Abstract
It is common to test medical devices in large animal studies that are or could also be used in humans. In this short report we describe the use of a ureteral J-stent for the evaluation of biodegradable tubular constructs for tissue reconstruction, and the regeneration of ureters in Saanen goats. Similarly to a previous study in pigs, the ureteral J-stent was blindly inserted until some resistance was met. During evaluation of the goats after three months, perforation of the renal cortex by the stent was observed in four out of seven animals. These results indicated that blind stent placement was not possible in goats. In four new goats, clinical protocols were followed using X-ray and iodinated contrast fluids to visualize the kidney and stent during stent placement. With this adaptation the stents were successfully placed in the kidneys of these four new goats with minimal additional effort. It is likely that other groups in other fields ran into similar problems that could have been avoided by following clinical protocols. Therefore, we would like to stress the importance of following clinical protocols when using medical devices in animals to prevent unnecessary suffering and to reduce the number of animals needed.
The application of new surgical techniques often requires evaluation in large laboratory animals to study the effectiveness and safety. 1 This is especially important in emerging fields such as tissue engineering and regenerative medicine. Tissue engineering aims to replace damaged organs and tissues by providing an alternative to autologous donor tissue or when standard surgical procedures are not possible. 2 One tissue engineering application that we are interested in is the replacement of a long segment of the ureter with a tubular template after trauma or iatrogenic injury. 3 To prevent obstruction and kidney damage a ureteral stent is placed during surgery. This stent facilitates sufficient urine transport during the initial wound healing phase, which is accompanied by tissue contraction and scar formation. A ‘J’ tail curl at the ends helps to keep the stent in the correct place. 4 In previous pig studies by our group, blind insertion of this stent into the proximal ureter/pyelum of the kidney was uneventful. 5
Recently, we switched from young Landrace pigs to adult goats as our animal model for ureter reconstruction because the pigs used were growing at a fast rate (30 kg weight gain in 3 months). The rapid growth was also associated with fast wound healing as evidenced by the fast skin closure in a stoma model, which may have influenced the regenerative outcome. 6 Despite the smaller overall size of the kidneys, pyelum and ureteral diameter, stenting was easily performed in the cadaver material in preparation for the animal experiment.
Initially, seven female Saanen goats (Fam. Van Dijk, Achterberg, The Netherlands) weighing 50–70 kg were used in this study (based on power analysis). The study was approved by the Nijmegen Medical Center animal ethics committee (RU-DEC-2014-223), and all procedures were performed according to the Institute of Laboratory Animal Research guide for laboratory animals. 7 As catheterization was required, only female goats were used due to the curved urethra in male goats. The goats were housed in groups at the Institute’s farm with free access to water and a restricted diet. They were sedated according to the Institute’s protocol prior to surgery, and were treated with analgesia and antibiotics afterwards. In all goats, the right ureter was located and mobilized through a flank incision. The ureter was cut, spatulated and the ureteral stent (4.7 Fr/22–32 cm multi-length double J stent, Inlay Optima; Bard Medical, Covington, GA, USA) was blindly inserted into the proximal ureter until some resistance was met as an indicator of its correct positioning. At that point, the guide wire was removed to allow the proximal ‘J’ to curl inside the pyelum. The tissue-engineered template was positioned over the stent and the stent was then inserted into the distal ureter and urinary bladder. Finally, the tissue-engineered template was connected to both spatulated openings of the ureter by end-to-end anastomosis.
After three months the goats were killed by an overdose of pentobarbital for evaluation. We noticed that the stents had perforated the kidneys in four out of seven treated goats (Figure 1). In one case this resulted in a large urinoma on top of the kidney, while in the other three cases inflammation in the kidneys was noted. This clearly was an unwanted result; the wound healing results were unreliable and this may have caused unnecessary suffering for the goats. Upon close inspection of the harvested tissue, it was obvious that the kidney tissue of the goats was much softer than that of the pigs. This may have been missed in the cadaver study due to the room temperature and the clotting of blood in the highly vascularized kidney, which may have increased the stiffness and resistance of the kidney. In addition, due to the small size of the renal calyces and pyelum, it was difficult for the ‘J’ end of the stent to correctly curl inside the kidney. Therefore, it was more likely that the stent had penetrated the kidney cortex during placement.
(a) Macroscopic view of the right kidney after perforation by the ureteral catheter (white arrowhead). (b) X-ray scan without iodinated contrast fluids. Stent was clearly visible in the system and perforated the renal cortex (white arrowhead). (c) X-ray scan with iodinated contrast fluids which filled the kidney calyces, and the stent could be seen in the renal cortex outside the calyces (white arrowhead).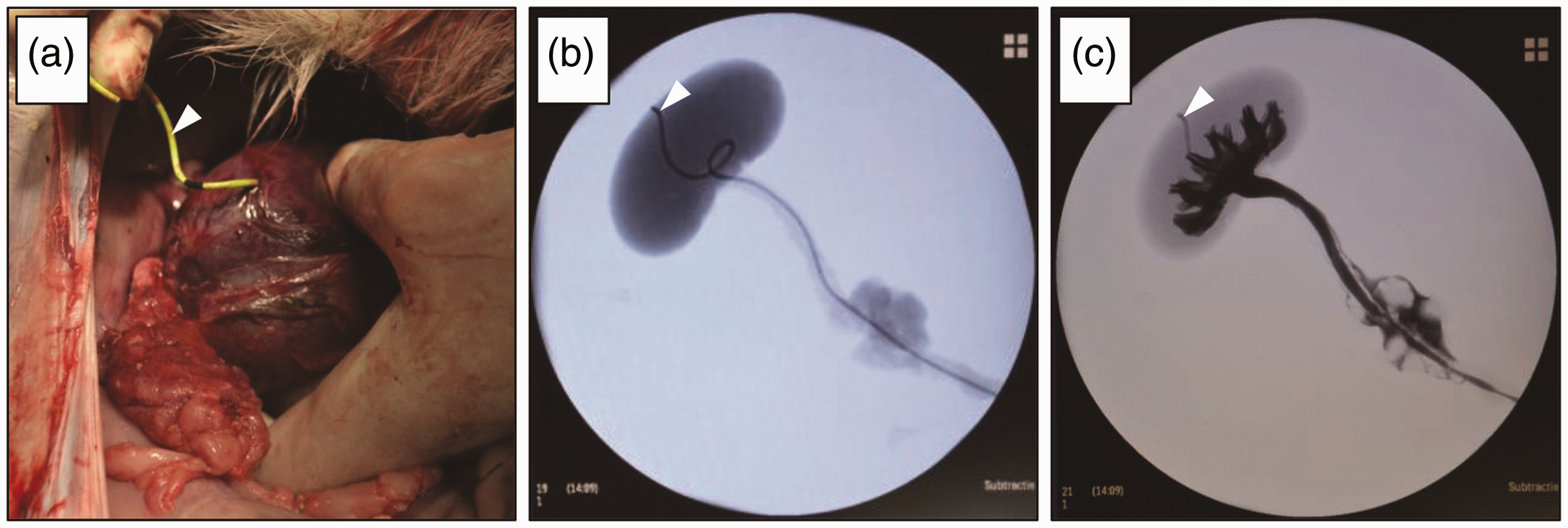
In routine human clinical practice, ureteral stents are mainly inserted using contrast fluids and with X-ray guidance.
8
This allows visualization of the kidney, and correct positioning of the stent inside the pyelum can be verified. In view of the observed kidney perforations, we used iodinated contrast fluids (Iomeron 300, Bracco Imaging, Konstanz, Germany) and C-arm X-ray (Veradius, Philips, Amsterdam, The Netherlands) guidance to position the ureteral stent in an additional four goats (Figure 2). By following this clinical protocol, we managed to successfully position the stent with a minimal increase in surgery time. When the ureteral stent penetrated the renal cortex (Figure 2b), we were able to adjust the position until the stent was correctly placed (Figure 2c). This way we avoided unwanted side-effects, and the regeneration of the ureter progressed as was initially intended in the experiment.
(a) X-ray scan of the right kidney (white circle) with iodinated contrast fluids to visualize the renal calyces before inserting the stent during surgery. (b) After inserting the stent, the tip perforated the renal cortex (white arrowhead), indicating a misplaced stent. (c) The stent was retracted and repositioned. The stent now curled nicely inside the calyces (white arrowhead) and no penetration of the renal cortex was seen, confirming a successful stent placement.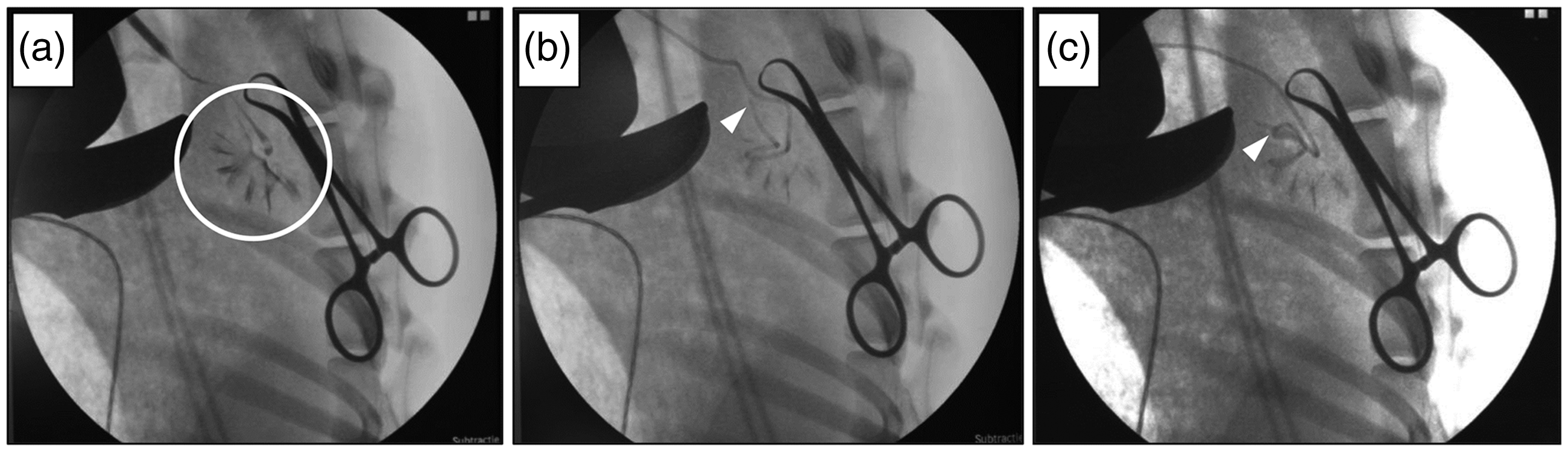
It is likely that other groups in different fields have similar experiences with the use of medical devices in their experiments. Based on our experience with ureteral stenting in goats, we would like to advise the use of routinely applied clinical techniques when using medical devices in animals. This refinement is often fast and easy to perform and should prevent unnecessary suffering in animals and reduce the number of animals required due to technical failures.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research received support from the Radboudumc Research Program Reconstructive and Regenerative Medicine, and no other specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
