Abstract
The surgical procedure of two-thirds partial hepatectomy (PH) in rodents was first described more than 80 years ago by Higgins and Anderson. Nevertheless, this technique is still a state-of-the-art method for the community of liver researchers as it allows the in-depth analysis of signalling pathways involved in liver regeneration and hepatocarcinogenesis. The importance of PH as a key method in experimental hepatology has even increased in the last decade due to the increasing availability of genetically-modified mouse strains. Here, we propose a standard operating procedure (SOP) for the implementation of PH in mice, which is based on our experience of more than 10 years. In particular, the SOP offers all relevant background information on the PH model and provides comprehensive guidelines for planning and performing PH experiments. We provide established recommendations regarding optimal age and gender of animals, use of appropriate anaesthesia and biometric calculation of the experiments. We finally present an easy-to-follow step-by-step description of the complete surgical procedure including required materials, critical steps and postoperative management. This SOP especially takes into account the latest changes in animal welfare rules in the European Union but is still in agreement with current international regulations. In summary, this article provides comprehensive information for the legal application, design and implementation of PH experiments.
Keywords
Historic background of the model
The liver has the unique ability to regenerate itself after injury and cell loss. This strong regenerative property of the liver has been known since ancient times and is reflected in the myth of Prometheus. The amazing regenerative capacity of the liver is most clearly demonstrated by the experimental two-thirds partial hepatectomy (PH) model in rats, which was pioneered by Higgins and Anderson in 1931. 1 In this model, 70% of the liver is surgically removed in rats or mice. The residual liver lobes restore the original liver mass within approximately one week after surgery, though the resected lobes never grow back. 2 This model has been quoted as a technique in practically all papers reporting on PH. Unfortunately, it very often fails to take into account the distinct anatomical differences among species, for example the absent of a gallbladder in the rat. Additionally, alternative techniques for hepatic resection have been described, such as ‘one knot’ (ligature en-bloc),1,3,4 ‘two knot’, 5 or ‘three knot’ methods, and the vessel-oriented anatomical microsurgical technique, etc.6,7
Induced pathways after PH and translational aspects
Adult hepatocytes are normally quiescent, highly differentiated cells with an extremely low proliferative index. However, following PH in mice (and also in rats and humans), the remaining hepatocytes leave quiescence in a highly synchronized way and switch to a pre-replicative state (termed G1) within approximately 24 h, followed by DNA replication (termed the S-phase) and subsequent mitosis and cell division (the M-phase). Therefore, PH is a powerful model for studying cell cycle regulation in vivo.
The events of liver regeneration can be roughly divided into three phases, referred to as ‘priming’ (the early period of 0–6 h after PH, where hepatocytes prepare for cell cycle re-entry), ‘progression’ (the actual phase of DNA replication and hepatocyte proliferation), and ‘termination’ (the process that stops hepatocyte proliferation once the liver mass is restored).8,9 The priming phase involves the activation of several proinflammatory cytokines (e.g. tumour necrosis factor [TNF], TGF-ß, interleukin [IL]-6) and the so-called immediate-early response factors such as nuclear factor (NF)-κB, c-Jun, STAT3 and c-Myc, as described elsewhere, 10 while the progression phase activates the complete cell cycle machinery of hepatocytes. Thus, PH in mice and the subsequent investigation of the remaining liver tissue allows the comprehensive analysis of all these mechanisms. It should be noted that the key step after PH – the switch from a resting hepatocyte into a proliferating one – also reflects the starting point of cancer development in the liver. Therefore, the study of liver regeneration following PH also contributes to a better understanding of hepatocarcinogenesis. Besides this, understanding, manipulating and improving liver regeneration after PH in rodents also has a significant practical use for the treatment of human disease, as it could improve the recovery of patients after partial liver resection and definitely has future potential for efficient and safe partial liver transplantation.
Experimental procedure
General considerations
In general, the procedure is not difficult, requires only basic surgical skills and good hand–eye coordination, and takes only a little practice to master. Indeed, the outcome of the experiment and reproducibility of results can greatly vary if the operation is not performed properly. Therefore, drawing on the seminal publications3,5,11–15 and summarizing our experience of more than 10 years, we standardize and describe here a detailed surgical protocol for two-thirds PH with three separate ligatures. In our opinion, this method is fast, relatively easy to perform, not associated with mortality and gives highly reproducible results which we have successfully described in several publications.16–22 In addition to the surgical technique, the impact of several factors also has to be taken into account before surgery, as specified below.
Gender: It should be noted that distinct gender differences have been reported in experimental studies with wild-type animals. Such as, female mice show a significantly delayed or decreased rate in the processes of hepatocyte replication and liver mass reconstruction.
23
Additionally, there is a possibility that certain genetic modifications could induce sex-dependent effects and produce different outcomes after PH.
24
Finally, data obtained from clinical studies suggest that female patients with hepatocellular carcinoma have a better prognosis than males after surgical resection of the liver.
25
It is therefore advisable to design PH studies using animals of the same gender.
26
Age: Several studies have shown that the regeneration capacity decreases with age in rodents and humans. In addition, ageing represents an important risk factor which may lead to increased susceptibility to oxidative stress, an increase of cell death, reduced cell proliferation rate and, thus, lower regeneration with higher hepatocyte damage.27,28 Clinical studies have consistently reported higher postoperative mortality rates with age-related comorbidities.29,30 Therefore it is highly recommended to perform PH with 8–14-week-old mice, unless age-dependent studies on liver regeneration are intended. Genetic background: Although it seems that genetic background has no important effect on the regenerative process in wild-type animals, mutations can have very different phenotypes in different backgrounds. Indeed, it has been reported that the level of fibrosis and severity of hepatitis B virus (HBV) infection depends on the genetic background of transgenic mice.31,32 Thus, it is important to use mice with the same genetic background throughout experiments.
33
Anaesthesia: All animals must be maintained under standard general anaesthesia throughout the procedure. There is little evidence to favour any particular type of anaesthesia, although the fact that hepatic clearance will be reduced after resection should be taken into consideration. Ideal anaesthetic effects (loss of consciousness, loss of sensation, analgesia, and muscle relaxation) can be produced by using an inhalative application (i.e. isoflurane) or injection of drugs (i.e. ketamine and xylazine).
1
The combination of ketamine and xylazine is the most commonly used anaesthetic drug for inducing sedation, immobility and analgesia in mice and is very reliable. The most widely used dose of ketamine/xylazine for mouse surgery is 80–110 mg/kg and 5–10 mg/kg body weight, respectively.34,35 Drugs can be mixed together and provided by the intraperitoneal (IP) route. A suitable formulation for mice would be 1 mL ketamine (100 mg/mL), 0.25 mL xylazine (20 mg/mL) in 8.75 mL normal saline 0.9% or phosphate-buffered saline (PBS), given that 0.1 mL per 10 g of body weight will provide a 100 mg/kg ketamine/5 mg/kg xylazine dose.
36
Isoflurane is another drug that has been considered as an anaesthetic of choice for two-thirds PH surgery. Isoflurane is an inhalant anaesthetic that is widely used in veterinary medicine because of its good safety record and the rapid recovery of the animal after surgery. Anaesthesia is best maintained by inhalation of the isoflurane vaporized at concentrations of up to 4 vol% in the induction phase, and at 1.5 vol% during acute surgical procedure. 37
However, the use of isoflurane as an anaesthetic requires the additional application of appropriate analgesia such as buprenorphine hydrochloride (see ‘Postoperative management’ below). It should be noted that its use has some disadvantages which include a risk of fatal overdose, depressed respiratory rate and decreased blood pressure. In addition, once animals awaken from the gas anaesthesia, there is no residual analgesic activity.
5
Inhalational anaesthesia also requires special equipment such as a vaporizer which may not be available in every institution.
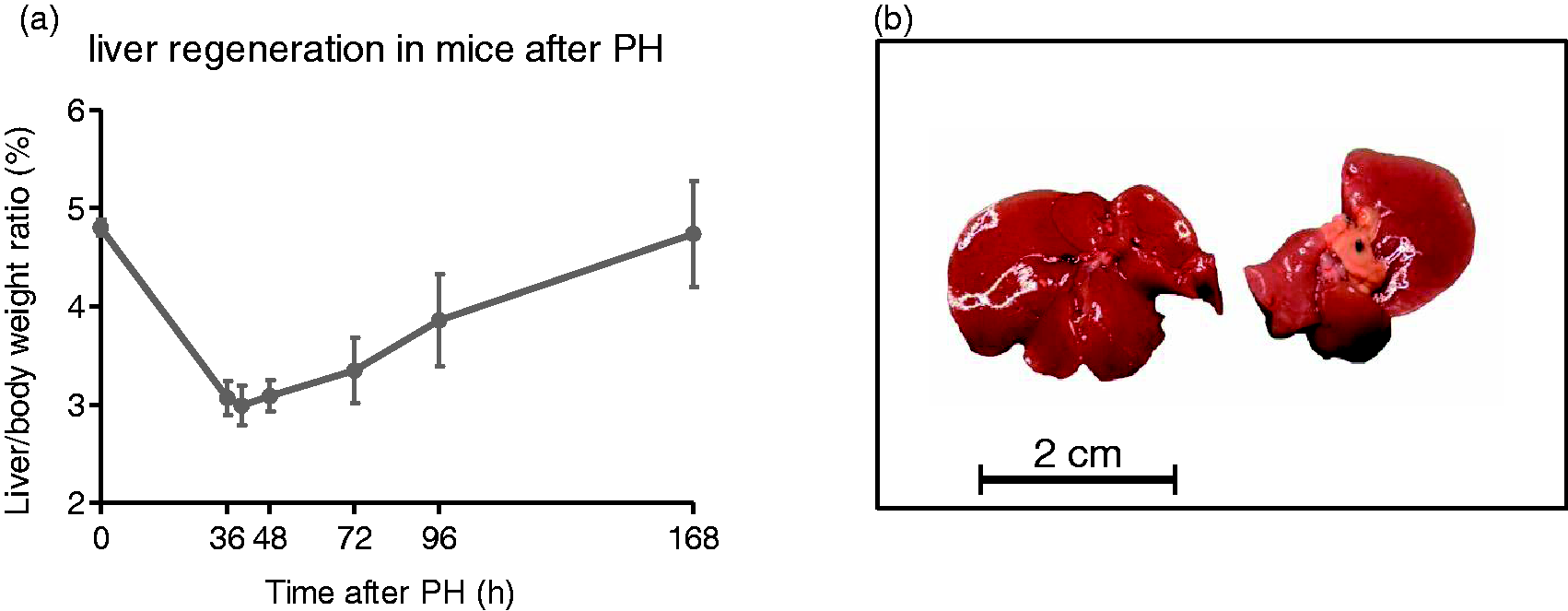
Observation time: The appropriate time points for euthanizing hepatectomized mice are determined by the signal pathways of interest. Typical time points for the investigation of the priming phase are 0.5, 2, 4 and 6 h post-hepatectomy which covers the induction of most immediate response factors such as NF-κB, pSTAT3, JNK, c-Jun and others.17,19 If investigation of the progression phase is intended, mice should be sacrificed at 36 (onset of S-phase), 40, 48 (peak of DNA replication) and 60 h (termination of cell cycle activity) after liver resection. Significantly, the PH procedure enables the collection of quiescent liver tissue (resected material at time point zero) and regenerating liver at the time point of interest from the same animal, thereby allowing a direct comparison of both states in one individual. For the monitoring of the actual liver mass restoration, mice should be investigated at a minimum after two, three, four and seven days after PH, respectively as shown in Figure 1a and published previously.
18
Typically, liver mass and liver size is almost completely restored after seven days (Figure 1b). Kinetics of liver regeneration in mice following partial hepatectomy (PH). (a) Representative time course of liver mass restoration in wild-type mice following PH. The peak of cell cycle activity in the remaining hepatocytes usually occurs between 36 and 48 h post-surgery. Original liver mass is almost completely restored seven days after hepatectomy. (b) Morphology and size of mouse liver before and seven days after PH. Please note that liver regeneration is a pure reconstitution of liver mass while the resected liver lobes are not restored.
Animal burden/default rates
Mice subjected to PH are only moderately burdened due to laparotomy and do not develop severe long-term complications. Usually the animals already show normal behaviour including food and water intake a few hours after recovery from anaesthesia. Postoperative pain is avoided due to analgesic treatment after surgery (see below). If the hepatectomy is performed properly, mortality rates in wild-type mice due to surgery will be below 5%. However, this does not exclude the possibility that genetically-modified mice may exhibit higher mortality rates if the manipulated gene of interest directly interferes with the liver regeneration process.
Classification of severity of procedure
According to Article 15 of the EU Directive 2010/63 (http://eurlex.europa.eu/LexUriServ/LexUriServ.do?uri=OJ:L:2010:276:0033:0079:en:PDF) the estimated degree of pain, suffering, distress or lasting harm of the animals subjected to liver resection should be classified as ‘non-recovery’, ‘mild’, ‘moderate’ or ‘severe’. Details about the classification criteria that underlie this assessment have been established by the Expert Working Group on severity classification of scientific procedures performed on animals. These can be found at: http://ec.europa.eu/environment/chemicals/lab_animals/pdf/report_ewg.pdf. The partial liver resection with no major impairment of liver function is classified as a moderate procedure according to Article 15 of the above-mentioned EU Directive 2010/63.
Practical implementation
The following procedures need to be implemented and equipment needs to be used:
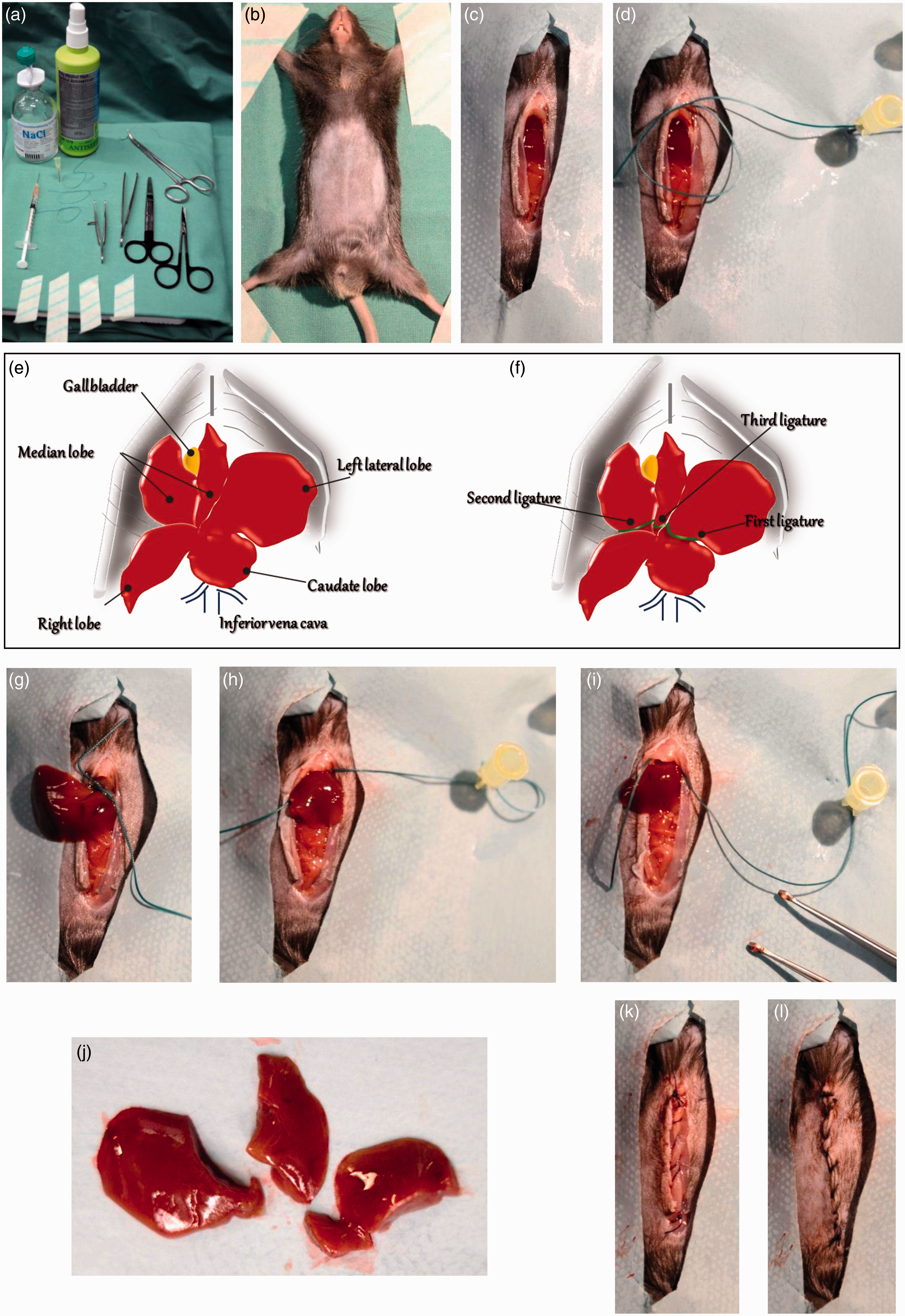
Prepare sterile surgical instruments (needle holder, microsurgery scissors, surgical forceps) and three suture lassos for ligation of the liver lobes (Figure 2a). For anaesthetization choose one of the methods described above. A suitable mask should be used to maintain anaesthesia by isoflurane inhalation. Check that the mouse is deeply anaesthetized. Shave the abdominal area using an appropriate electric fur shaver (e.g. Shaver Favorita II; Aesculap, Braun Melsungen AG, Melsungen, Germany, Figure 2b). Lay the anaesthetized mouse on its back onto a heating pad at 37 ℃ (e.g. HP062; Labotect, Göttingen, Germany) to prevent hypothermia and fix in an X-shaped supine position using tape (Figure 2b). Critical step: test the tail and toe reflexes by a gentle pinch of the toe. Any observed movement (withdrawing the paw) indicates that the animal is not sufficiently anaesthetized for surgery (surgical tolerance state).
Practical implementation of partial hepatectomy (PH). (a) Material and equipment required for the surgery: disinfectant, pre-warmed saline, surgical needle holder, microsurgery scissors, surgical forceps, tape for fixation of animal, three suture lassos for ligation of liver lobes. (b) Preparation of anaesthetized mouse. For midline laparotomy, the abdominal area is shaved and disinfected. (c) Midline laparotomy. (d) Preparation and fixation of suture lasso/loop prior to ligation of liver lobes. (e) Schematic representation of murine liver anatomy. Please note that for PH the left lateral lobe, the right portion of the median lobe and the left portion of the median lobe are resected. (f) The locations for optimal ligature of the three liver lobes are highlighted within the scheme. (g–i) Ligation of left lateral lobe (g), left median lobe (h) and right median lobe (i), respectively. (j) Size and morphology of resected liver lobes. From left to right: left lateral lobe, left median lobe, right median lobe. (k) Closure of the peritoneum. (l) Mouse post-surgery with separate closure of the skin layer. Apply the ophthalmic lubricating ointment (Puralube®, Lacrilube® or equivalent) to the eyes of the animal to protect the corneas from drying out. Disinfect the skin with 70% ethanol or polyiodine. Using blunt microsurgery scissors, make a midline laparotomy via a 3 cm long abdominal skin and muscle incision exposing the xiphoid process, the abdominal organs and liver (Figures 2c and 2d). Identify the relevant hepatic lobes (Figures 2e and 2f). Critical step: avoid accidental penetration into the chest cavity. Prepare three suture lassos/loops (EXCEL 2-0 non-absorbable suture; Ethicon Inc, Cincinnati, OH, USA) with an approximate outer diameter of 1.5 cm. Stick one needle into the styrofoam pad on the left upper side of the animal and fix one free end of the loop (Figure 2d). Place a loop of suture around the protruding margin of the left lateral liver lobe. Gently pull down the lateral lobe through the loop with non-traumatic tissue forceps or with cotton wool swaps. Try to place the knot as close to the base of the lobe as possible (Figure 2g). Pull the ligature tight, and allow it to gently crush through the hepatic parenchyma. As the suture tears through the soft hepatic tissue, vessels and biliary ducts are ligated. Make a knot and tie the two ends of the suture. The colour of the lobe will change (to dark red) due to disrupted blood outflow. Hold the liver gently between the forceps and, using microsurgery scissors; sharply cut the tied segment 2–3 mm distal to the placed ligature. Place the next suture lasso for the second knot (Figure 2h). Using tissue forceps, pull down the right portion of the median lobe extremely gently and carefully place the suture around the lobe. Pull the loop and firmly tighten the suture ends. Make a knot and excise the hepatic parenchyma distal to the ligatures. Similarly, place the third suture loop encircling the left portion of the median lobe, tighten and ligate the suture (Figure 2i). Resect the parenchymal tissue distal to the ligature. Critical step: take care not to harm the gallbladder, extrahepatic biliary tree or suprahepatic vena cava, or this will lead to necrosis of the remaining lobes and failure to regenerate. Check carefully for the presence of bleeding points using a cotton swap: minor bleeding could occur from the resected lobe. Wash the intraperitoneal cavity and organs with warm saline. Close the peritoneum and fascia first with a continuous suture using absorbable thread (4-0 Vicryl; Ethicon Inc, Figure 2k). The skin layer should be closed with single stitches using the same method (Figure 2l). After closing the abdomen, wipe the skin surrounding the suture with antiseptic (Betadine). Inject 0.5 mL warm sterile isotonic fluids subcutaneously in order to replace fluid loss from intraoperative evaporation and bleeding.
Postoperative management
Animal care
Mice have a greater body surface area to body mass ratio than larger animals; thermal support is therefore critical to their survival and successful anaesthetic recovery. Hypothermia results in prolonged recovery, adding additional stress to the animal and increased mortality due to anaesthetic side-effects. Therefore supplemental heat should be provided to mice awakening from anaesthesia, such as hot water blankets, temperature controlled warming pads or heat lamps (but avoid burns).38,39 Mice should be allowed to recover from anaesthesia in a clean cage with constant observation until they are stable and fully recovered.
Analgesia
One of the most important components of animal care is the prevention or recognition and minimization of pain and distress after an operation. Recent studies have demonstrated that unrelieved pain can have profound negative physiological consequences and affect locomotor activity, body weight, food and water intake and, subsequently, alter research results.40,41 However, experimenters should be advised that most analgesics may affect liver metabolism or inflammatory pathways as specified below and therefore might influence the regenerative process. Commonly used analgesics include buprenorphine hydrochloride (Buprenex®, recommendation: 0.05–0.1 mg/kg), meloxicam (Metacam®, 1–2 mg/kg) and carprofen (Rimadyl®, 5 mg/kg). These drugs should be injected at the recommended doses subcutaneously in the flank shortly before surgery and immediately after the mice have recovered from anaesthesia. 42 Buprenorphine is an opiate agonist and has a maximum biological effect achieved regardless of the maximal dose given (ceiling effect). The duration of analgesia is around 6 to 12 h. 43 In fact, it has also been reported that buprenorphine can disrupt normal liver metabolism and be hepatotoxic. 44 Hence, if possible, in mouse models with impaired liver function a reduced dosage should be considered (0.05 mg/kg). 44 Additionally, this drug also has sedative properties; therefore animals must be observed more carefully for possible cumulative effects.45,46 Meloxicam (Metacam) and carpofen (Rimadyl) are non-steroidal anti-inflammatory drugs (NSAIDs) that reduce inflammatory pain by the inhibition of prostaglandin synthesis. The duration of the effect in mice has been documented as 12 to 24 h. Although the drugs are considered safe, NSAID-associated gastrointestinal bleeding is listed as a side-effect reported with chronic use. As opposed to opiate agonists, NSAIDs are not controlled substances, and do not require federal licensing, strict storage and record keeping.47,48 To assess the adequacy of analgesia and the potential need for its extension, an appropriate and easy method for monitoring mice for signs of pain throughout the postoperative period should be used, for example facial expression coding. 49 It is strongly recommended that analgesia should be maintained until the end of postoperative day 2 or 3.5,48
Biometric calculation of requested cohorts
The most frequently used readout parameters for measuring liver regeneration following PH are nuclear expression of the proliferating cell nuclear antigen (PCNA) and the incorporation of the nucleoside analogue BrdU, which is typically injected into mice 2 h before the end of the experiment. Both parameters are excellent indicators of DNA synthesis preceding hepatocyte division and are maximal in mice 40–48 h after surgery. In our studies we usually observe a maximum of 20–30% BrdU positive hepatocytes in wild-type mice with a standard deviation of approximately 25%. 18 In addition, we and others have shown that a difference in hepatocyte proliferation of 30% already has strong implications for liver regeneration (increase: hepatomegaly; decrease: impaired liver mass reconstitution),16,18 and should be considered to be biologically relevant. Using the software G Power V.3.1.5., 50 a statistical power of 80%, a specific α error probability of 0.05 and a dropout rate of 5% due to postoperative complications, the minimal required sample size calculated is n = 8 mice per group.
Concluding remarks
More than 80 years after the key publication from Higgins and Anderson1 PH in rodents is still a state-of-the-art method for the community of liver researchers, though today's opportunity to modify the expression of individual hepatic genes by gene targeting in combination with this method will enable us to further improve the treatment of human liver disease. However, highly standardized operation techniques together with responsible postoperative management are essential to meet the criteria outlined in modern animal welfare standards.
Footnotes
Ethical statement
All experimental animal procedures were carried out according to German legal requirements and animal protection laws and were approved by the authority for environment conservation and consumer protection of the state of North Rhine-Westfalia (LANUV, Recklinghausen, Germany).
Funding
This work was supported by grants of the German Research Foundation (SFB/TRR57, P04, Q1, DFG LI1045/2-4). Yulia Nevzorova is supported by a grant from the Wilhelm-Sander Foundation for Cancer Research.
Conflict of interest statement
The authors have no conflicts of interest to declare.
