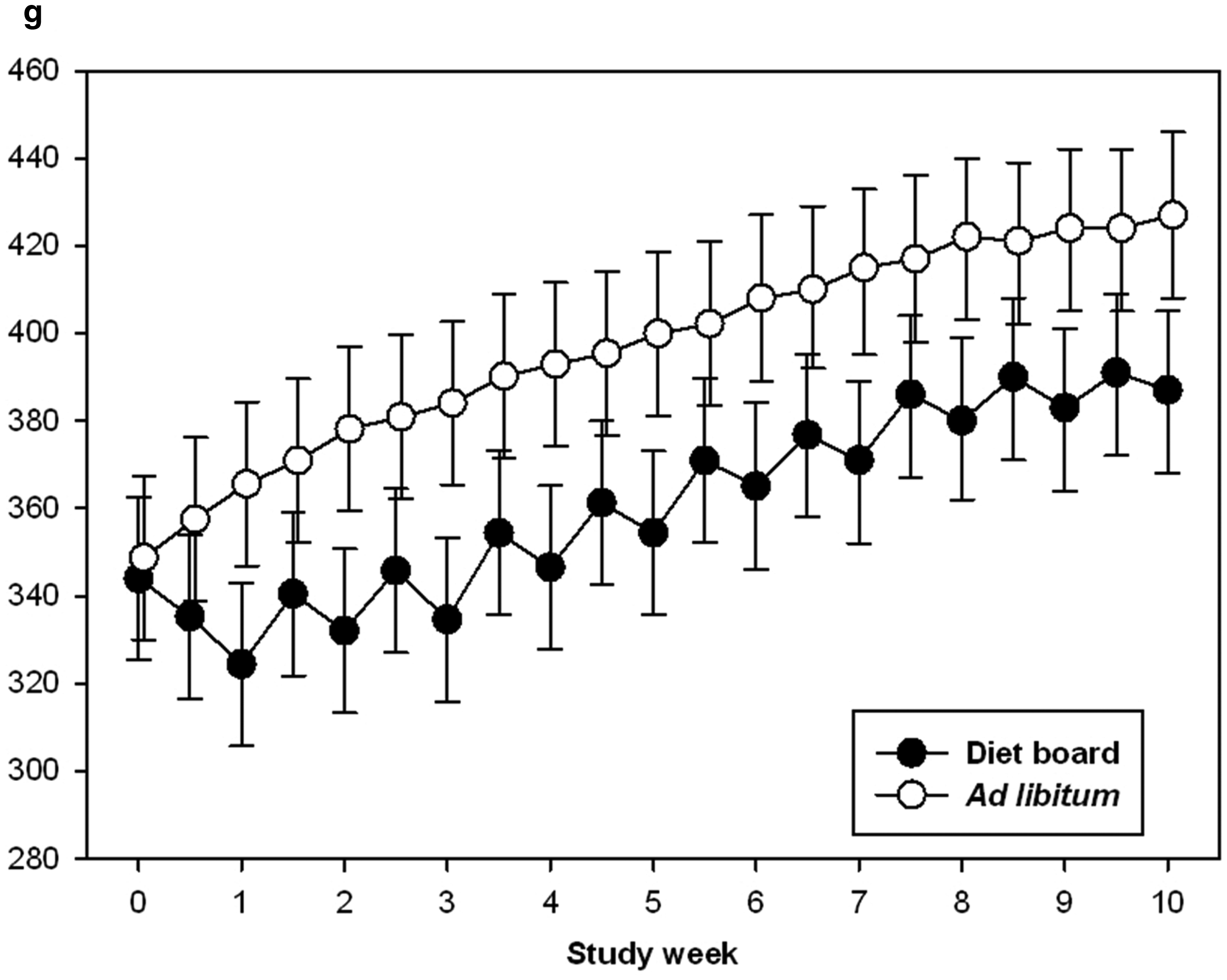
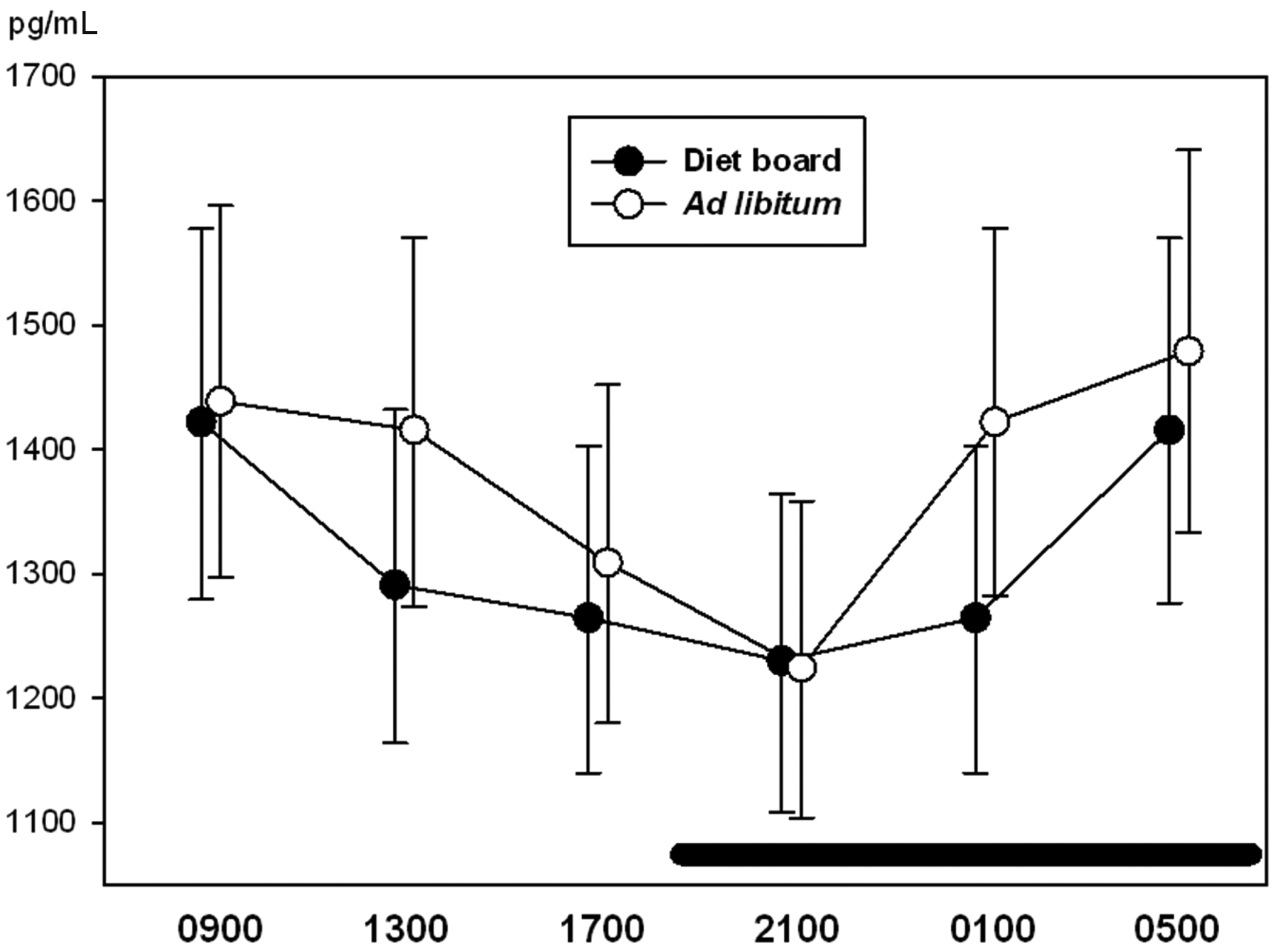
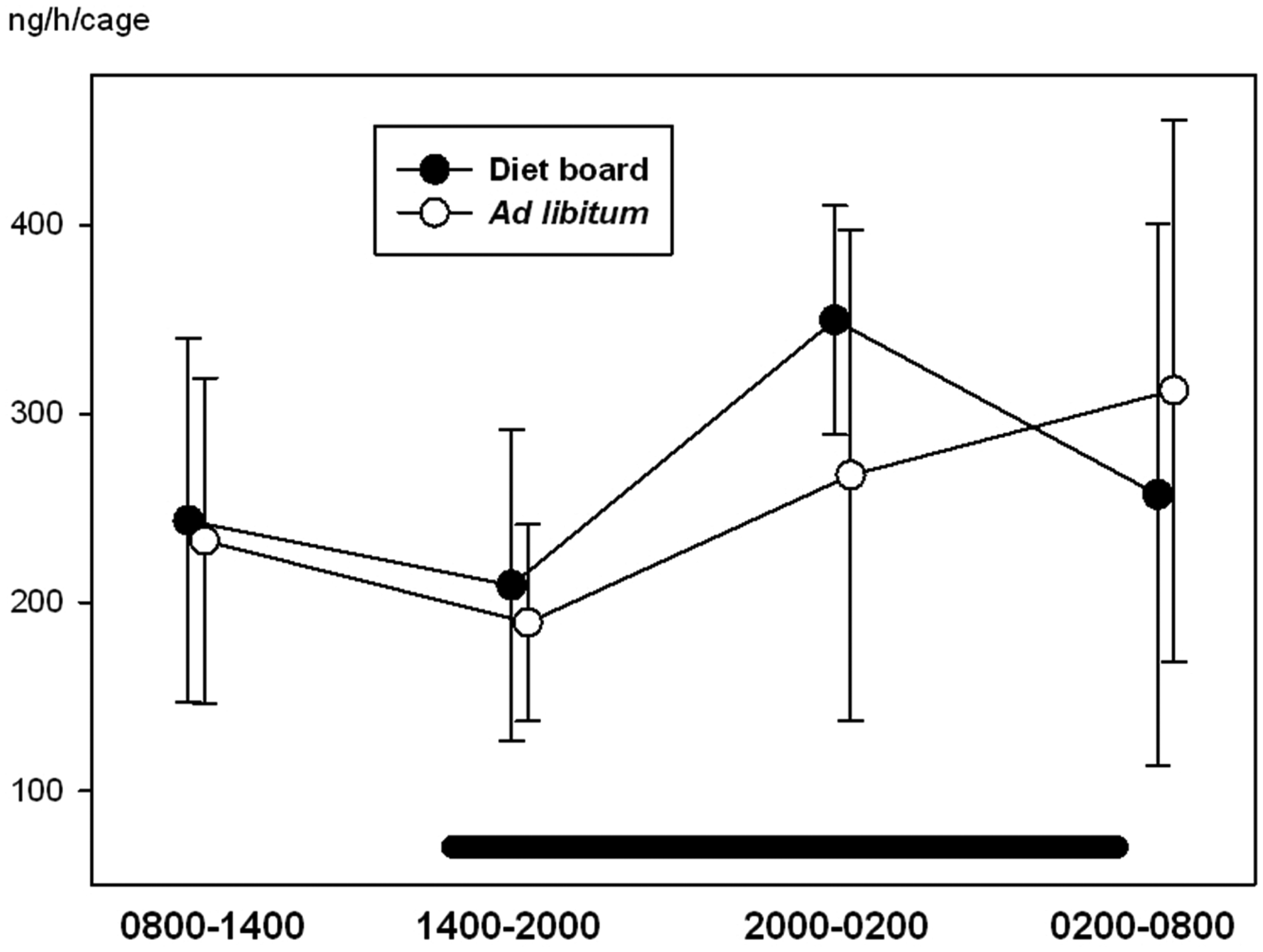
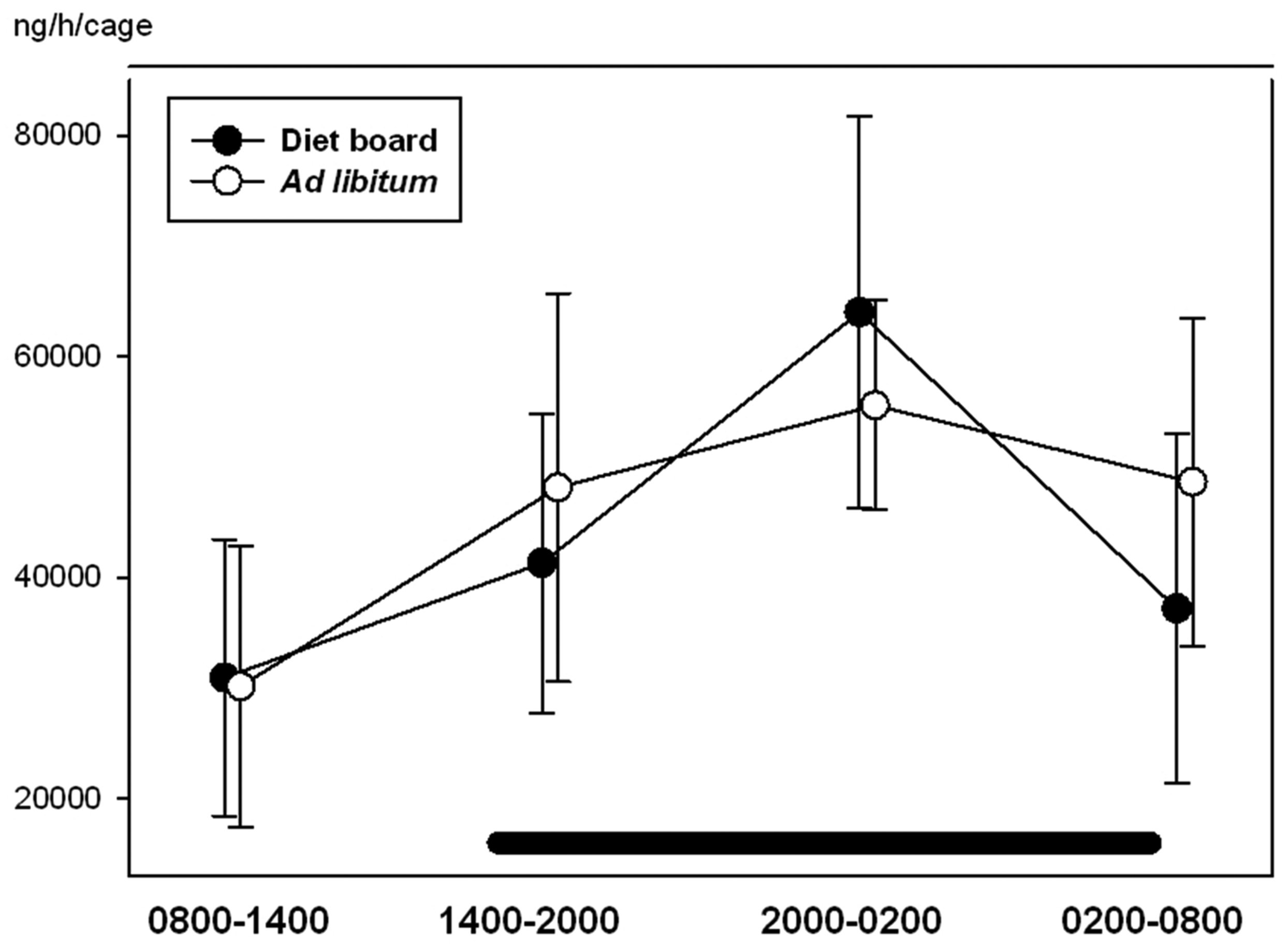
With ghrelin, glucose, IgA and corticosterone, time was a significant factor (P < 0.05), i.e. the variables showed a diurnal rhythm. There were no statistically significant differences between the ad libitum and the diet board groups in any of the measured variables (P > 0.05). The interactions between time and treatment were not significant in any of the variables (P > 0.05), indicating that there were no significant differences in the diurnal rhythms of ad libitum and diet board fed rats (Figures 2–5). The model-based estimates of the means and their 95% CI at each time point are presented in Table 1.
Diurnal rhythms of blood glucose concentration in diet board and ad libitum fed rats on study week 9. The error bars represent standard deviations from the raw data. The horizontal black bar represents the dark period during the 24 h sampling. The figure is based on data from 48 rats. Diurnal rhythms of serum ghrelin concentration in diet board and ad libitum fed rats on study week 9. The error bars represent the 95% confidence intervals of the means of the raw data. The horizontal black bar represents the dark period during the 24 h sampling. The figure is based on data from 48 rats. Diurnal rhythms of faecal corticosterone secretion (amount of corticosterone secreted per cage per hour) in diet board and ad libitum fed rats on study week 6. The error bars represent standard deviations from the raw data. The horizontal black bar represents the dark period during the 24 h sampling. The figure is based on data from 16 rat cages. Diurnal rhythms of faecal immunoglobulin A (IgA) secretion (amount of IgA secreted per cage per hour) in diet board and ad libitum fed rats on study week 6. The error bars represent the 95% confidence intervals of the means of the raw data. The horizontal black bar represents the dark period during the 24 h sampling. The figure is based on data from 16 rat cages. Values of blood glucose, serum ghrelin, faecal IgA and faecal corticosterone. The means are presented as model-based estimates of the means (arithmetic for glucose and corticosterone, geometric for ghrelin and IgA) and their 95% confidence intervals. The overall P value is the significance of the repeated measures test taking all time points into account. The P value in the ‘treatment’ column represents the comparison of ad libitum feeding to diet board feeding. The ‘time’ is the significance of time with both treatment groups together and ‘time * treatment’ is the significance of the interaction of time and the feeding method. CI: confidence interval, CORT: corticosterone, IgA: immunoglobulin A.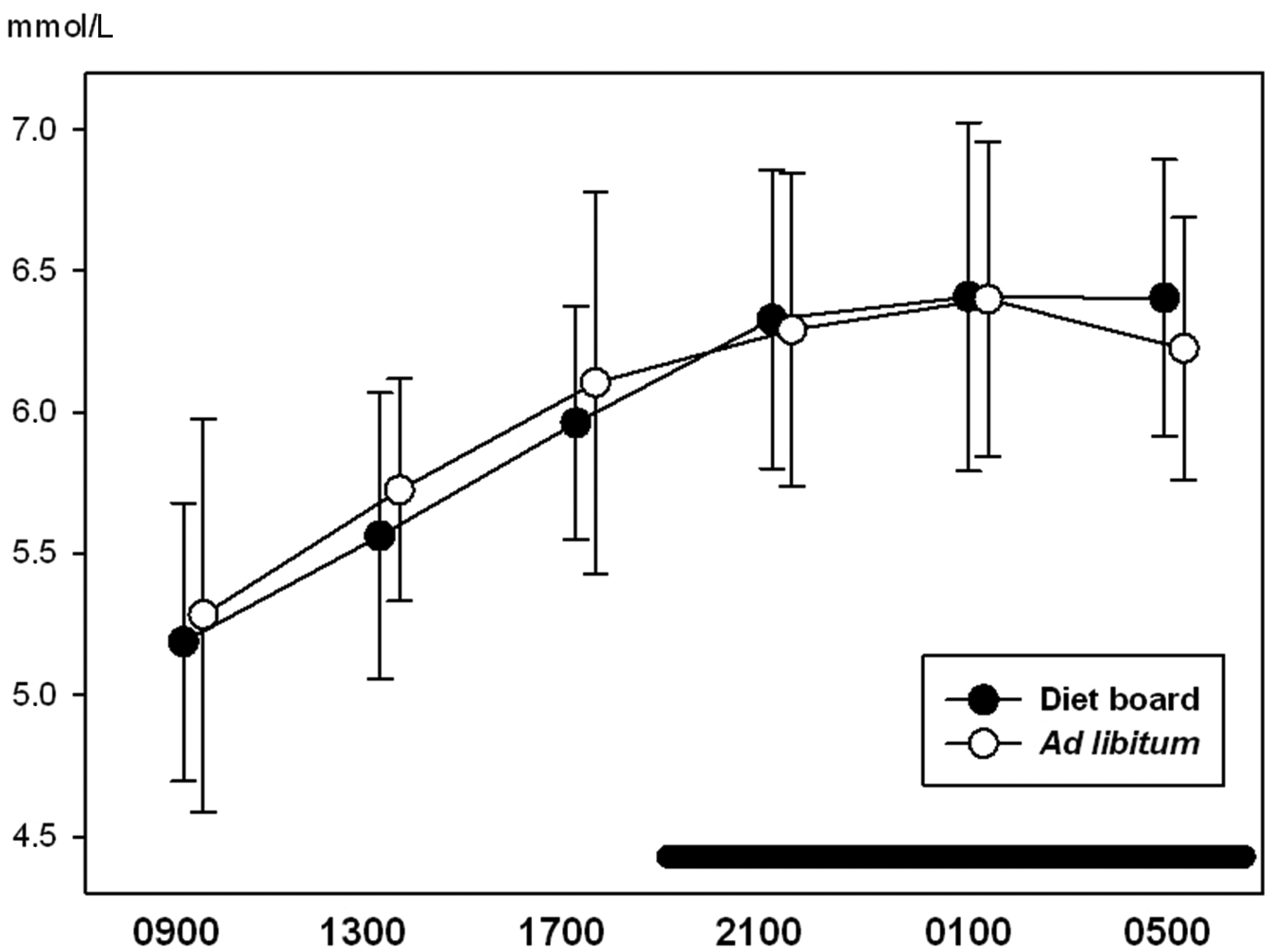

Discussion
The diet board was developed as a refinement alternative to traditional methods of dietary restriction. Novel methods should always be validated before they can be recommended for wider use or offered for commercial production. Methods intended for laboratory animal use should also be evaluated with respect to their impact on the 3Rs (replacement, reduction and refinement). 19 The effects on the quality and relevance of the research data obtained from rats subject to diet board feeding should also be established. We have previously shown the diet board’s efficacy, safety and reproducibility in providing moderate caloric restriction for laboratory rats.15–17 The most important refinement the diet board can offer compared with traditional methods of dietary restriction is the possibility of group-housing the animals. Preference tests indicate that the company of conspecifics is valued more by rats than any other form of environmental enrichment, 25 and both European and American guidelines clearly state that rodents should be group-housed by default, and single housing should only be practised for veterinary, welfare or scientific reasons for a limited time period.26,27 Furthermore, the diet board also provides a recommended structural complexity into the cage. 27
We have previously shown that the diet board elicits similar, but somewhat milder, endocrinological effects as using traditional methods of dietary restriction. 16 This further proves that the diet board provides true caloric restriction, and not merely as an enrichment for the animals.
The purpose of this study was to assess the effects of diet board feeding on diurnal rhythms of blood glucose, serum ghrelin, faecal IgA and faecal corticosterone. These are variables known to have a clear diurnal rhythm that can be altered by changes in feeding schedules.28–34 Disruption of diurnal rhythms is one of the most important threats to research quality when using traditional methods of dietary restriction. Our hypothesis was that since the animals are free to eat when they choose, the diet board would not alter diurnal rhythms. The results of this study proved our hypothesis to be correct. Diet board feeding did not cause significant changes in diurnal rhythms or the actual levels of the analysed variables (Figures 2–5). The diet board is thus a refinement alternative in the best sense of the word, providing both enhanced welfare and better science.
The reduction potential of using the diet board has also been evaluated. We have investigated the equality of variances from residual variance terms in more than 20 different variables from different experiments with the diet board. No significant differences were found in the variances of any of the variables. Compared with ad libitum feeding, diet board feeding does not alter the number of animals needed in experiments in order to maintain the same statistical power.
In conclusion, the diet board is now sufficiently validated as a method of dietary restriction, as well as providing a structural complexity to the cage environment. It can therefore be recommended for group-housed rats when dietary restriction is needed.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The authors received funding from the Academy of Finland and the Ministry of Education, Finland.
