Abstract
Intravenous drug administration in adult swine is difficult to perform due to inaccessible superficial veins and thick subcutaneous fat layers. However, successful intravenous drug administration is critical for many biomedical applications including pharmacokinetic studies as extravascular drug administration can influence the drug’s absorption and elimination rate. The purpose of this study was to develop and refine an effective technique for indwelling auricular vein catheter placement in the conscious mature sow. We developed a protocol using a topical anesthetic cream and minimal physical restraint to place indwelling catheters in the auricular vein of six multiparous sows. This method was quick (3 min 20 s ± 8 s [mean ± SE per catheter]), effective (11/12 catheters successfully placed) and reliable, allowing a large drug volume (20–22 mL) to be administrated successfully during the trial without relying on prolonged restraint or general anesthesia of the sow.
Intravenous access is important in veterinary medicine to administer drugs for treatment of clinical disease. However, intravenous drug administration is difficult to perform on swine due to inaccessible superficial veins and thick subcutaneous fat layers. 1 For all swine, ages and weights, the two most common methods for intravenous administration include: (1) temporary catheterization 2 or (2) jugular vein injection using a ‘blind-stick’ approach (defined as drug administered without vein visualization3,4). Blind-stick injection is unreliable as it is difficult to ensure that the drug is not administered extravascularly. Thus, access to the vein using an indwelling catheter is the most appropriate method for intravenous drug administration.
To the authors’ knowledge three procedures for auricular vein catheterization for swine have been reported in the literature.5–7 Phillips and colleagues 5 successfully placed auricular vein catheters for blood collection while sows were anesthetized. General anesthesia use can contribute to increased variation when conducting pharmacokinetic studies because anesthetic agent systemic effects on clearance, absorption time and volume of distribution for the drug of interest are most often unknown. Porter and colleagues 6 placed catheters in restrained sows without local or general anesthesia being used. Single catheter placement took on average 30 min, requiring prolonged sow restraint. In clinical cases where an animal may have cardiovascular or respiratory complications, prolonged restraint is inappropriate as it may cause the animal to go into distress. Rushen and colleagues 7 did not provide any details concerning the catheter placement process itself. Although catheterization of other veins including the jugular vein,8,9 subcutaneous abdominal vein, 10 and uterine vein 11 has been successfully conducted in swine, catheterization of these veins requires anesthesia or prolonged restraint of the patient. This indwelling ear vein catheter placement technique is novel because we developed and refined an effective access route to confidently administer drugs intravenously in adult swine without relying on prolonged restraint or general anesthesia. Prolonged restraint may result in both physical and emotional distress for a compromised animal and general anesthesia may influence drug metabolism, absorption and efficacy. Furthermore, this novel auricular vein catheter placement technique utilized local anesthetic to minimize pain sensitivity during placement. The combination of sow welfare during placement and the resultant success of the intravenous access reinforces the novelty of this procedure.
This laboratory technique was carried out on healthy female sows (Sus scrofa domestica; age: 1–3 years, weight: 202–222 kg) concurrently enrolled in a pharmacokinetic study in which they received either an oral or intravenous meloxicam dose (Loxicam®; Norbrook Inc, Lenexa, KS, USA). Health status was determined by physical examination of each sow. Physical examination included lung and heart auscultation, rectal temperature and reproductive tract ultrasonography. The animals were cared for in accordance with the Animal Welfare Act and the Guide for the Care and Use of Laboratory Animals (8th edition). This work was performed in an Association for Assessment and Accreditation of Laboratory Animal Care (AALAC) accredited facility at the Iowa State University College of Veterinary Medicine. Sows were housed individually in pens (3.7 m length × 1.4 m width × 1.2 m height) with a rubber mat (3.5 m length × 1.3 m width). Lights were on a 12:12 light–dark cycle (light hours [06:00 to 18:00 h]) and room temperature ranged between 12.5℃ and 20.5℃. No other additional bedding material (straw, hay, etc.) was provided but the sows were provided with cotton rope for environmental enrichment. The animals had free access to drinking water and were hand-fed a custom-mixed diet composed of corn, soybean meal and soy hulls. They were allowed a 14-day acclimatization period before the beginning of the study. Experimental procedures were approved by the Institutional Animal Care and Use Committee at Iowa State University (USA).Two sows were used to refine the catheterization technique prior to study commencement, and the remaining six sows were catheterized twice, once in each ear during the trial.
One hour prior to catheterization, a topical anesthetic cream (15 g, EMLA cream; Hi-Tech Pharmacal Co Inc, Amityville, NY, USA) was applied to each sow’s ear pinna covering the entire length of the ear from base to tip (Figure 1). After one hour, the excess cream was wiped off using a paper towel. Once sows were manually restrained with a pig snare, the ear was cleaned with 70% isopropyl alcohol and 0.75% iodine scrub. An indwelling catheter (22 gauge, 2.54 cm long; manufacturing code: SMIT-9986, Smiths Medical, Dublin, OH, USA) was inserted into the distal portion of the largest and most prominent auricular vein (per visual assessment) until blood was present in the catheter hub. Once placed, the stylet was removed and transparent tape (Dermiclear Tape; Johnson & Johnson, New Brunswick, NJ, USA) was placed over the catheter to temporarily secure it. The catheter was capped with a threaded injection cap (Hospira Inc, Lake Forest, IL, USA) and the skin surrounding the catheter was dried with gauze to absorb any blood that may otherwise have prevented proper tape adherence to the skin.
Topical anesthetic cream (15 g, EMLA cream; 2.5% lidocaine and 2.5% prilocaine) was applied to each sow’s ear pinna covering the entire length of the ear from base to tip.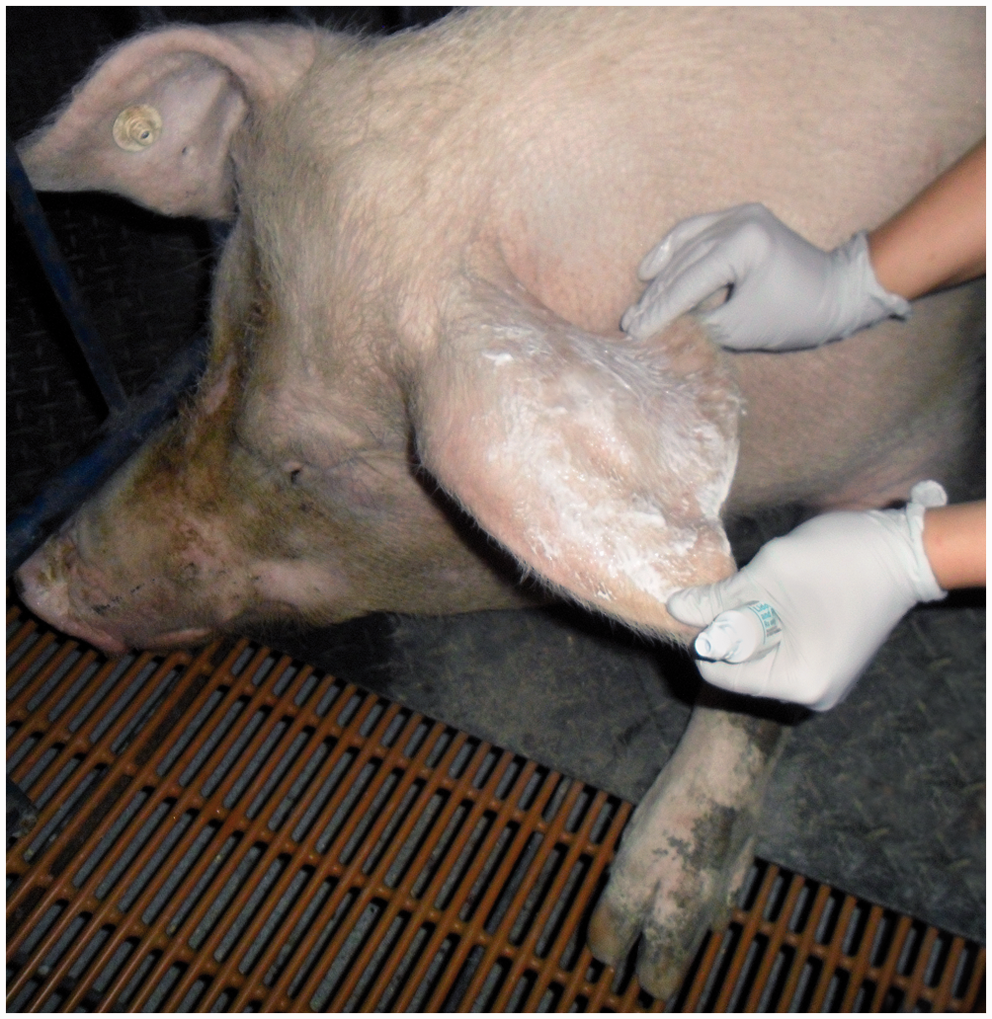
The catheter hub was glued to the ear skin using surgical glue (3 M Vetbond Tissue Adhesive; 3 M Animal Care Products, St Paul, MN, USA) applied underneath the transparent tape for further security. Two transparent dressings (Tegaderm; 3 M Animal Care Products) were placed over the entire catheter hub and injection port. Special care was taken to orientate the dressing where the distal edges could be folded over the ear for further security (Figure 2). A 30.5 cm butterfly catheter (SurFlo Plastics & Engineering Inc, Warren, MI, USA) was inserted through the transparent dressing perforating the injection cap and flushed with 6 mL heparinized saline (Hospira Inc; 2 IU heparin/mL saline) to ensure patency.
Final layer of transparent dressing is placed on the catheter hub and injection port, which prevented displacement of the catheter from the vein.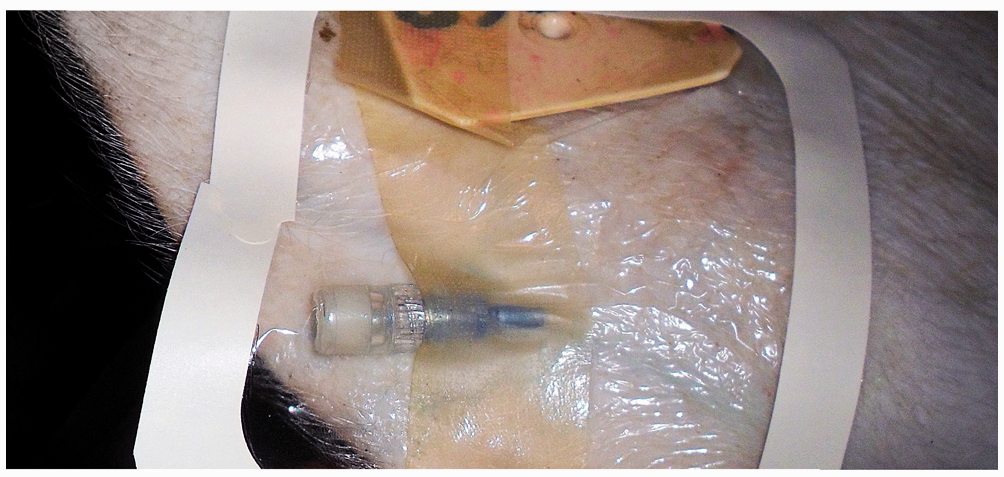
Three hours after drug administration, the transparent dressing, tape and catheter were removed from the auricular vein. Gauze was taped over the catheter site providing pressure to prevent bleeding which was removed one hour later and the ear was checked for bleeding. If excess surgical glue was difficult to remove, hydrogen peroxide was applied to the ear to assist in removal. Ears were cleaned with isopropyl alcohol and evaluated for inflammation or hematoma formation.
Prior to the study commencement, two sows were used to refine catheter placement and access technique. The catheters were placed as described above with the exception that the sows were not physically restrained. For these efforts, they were controlled by providing feed in a small pan to limit excessive movement. Success rate for catheter placement was low (25%) resulting in multiple hematoma formation and failed patency. Therefore manual restraint with a snare was required.
The original goal for this project was to develop a technique for indwelling catheter placement without the use of physical restraint. However, during our pilot study using only two sows, catheter placement without restraint was difficult to perform and had a low success rate. Based on these difficulties, the six sows on trial were restrained using a pig snare for catheter placement and drug administration. Although restraint may have caused temporary stress, the success rate improved dramatically (92%); hematoma formation was rare and overall handling and interaction time per sow decreased. Placement time was also less compared with previous published techniques using physical restraint. 6 Although the use of a sedative to restrain the sow may have caused less temporary stress compared with the snare, the sedative drug’s unknown systemic effects on clearance, absorption time and volume of distribution made its use problematic in the context of a pharmacokinetic study.
Intravenous catheters were successfully placed in both ears of six on-trial restrained sows for a total of 12 successful auricular vein catheter placements. The catheters were placed in the auricular vein within 3 min 20 s ± 8 s (mean ± SE per catheter) from the time sow restraint began. Catheter patency was evaluated with a sterile saline flush and all catheters, with one exception, remained patent for drug administration. Each sow was administrated 20–22 mL meloxicam over 90 s. Total drug volume was successfully administered to each sow while restrained using a pig snare.
No permanent physical alterations were noted with this technique. The most prominent change to the ear was temporary pinna discoloration at the cream application site and mild epithelial bruising around the catheter placement site. Full recovery of the ear occurred one week after placement.
The catheter that failed to maintain patency was removed by a neighboring sow through pen bars. After this incident, the sows were moved before the trial start into housing that minimized sow-to-sow contact until after the drug was administered via the catheter. Although the catheters could hypothetically be removed by physical force (rubbing, scratching), no such behavior was noted during the trial. No sows appeared to react to catheter presence and no sows attempted to remove their own catheter.
We found the described catheter placement technique to be an effective and quick way to administer drug intravenously. This technique provides secure intravenous access and can be utilized for studies or treatments requiring intravenous drug administration. Blood was not collected from the auricular vein catheter and further evaluation of the efficacy of this type of catheter for blood collection is needed. The application of this method contributes to the ‘refinement’ of experimental procedures, according to the principle of ‘3Rs’, by reducing unnecessary sow stress and pain associated with prolonged restraint and catheter placement.
Footnotes
Declaration of conflicting interests
Actual or potential conflicts of interest do not exist.
