Abstract
The partial hepatectomy (PH) model is widely used to study liver regeneration. Currently, the extent of regeneration is analyzed by measuring the weight of the liver post-mortem or by magnetic resonance imaging. In this study we aimed to determine whether liver volume gain can be accurately measured using micro-computed tomography (microCT). Approximately 42% of the liver was removed by ligation in C57BL/6 N mice. Mice were divided into two study groups. In group 1 conventional characterization of liver hyperplasia was performed by weighing the liver post-mortem. In group 2, liver volume gain was determined by microCT volume estimation. MicroCT results showed equivalent regeneration rates compared with the conventional method without the need to mathematically determine initial liver weights before PH. This parameter is strongly influenced by the age, strain and sex of the mice. In addition non-invasive microCT determination of volume gain over multiple time-points using the same animal reduces the number of animals needing to be used (in line with the 3R principle of replacement, reduction and refinement).
The liver is an organ with remarkable regenerative capabilities. Partial hepatectomy (PH) is one of the classical models used in the study of liver regeneration. The surgical methodology is well described and widely standardized. 1 One of the crucial issues with this method is the estimation of liver weight before surgery, expressed as the percent of the liver-to-body weight ratio. This ratio, however, is specific for each mouse strain (e.g. 4.53% for C57BL/6 J, 4.08% for BALB/cJ and 3.37% for 129S1/SvlmJ) and varies with age. The classical method used in liver regeneration calculations is to determine the weight–percentage of the liver after PH compared with the estimated pre-PH weight. 2
Three-dimensional micro-computed tomography (microCT) opens up opportunities for longitudinal studies of liver structure, lesions and regeneration by facilitating volume estimation of the liver after PH. In this study we tested the applicability of microCT in order to reduce the number of animals used in a PH study. We performed the conventional procedure 3 in parallel with longitudinal microCT imaging. Our findings revealed that volume estimation strongly correlated with post-mortem weight. Therefore, this procedure allows for a reduction in the number of animals required per experiment which is in accordance with the 3R (replacement, reduction and refinement) principle.
Materials and methods
PH was carried out as previously described 3 in 11–12-week-old C57BL/6 N female mice, with a body weight of 18–26 g (Charles River Laboratories, Sulzfeld, Germany). The mice were acclimatized for 10 days before surgery, and were housed under standard conditions. The liver was removed by ligature of the upper left posterior hepatic lobules and the left hepatic anterior lobes, and weighed. A long-acting analgesic, carprofen (Rimadyl® 5 mg/kg ad us vet; Zoetis Schweiz GmbH, Zurich, Switzerland), was subcutaneously injected immediately after the operation and while the animal was still under anesthesia, and subsequently every 8–12 h for a period of 48 h. Group 1 animals (n = 24) were humanely killed by cervical dislocation on days 1 (n = 6), 2 (n = 6), 3 (n = 6) and 7 (n = 6). Group 2 (n = 3) animals were imaged by microCT with their liver volumes determined before surgery and at the same four time-points as in group 1, and were humanely killed by cervical dislocation on day 7. In both groups, the entire liver was then removed, weighed and stained for Ki67 to determine the influence of radiation on the proliferation of hepatocytes in group 2. All experiments were carried out in accordance with the guidelines for animal care at the University of Kiel, Germany, and were approved by the local animal ethics committee (MELUR).
In a preliminary experiment, the mouse liver weight was determined to be 4.9 ± 0.7% of the body weight (n = 6). Pre-surgical liver weights were calculated using this value. Regeneration of the liver in group 1 was calculated using the extracted liver at each time-point (n = 6). To reduce the influence of body growth, the relative liver regeneration was calculated as follows: (calculated initial liver weight relative to body weight)/(measured liver weight relative to current body weight). Using microCT in group 2, liver volumes were determined before surgery and at four further time-points in the same animals (n = 3). A contrast agent, Viscover™ ExiTron™ nano6000 (Miltenyi Biotec, Bergisch-Gladbach, Germany) consisting of 110 nm particles which accumulate in the liver and spleen was used, and provide X-ray contrast for up to several weeks after application. A dose equivalent to 640 mg iodine per kg of body weight (100 µL intravenously) was applied one day prior to surgery. The mice were anesthetized with 100 mg/kg of ketamine and 16 mg/kg of xylazine intraperitoneally and scanned by microCT prior to surgery, and 24, 48, 72, and 168 h after surgery (vivaCT 40; Scanco Medical, Brüttisellen, Switzerland) (Figure 1a). An area beginning at mid thorax and extending to the upper abdomen including the entire liver was selected and scanned at a voxel size of 38 µm (70 kVp, 114 µA, 200 ms integration time, 500 projections per 180°). Radiation dose was 116.1 mGy per scan.
(a) Illustrative 3D liver volume surface reconstructions. (b) Liver mass restoration in mice over seven days after partial hepatectomy (PH). For all time-points, comparisons made by the two estimation methods show no significant differences. Each bar represents the mean ± SEM. (○): conventional method with six mice per time-point. (•): longitudinal micro-computed tomography (microCT)-based volume estimation using three mice and repeated measurements. Mean values of the two groups correlate linearly with R2 = 0.95 and P = 0.024 (Prism 5.03; GraphPad Software, Inc, San Diego, CA, USA).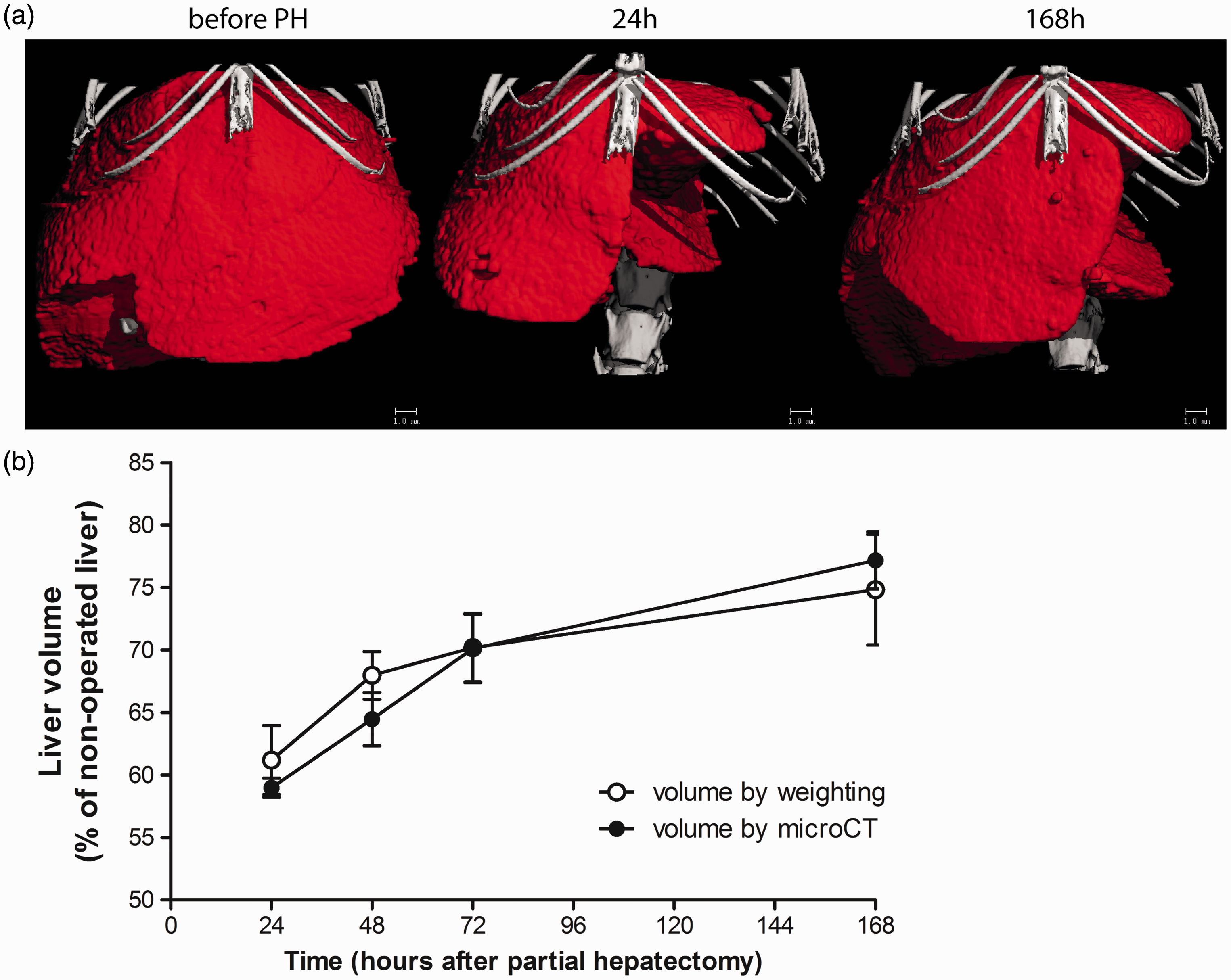
The automatic segmentation script was created using Image Processing Language version 5.15 (Scanco Medical). In each image, a binary ‘mask’ was generated, which defined the outer border of the liver tissue. This was accomplished by first applying a Gaussian filter to the greyscale images (σ = 2.6, supp = 3) followed by a threshold (8.4–11.0% of maximal greyscale value) such that each voxel in the image was labeled as either liver tissue or background. A dilation and subsequent erosion of five voxels were then applied to the images to ensure that all parts of the liver tissue were connected. Finally, a component-labeling filter was applied to remove any tissue not belonging to the liver. The binary mask was then converted into a contour that could be visualized in each 2D slice for manual adjustment.
Results and discussion
Excision of liver volume per mouse was 42.3 ± 3.5% of the total volume. A significant correlation between conventional and microCT mean values at each time-point was obtained (R2 = 0.95, P = 0.024, Figure 1b). Both groups showed similar numbers of proliferating cells (Figure 2) based on expression of the proliferation marker Ki67.4 Previous studies using magnetic resonance imaging (MRI) liver volume estimation have been performed in non-invasive longitudinal experiments.5,6 However, this technique is complicated and challenging compared with microCT measurements.
Evaluation of Ki67+ proliferating liver cells. (a) Quantitation of Ki67-positive cells (mean ± SD). Representative immunohistochemistry for Ki67 staining at 168 h (b) without and (c) micro-computed tomography (microCT) scans.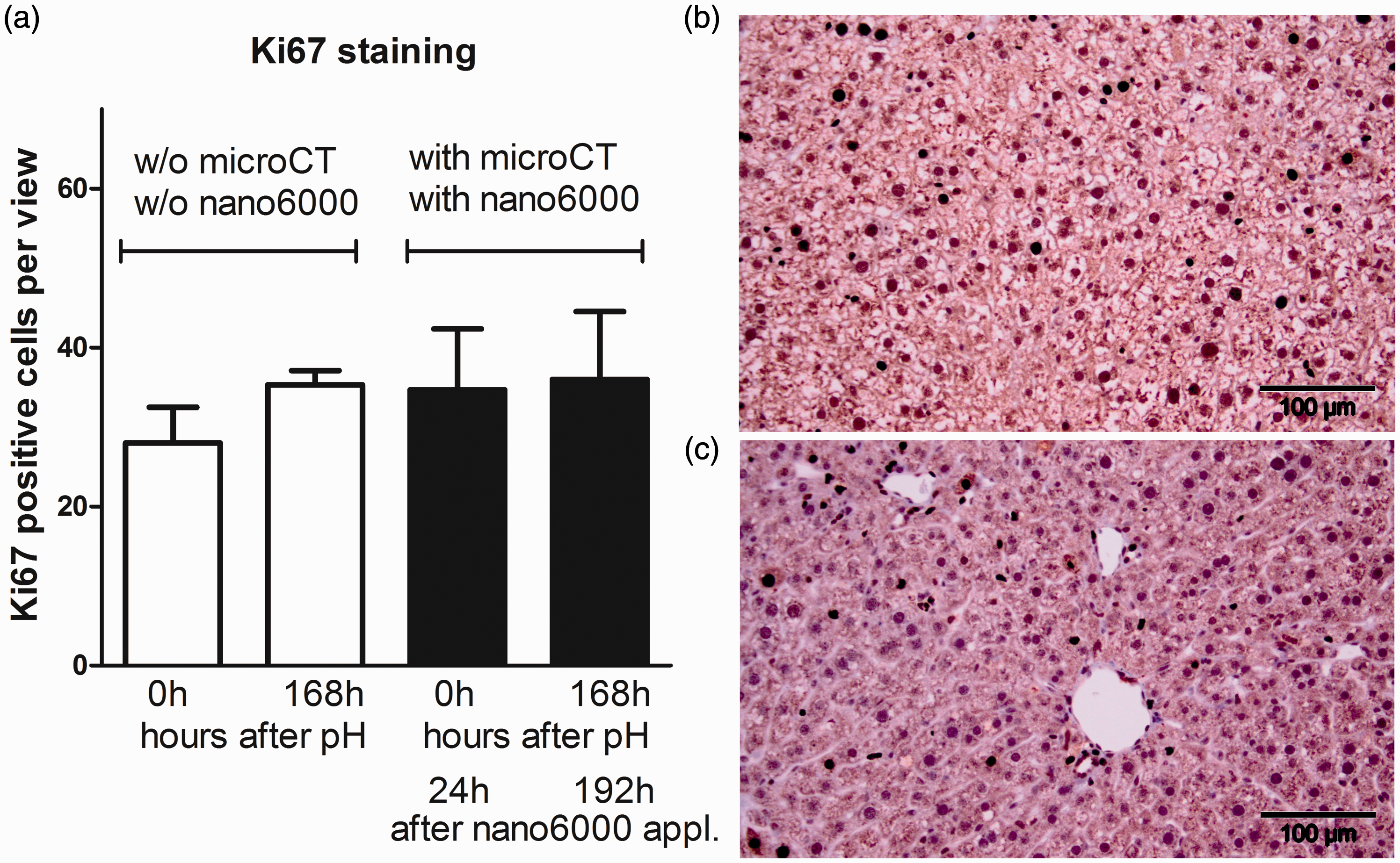
Although a high radiation dose (3.4 Gy) causing macroscopic damage to the liver has been reported, no detectable macroscopic damage was observed using a smaller radiation dose (0.5 Gy). However, the liver and jejunum showed some changes in γH2AX leukocyte staining.7 A radiation dose of 0.2 Gy showed neither DNA damage of leukocytes nor macroscopic damage to the liver and intestine. In our study the radiation dosage was around 0.12 Gy per single measurement and the proliferation of hepatocytes was not affected between the two groups (Figure 2).
In summary we demonstrate that longitudinal microCT estimation of liver volume after PH does not affect liver cell proliferation, and correlates with post-mortem liver weight. This procedure requires fewer animals than the classical approaches.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
