Abstract
Group housing is considered to be important for rats, which are highly sociable animals. Single housing may impact behaviour and levels of circulating stress hormones. Rats are typically used in the toxicological evaluation of insulin analogues. Human insulin (HI) is frequently used as a reference compound in these studies, and a comparator model of persistent exposure by HI infusion from external pumps has recently been developed to support toxicological evaluation of long-acting insulin analogues. However, this model requires single housing of the animals. Developing an insulin-infusion model which allows group housing would therefore greatly improve animal welfare. The aim of the present study was to investigate the suitability of implantable infusion pumps for HI infusion in group-housed rats. Group housing of rats implanted with a battery-driven pump proved to be possible. Intravenous infusion of HI lowered blood glucose levels persistently for two weeks, providing a comparator model for use in two-week repeated-dose toxicity studies with new long-acting insulin analogues, which allows group housing, and thereby increasing animal welfare compared with an external infusion model.
Several studies have investigated different housing conditions, as well as the impact of single housing in rats, and there is a consensus that rats are social animals preferring group housing which enables normal social interaction.1–3 When this need for social interaction is not met, it has both behavioural and physiological consequences for the animals.2,4 The importance of group housing on animal welfare in rats has, furthermore, been acknowledged in the guidelines for the accommodation and care of animals by the European Convention for the Protection of Vertebrate Animals used for Experimental and other Scientific Purposes (Appendix A, ETS123 2007).
The rat is often chosen as the most appropriate rodent species for the toxicological evaluation of insulin analogues during preclinical drug development, due to its pharmacological responsiveness to insulin. 5 Because of the intended effect of insulin analogues, i.e. to lower blood glucose levels, rats in repeated-dose studies become recurrently hypoglycaemic. In toxicological studies with long-acting insulin analogues rats are rendered persistently hypoglycaemic due to the prolonged exposure between dosing. Little is known about the effects of persistent insulin-induced hypoglycaemia in rats, challenging differentiation between the exaggerated pharmacological effect (i.e. the hypoglycaemia) and the off-target effects of the new analogue. This differentiation is usually accomplished by including an in-study human insulin (HI)-dosed comparator group which serves as a reference. 5 A comparator model using a continuous HI infusion to rats by an external infusion system has recently been developed to mimic the prolonged exposure and persistent hypoglycaemia seen with long-acting insulin analogues. 6 However, the use of external infusion devices requires single housing to avoid interference with the infusion line. Besides the welfare issues for these highly social animals, single housing has also been shown to increase levels of stress hormones, which are involved in the regulation of glucose homeostasis.4,7 Thus, results from studies investigating the effects involving glucose homeostasis in rats may potentially be affected by single housing. Therefore, the development of a new HI infusion model in rats, which allows for group housing, would not only increase animal welfare, but also reduce stress-related effects on the study results.
Implantable infusion pumps allow unrestrained movement of the animal as well as the potential for group housing. The aim of the present study was to investigate the suitability of group housing of rats receiving HI infusion by implantable infusion pumps to provide a preclinical comparator model of persistent exposure; which, in contrast to an already established model, allows for group housing and unrestrained movement of the animals. This improves the experimental conditions for the animals and reduces the stress caused by single housing. Initially, an in vitro study was carried out to assess the stability of HI when stored at body temperature in two different types of implantable infusion pumps (a battery-driven and an osmotic pump). The pump most compatible with HI in vitro was then evaluated in two in vivo studies. The number of animals used in the in vivo studies was kept to a minimum due to the exploratory nature of the studies.
Animals
All in vivo procedures were conducted under the Project Licence authorized by the UK Secretary of State and in compliance with EC Commission Directive 2004/10, 8 OECD principles and good laboratory practice, 9 and the Good Laboratory Practice (Codification Amendments etc.) Regulations 2004. 10 Sprague–Dawley (Crl:CD (SD)) rats (Charles River Ltd, Margate, UK) were housed in solid bottom plastic cages with a stainless steel mesh lid, sawdust litter, and pieces of wood and tunnels as cage enrichment under standard conditions (19–23℃, 40–70% humidity, 12 h light–dark cycle). They received a standard laboratory diet and water ad libitum. Body weight and food consumption were monitored.
Materials and methods
The test substance used comprised recombinant HI formulated in vehicle (Novo Nordisk A/S, Måløv, Denmark). The composition of the HI stock solution (pH 7.4) consisted of 600 nmol/mL of HI, 19.6 µg/mL of zinc (as chloride), 1.50 mg/mL of phenol, 1.72 mg/mL
The infusion pumps used were the iPRECIO micro infusion pump (model SMP-200; Primetech Corp, Tokyo, Japan) and the Alzet osmotic pump (2ML4; DURECT Corp, Cupertino, CA, USA).
In vitro study
Details are listed in the Supplementary information (all Supplementary material can be found online at http://lan.sagepub.com). In short, HI was incubated in the two different types of pumps for up to four weeks. After incubation, recovery of the total peptide content and weight change of pumps were measured to evaluate stability of the peptide content and evaporation from the pumps, respectively.
Total peptide concentrations were closer to nominal concentrations after incubation in iPRECIO pumps compared with Alzet pumps (±3–4% versus ±6–15%). Furthermore, evaporation was higher in the Alzet compared with the iPRECIO pumps (≤12.6% versus ≤3.5%). Consequently, based on these results, it was decided to move forward with the iPRECIO pump for HI infusion in vivo.
Subcutaneous infusion study
Experimental design
Study design of in vivo studies.
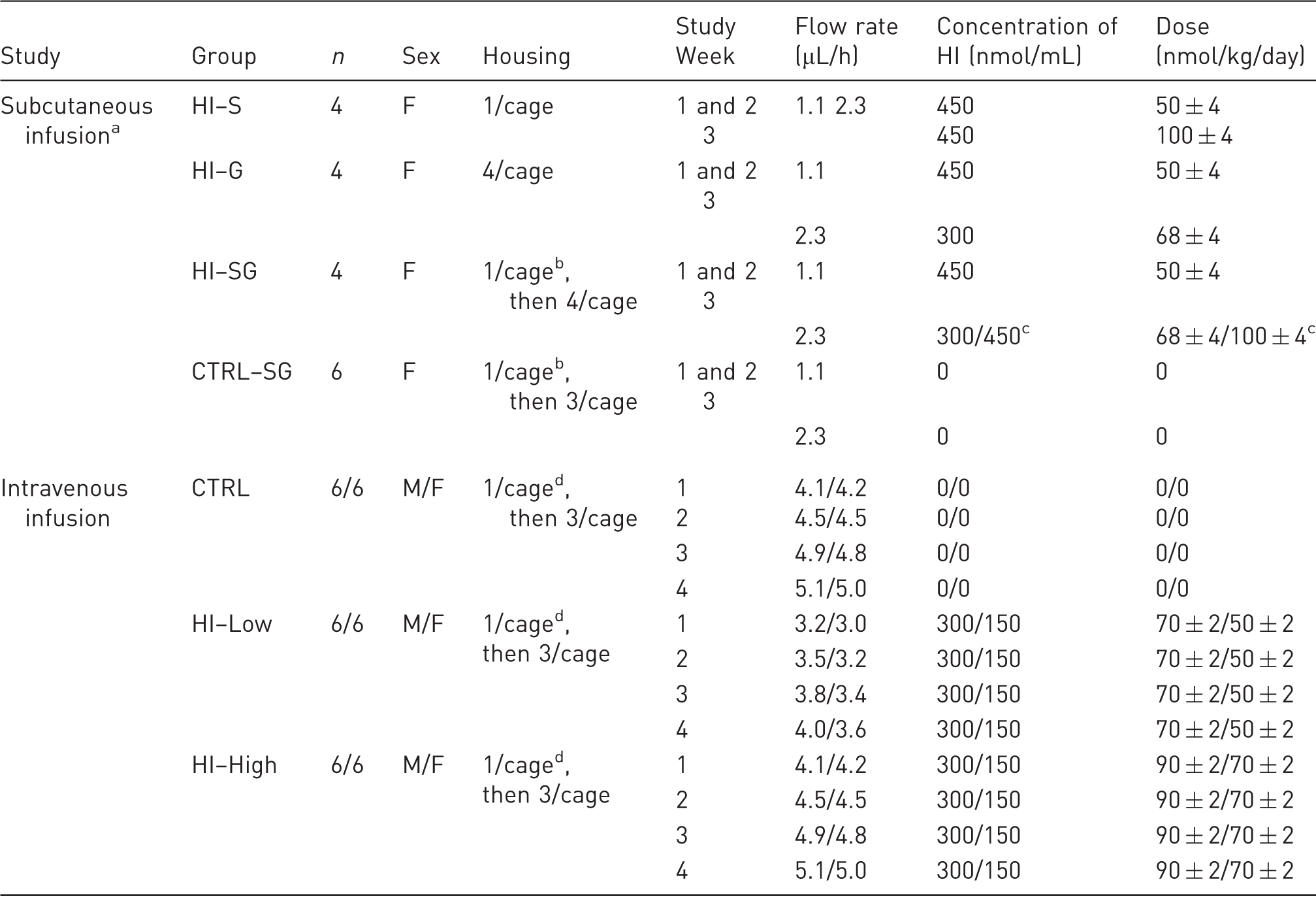
aGroups listed in chronological order of study start for each group (staggered start of 10 days apart for each group). bFor three days. cDays 15-16/Days 17–22. dFor seven days. F: females, M: males, HI: human insulin, S: single housing throughout the study, G: group housing throughout the study, SG: single housing for three days post-surgery followed by group housing for the remaining study period.
Blood samples (max 0.9 mL/animal/sampling day, sublingual vein, isoflurane anaesthesia) for plasma glucose and HI quantification were taken on days 1 and 22 in the CTRL–SG and HI–S groups, and on days 15 and 22 in the HI–G and HI–SG groups, to minimize blood sampling of the individual animals. Sampling was performed at 0.25, 0.5, 6, and 12 h time-points relating to commencement of infusion on day 1 (time-point zero). Spot glucose measurements (Accu-Chek Aviva, Cat/Type 05911974002; Roche Diagnostics, Burgess Hill, West Sussex, UK) were used for any additional monitoring of blood glucose levels (tail vein).
Pump handling and surgical implantation
Pumps were programmed (iPRECIO management software; Primetech Corp) prior to implantation, and pump catheters were shortened to 110 mm. Prophylactic antibiotic cover (enrofloxacin, 5 mg/kg subcutaneously) and analgesics (meloxicam, 1 mg/kg subcutaneously) were administered prior to induction of general anaesthesia (isoflurane). Using aseptic techniques, the pumps were implanted subcutaneously through incisions in the right flank. The pump catheters were tunnelled subcutaneously to the nape of the neck where they were sutured in place. Incisions were closed using sutures or clips. Postoperative analgesia and antibiotic treatment (as above) were provided for three days postoperatively.
Intravenous infusion study
Experimental design
On arrival, female and male (
Blood samples (sublingual vein, isoflurane anaesthesia) were obtained on days 1, 8, 15 and 29 for glucose and on days 1 and 29 for HI quantification. On day 8, sampling was performed at the 6 h time-point only for all animals; on the remaining sampling days, sampling was performed pre-dose (only day 1), 0.5, 6, 12, and 24 h (2 animals/sex/time-point for each group). Time-points refer to hours after infusion start (time-point zero) on day 1. Scheduled sacrifices took place on day 30 immediately after the last blood sampling, performed as for the subcutaneous infusion study. Furthermore, tail vein blood was sampled every other day from day 6 or day 7 for measurement of whole blood glucose levels. After sacrifice, the skin overlying the pump injection port as well as the jugular vein including the catheter tip were isolated, fixated in 10% neutral buffered formalin, processed, and sections of a nominal thickness of 4–5 µm were haematoxylin and eosin (H&E)-stained and microscopically evaluated.
Quantification of glucose and HI in plasma
Blood samples were centrifuged (3000 rpm, 1400–1942 g, 15 min at 2–8℃), and the plasma was separated and stored at 2–8℃ for plasma glucose and frozen (−20℃) for plasma HI, prior to analysis. Plasma glucose concentration was determined by the hexokinase method (Roche Modular P system; Roche Diagnostics, Indianapolis, IN, USA). HI was quantified using an enzyme-linked immunosorbent assay (ELISA) kit (K6219; Dako, Glostrup, Denmark) according to the manufacturer’s instructions. The lower limit of quantification (LLOQ) was 10–20 pmol/L. Samples were analysed in duplicate and the mean concentrations were reported.
Data analysis
Data for body weight, food consumption, and whole blood glucose levels were analysed using a two-way analysis of variance (ANOVA) with ‘group’ and ‘sex’ as fixed variables followed by a Tukey multiple comparison post hoc test for each time-point separately in case of interaction. Plasma glucose results from day 8 were analysed first using a two-way ANOVA to test for effect of dose (group) and sex, followed by a one-way ANOVA post hoc test for each sex separately to test for a dose response.
Results
Subcutaneous infusion study
Animals and housing
Increased interference with the sutures/wound sites was observed especially during the first 48 h post-surgery in the HI–G group versus the HI–S group. Consequently, animals in groups HI–SG and CTRL–SG were singly housed for three days post-surgery followed by group housing, which reduced wound interference by cage-mates. Changing from sutures to clips for these groups further reduced wound interference.
Post-surgery complications.
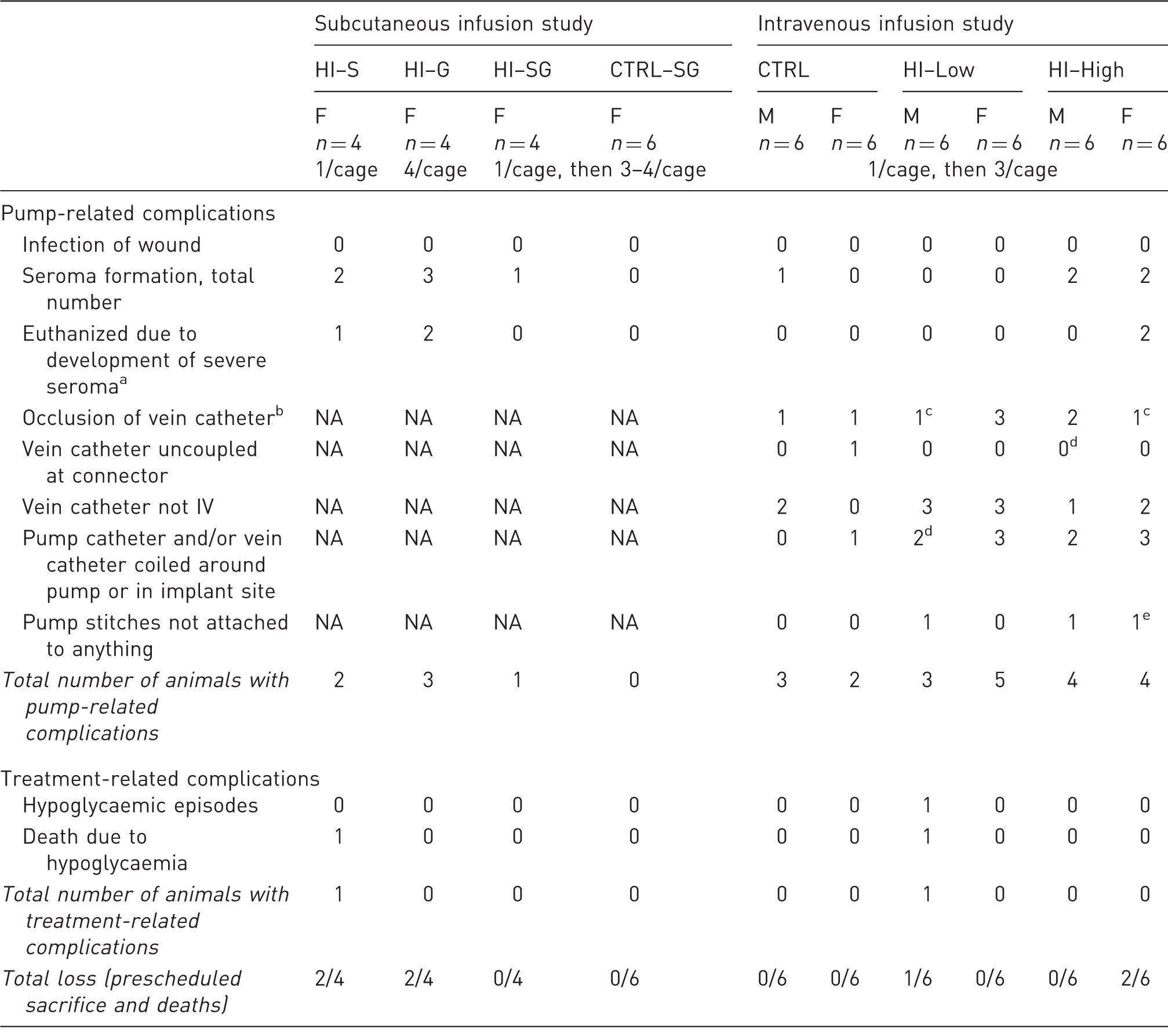
Severe seroma is defined as a seroma resulting in the skin becoming weakened over the pump. bAssessed by flushing the catheter with saline at necropsy. cOnly 4/6 animals were tested for vein catheter patency. dOne animal was not checked. eTwo animals were not checked. F: females, M: male, NA: not applicable, HI: human insulin, S: single housing throughout the study, G: group housing throughout the study, SG: single housing for three days post-surgery followed by group housing for the remaining study period.
Body weight and food consumption were not affected by HI infusion. Body weight gain among all groups was as anticipated for animals of this age and strain during the recovery period and post-surgical food consumption was similar to that of the dosing phase (data not shown). Necropsy revealed no macroscopic findings related to HI infusion. At the pump implant site thickened dermal tissue, abnormal contents (pale, caseous material), and scabs were observed in three animals, one each from the HI–S, HI–G, and HI–SG groups, respectively. No macroscopic changes were seen at the catheter tip site.
Plasma glucose and HI levels
On day 1, the animals in the HI–S group showed clear evidence of a glucose-lowering effect, with values being generally below the levels of the controls from 0.25 h after infusion start (3–6 mmol/L versus 8–11 mmol/L in controls). On day 22, the HI–S group had plasma glucose values below the CTRL group values at all sampling points with similar values to day 1 in both groups. In the remaining HI-infused groups, plasma glucose levels were generally not different from those in the CTRL group, with the exception of a few measurements from the HI–SG group of approximately 5 mmol/L.
All the HI-infused animals were systemically exposed to HI except for one animal in the HI–SG group. Plasma HI concentrations displayed high variability between the animals, but clearly correlated inversely with blood glucose levels on an individual basis, i.e. high plasma HI levels were seen in animals with low blood glucose levels (data not shown).
Aspirated residual formulations
In all four groups, observed dosed volumes were generally as expected, i.e. ±25%. The CTRL–SG group samples had a total peptide content below LLOQ (i.e. <1 nmol/mL). Recovery of total peptide content in aspirated samples was 85–119%, with all analysed aspirated samples (
Intravenous infusion study
Animals and housing
No clinical signs associated with HI infusion were observed during the study (Table 2). On day 7 a HI–Low group male died, most likely due to an unobserved episode of severe hypoglycaemia, since an antemortem blood sample revealed a whole blood glucose level of only 0.7 mmol/L, suggesting the maximum tolerable dose had been exceeded. Seroma formation overlying the pump was observed in a total of five animals (Table 2), of which two were euthanized as the seromas progressed in severity.
Post-surgical mean body weight gain in all the animals was comparable to that anticipated for animals of this age and strain and with no effect of HI infusion. Macroscopic findings related to the pump as well as pump catheter positioning and patency are listed in Table 2. Procedure-related lesions such as thickened tissue and abnormal content (pale, caseous material) were observed at the implant and injection sites with a similar incidence in all groups.
Blood glucose and HI levels
After the death of one HI-infused animal, daily whole blood glucose measurements were implemented allowing close monitoring of blood glucose levels. Results revealed a general lowering of glucose concentrations in HI-infused animals (Figure 1). Approximately after day 14, blood glucose values began to normalize in HI-infused animals, and from day 21 and onwards glucose levels were similar between groups.
Whole blood glucose concentrations, mean ± SD. (a) Males and (b) females (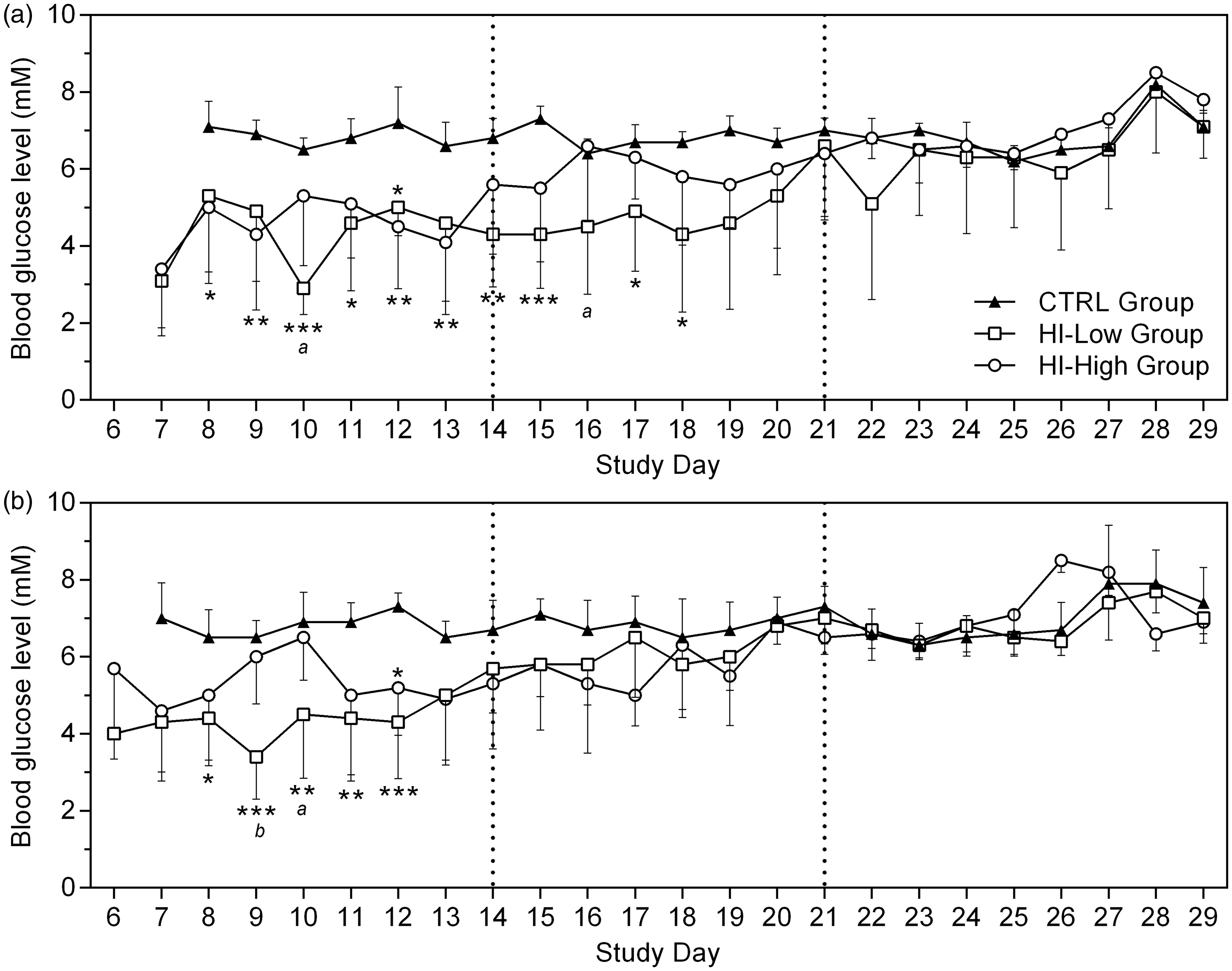
Plasma glucose profile on day 1 showed a clear lowering of plasma glucose concentration in HI-infused animals (approximately 5–8 mmol/L versus 9–11 mmol/L in controls). On day 8, single time-point plasma glucose levels were generally lower in HI-infused groups compared with controls (Figure 2), with no evident dose response. A similar tendency was also apparent on day 15, though on this occasion the plasma glucose values in HI–High groups were only marginally lower than those of the control group (approximately 7–9 mmol/L versus 9–11 mmol/L in controls). On day 29, individual plasma glucose values were generally similar between groups (approximately 7–11 mmol/L).
Plasma glucose concentrations at the 6 h time-point study day 8, individual and mean ± SD. **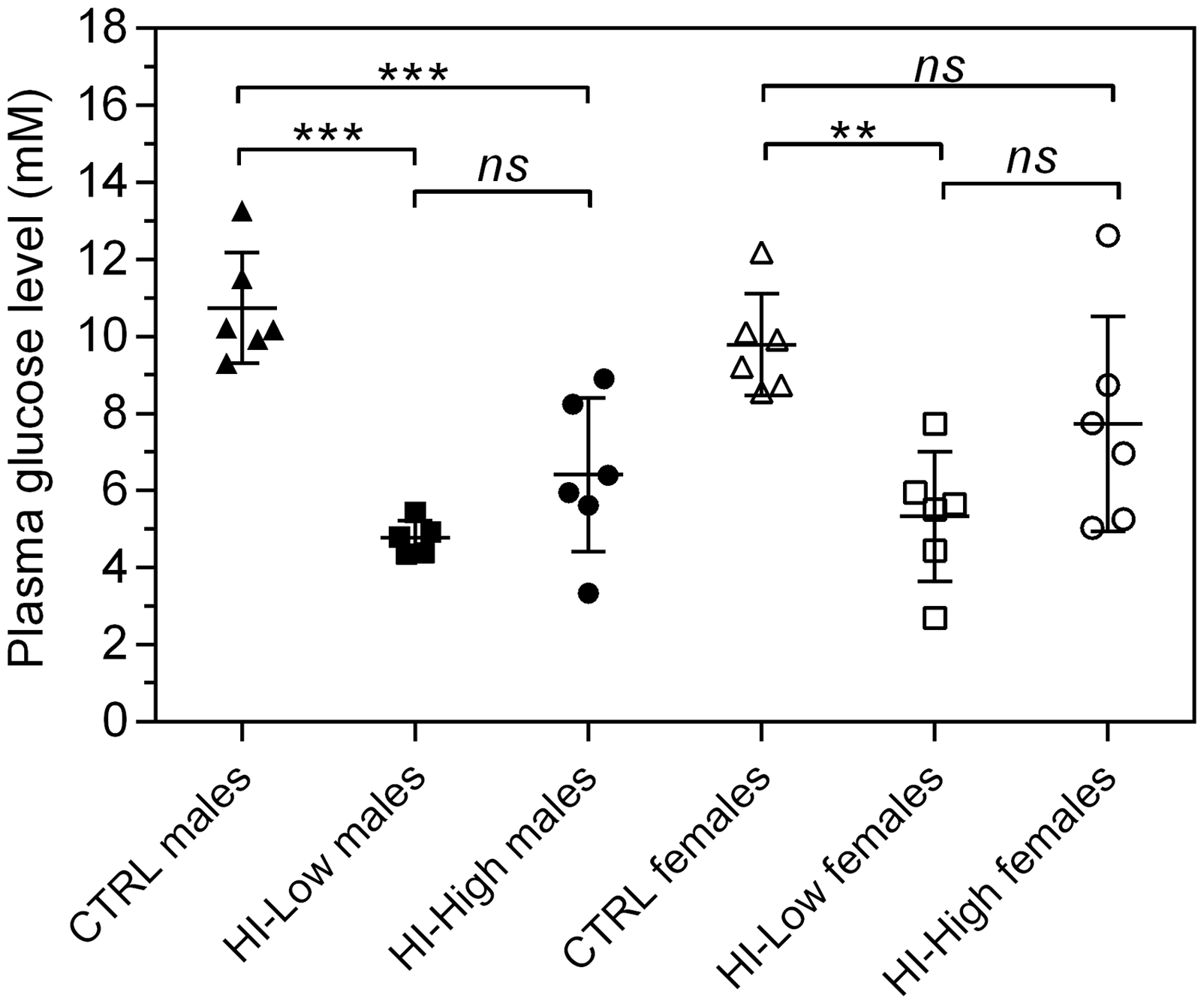
All the HI-infused animals were systemically exposed to HI, although not consistently over time. On day 1, all sampled animals from the HI-infused groups were exposed to HI from 6 h after commencement of infusion, with high inter-individual variations and similar levels between the HI–Low and HI–High groups and between males and females (Figure 3). At day 29, plasma HI levels were generally low, with levels being <LLOQ in many of the samples.
Individual (symbols) and mean (lines) plasma human insulin (HI) concentrations versus time (semi-log). Showing days 1 and 29 following continuous intravenous infusion with HI. Plasma HI concentrations reported as being < 10 pmol/L (i.e. below the lower limit of quantification [LLOQ]) is depicted as the nominal concentration of 10 pmol/L for illustration purposes. Pre-dose sample is set as time zero. In the CTRL–SG group, HI concentrations in all plasma samples were below the LLOQ (not depicted). In the HI–Low and HI–High groups, five out of eight pre-dose samples had unexpected values above the LLOQ. Day 1 Males: HI–Low (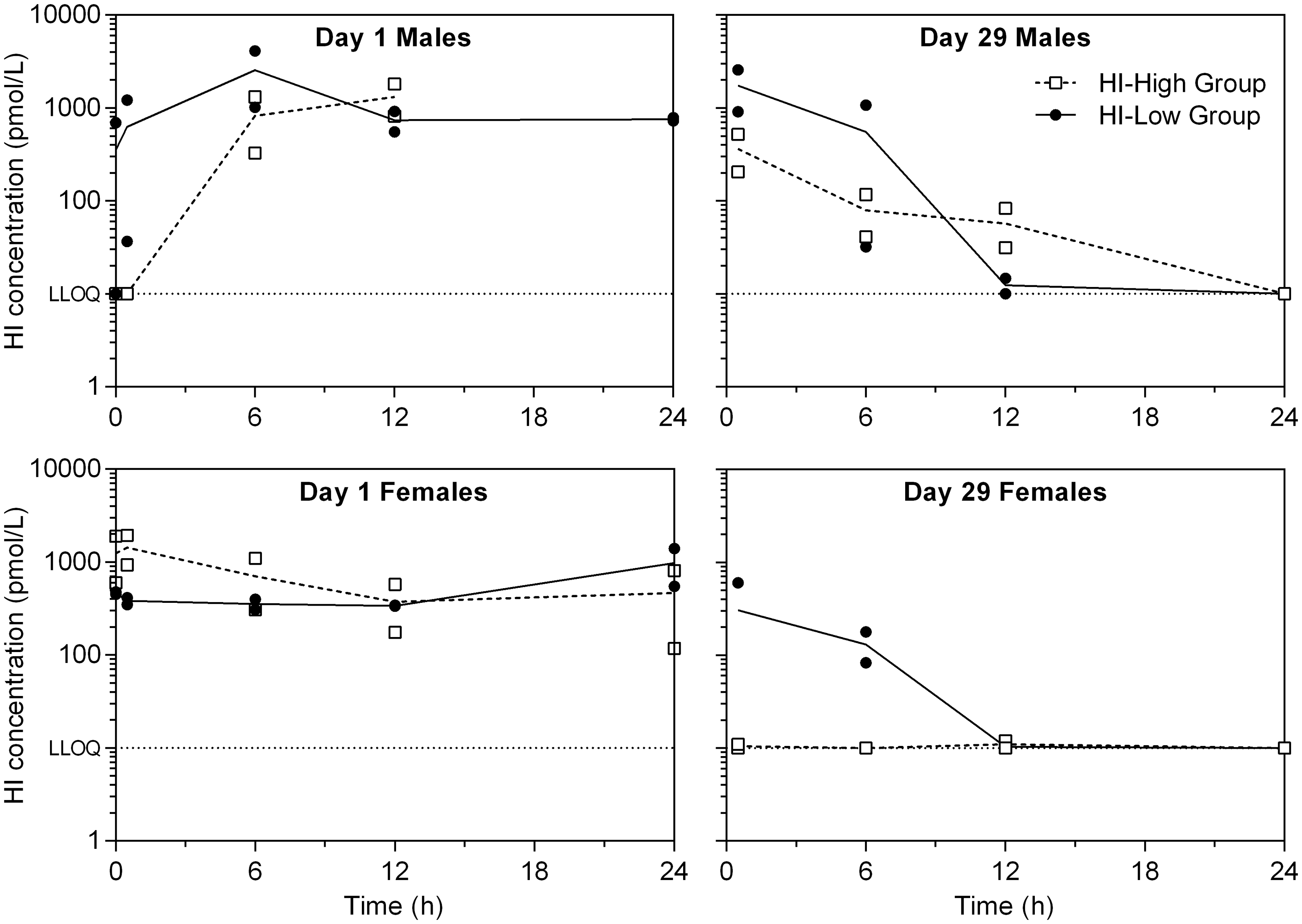
Aspirated residual formulations
Dosed volumes were generally close to expected on day 8 (±25% of expected), but lower than anticipated from day 15 and onwards (Figure 4), especially in the HI–High group. Recovery of total peptide content in aspirated samples from all the HI-infused groups was 93–101%, except for one sample with 49% recovery (HI–High female, day 8), considered to be sampling-related. Total peptide content was <LLOQ in the CTRL group samples. Purity of aspirated samples from the HI-infused groups was 93.2–98.0%.
Relative dosed volumes (observed/expected), mean (lines) and individual (symbols) values. A relative dosed volume of 100% indicates that the observed dosed volume was equal to that expected.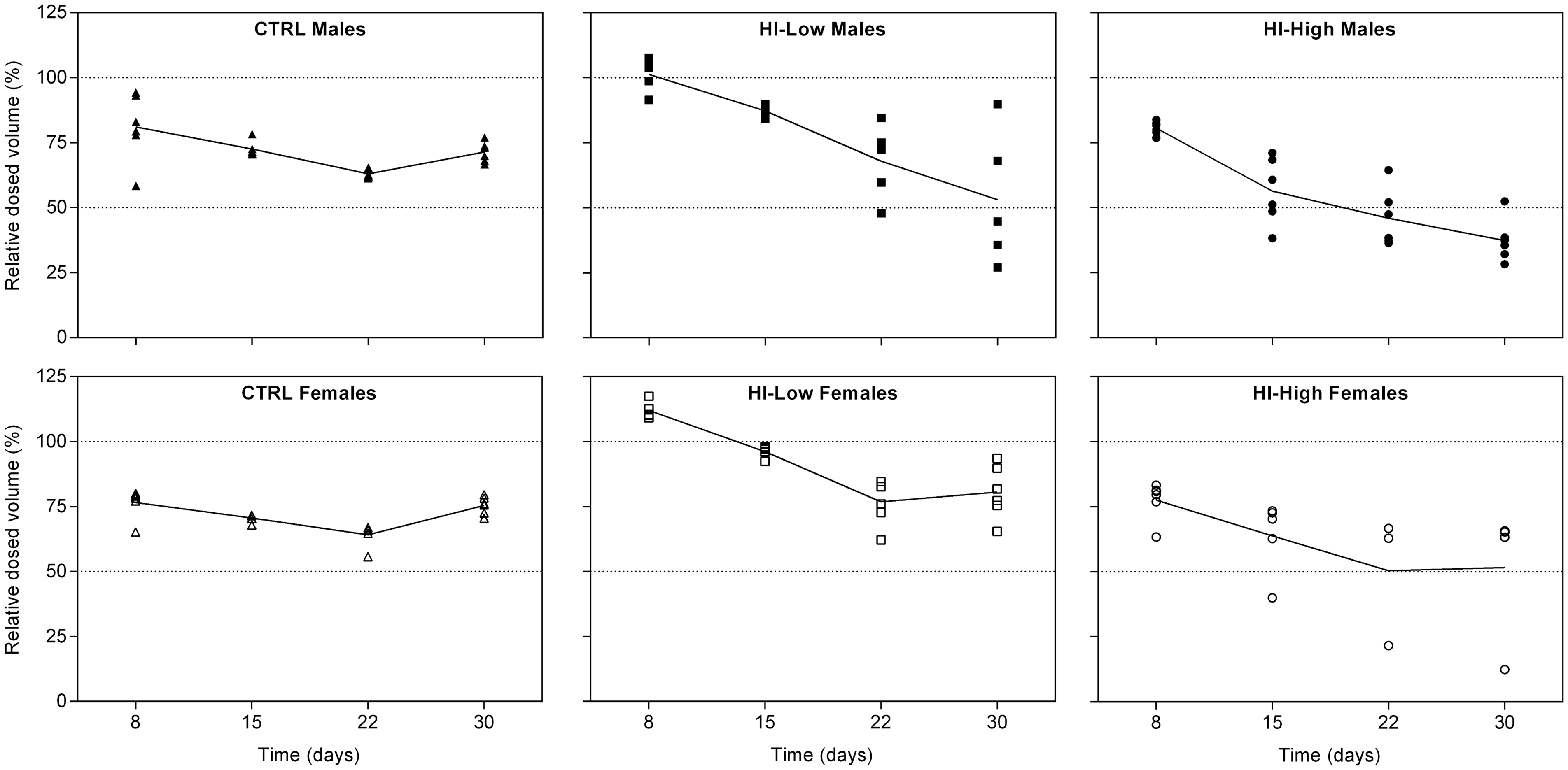
Histopathology
Findings at the implantation site included dermal inflammation (3 out of 33 examined), eosinophilic material (2/33), granulation tissue in all animals, scabs (3/33) and suture granulomas (9/33). Findings at the infusion site (jugular vein including catheter tip) comprised fibrinous/fibrous cuff formation at the catheter tip (15/32), intimal proliferation in the vein wall (2/32), suture granulomas (13/32), thrombus formation at the catheter tip (9/32), vascular/perivascular granulation tissue (8/32), and vascular/perivascular inflammation (3/32). Distribution of the histopathological findings was similar across all groups and was considered to be foreign body reactions towards the pump and catheter themselves.
Discussion
Our findings show, for the first time, the applicability of an implantable battery-driven pump for the group housing of rats which are continuously infused with HI. The potential for group housing after pump implantation was evaluated. Group housing of the rats immediately post-surgery proved to be problematic due to wound/suture interference by cage-mates. However, this was circumvented by isolating the animals for three days post-surgery, allowing pump-implanted animals to be group-housed, providing a major welfare advantage for this infusion pump, by enabling normal social behaviour and reducing stress.2,3 A post-surgery period with seven days of saline infusion was included, allowing animals to recover after surgery before initiating HI infusion, as growth rate has been shown to decrease post-surgery, then normalize after seven days, in young catheter-implanted rats. 14 In the present study the animals recovered quickly from surgery, demonstrated by there being no changes in food consumption or body weight gain post-surgery. Generally, the animals displayed normal behaviour and unrestricted movements in both studies, providing a clear advantage over external infusion devices, which may restrict the movements of the animal. In the subcutaneous infusion study, severe seromas formed over pumps in 17% of the animals; however, this was ascribed primarily to surgical technique and not considered to be related to housing conditions. Continuous adjustments, with emphasis on the size of the subcutaneous pocket for the pump, were implemented, reducing occurrence of severe seromas to 6% in the intravenous study. Thus, the number of animals sacrificed prematurely due to pump-related issues was reduced compared with HI infusion from external pumps (6% versus 11%). 6
Initially, compatibility of HI with the battery-driven iPRECIO pump and the osmotic Alzet pump was evaluated in vitro to avoid unnecessary use of animals. Though the Alzet pump has been used in long-term rat studies for subcutaneous insulin infusion,15,16 it has failed to lower blood glucose levels adequately in several studies,15,17,18 possibly due to insulin aggregation in the pumps.15,17 Additionally, infused volumes by the Alzet pumps over two weeks have been shown to be only 79% of expected values, compared with 96% by the iPRECIO pumps. 19 Our in vitro results indicated evaporation from Alzet pumps, resulting in increased peptide concentrations of the remaining formulation in the pumps. Furthermore, in contrast to the iPRECIO pump, the Alzet pump cannot be refilled, eliminating the possibility of a post-surgery recovery period. Therefore the decision was made to move forward with the iPRECIO pump.
In the intravenous infusion study, the normalization of blood glucose levels after two weeks in HI-infused animals coincided with a simultaneous general decrease in dosed volumes, suggesting clotting of catheters from approximately day 15 and onwards. Supporting this, necropsy revealed a considerable number of animals with coiled catheters and/or loss of intravenous access with a concurrent lack of exposure in most of the HI-infused animals. As the flank and neck, where the pump and catheter were implanted, respectively, represent areas of large mobility, small vibrations of the pump itself might slowly have pulled the catheters out of the veins. This is in contrast to a previous study in which the use of the pump for intravenous infusion for up to seven weeks in rats seemed to be unproblematic. 20 Clotting of catheters might be attributable to the very low infusion rates used (3–5 µL/h); however, these infusion rates were comparable with those used in the latter study. Formation of neutralizing anti-insulin antibodies should be considered. The presence of antibodies was not evaluated, as all the data corroborated the explanation of lack of infusion due to catheter-related complications. Presence of antibodies would not explain the decreased dosed volumes and clotted/coiled catheters.
Thus, successful group housing of HI-infused rats with a persistent hypoglycaemic effect was achieved for two weeks following post-surgical recovery. Refining the surgical procedure, increasing the fixation points of the pump catheter might prevent coiling of catheters and loss of intravenous access prolonging duration of infusion. Also, including a prosthetic semi-resorbable mesh for example to promote tissue adherence, as used in hernia repair in humans, 21 might improve fixation of the pump itself. Furthermore, the pump has been used for infusion through the femoral vein in rats. 22 Using the caudal vena cava via the femoral vein rather than the cranial vena cava via the jugular vein might prolong catheter patency, due to the larger vessel diameter, and so improve blood flow around the catheter tip and retard the growth of reactive tissue.23,24 Such adjustments might have the potential to prolong duration of intravenous HI infusion in growing rats, and provide a more versatile comparator model.
With subcutaneous HI infusion over a three-week period, HI exposure and hypoglycaemic effect were highly variable, with some of the plasma samples from HI-infused animals showing no detectable exposure. This was despite the fact that the pumps performed well in this study with regard to dosed volumes, and with no apparent clotting of catheters and high stability of HI in the pumps. This is further discussed in the Supplementary information.
In summary, with the presented technique, the implantable pump offers an opportunity for continuous intravenous delivery of HI to group-housed rats, allowing complete freedom of movement as well as social interaction, and so provide a clear welfare advantage over external infusion devices. Additionally, it eliminates the stress related to single housing, which may impact on the regulation of glucose homeostasis. This provides a new group-housed rodent model of persistent insulin-induced hypoglycaemia for use as a comparator in repeated-dose toxicity studies of two-week duration with new long-acting insulin analogues. However, longer duration of infusion requires surgical refinements.
Footnotes
Acknowledgements
The author(s) would like to thank Line O Bertelsen, Estelle Watson, and Alan Broadmeadow for their contributions to the studies.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: VFHJ and JL were supported by the LifePharm Centre for in vivo pharmacology. Studies were funded by Novo Nordisk A/S.
