Abstract
Intratracheal injection is a traditional technique used in small animal studies of highly contagious airborne pathogens such as Mycobacterium tuberculosis. However, current techniques of intratracheal injection generally involve procedures that pose a risk of incident injury and infection for researchers, and may also cause collateral damage to experimental animals during the installation process. Here we describe an intratracheal injection method that was enabled by a three dimensional printing of a custom platform. This updated technique improved the overall ergonomics of intratracheal injection in mice, minimizing the risk of human injury and implementing the 3R (replacement, reduction and refinement) principle in mouse infection studies.
Intratracheal injection is one of the primary experimental methods used to deliver precise quantities of agents, such as infectious organisms or drugs, into the lungs of experimental animals. Classical methods require the animals to be under anesthesia for extended periods due to the time needed to properly administer a given agent into the mouse airway. Since the procedure involves the airway itself, this precludes the use of inhalation anesthetics and requires injectables, such as ketamine, delivered via an intraperitoneal (IP) injection, which potentially increases the animal’s pain, suffering and distress. 1 Additionally, due to the presence of sharp materials such as needles, intratracheal injection poses a risk of infecting researchers, especially when contagious pathogens, such as Mycobacterium tuberculosis, are used. Our new technique achieves the 3R (replacement, reduction and refinement) principle and minimizes the risk of incident infection for researchers.
Three-dimensional (3D) printing technology, on the basis of a process known as additive manufacturing (AM), is becoming more widely accepted in creating custom-designed and sometimes difficult to manufacture products. Using computer-aided drafting (CAD) software, a user may create, modify or download a digital 3D model of an object, and initiate the process of creating it with the required specifications. Further, the widespread availability of 3D printing technology ensures that when new models are shared, the broader scientific community may replicate the object. Here we detail the creation and use of a mouse platform for intratracheal instillation, and share this file such that it may be used by other preclinical researchers. The device was designed using Autodesk Inventor Professional 2013 (Autodesk Inc, San Rafael, CA, USA). The design has the following dimensions, 80 mm × 235 mm × 80 mm, with a 25° angled surface on which the mouse would lie within a depression size of 30 mm × 100 mm × 5 mm. The device is compatible with cross bars that have a diameter of 4 mm. Once the design was completed, it was exported as a ‘.stl’ file. This file was then opened in MakerBot Desktop v2.7.0.108 (MakerBot, Brooklyn, NY, USA), and configured for the Replicator 2 by going to Devices > Selected Type of Device and selecting Replicator 2. A ‘.x3g’ file, or print file, was then created using 100% infill, a raft, and support. The remaining settings were configured by selecting MakerBot PLA as the desired filament. The ‘.x3g’ file was then loaded onto a MakerBot Replicator 2 and the file was printed using white PLA (Figure 1a). The original ‘.stl’ file is available for free download at http://www.thingiverse.com/thing:916853. The final prepared platform included a cross bar, a mouse incisor hanger and a metal bar which could be used to adjust the height of the cross bar by inserting it into one of three holes based on experimental needs (Figures 1b–d).
Assembly of the intratracheal injection system. (a) 3D print of the main part of the platform. (b) Two metal bars, one to be used for holding the mouse through the attachment of the incisor hanger and a second to adjust the bar height. (c) Assembled platform. (d) A lateral view of the platform. (e) An entire assembled intratracheal injection system. (f) Front view of injection platform. (g) Eppendorf Research® pipette and BioDot Standard Gel LoadingTip.
We assembled the entire injection system as shown in Figures 1e and f. Mice were anesthetized using inhalation isoflurane (NDC 66794-013-25; Piramal Healthcare, Mumbai, India) in a heavy glass desiccator (Cat. 08-595A; Fisher Scientific, Pittsburg, PA, USA). They were then monitored to ensure sufficient depth of anesthesia. Each mouse was gently suspended by its superior incisors on the ‘mouse incisor hanger’ and its body placed into the slot on the bevel of the platform as shown in Figure 2a. The Schott Fostec Ace 20500 adjustable illuminator with double goosenecks (Schott North America, Inc, Elmsford, NY, USA) was positioned near the pharyngoepiglottic region of the mouse neck (Figure 2a). The tongue of the mouse was gently pulled out to the left side using small tweezers and then pushed down with a micro spatula. The trachea was visualized by percutaneous transillumination using the light emitted from the end of a gooseneck fiber-optic rod (Figure 2b). Thirty microliters of samples were gently administered through the trachea using an Eppendorf Research® pipette with BioDot Standard Gel Loading Tip (1–200 uL) (Cat. 010; Dot Scientific Inc, Burton, MI, USA) (Figure 1g). After injection, the mice were wrapped with a clean paper towel, as bedding materials could stick to their eyes or be inhaled while they are recovering from the anesthesia. The mice were monitored until they were fully recovered from the anesthesia. This procedure generally took 2–3 min. The mice were then returned to the animal housing rooms when they were alert and fully ambulatory.
Demonstration of an intratracheal injection in a mouse. A biological safety cabinet is required when working with infectious organisms. (a) Restraint of mouse on the platform. (b) Front view of the of mouse trachea with the Schott Fostec Ace 20500 adjustable illuminator. (c and d) Injection of agents into the mouse trachea using an Eppendorf Research® pipette and BioDot Standard Gel LoadingTip. (e) Quantitation of Mycobacterium bovis BCG in the mouse lung one day after intratracheal injection (n = 3 mice). Error bar indicates standard deviation.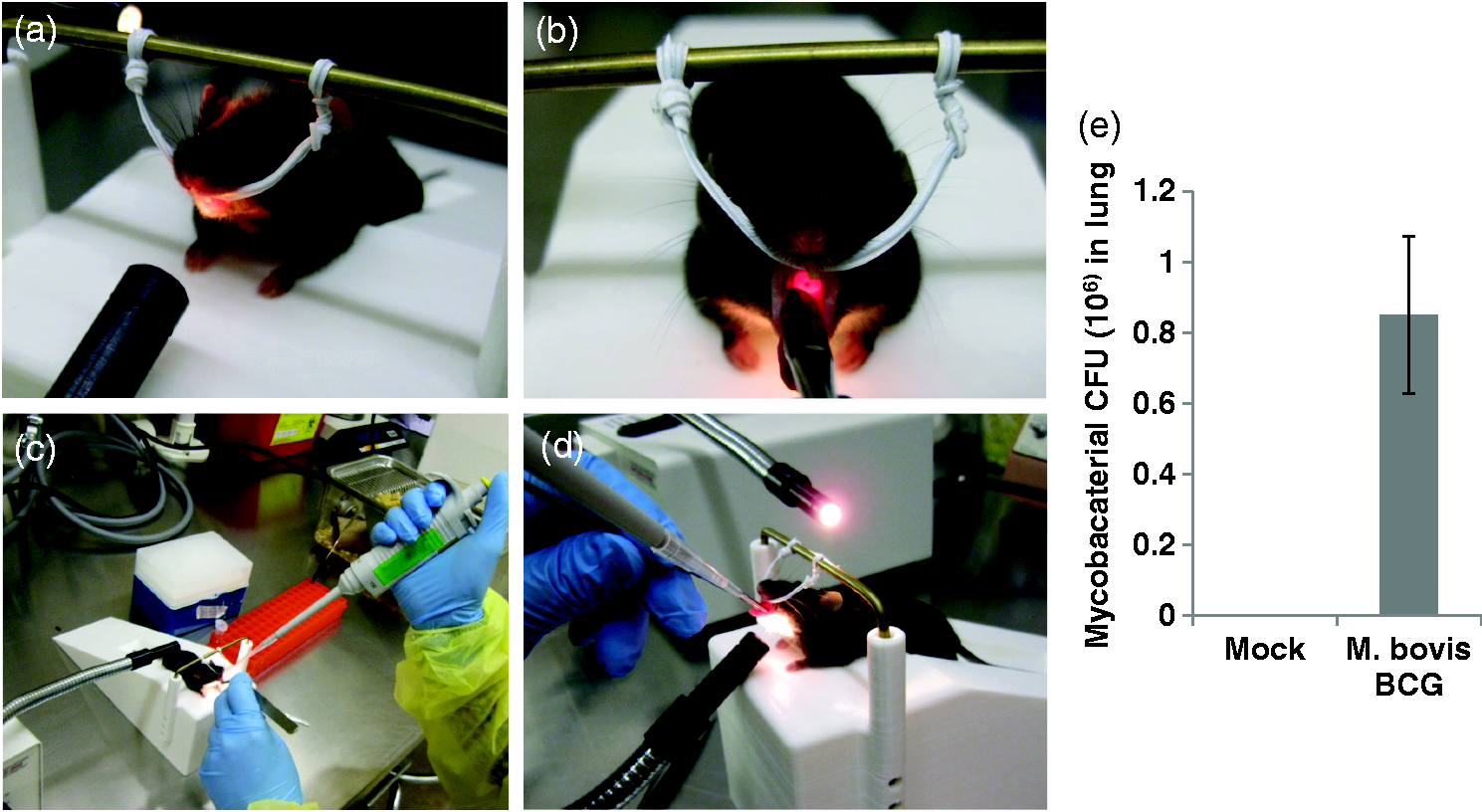
This new method of intratracheal injection was verified in wild-type C57BL/6 mice challenged with Mycobacterium bovis BCG. All wild-type C57BL/6 mice (female, 8 to 10 weeks old) were housed at the institutional animal facility under specific pathogen-free conditions during the experiments. The University of Notre Dame is accredited through the Animal Welfare Assurance (#A3093-01). All animal procedures were approved by the institutional animal care and use committee. The bacterial suspension was prepared as described previously. 2 A frozen M. bovis BCG stock vial was taken and thawed at 37℃. The mycobacterial cells were spun at 8000 rpm for 5 min at room temperature and the pellet washed with 1 × phosphate buffered saline (PBS) (SH30013.02; Thermo Scientific, Waltham, MA, USA). The cell pellet was resuspended in 660 µL of PBS, generating a final concentration of 106 CFU/30 µL. The mycobacteria were de-clumped using a sonicator (Model CL-18; Fisher Scientific, Pittsburg, PA, USA) for 5–15 s, and then vortexed briefly. The mice were infected by intratracheal injection as described above in a Class II biosafety cabinet. The mice were killed by cervical dislocation following isoflurane inhalation one day after infection, and placed on their backs on clean, dry, absorbent paper towels covering a dissection board. The area was sterilized with 70% ethanol to reduce the possibility of contamination while making incisions. To remove the lung, incisions were made through the thoracic cavity with surgical scissors. Using small toothed forceps, the bronchial tree close to the heart was grasped and the lung lobes cut away. The lungs were placed in a 10 mL Glas-Col homogenizer (Glas-Col, Terre Haute, IN, USA) containing 5.0 mL sterile PBS, and homogenized gently. Serial dilutions were made for each homogenate in PBS and plated onto 7H10 agar (Cat. 262710; BD, Franklin Lakes, NJ, USA) Petri dishes (Cat. 25384-302; VWR, Radnor, PA, USA). The plates were incubated at 37℃ in the dark for 3–4 weeks. The results showed that the mean value of the mycobacterial burden was 0.85 × 106 in the lungs of M. bovis BCG-infected mice, indicating an approximately 85% infection efficacy. As expected, no mycobacteria was detected in the lungs of mice administered with PBS (Figure 2e).
Mycobacterium tuberculosis, the etiologic agent of tuberculosis, causes approximately 1.3 million deaths annually. 3 In laboratory research involving live organisms, a Biosafety Level 3 environment is mandatory and all researchers are required to wear personal protective equipment (PPE). In this situation, any sharp instruments and complicated procedures are risk factors in incident infection for researchers. With this new method, we developed a technique free of sharp instruments for the delivery of infectious organisms into the lungs of mice by intratracheal injection. Moreover, our technique would decrease mouse pain, suffering and distress when compared with standard methods of intratracheal injection in M. tuberculosis studies, with the added bonus of implementing the 3Rs along the way.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Funds for this work were provided by grant AI052439 from the National Institute of Allergy and Infectious Diseases.
