Abstract
Orogastric gavage, while a common method for delivering experimental substances in mice, has been shown to induce stress. To minimize the associated stress with this procedure, sham gavage prior to the start of experiment is a common method for habiutating mice. We investigated whether handling and restraint could replace sham treatment in the acclimatization protocol. Mice were either undisturbed, hand-restrained for 10 s or sham-gavaged daily for six days prior to eight days of twice daily gavage. The results showed that repetitive restraint and gavage had no differences in body weight after eight days of treatment compared with the body weights at the start of treatment, whereas animals left undisturbed lost significant weight once treatment began. These data suggest that procedure refinement by replacing sham treatment with hand restraint is sufficient to acclimatize mice to the stress associated with gavage.
Multiple studies have shown that enteral substance administration by oral gavage, even when performed by experienced technicians, is a stressful procedure in unhabituated rats.1–4 Restraint required for gavage is stressful in rodents,5,6 and habituation to handling reduces the stress associated with gavage in rats. 7 High fat diet-induced obese (DIO) mice are an important animal model for type II diabetes mellitus research.8,9 This model has been shown to be more sensitive to stress-related behaviours than genetic models of obesity. 10 This stress results in weight loss from decreased food intake and/or increased catabolism. 11 To prevent this stress-induced weight change animals are often habituated before the start of experiment by sham gavage without substance administration. This study was designed to address if restraint alone could prevent the weight loss seen in the absence of habituation.
Twenty-three female C57BL/6JHsdOla mice (Harlan Laboratories, Horst, The Netherlands) were given ad libitum tap water and a 60 kcal % fat diet (D12492; Research Diets, New Brunswick, NJ, USA) supplemented with a high-fat confectionery (Delicato Bakverk AB, Kungens Kurva, Sweden) beginning once animals reached 20 g body weight. Mice were group-housed (n = 5–6/cage) and acclimatized for at least two weeks to a reverse light cycle (light hours: 21:30 to 09:30) before being assigned to treatment groups. All experiments were conducted using protocols approved by the regional animal welfare committee (Göteborg Djurförsöksetiska Nämnd) in accordance with Swedish regulatory requirements. Mice were housed in non-sterile open cages (Makrolon III; Scanbur, Karlslunde, Denmark) containing hardwood bedding with nesting material (Papyrus, Mölndal, Sweden), gnawing sticks, and a shelter (Brogaarden, Lynge, Denmark). Animal holding room temperatures were maintained between 21℃ and 22℃ with relative humidity of 50 ± 5% and 20 air changes per hour. All animals were free from adventitious murine viruses, as well as Pasteurella and Helicobacter spp.
Animals (63.0 ± 0.75 g) were randomly placed in two cages per treatment group (undisturbed, 10 s restraint, sham gavage) 16 days before the start of treatment. The day before the start of experiment (day −7) cages were randomly assigned to treatment groups based on the weight of the cage containing the animals. Animals in the undisturbed group (n = 8) were handled only during weighing at the start of pre-treatment (day −6). Mice assigned to the restraint group (n = 8) were restrained by scruffing, with the front feet away from contact with the gavage needle. The mice were restrained by hand for 10 s once a day and weighed daily from days −6 to −1. Mice in the sham gavage group (n = 7) were restrained by hand once a day for insertion of a dry oral gavage needle into the stomach without substance administration and weighed daily. Pre-treatment (day −6 to day −1) was carried out at 08:30 to 09:00 h every day. All groups received twice daily (08:30 and 14:30 h) gavage and daily weighing (08:30 h) from days 0 to 8. The gavage procedure was performed by restraining the mouse with a non-dominant hand and insertion of a blunt-tipped gavage needle (AgnThos AB, Lidingö, Sweden) into the oral cavity with the tip positioned within the stomach. Room temperature sterile water (5 mL/kg) was administered over a 2 s period, followed by needle removal and return to the home cage. All animals were fasted for 4 h in the morning on day 8 prior to conscious blood collection from the vena saphena to measure basal glucose and insulin levels. Glucose levels were measured using the AccuCheck glucometer (Roche Diagnostics, Indianapolis, IN, USA). Insulin measurements were analyzed using a radioimmunoassay kit (Millipore, Billerica, MA, USA). Homeostatic model assessment (HOMA), a model for assessing β-cell function and insulin resistance, was calculated from the glucose (mM) and insulin (nM) levels.
Body weight change (days 0 and 8), glucose, insulin and HOMA levels (day 8) were analyzed by one-way analysis of variance (ANOVA) followed by Tukey multiple comparison test (Minitab 15; Minitab Ltd, Coventry, UK), with P < 0.05 considered to be significant. The results (Figure 1) showed that the 2.4% and 1.4% weight loss seen during habituation (days −6 to 0) in the restraint and sham gavage groups, respectively, was significantly more than the undisturbed animals (P < 0.01). The weight loss was most significant in the first four days, with the rate of loss plateauing from days −2 to 0. Animals in the undisturbed group showed a slight weight gain during this period. The 4.4% weight loss in the undisturbed group during water gavage treatment (days 0 to 8) was statistically different from the other groups (P < 0.05). The HOMA analysis on day 8 showed no statistically significant differences between treatment groups (P = 0.41). Similarly, the basal insulin and glucose levels were not significantly different between treatment groups (P = 0.43 and P = 0.49, respectively).
Percentage body weight change from day 0. The daily body weight average for each treatment group (undisturbed, n = 8; restraint, n = 8; sham gavage, n = 7) is shown with the standard error of the mean SEM.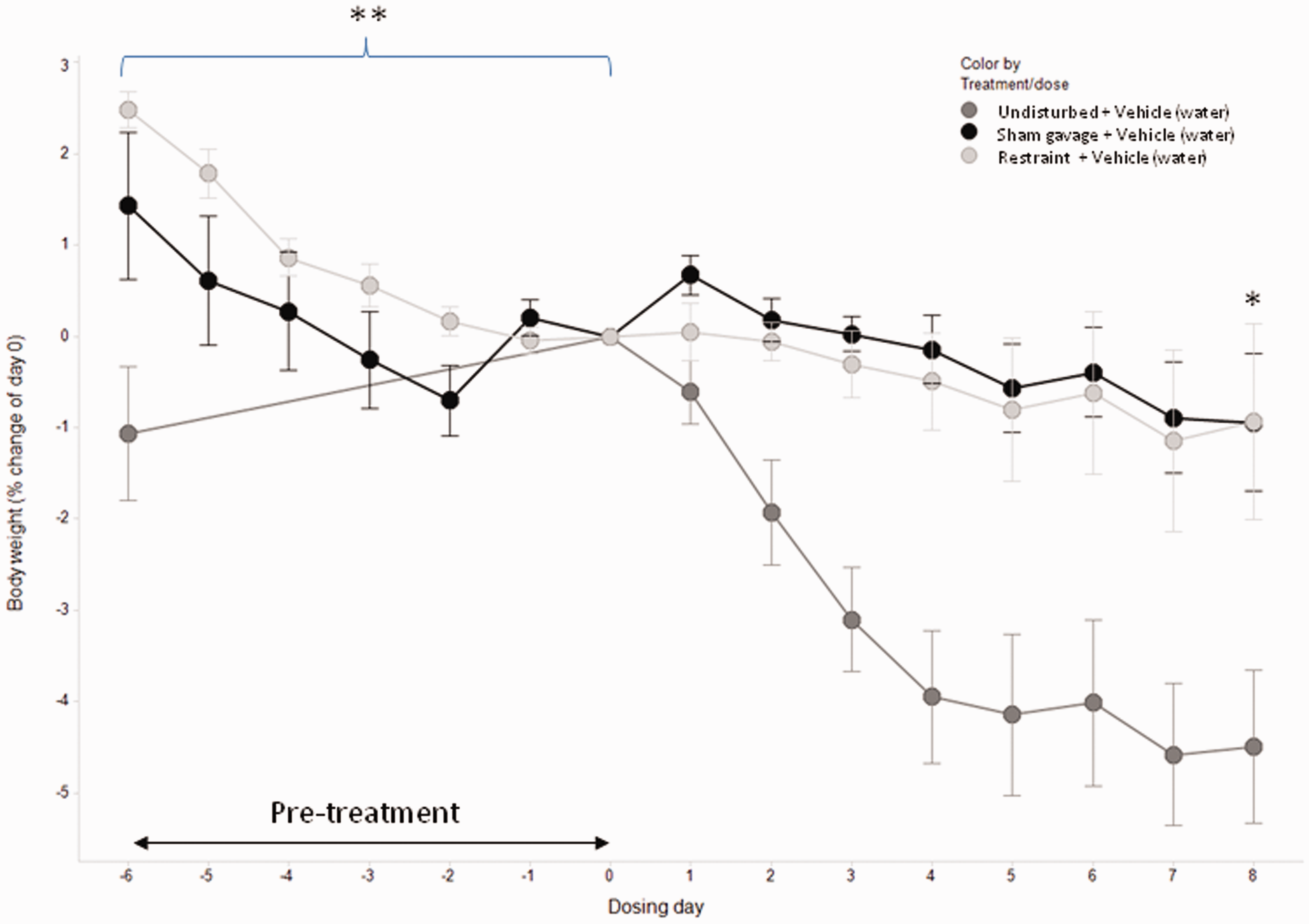
Animal stress associated with experimental manipulation can impact the scientific outcome by lessening the difference between drug- and control-treated animals, and requiring more animals to provide greater statistical power.2–7 Refinements that lessen procedural stress provide a stable baseline and may ultimately reduce the number of animals required. 5 In DIO mice, body weight change has been shown to be a sensitive non-invasive indicator of stress. 10 By habituating animals to substance administration, stress-associated weight change can be eliminated. Animals that received the sham administration did not show further weight loss when the dosing with water commenced suggesting that the handling and insertion of the gavage needle alone was the stressful event rather than the liquid administration to the stomach.
To further understand the stress associated with gavage, a comparison between restraint and sham gavage was conducted. This study suggested that the more profound stress was due to restraint rather than to the insertion of the gavage needle since the weight loss associated with restraint alone was not statistically significant from restraint and gavage. Correspondingly, the HOMA values were not statistically different among the groups, suggesting that all animals habituated to the procedures after multiple days of handling either before or after the start of the study. By reducing the period of repeated gavage, the potential for traumatic injury should be lessened and a significant refinement in animal well-being should be developed.12,13 The methodology used in this study provides a basis by which to assess refinements to habituation by substituting hand restraint for more invasive procedures.
Footnotes
Acknowledgements
We wish to thank Charlotte Lindgren for her support with bioanalytical analysis and the Laboratory Animal Sciences staff for assisting with these studies.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: this authors are employees of AstraZeneca.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: this work was funded by AstraZeneca.
