Abstract
Unlike non-steroidal anti-inflammatory drugs (NSAIDs), metamizole has poor anti-inflammatory effects; and is suitable for models where analgesia, but not anti-inflammatory effects, is desirable. Like opioids, these drugs produce perioperative analgesia while reducing anaesthetic requirements, but it remains unclear whether they may develop tolerance or hyperalgesia, and thus decrease in analgesic efficacy. The aim was to determine whether tolerance or hyperalgesia to metamizole occurred in rats, and whether the sevoflurane minimum alveolar concentration (MAC) was affected. In a randomized, prospective, controlled study, male Wistar rats (n = 8 per group) were administered metamizole (300 mg/kg, day 4). Previously, the following treatments were provided: daily metamizole for four days (0–3), morphine (10 mg/kg; positive control, day 0 only) or saline (negative control). The main outcome measures were mechanical (MNT) and warm thermal (WNT) nociceptive quantitative sensory thresholds. The baseline sevoflurane MAC and the reduction produced by the treatments were also determined. The mean (SD) baseline MAC [2.4(0.2)%vol] was decreased by morphine and metamizole by 45(11)% and 33(7)% (P = 0.000, both), respectively. Baseline MNT [35.4(4.5) g] and WNT [13.2(2.4) s] were decreased by morphine and metamizole: MNT reduction of 22(6)% (P = 0.000) and 22(7)% (P = 0.001), respectively and WNT reduction of 34(14)% (P = 0.000) and 24(13)% (P = 0.001). The baseline MAC on day 4 was neither modified by treatments nor the MAC reduction produced by metamizole (days 0 and 4; P > 0.05). In conclusion, repeated metamizole administration may produce hyperalgesia, although it may not modify its anaesthetic sparing effect. The clinical relevance of this effect in painful research models requiring prolonged analgesic therapy warrants further investigation.
Metamizole (dipyrone) is one of the most commonly employed analgesic drugs, used in both the perioperative period 1 and for reducing postoperative opioid consumption in humans.2,3 Compared with non-steroidal anti-inflammatory drugs (NSAIDs), metamizole has low renal, cardiovascular and gastrointestinal toxicity. This relative absence of anti-inflammatory effects has encouraged its use in research in animal models of coagulation and inflammation, 4 including adjuvant-induced arthritis. 5 Metamizole has also been employed in neuropathic pain models, such as sciatic pain or acute lumbalgia, and in the postsurgical period. 6 Due to its unique actions, metamizole can be administered in combination with NSAIDs 7 or opioids. 8
Compared with NSAIDs, an alternative mode of action has been proposed for metamizole. 9 This drug is a weak inhibitor of the cyclooxygenase isoforms 1 and 2 (COX-1 and COX-2), but is more potent with COX-3, 10 and may also act through the cannabinoid,11,12 transient receptor potential vanilloid (TRPV) and opioidergic systems. 13 This latter effect has been associated with opioid tolerance and increased risk during opioid withdrawal,14,15 which may decrease the efficacy of repeated metamizole administration15,16 and may modify perioperative inhalational anaesthetic requirements. The anti- or hyperalgesic actions of metamizole remain unclear, although an antihyperalgesic effect has been proposed, 17 but it may also produce an indirect activation of opioid receptors by releasing endogenous opioids. 18 Tolerance has been observed within a few days of repeated drug administration in rats. 19
We hypothesized that metamizole reduces the minimum alveolar concentration (MAC) of sevoflurane; but its repeated administration might be associated with the development of tolerance and hyperalgesia, similar to opioid-induced hyperalgesia, 20 which might modify sevoflurane MAC in rats. Therefore, the aim was to determine the sevoflurane MAC sparing action of metamizole and to assess whether it could induce hyperalgesia or tolerance, which might modify sevoflurane MAC in rats.
Animals
Twenty-four adult male Wistar rats (9–11 weeks old, originally purchased from Charles River Laboratories, Barcelona, Spain) were used in this research and were housed in groups of four animals per cage (Makrolon type IV; Panlab, Barcelona, Spain) with a 12:12 h light–dark cycle at a relative humidity of 40–70% and 22 ± 2℃ ambient temperature. Food (A03; SAFE, Augy, France) and water were provided ad libitum. Enrichment was provided by adding cardboard houses and aspen bricks (Sodispan Research, Madrid, Spain). The animals were allowed to acclimatize to the animal facility for at least one week. All of the studies started at 10:00 h and were performed during the light period. The study was approved by the institutional ethics committee of the Complutense University of Madrid (UCM), Madrid, Spain (approval date: 17 March 2011). The study was performed in accordance with EU Directive 2010/63, and the Animals in Research: Reporting in Vivo Experiments (ARRIVE) statement.
Materials and methods
Experimental design and drug groups
To determine the impact of previous analgesic therapy (metamizole, morphine or placebo) on the metamizole analgesic effects and anaesthetic sparing action, 24 rats were randomly assigned to one of three treatment groups (n = 8 per group). All the animals were evaluated for quantitative sensory thresholds (QSTs), using the Von Frey (mechanical) and the tail flick (warm thermal) tests (days 0, 2 and 4), and were anaesthetized under sevoflurane inhalant anaesthesia twice (days 0 and 4) for MAC determination where two consecutive MAC values (MAC-A and MAC-B) were determined in every animal. Briefly, once rats were anaesthetized and instrumented (30 min), the baseline sevoflurane MAC (MAC-A) was determined and the rats were administered the treatments, and a second MAC (MAC-B) was calculated starting 30 min following drug administration. Each MAC determination lasted between 40 and 60 min, and the whole experiment lasted 2.5–3.0 h. After the last MAC determination on day 4 (MAC-B), the rats were euthanized with an overdose of potassium chloride given intravenously while still deeply anaesthetized.
Drug groups
The experiment lasted five days and the three treatments were administered randomly. In a first group, rats were administered metamizole (300 mg/kg; Nolotil, Boehringer Ingelheim, Sant Cugat del Vallès, Spain) daily for four days (day 0 to day 3). In a second group rats were administered a single dose of morphine on day 0 (positive control; 10 mg/kg; Morfina 2%, B. Braun Medical, Terrasa, Spain. A final group was administered saline (negative control). In the two latter groups sham treatment with saline was administered instead of metamizole from day 1 to day 3. On day 4 the three groups were administered metamizole (same dose and route) and the combined effects of previous treatments and metamizole on nociceptive thresholds and MAC were assessed (see below). Treatments on day 0 were randomly administered intraperitoneally. All drugs were administered in a total volume of one millilitre (Figure 1).
Experimental design. Mechanical (MNT; Von Frey test) and warm thermal (WNT; tail flick test) quantitative nociceptive thresholds (QST) determined on days 0, 2 and 4, and the minimum alveolar concentration (MAC) at baseline (MAC-A) and following drug administration (MAC-B), determined on days 0 and 4. Drugs administered on day 0 were morphine, metamizole and saline (control). Then, from days 1 to 3, animals were injected daily with saline, in the control and morphine groups, and with metamizole in the metamizole group. On day 4 metamizole was administered in the three groups. Hyperalgesia was defined as a decrease in mechanical nociceptive thresholds and delayed tolerance was considered to be the decrease in the MAC reduction (MAC-B vs MAC-A) produced by metamizole on day 4 in all groups. NT: nociceptive threshold.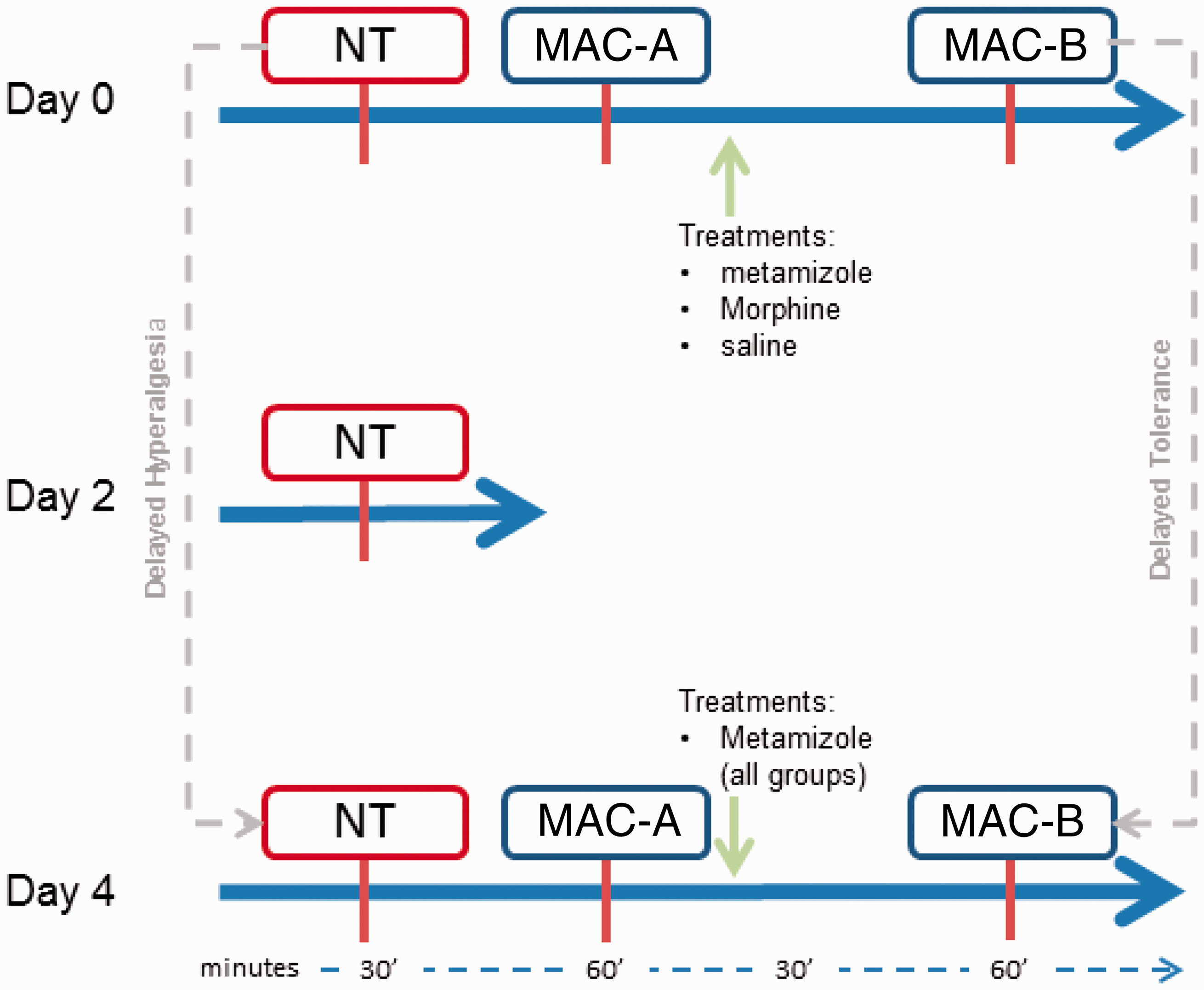
Behavioural tests of nociception
Mechanical nociceptive thresholds (MNT, Von Frey test) and warm thermal nociceptive thresholds (WNT, tail flick test) 21 were determined on days 0, 2 and 4, and always before the MAC determination on days 0 and 4 (see below and Figure 1). Hyperalgesia was defined as a decrease in MNT force values and as a decrease in WNT time values. Tolerance was considered as a decrease in the ability of metamizole to reduce the MAC on day 4 compared with day 0.
Von Frey test
An MNT test was performed by evaluating and measuring the left and right hind paw withdrawal force responses to the application of electronically calibrated Von Frey filaments (Electronic Von Frey Anesthesiometer, Model 2393; IITC Inc, Woodland Hills, CA, USA). Animals were acclimatized to the testing procedure for 5–7 days before baseline values (day 0) were obtained. On day 0, they were placed in a methacrylate cylinder (25 × 15 × 15 cm) with a wire grid bottom (1 cm2 perforations) 10 min before the procedure started. Filaments were provided with the electronic Von Frey device with a tip surface of 0.5 mm2 and were chosen to cover a pressure range within 10–50 g as indicated by the manufacturer. The pressure range produced by every filament when applied was ascertained after 10 tests before the study. Filaments were applied, starting from the thinnest to the thickest, to both hind paws for 5 s or until a positive response was observed. Locomotion was not considered to be a withdrawal response. After a negative response was observed, the pressure was increased using the next thicker filament until the rat showed a positive response. The maximum and minimum pressures applied, as determined by the device, were recorded in grams as a measure of the threshold. When this latter event was observed the filament with the next lesser thickness was used. The maximum and minimum pressures applied, as determined by the device, were recorded. Filaments were applied in one hind paw and then in the contralateral paw and the measurements on the same side were always done with an interval of at least 5 min. The overall threshold force in grams of each hind paw was calculated as the mean of the five applied pressures from the first crossover, and the mean of these two values (left and right paws) was recorded as the MNT of each animal.
Tail flick test
A WNT test was performed by measuring the tail withdrawal response, latency in seconds, to the application of an electronically calibrated tail flick device (Tail Flick Analgesia Meter, Model 336; IITC Inc). 21 Similarly, to minimize the influence of stress during the experimental procedure, the animals were acclimatized for 5–7 days five days before baseline values were determined on day 0 and for 10 min before the procedure started (day 0) by introducing them in a methacrylate cylinder (7 × 20 cm) for tail flick measurements. During the testing, a heating light beam pointed on the dorsal skin surface in the rat’s tail was used to elicit a noxious response. When the animal flicked its tail, the time in seconds was recorded as a measure of the threshold. This test was repeated five times at different zones along the tail and the threshold time in seconds from each rat was calculated as the mean of those five measures. The MNT and WNT determinations were performed and evaluated by the same investigator (DR).
Anaesthetic procedure
The rats were induced with sevoflurane 8%vol vaporized in a continuous oxygen flow of 2 L/min (Sevoflurane Vaporizer, Sevorane Dragër Vapor 2000; Dragër, Lübeck, Germany). All the rats were intubated using an otoscope to introduce a flexible, blunt-tipped wire into the trachea, which was also used to direct a 14-gauge polyethylene intravenous catheter (Terumo Surflo; Terumo Europe NV, Leuven, Belgium). To ensure that the catheter was well positioned, a capnograph was used to determinate the presence of exhaled carbon dioxide. The catheter was then connected to a small T-piece breathing system with minimum dead space (2 mL). Fresh gas flow to the T-piece was adjusted to 0.50–1 L/min of oxygen (100%), and the sevoflurane concentration was adjusted to 1 × MAC (2.4–2.6%vol). Anaesthetic monitoring was performed: pulse rate, arterial oxygen haemoglobin saturation (via pulseoximetry), and respiratory rate (Datex-Ohmeda SA5 monitor; General Electric, Helsinki, Finland). Rectal temperature was also monitored and maintained between 37.0℃ and 38.0℃ using a water-circulating warming blanket (Model TP-220; Gaymar, Orchard Park, NY, USA) and a heating light. Ventilation of all rats was maintained spontaneously.
Determination of MAC
MAC is a standard measure of volatile anaesthetic potency and is defined as the concentration required to prevent gross purposeful movement in 50% of subjects in response to a noxious stimulus. In this study the individual MAC value from every rat was determined on days 0 and 4 by means of intratracheal gas sampling to measure the anaesthetic gas concentration. A 20-gauge needle was inserted through the endotracheal catheter with the needle tip located at the entrance of the endotracheal catheter, and gas samples were assayed using a side-stream infrared analyser (Datex-Ohmeda SA5 monitor).
MAC determinations were evaluated by the same investigator (DR) who was not blinded to the drugs administered. A supramaximal noxious stimulus was applied with a long haemostat (8-inch Rochester Pean Hemostatic Forceps; Martin, Tuttlingen, Germany ) clamped to the first ratchet lock on the tail for 60 s, or until a positive response was observed. The tail was always stimulated proximally to a previous test site when the previous response was negative, or it was stimulated more distally if the response was positive, starting 6 cm from the tail base. A positive response was considered to be a gross purposeful movement of the head, extremities or body. A negative response was considered to be a lack of movement or grimacing, swallowing, chewing or tail flicking. When a negative response was seen, the sevoflurane concentration was reduced in decrements of 0.2%vol until the negative response became positive. Similarly, when a positive response was seen, the sevoflurane concentration was increased by 0.2%vol until the positive response became negative. MAC was considered as the mean of the two highest concentrations that did not prevent movement when the stimulus was performed and the two lowest concentrations that prevented such movement. Two independent crossover positive–negative or negative–positive responses were used to set the MAC value as the mean of these four calculations. Determination of the MAC was performed in a laboratory 650 m above sea level, which lowered barometric pressure and resulted in MAC values that would have been higher than those obtainable at sea level. Therefore, MAC values were corrected to the barometric pressure at sea level using the following formula: MAC (%) at sea level barometric pressure (760 mmHg) (altitude adjusted MAC) = measured MAC (%) × measured ambient barometric pressure (700 mmHg in Madrid)/sea level barometric pressure (760 mmHg).
Statistical analysis
Previous data from our lab and sample size calculations indicated that an n value of 7–8 was necessary to detect changes in the MAC of at least a 10%, with a power of 80% and a P value of 0.05. Data are presented as the mean (SD). Rats in each experiment were randomly allocated to the different treatment groups using a random number generator (www.randomization.com). To assess the effects of drugs on the MAC and MNT values (absolute and relative percentage of variation), the repeated measures analysis of variance (ANOVA) test was used, and the treatment (group) was the between-subjects factor. The one-way ANOVA test was employed to compare differences between groups on each day, but also to assess the appearance of hyperalgesia within the same treatment group by comparing the MNT, WNT and MAC-A (day 4 only) on days 2 and 4 with those on day 0. This test was further used to assess the appearance of delayed opioid tolerance by comparing the MAC-B on days 0 and 4. The Bonferroni post-hoc test was employed to compare groups. To assess the appearance of acute opioid tolerance, the paired Student's t-test was employed to compare the MAC-A and MAC-B of each treatment group on each day of the study (days 0 and 4). The analyses were two-tailed with a P value of < 0.05 to indicate statistical significance. All analyses were performed using the SPSS statistical package (SPSS Inc, Chicago, IL, USA).
Results
Animals
The rats in the different groups had similar body weights on day 0, with no differences between groups or study days [368(28) g on day 0, n = 24, ANOVA, P = 0.789]. No adverse effects were observed during the experimental-treatment days; however urinary flow rate (monitored but not recorded) seemed to be increased during MAC determinations under anaesthesia when metamizole was administrated, compared with the other two treatments.
Behavioural tests of nociception (Table 1, Figure 2)
Von Frey test
Quantitative sensory thresholds.
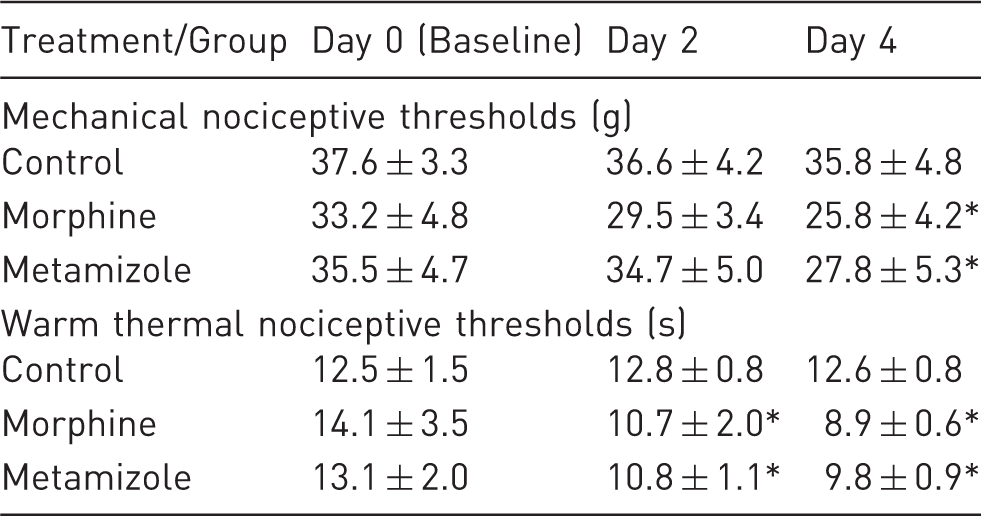
Mechanical (MNT; Von Frey test; force in grams) and warm thermal (WNT; tail flick test; time in seconds) nociceptive thresholds at baseline and following morphine and metamizole treatments on days 2 and 4. Data are expressed as mean (SD). The n value is eight per group (see Methods for details). *Indicates significant differences compared to control group, same day, P < 0.05.
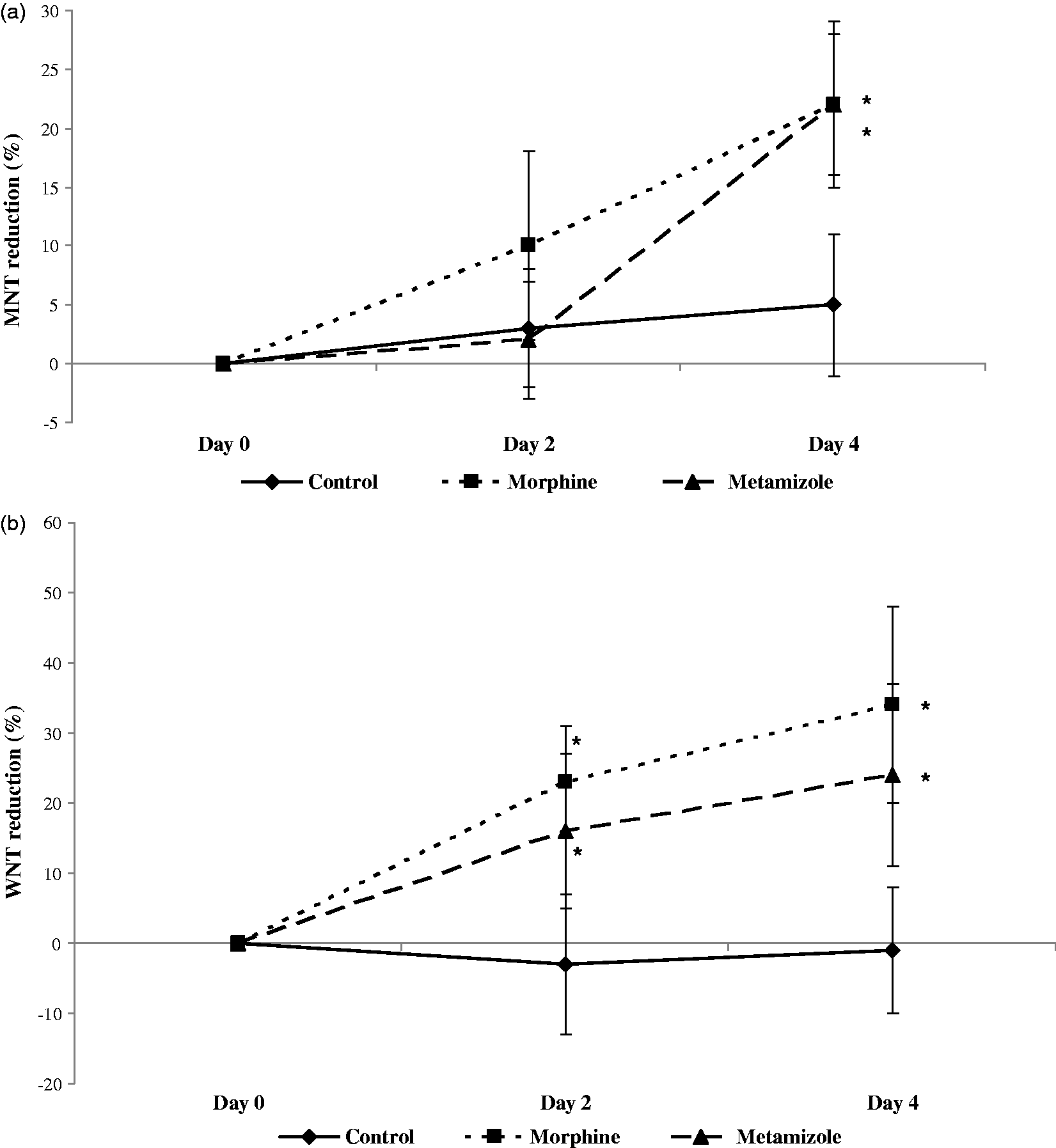
Quantitative sensory thresholds. Maximum reduction in the (a) mechanical (MNT; Von Frey test) and (b) warm thermal (WNT; tail flick test) thresholds produced by a single dose of morphine and saline administered on day 0, or metamizole administered daily on days 0 to 3, and determined on days 2 and 4. Data are expressed as mean (SD); n = 8 per group. *Indicates significant differences compared with the control group, same day, P < 0.05.
Tail flick test
The baseline WNT on day 0 from all rats was 13.2(2.4) s (n = 24), with no differences between treatment groups (P = 0.45). Rats in the groups administered morphine (on day 0) or metamizole (all days) presented similar decreased time thresholds on day 4 compared with the control group [8.9(0.6) s and 9.7(0.9) s, respectively; control group = 12.6(0.8) s; P = 0.000 and P = 0.001, respectively]. Similarly, WNT were decreased on day 2 in the morphine and metamizole groups compared with the control group [10.7(2.0) s and 10.8(1.1) s; control group = 12.8(0.8) s; P = 0.00 and P = 0.002, respectively]. No differences were determined between the morphine and metamizole groups on days 2 and 4 (P = 1.00 and P = 0.09, respectively).
MAC (Table 2, Figure 3)
Minimum alveolar concentration (MAC) values.
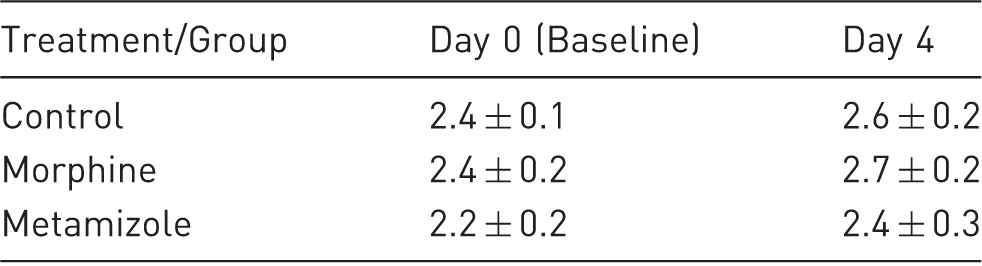
Baseline sevoflurane MAC (day 0, MAC-A; %vol) and at day 4 from each group. Data are expressed as mean (SD). The n value is eight per group (see Methods for details).
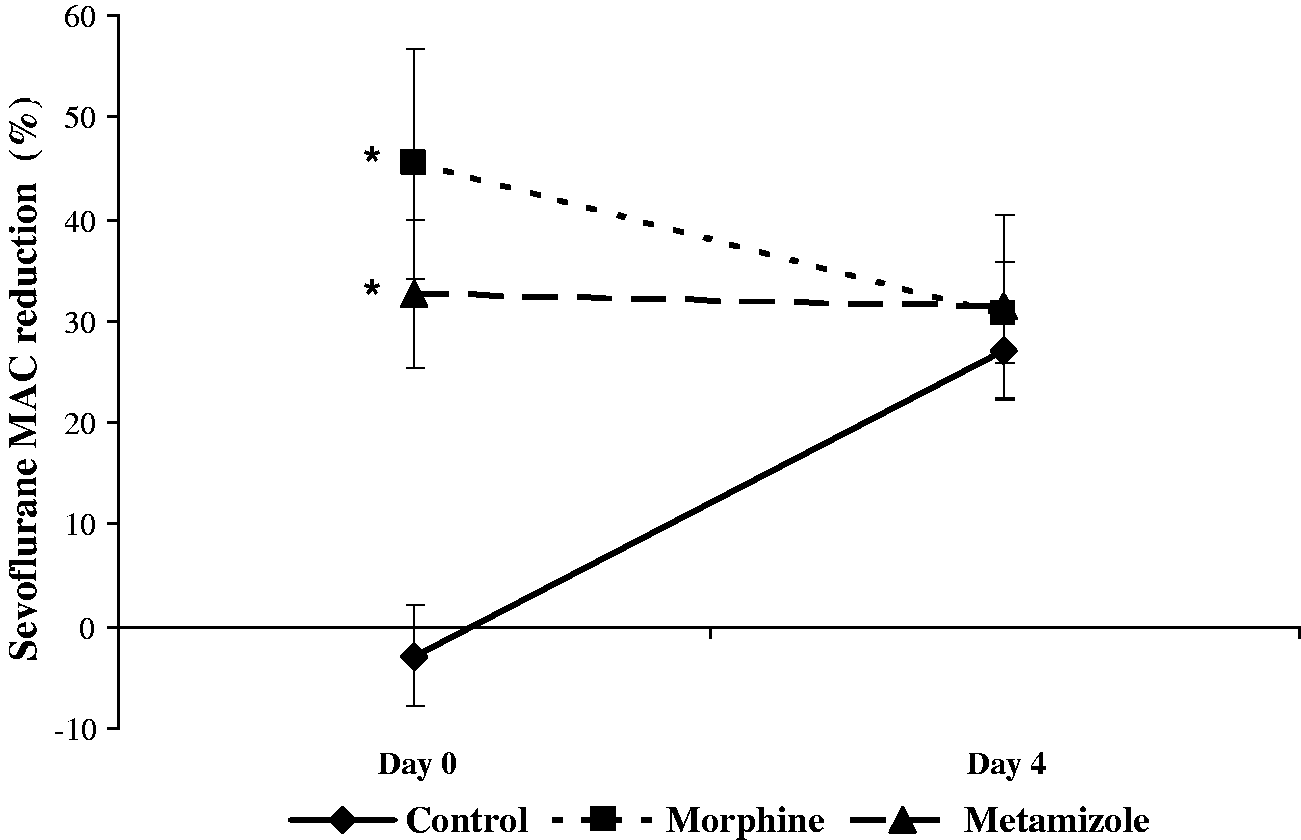
Sevoflurane minimum alveolar concentration (MAC). Maximum reduction in the sevoflurane MAC produced by a single dose of morphine and saline administered on day 0, or metamizole administered daily on days 0 to 3, and determined on day 0, and maximum reduction in the sevoflurane MAC produced by the same dose of metamizole in the same three treatment groups on day 4 (P = 0.37). Data are expressed as mean (SD); n = 8 per group. *Indicates significant differences compared with the control group, same day, P < 0.05.
Discussion
Metamizole reduced the sevoflurane MAC in rats at clinically relevant levels, and this reduction was not blunted when the same drug with the same dose was administered four days later, suggesting the absence of a tolerance effect. However, previous repeated administration of metamizole produced a decrease in the MNT and WNT, suggesting a hyperalgesic effect. As expected, this effect was also observed following a single dose of morphine. 22
Tolerance is defined as a decrease in the response to a substance due to previous exposure, while hyperalgesia is an increased sensitivity to pain. According to these definitions, both metamizole and morphine caused hyperalgesia but not tolerance. These results do not agree with previous findings suggesting a tolerance effect of metamizole when injected in the periaqueductal grey matter of rats. 15 Besides, findings in our study showed an increase in MNT and WNT 21 following both metamizole and morphine administration. Again, these findings are in contrast to a potential antihyperalgesic action of metamizol in carrageenan-evoked hyperalgesia and oedema agent, 17 which may be attributable to the hyperalgesia model employed and activating different pathways or systems. An analgesic potentiation of morphine by metamizole has been observed in rats (tail flick test), which might be a consequence of a common action on opioid-like receptors13,18 and may suggest that the hyperalgesic effect produced by metamizole might be promoted through opioid activation. A similar and early effect has been observed following a single administration of remifentanil (intravenously) 23 and morphine (intrathecally) in rats, 24 or fentanyl in mice. 25 Furthermore, previous reports have shown cross-tolerance to opioids such as morphine by non-opioid drugs such as the NSAID celecoxib in rats, 19 while a delayed development of tolerance has been determined with a simultaneous administration of metamizole and morphine. 26
The effects of hyperalgesia and tolerance have been investigated and determined with two different QST methods, behavioural nociceptive method and the MAC. Although not necessarily linked to their analgesic efficacy, analgesics such as opioids typically decrease the MAC of inhalational anaesthetics in a dose-dependent fashion. Therefore, a delayed hyperalgesic state like that produced by opioids23,25 or metamizole within a few days might also modify the MAC, albeit in an opposite direction by increasing it. Although not demonstrated in the present study, a relationship between hyperalgesia and increased inhalational anaesthetic requirements has been suggested.23,27 This may be explained by the degree of hyperalgesia determined being lower to that previously reported, which might have led to subtle increases in the MAC. Changes in the MAC below 10% fall within the variability of the MAC method itself 28 and a higher statistical power is likely to be required. In addition, the repetition of the MAC method, and thus the application of the noxious stimuli, may increase the MAC value by some 5%. 23 Finally, the expected individual variability in the MAC within a given species is close to 10%. 29 Overall, a reduction in nociceptive thresholds was not associated with changes in the MAC in rats, and furthermore, such decrease did not reduce the efficacy of metamizole in order to decrease the MAC immediately following its administration, being similar to that produced by the same drug and dose administered by the first time (MAC-B vs MAC-A on day 0).
A slightly lower MAC reduction of 20% has been determined in rats with a low dose of metamizole (15 mg/kg), 20-fold lower to that employed in this study (300 mg/kg) to produce a MAC reduction of 30%, suggesting that this latter high dose may actually not be necessary or that it does not increase the maximum MAC reduction that could be achieved in the rat with lower doses of the latter. 30 In the rat, opioids such as morphine produce a clear dose-response relationship, with high doses producing a reduction in the MAC above 50%. Therefore, it is unlikely that metamizole would further promote an anaesthetic sparing action with higher doses 13 than those employed in this study (300 mg/kg). The observed effects of metamizole in the rat should be determined in other species since great variations in the effects of analgesics on MAC have been determined between them, 31 and also between strains and individuals, 29 besides variations in the methodologies employed. 32 This MAC sparing effect is similar to that produced by relatively high doses of morphine in sevoflurane-anaesthetized Sprague–Dawley rats, 33 and isoflurane-anaesthetized Wistar rats. 34
One of the limitations of the study may be that a single dose was employed, although this was within the effective dose range reported in this species. 35 Another limitation is that QST (MNT and WNT) and MAC determinations were performed by a person not blinded to the drugs administered. However, the movement responses to behavioural tests of nociception, MNT and WNT, or the supramaximal noxious stimulus for MAC determination are highly objective and reproducible measures. Besides, all QST and MAC determinations were assessed by the same person, thus reducing bias in the results. Another potential limitation of the study is the naïve rat model employed, which may differ in potentially painful disease models. Despite conflicting results36,37 hyperalgesia may occur following opioid administration, with most studies suggesting this phenomenon when performed in volunteers.38–40 Therefore, it remains unclear whether the hyperalgesia observed might be of clinical relevance in patients with painful conditions such as in rodent models of arthritis.
In conclusion, metamizole administered to naïve rats reduced the MAC to a clinically significant level but may produce hyperalgesia. However, the hyperalgesia was not associated with changes in the sevoflurane MAC or in the anaesthetic sparing action of metamizole or morphine. Moreover, cross-tolerance between morphine and metamizole was not demonstrated, and both drugs may be combined perioperatively. These results further suggest the perioperative usefulness of metamizole in rats, although the clinical relevance of the hyperalgesia produced by its repeated administration should be determined.
Footnotes
Acknowledgement
The authors wish to thank the staff from the Experimental Surgery Unit, La Paz University Hospital for their assistance with the animals and the study performed.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by a grant from the Spanish Health Ministry (Grant PI11/01241).
