Abstract
The standard housing temperature in animal facilities is substantially below the lower critical temperature of mice. This does not only endanger animal welfare, it can also jeopardize scientific research as cold stress has a major impact on mouse physiology. There is some evidence that deep bedding, comparable to nesting material, can help mice to reduce heat loss. Whenever changes are applied to the cage environment, the potential impact on experimental results, including variation, needs to be assessed. An increased variation can result in a conflict between reduction and refinement, when more animals are needed for significance due to the housing design. The aim of this study was to assess the impact of different bedding volumes (0.5 L, 1.5 L and 6 L per type III cage) on mean values and coefficient of variation (CV) of physiological (pentobarbital sleeping time, blood and anatomical parameters) and behavioural parameters (open-field and novel object recognition tests) of group-housed female and male BALB/c and C57BL/6 mice. A larger bedding volume did not interfere with the CVs, but influenced mean values of organ weights and tail lengths. Mice housed on deeper bedding showed a significant reduction in adrenal, liver, kidney and heart weights as well as an increase in tail lengths; these anatomical changes are akin to warm adaptation, and were previously observed for mice housed under warmer environments. A larger bedding volume appears to be a sensible way to reduce cold stress for laboratory mice without increasing variation in experimental results.
Refinement in the husbandry of laboratory animals includes a cage environment adapted to their species-specific needs. 1 The standard housing temperature of 22℃ is below the lower critical temperature of mice (≈ 30℃) 2 and can result in various physiological changes due to cold stress. 3 Nesting material can at least partially compensate for a colder environment as it helps to reduce heat loss for mice, especially during resting periods. 4 Although the positive effects of nesting material are widely accepted, 5 some research facilities might waive its use to avoid interaction with specific mouse models 6 and experimental results 7 or simply due to financial and practical reasons. We aim to investigate whether a larger bedding volume can also alleviate thermal stress for laboratory mice. Preference tests have recently demonstrated that female mice prefer cages with deep bedding compared with shallow bedding. 8 Deeper cage bedding has already been associated with a stable core body temperature during the resting phase, as it allows mice to burrow and create a microclimate according to their needs. 9 In search of more evidence to support the hypothesis that deep bedding enables mice to reduce heat loss, we analysed the impact of bedding volume on anatomical and blood parameters that are known to be influenced by ambient temperature.10,11
Apart from the potential to alleviate cold stress, bedding volume might interact with other experimental parameters, including behaviour and drug metabolism. In contrast to nesting material, 12 the influence of bedding volume on learning or anxiety-like behaviour has not yet been evaluated. The metabolic rate of pentobarbital is known to be influenced by ammonia concentration. 13 Bedding volume interacts with ammonia concentration, 14 therefore we hypothesized that bedding volume might interfere with pentobarbital sleeping time.
Any changes in housing conditions can endanger reproducibility and comparability of experimental results. 5 Due to interaction with the sample size needed for significance, particular attention should be drawn to the influence of cage designs on variation.15–18 A more complex cage environment has been reported to increase within-group variability, 15 making it necessary to analyse the impact of bedding volume on variations of the obtained outcome measures.
Our aim was to document the effect of deeper bedding on experiments, therefore mean values and variation of behavioural (performance in open-field and novel object recognition tests) and physiological data (pentobarbital sleeping time, anatomical and blood parameters) of laboratory mice housed on different bedding volumes were analysed.
Animals
Female and male BALB/cByJRj and C57BL/6NRj (108 female and 36 male mice per strain) were obtained from Janvier Labs, Le Genest Saint Isle, France. The animals’ microbiological status was determined to be specific pathogen-free (SPF) in line with Federation of European Laboratory Animal Science Associations (FELASA) recommendations.
19
On arrival, the mice were three weeks (average weight at 4 weeks: BALB/c, female 14.0 ± 1.5 g, male 14.8 ± 1.8 g; C57BL/6, female 11.7 ± 1 g, male 12.2 ± 1.8 g) or 12 weeks of age (females only, average weight at 13 weeks: BALB/c, 21.4 ± 1.9 g; C57BL/6, 21.2 ± 1.0 g), and were killed at 15 weeks of age. The mice of each batch were housed in groups of four and were assigned to three experimental groups (0.5 L [≈0.5 cm bedding depths], 1.5 L [≈ 1.5 cm bedding depths] and 6 L [≈ 6 cm bedding depths] per type III cage). A Latin square design was used to make sure that the order in which the mice were assigned to the bedding volumes was different for every batch. Performance in open-field and novel object recognition tests, blood parameters (animals housed for 12 weeks), intra-cage ammonia levels (animals housed for 3 weeks), pentobarbital sleeping time as well as anatomical parameters (all mice) were analysed. A timeline for when each test was performed is provided in Figure 1. For every procedure one cage of each volume was tested per day. To minimize the potential impact of the time of day on outcome measures, the order in which the bedding volumes were tested varied between the replicates.
Experimental design. BALB/c and C57BL/6 mice housed on 0.5 L, 1.5 L or 6 L for 12 weeks (females and males; three cages/bedding volume/strain/sex) or three weeks (females; three cages/bedding volume/strain for narcosis on day 1 respectively day 14).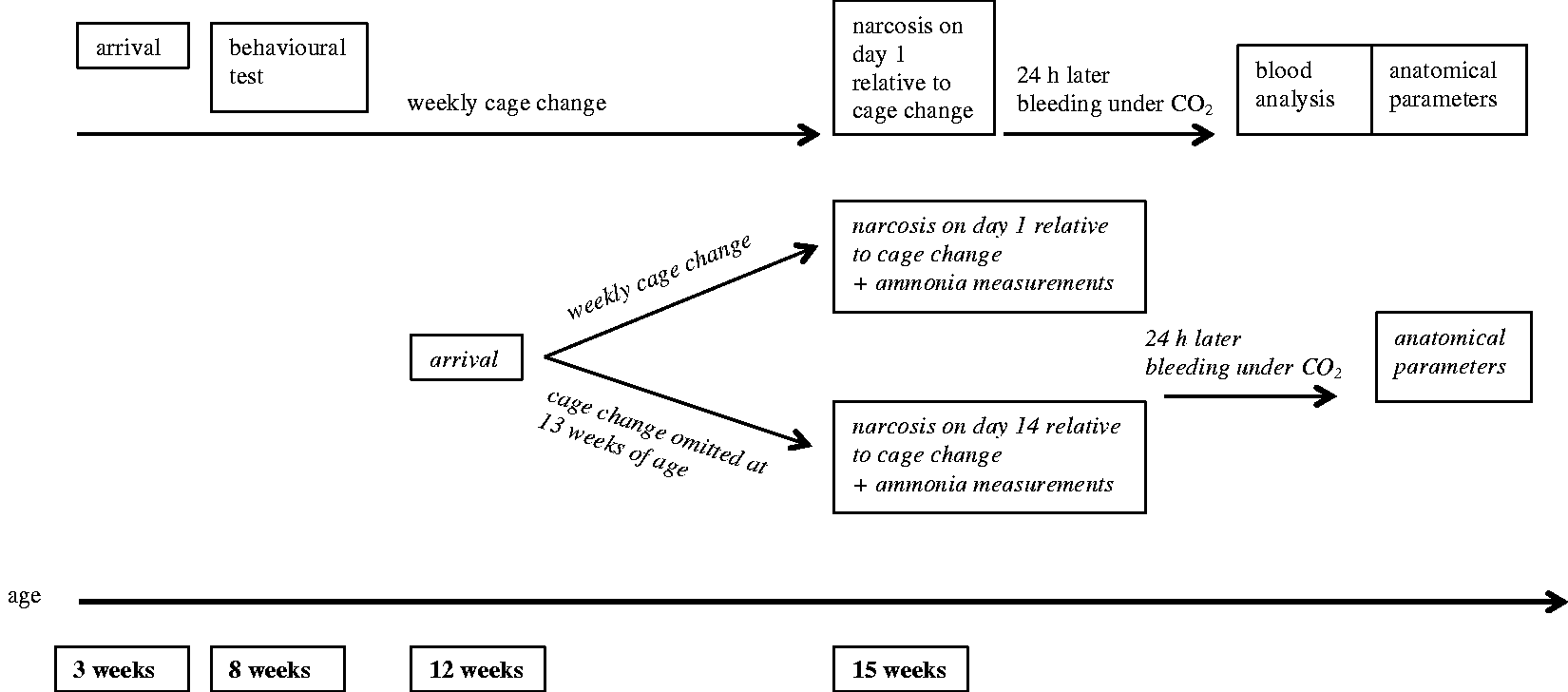
The animals were housed in type III (375 mm × 215 mm × 150 mm) Makrolon cages (Bioscape, Castrop-Rauxel, Germany) in an open rack at a temperature of 22 ± 2℃ with 55 ± 10% relative humidity and 10–16 air changes per hour. The room was maintained on a 12/12 h light/dark cycle (lights on 06:00 h central European time [CET]) with artificial light (140 ± 10 lux in the rack, 1 m above the floor). The position of the cages in the rack was rotated weekly to reduce potential influence of environmental conditions on outcome measures. The three different bedding volumes were placed on one shelf, initially the position of the cages was rotated within the shelf, then all three cages were moved to a different shelf. For the animals that arrived at three weeks of age 12 positions in the rack (four shelves with three positions each) were available. Consequently each cage occupied each position in the rack before the animals were killed. For animals that arrived at 12 weeks of age only three positions (one shelf in the rack) were used and the volumes were rotated within the shelf.
The same amount of food (Ø10 mm pelleted diet, Altromin No. 1324; Altromin, Lage, Germany) and tap water were available ad libitum in every cage. Coarse-grained aspen chips were used as bedding (ABEDD; Lab & Vet Service GmbH, Vienna, Austria), the amount of bedding varied according to the experimental group.
Materials and methods
Ethics statement
Housing and experimental procedures of all experiments conform to the German Animal Welfare Act and the European Convention for the Protection of Vertebrate Animals used for Experimental and Other Scientific Purposes (Council of Europe, ETS No. 123, Strasbourg 2006), and were approved by German authorities.
Behavioural test
These tests were chosen because they are commonly used methods, especially in pharmacology research, to assess anxiety-related behaviour and cognitive functions in mice.
Open-field test
The open-field test was carried out between 09:00 and 12:00 h CET with lights turned off using a camera with night shot function (Sony HDR-CX 900EB; Sony, Tokyo, Japan). Further procedures corresponded to methods described by Tsai et al. 16 Freezing frequency and travel distance of each animal were obtained.
Novel object recognition test
The open-field test was followed by a two-day novel object recognition test using the same experimental device as described above. On day 1, two identical looking wood cubes (2.4 × 2.4 × 2.4 cm; VEDES Großhandel GmbH, Nümberg, Germany) were placed in the central area with a distance of 10–12 cm from each other. On day 2, a wood cube and a novel object of the same height (wood triangle) were placed in the central area. Exploration time (T=touching the object with nose and/or directing the nose at a distance ≤2 cm to the object) 20 on days 1 and 2 was recorded. A discrimination index (DI=[Tnovel – Tfamiliar]/[Tnovel + Tfamiliar]) was used to assess the discrimination between the novel and familiar objects. 20 Animals that did not explore either of the objects on day 1 or day 2 were excluded from the analysis. Female BALB/c mice needed to be excluded completely from analysis as only a few of the animals explored the objects.
Pentobarbital metabolic rate
Sleeping time was used as an indicator of metabolic rate. 21 To assess the interaction between bedding volumes, ammonia levels and pentobarbital metabolism, sleeping time was measured on day 1 (animals housed for 12 weeks and half of the animals housed for 3 weeks) or day 14 (half of the animals housed for 3 weeks) after cage change (Figure 1).
The animals were anaesthetized with 60 mg/kg of pentobarbital sodium (Narcoren®; Merial GmbH, Hallbergmoos, Germany), diluted with isotone saline solution (0.9%). Mice were put in a small cage for the onset of sleep and then placed in a supine position on a heating plate (Haake; Omnilab, Bremen, Germany) 10 min after injection. The heating plate consisted of a water pump (water temperature 42℃) and a thermostat to ensure a constant temperature on the heating plate. Metal attachments on top of the heating plate divided it into separate sleeping berths to avoid contact between animals. Narcosis started at 08:00 h CET, the animals within one cage were injected sequentially at 5 min intervals. The sleeping time was recorded using a camera (Sony HDR-CX 900EB) to assess the first move (of head, fore or hind legs) and then movement into a lateral position of each mouse. The animals were returned to their home cage after narcosis.
Ammonia
Prior to the pentobarbital narcosis, ammonia levels in the cage environment were obtained for all the groups housed for three weeks. Measurements were carried out at the nesting site (pocket within the bedding where the mice huddled during daytime) and the area of faeces and urine disposal of each cage (approximately 1 cm above the bedding) using Draeger-Tubes® (Ammonia 0.25/a; Draeger Safety AG & Co. KGaA, Hamburg, Germany) and a hand-operated pump (Accuro® gas detection pump; Draeger Safety AG & Co. KGaA).
Blood sampling and analysis
All mice were anaesthetized with carbon dioxide
22
between 08:00 and 12:00 h CET, and bled with a shortened lithium–heparin capillary tube (32 µL Hirschmann®; Hirschmann Laborgeräte, Eberstadt, Germany). Only blood from animals housed for 12 weeks were analysed. The blood was collected in dipotassium–EDTA tubes (300 µL Microvette® CB300K2E; Sarstedt, Nümbrecht, Germany) for assessment of haematological parameters, and in lithium–heparin tubes (300 µL, Microvette® CB300LH; Sarstedt) for clinical chemistry parameters, including electrolytes. Vet ABC (SCIL Animal Care Company GmbH, Viernheim, Germany) was used to analyse white blood cells (WBC), red blood cells (RBC), haemoglobin (HGB) and haematocrit (HTC); sodium (Na+), potassium (K+), calcium (Ca++) and chloride (Cl–) were analysed with Rapidlab 1260 (Siemens Healthcare Diagnostics GmbH, Eschborn, Germany); alanine aminotransferase (ALT), alkaline phosphatase (ALP), aspartate aminotransferase (AST), urea, creatinine (Crea), cholesterol (Chol), triglycerides (Tri), total protein (TP), albumin (Alb), total calcium (Ca total), and lactate dehydrogenase (LDH) with the autoanalyser Cobas® c311 (Roche Diagnostics GmbH, Mannheim, Germany). Haematological data and electrolytes were determined between 1 and 2 h after sampling using whole blood anticoagulated with EDTA or lithium–heparin. The residual lithium heparinized blood was centrifuged (10,000 ×
Anatomical parameters
Final body weight was measured prior to carbon dioxide anaesthesia. After blood sampling the animals’ body and tail lengths as well as relative organ weights (adrenal, spleen, kidney, liver and heart) were obtained. The organs were kept in a moist chamber and weighted using a sensitive electronic balance (BP 211D; Sartorius AG, Göttingen, Germany).
Statistical analysis
Data were analysed with IBM
Mice housed for 12 weeks
Initially the data were analysed for strain and sex effects. The data were later split by strain and sex and the impact of bedding volume on travel distance and freezing (open-field test), exploration time of novel and familiar objects (novel object recognition test), first move and move into lateral position (pentobarbital metabolic rate) as well as on blood and anatomical parameters was analysed using the Kruskal–Wallis H-test.
The coefficient of variation (CV) of the data obtained from each cage was calculated. For open-field test, pentobarbital narcosis, haematological and clinical chemistry data as well as anatomical parameters significant strain or sex differences regarding the CVs were not detected. Therefore, the average CVs of each item of both strains and sexes were pooled and compared using the Friedman test. No CVs were calculated for the novel object recognition test due to the small sample size.
Mice housed for three weeks
Intra-cage ammonia levels were grouped into eight categories (0 ppm = score 0; > 0 – ≤ 0.1 ppm = score 1; > 0.1 – ≤ 0.25 ppm = score 2; > 0.25 – ≤ 0.5 ppm = score 3; > 0.5 – ≤ 0.75 ppm = score 4; > 0.75 – ≤ 1 ppm = score 5; > 1 – ≤ 3 ppm = score 6; ≥ 3 ppm = score 7) and analysed using the Kruskal–Wallis H-test to detect differences between the bedding volumes. Additionally, ammonia levels were analysed with the Wilcoxon signed-rank test to detect differences between the nesting site and the area of faeces and urine disposal within one cage. Pentobarbital metabolic rates and anatomical parameters were analysed according to day relative to cage change (ammonia levels) and strain; the data were later split by the two factors and analysed according to bedding volume using the Kruskal–Wallis H-test. As the day relative to cage change did not affect the anatomical parameters, the data presented in the results were only split by strain.
Results
Anatomical parameters
Mice housed for 12 weeks
The anatomical parameters were significantly affected by the amount of cage bedding provided. Mice housed on deep bedding had smaller adrenal, kidney, liver and heart weights as well as larger body and tail lengths compared with groups kept on shallow bedding (Figure 2). Female BALB/c and C57BL/6 mice showed significant differences between the bedding volume for body (P = 0.012; P = 0.005) and tail (P = 0.006; P = 0.032) lengths; and for adrenal (P = 0.045; P = 0.005), kidney (P = 0.010 for C57BL/6 only) and heart (P = 0.002 for BALB/c only) weights. Tail lengths (P = 0.006), adrenal (P < 0.001) and heart (P = 0.001) weights of male C57BL/6 mice as well as kidney (P < 0.001; P = 0.002) and liver (P = 0.009; P = 0.002) weights of male BALB/c and C57BL/6 mice were significantly influenced by the amount of cage bedding provided. The difference became particularly obvious between groups housed on bedding of 0.5 L compared with 6 L (Figure 2). Female and male BALB/c mice exhibited larger body and tail lengths as well as higher relative organ weights compared with female and male C57BL/6 mice (P < 0.05, except for adrenal weights). No distinct gender differences were noted. While female BALB/c and C57BL/6 mice had significantly larger relative adrenal and spleen weights (P < 0.001), male mice had higher final body weights (P < 0.001), larger body and tail lengths (P < 0.001 female versus male BALB/c) as well as larger relative kidney weights (P < 0.001).
(a)–(h) Anatomical parameters of female (f) and male (m) BALB/c and C57BL/6 mice (mean ± SD); *P < 0.05; females: BALB/c 0.5 L and 1.5 L (n = 12), 6 L (n = 11), C57BL/6 0.5 L and 6 L (n = 11), 1.5 L (n = 12); males (n = 12).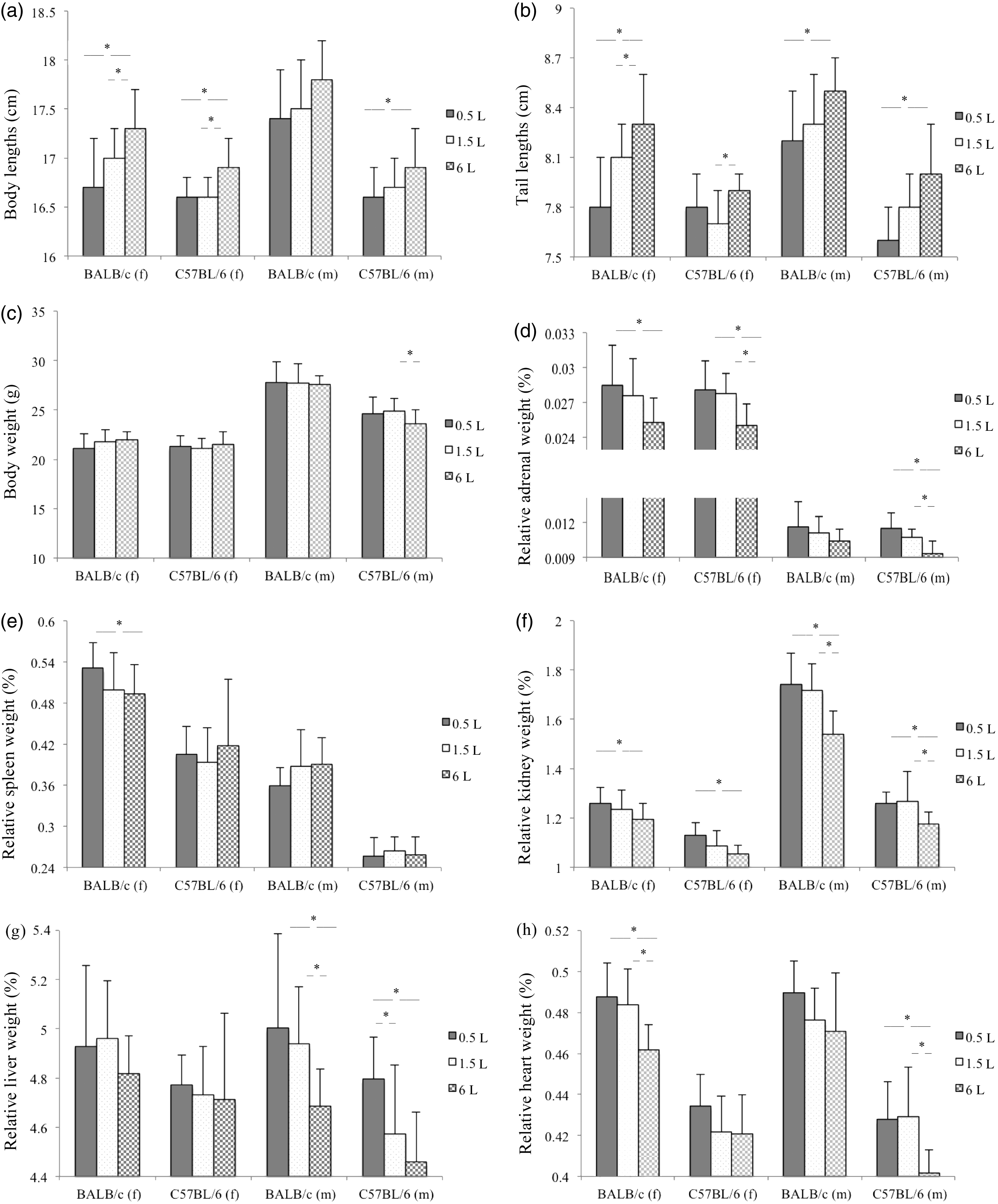
Mice housed for three weeks
Comparable to the results mentioned above, mice housed on shallow bedding had increased organ weights, however the differences between the bedding volumes were less pronounced. The bedding volume significantly influenced adrenal weights of BALB/c and C57BL/6 mice (P = 0.045; P = 0.026) as well as kidney (P < 0.001) and liver (P = 0.020) weights of BALB/c mice. BALB/c mice had larger relative organ weights compared with C57BL/6 mice (P < 0.05), however C57BL/6 mice exhibited larger body and tail lengths (P < 0.001).
Blood parameters
Blood parameters of female BALB/c and C57BL/6 mice (mean ± SD).
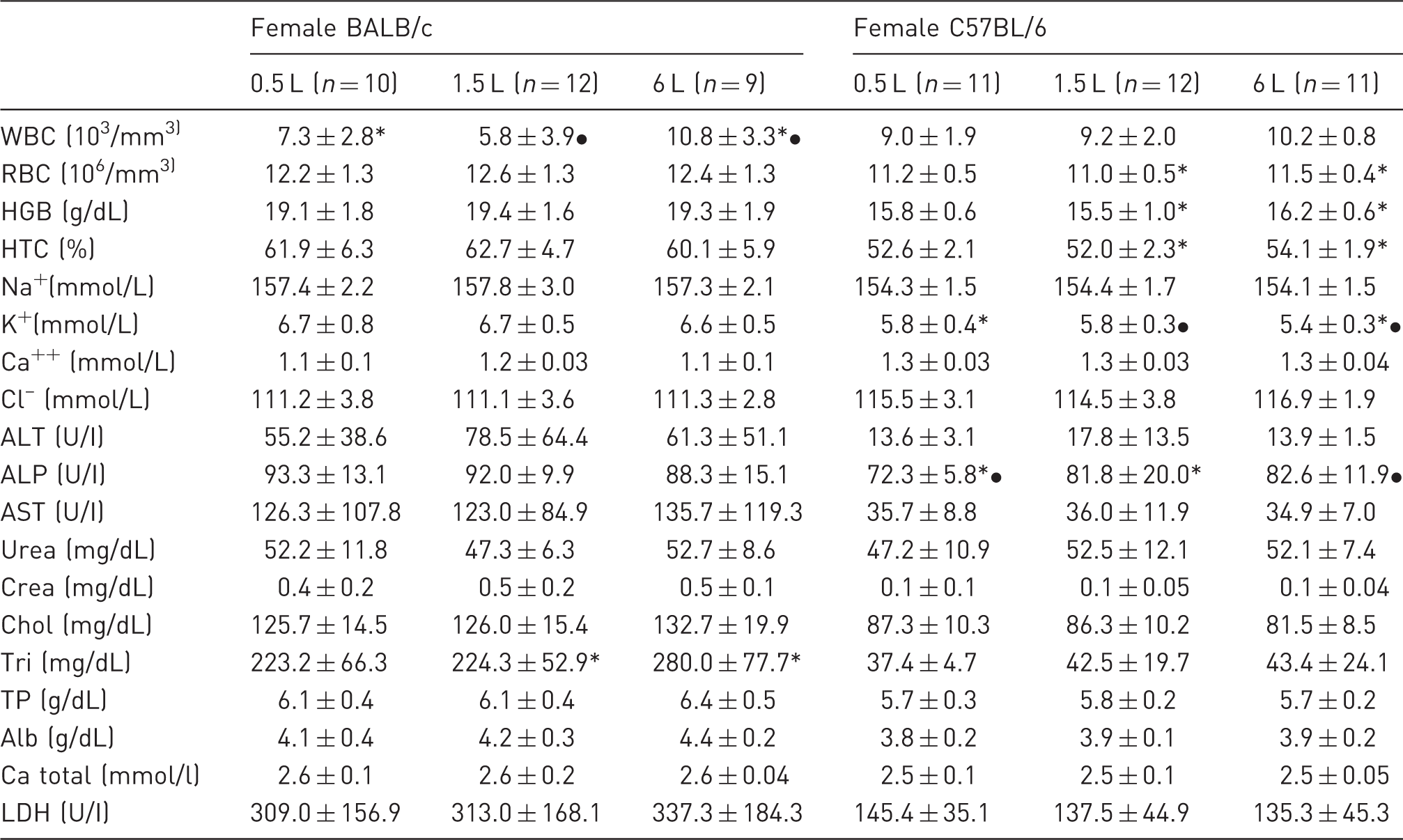
Corresponding symbols (* / *), (• / •) indicate P < 0.05 between the bedding volumes within the BALB/c or C57BL/6 groups. WBC: white blood cells, RBC: red blood cells, HGB: haemoglobin, HTC: haematocrit, Na+: sodium, K+: potassium, Ca++: calcium, Cl–: chloride, ALT: alanine aminotransferase, ALP: alkaline phosphatase, AST: aspartate aminotransferase, Crea: creatinine, Chol: cholesterol, Tri: triglycerides, TP: total protein, Alb: albumin, Ca total: total calcium, LDH: lactate dehydrogenase.
Blood parameters of male BALB/c and C57BL/6 mice (mean ± SD).
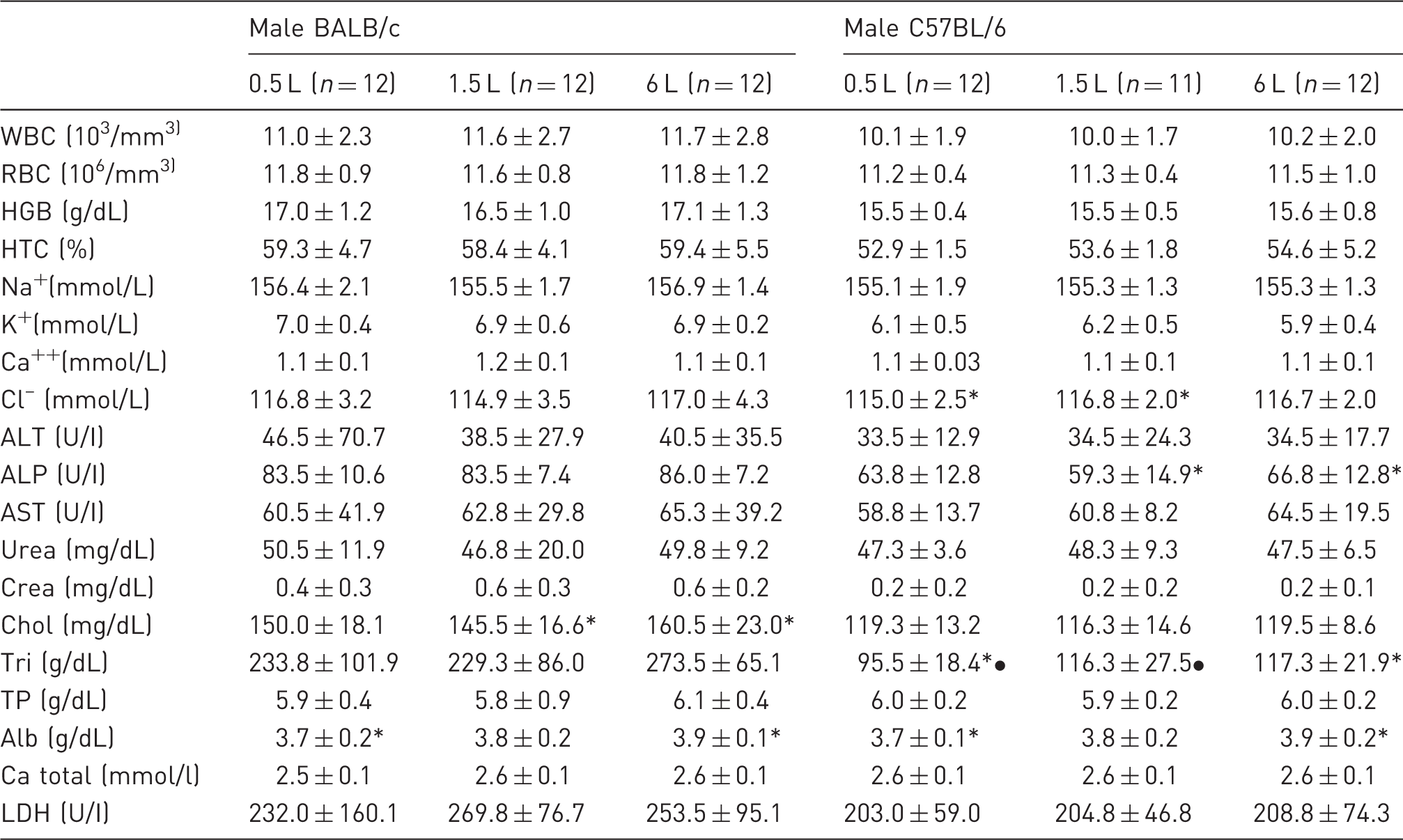
Corresponding symbols (* / *) indicate P < 0.05 between the bedding volumes within the BALB/c or C57BL/6 groups. WBC: white blood cells, RBC: red blood cells, HGB: haemoglobin, HTC: haematocrit, Na+: sodium, K+: potassium, Ca++: calcium, Cl–: chloride, ALT: alanine aminotransferase, ALP: alkaline phosphatase, AST: aspartate aminotransferase, Crea: creatinine, Chol: cholesterol, Tri: triglycerides, TP: total protein, Alb: albumin, Ca total: total calcium, LDH: lactate dehydrogenase.
Behavioural tests
Open-field test
Impact of the bedding volume on freezing and travel distance were unclear. Highest mean travel distance was noted in the 6 L bedding for female BALB/c mice, the 1.5 L bedding for male BALB/c mice, and the 0.5 L bedding for female and male C57BL/6 mice. A reduced travel distance corresponded with an increased freezing frequency (Figure 3). Female C57BL/6 mice showed a higher travel distance (P < 0.001) and less freezing (P = 0.002) compared with C57BL/6 males; no significant gender differences were found for BALB/c mice. In general C57BL/6 mice showed a significantly higher travel distance and less freezing compared with BALB/c mice (P < 0.001 for both parameters).
(a) Travel distance and (b) freezing frequency of female (f) and male (m) BALB/c and C57BL/6 mice (mean ± SD); *P < 0.05; females: BALB/c 0.5 L and 1.5 L (n = 12), 6 L (n = 11), C57BL/6 0.5 L and 6 L (n = 11), 1.5 L (n = 12); males (n = 12).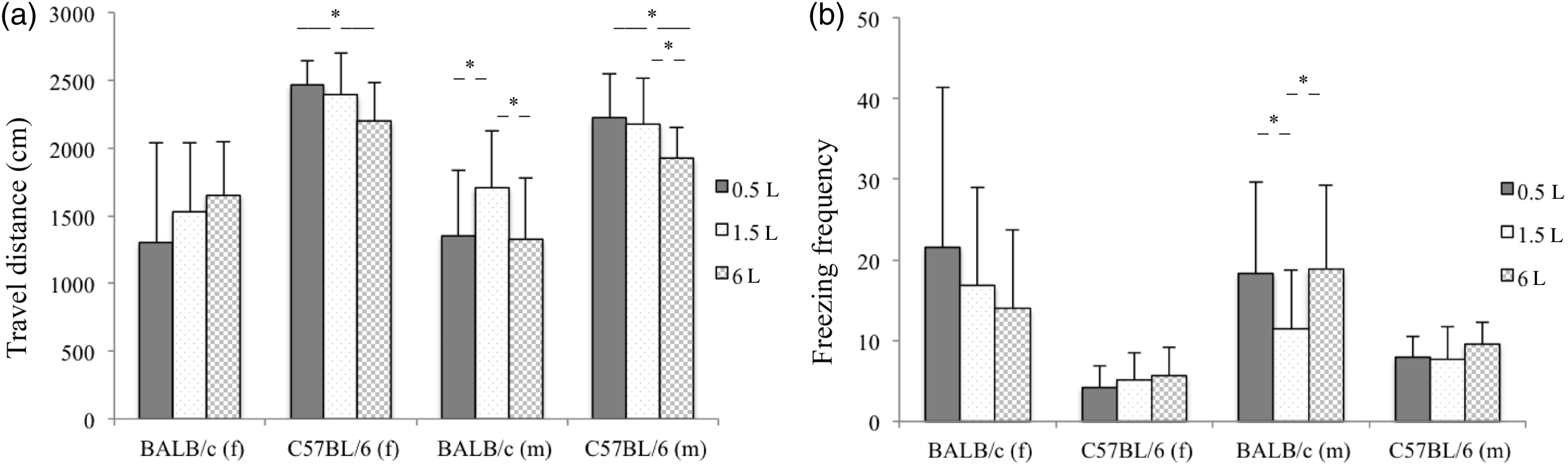
Novel object recognition
There were no significant differences between the bedding volumes regarding exploration time of familiar or novel objects. Female C57BL/6 and male BALB/c mice had a positive discrimination index for the 0.5 L (0.28; 0.14) and 1.5 L (0.04; 0.15) bedding, and a negative index for the 6 L bedding (–0.02; –0.1). Male C57BL/6 mice showed a reversed trend (–0.15 [0.5 L], –0.36 [1.5 L], 0.29 [6 L]). No clear gender or strain differences could be ascertained.
Pentobarbital metabolic rate
Mice housed for 12 weeks
The pentobarbital narcosis revealed significant sex and strain differences but no clear impact of the bedding volume was detected (Figure 4). BALB/c mice showed reduced pentobarbital sleeping times compared with C57BL/6 mice (1st move: P = 0.002 male BALB/c versus C57BL/6 mice; lateral position: P < 0.001 female BALB/c versus C57BL/6 mice). Female mice woke up earlier from pentobarbital narcosis compared with males. Significant differences were detected for lateral positions (P < 0.001 female versus male BALB/c mice).
1st move (1 m) and lateral position (lp) of BALB/c and C57BL/6 mice (mean); females: BALB/c 0.5 L and 1.5 L (n = 12), 6 L (n = 11), C57BL/6 0.5 L and 6 L (n = 11), 1.5 L (n = 12); males (n = 12).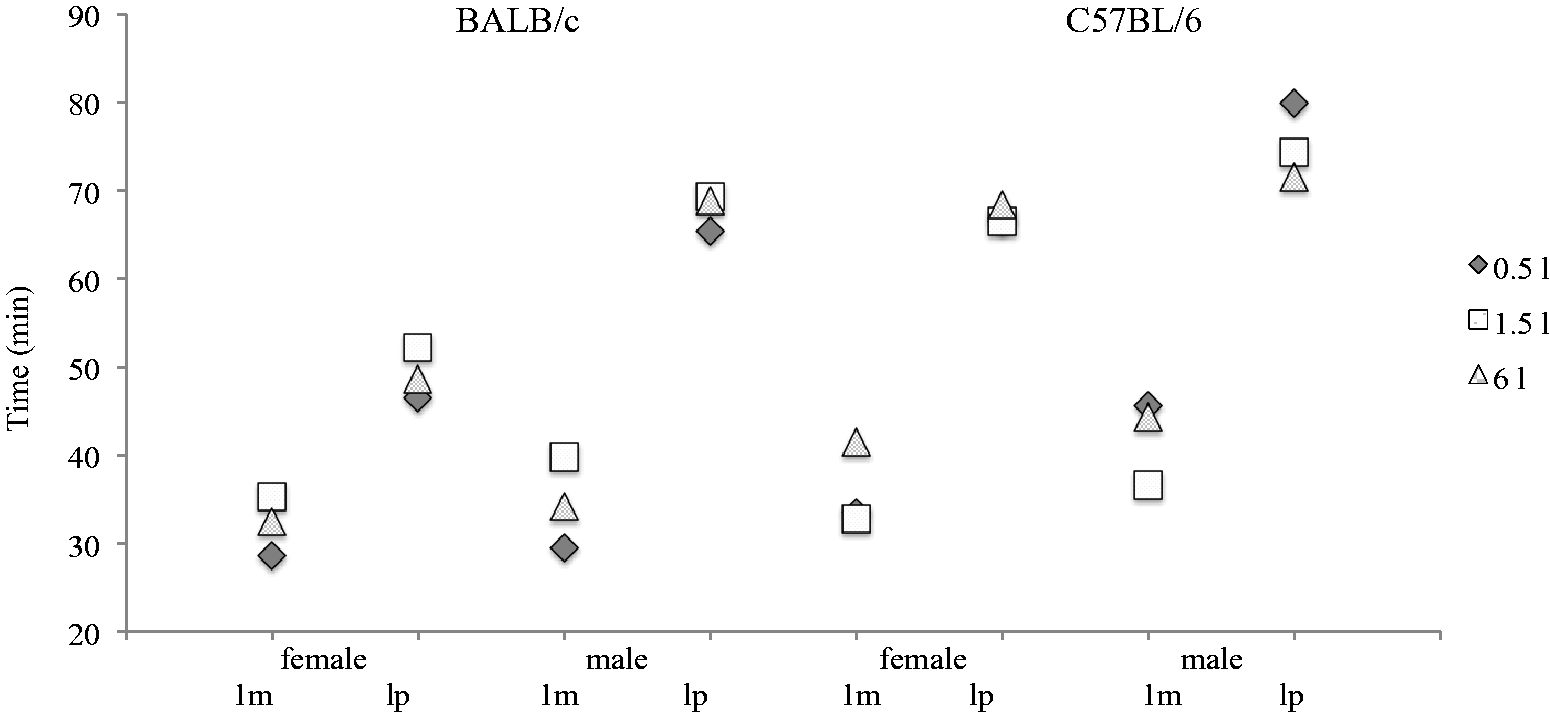
Mice housed for three weeks
Neither the day relative to cage change, nor the bedding volume had a significant effect on sleeping times in female BALB/c and C57BL/6 mice (data not shown). Regardless of cage change interval sleeping time of C57BL/6 mice was longer compared with BALB/c mice (1st move: P = 0.006 [day 1]; P = 0.009 [day 14]; lateral position: P < 0.001 [day 1/day14]).
Ammonia
On day 1, no ammonia was detected in any of the cages. On day 14, cages with larger bedding volumes received reduced scores in the nesting (BALB/c:C57BL/6 – 1:4 [0.5 L], 0:2.7 [1.5 L], 0:0 [6 L]) and the area of faeces and urine disposal (BALB/c:C57BL/6 – 2:6.7 [0.5 L], 1:5.3 [1.5 L], 0:0.3 [6 L]) compared with cages with smaller volumes. Significant differences between nesting areas and areas of faeces and urine disposal were noted for C57BL/6 mice between 0.5 L and 1.5 L (P = 0.034 for nesting areas only) or 6 L (P = 0.025; P = 0.043) as well between 1.5 L and 6 L (P = 0.034; P = 0.046). In general lower ammonia levels were detected in the nesting areas compared with the area of faeces and urine disposal for BALB/c and C57BL/6 mice (P = 0.046; P = 0.017). Overall higher ammonia levels were measured in cages with C57BL/6 mice.
Coefficient of variation
The impact of the bedding volume on CVs varied depending on the strain, sex and parameter studied. The Friedman test revealed an overall higher CV of anatomical parameters for mice housed on 0.5 L, followed by 1.5 L and 6 L. The pooled CVs of haematological data were higher on 0.5 L followed by 6 L and 1.5 L (0.5 L > 1.5 L > 6 L for clinical chemistry). Significant differences between the housing conditions were noted for pooled clinical chemistry variables (P = 0.018). The results of the open field followed a similar pattern with a higher CV on 0.5 L, followed by 1.5 L and 6 L. However, the pooled CVs of the pentobarbital narcosis were higher on 6 L and 1.5 L compared with 0.5 L.
Discussion
The bedding volume had a significant impact on anatomical parameters. Mice housed on larger volumes showed a reduction in organ weights as well as an increase in body and tail lengths. Mean values of pentobarbital sleeping times, blood parameters, performances in open-field and novel object recognition tests revealed sex and strain differences, but no clear impact of bedding volumes. The bedding depths had no significant impact on variation, while overall larger CVs were recorded on smaller volumes for open-field tests, blood and anatomical parameters. Pentobarbital sleeping times showed a reversed trend.
The impact of a larger bedding volume on freezing frequency and travel distance in the open-field test varied between female and male BALB/c and C57BL/6 mice. There are ambiguous results regarding the influence of cage enrichment on open-field activity. It appears that activity is increased or reduced depending on sex, strain, and cage design.16,23 In line with previous reports, 24 C57BL/6 mice were more active in the open-field test, but the current novel object recognition test did not confirm these strain differences. Nesting material enhanced learning in a water maze test in rodents, 12 but the present data suggest that bedding volume does not influence cognitive functions.
In line with previous results, pentobarbital metabolism was influenced by strain and sex, 25 however not by intra-cage ammonia levels 13 in the present study. Although the bedding volume and day relative to cage change did influence ammonia concentrations in the cage environment, 14 the sleeping time was not affected by any of these parameters. It can be assumed that the ammonia concentrations would have needed to be higher to result in a clear inhibiting effect on the hepatic microsomal system. 13
Mice housed on shallow bedding showed increased organ growths compared with animals housed on deeper bedding. Enlargement of especially heart, liver and kidney masses to compensate for the increased metabolic demands 26 is a well-documented phenomenon with cold ambient temperatures.10,11,27 Hypertrophy of these organs was already present in laboratory mice after a two-week exposure to 15℃ compared with animals housed at 23℃. 27 In line with these findings, changes in organ weights were already visible in animals housed on different bedding volumes for three weeks. Mice kept on deep bedding had smaller adrenal weights compared with groups on shallow bedding. Increased adrenal weights are associated with higher plasma corticosterone levels, 28 and are used as an indicator of endocrine stress. 29 It remains unclear whether potential thermal stress led to enlarged adrenals or whether the hypertrophy was associated with cold adaptation; this could have been answered by an assessment of the thymus weight involution. 30 Comparable to mice housed at colder ambient temperatures,10,31 mice kept on smaller bedding volumes showed reduced tail lengths, which can be considered as a reliable indicator for cold adaptation. 10 Appendages tend to be shorter from animals of the same species living in colder climates, as more compact body parts have less surface area and thus radiate less heat (Allen’s rule). All these results confirm the hypothesis that deep cage bedding helps mice to reduce heat loss. Regardless of the bedding volume provided, mice created pockets within the bedding where they huddled during resting. Especially in cages with deep bedding, these pockets most likely provided mice with an insulated environment that restricted heat loss and thereby reduced metabolic demands for thermoregulation. Bedding material was found especially on the floor beneath cages containing the 6 L bedding, indicating that mice must have performed quite vigorous digging behaviour.
Sex and strain are known to affect blood values, 32 apart from these differences no clear impact of the bedding volume on the blood parameters could be ascertained. Haematocrit and especially haemoglobin are known to increase in response to cold temperatures.11,33 However, these patterns became particularly obvious when mice were housed at extreme temperatures (such as 5℃), 33 which may explain why the trend was not visible in our study.
Pursuant to our data, the bedding volume did not have a significant influence on the CVs of physiological and behavioural parameters, however we observed a tendency of reduced variation on deeper bedding compared with shallow bedding. The impact of various cage designs on variation has been assessed, but with opposing results. While the mean absolute deviation was not affected by housing conditions, 34 an overall increased CV,15,16 respectively a higher number of animals needed to detect an arbitrarily chosen effect size,17,18 was reported for enriched cages. The influence of housing conditions on variation depended considerably on the parameters measured, as well as on the strain and cage enrichment chosen for the study. 15
The impact of bedding volume on mouse anatomy should not be underestimated, therefore we strongly recommend specifying the amount of bedding provided, especially when the outcome measure is likely to interact with thermoregulation.
Conclusions
Anatomical changes suggest that deep bedding helps mice to reduce heat loss under standard housing conditions without increasing variations in experimental results. A larger bedding volume is an easy and effective way to reduce cold stress for laboratory mice and can improve both animal well-being and the utility of mice as models in biomedical research.
Footnotes
Acknowledgments
The authors wish to thank Sabine Schmidt and Clemens Dasenbrock for their help with experimental planning and Dirk Menzel for his help regarding the analysis of the blood samples.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
