Abstract
Gabapentin has been used to treat a variety of conditions in both human and veterinary medicine, including seizures, neuropathies and chronic pain. However, little information is known about the effects of gabapentin on the minimum alveolar concentration (MAC) of volatile anaesthetics. In this study, we investigated the effect of intraperitoneal administration of gabapentin on isoflurane MAC in adult male rats and hypothesized that gabapentin would decrease MAC in a dose-dependent manner. Using a standard MAC study protocol, we compared five treatment groups (G) receiving 0 (G0), 30 (G30), 100 (G100), 300 (G300) and 1000 (G1000) mg/kg gabapentin intraperitoneally and compared post-drug MAC values among groups and with corresponding baseline MAC values determined in each group prior to drug testing. The average baseline isoflurane MAC value was 1.45 ± 0.17%, which did not differ significantly between groups (1.47 ± 0.23% [G30], 1.46 ± 0.23% [G100], 1.48 ± 0.18% [G300] and 1.42 ± 0.2% [G1000]). In the G300 and G1000 groups, the isoflurane MAC value decreased significantly by 19% and 18%, respectively, from corresponding baseline values (P< 0.05, when compared with G0). Linear regression analysis revealed a negative correlation between blood gabapentin concentration and percent change in MAC (R 2 = 0.43; P< 0.05) but not dose. In conclusion, high-dose intraperitoneal gabapentin decreased isoflurane MAC. However, the effect was small and not dose-dependent, and is unlikely to be clinically significant.
In 1993, gabapentin was introduced as a drug for the treatment of seizures. 1 Subsequent studies have shown that gabapentin is useful in treating a variety of other conditions in human patients, including postherpetic neuralgia 2 and diabetic neuropathies. 3 In a perioperative setting, the preoperative administration of a single dose of gabapentin (1200 mg or less) has been effective in reducing postoperative pain and opioid consumption in human patients. 4 However, despite its apparent anticonvulsive and antinociceptive properties, the exact mechanism(s) of action of gabapentin is still being determined. Many theories have been proposed, such as interactions at the GABAergic and glutamatergic systems, and more recently at the α 2 δ subunit of N-type Ca2+ channels. 5
In rat pain models, studies have demonstrated that intrathecal administration of gabapentin attenuates the nociceptive response to formalin injected into the animal's paw 6 or face. 7 Another study described a dose-dependent antinociceptive effect of systemic gabapentin on visceral pain produced by intraperitoneal acetic acid injection. 8 Utilizing a Brennan model of surgical pain, the intraperitoneal administration of gabapentin in male Sprague–Dawley rats resulted in a decreased expression of c-Fos, a marker gene for nociception. 9 In a recent feline study, intravenous gabapentin administration failed to significantly alter the minimum alveolar concentration (MAC) of isoflurane. 10 To our knowledge, there are no studies published that have investigated the isoflurane MAC-sparing effects of gabapentin in rats. In this study, we investigated the effect of intraperitoneal gabapentin on isoflurane MAC in adult male rats and hypothesized that gabapentin would reduce MAC of isoflurane in a dose-dependent manner.
Materials and methods
Animals
The study protocol comprising a total of 33 rats was approved by the University of Pennsylvania's Campus Animal Care and Use Committee and was in compliance with the Guide for the Care of Laboratory Animals (National Institutes of Health publication 86-23, revised 1985). Male Harlan Sprague–Dawley outbred rats (HSD; colony 217, Indianapolis, IN, USA) approximately 3–5 months in age and weighing 325–375 g with conventional microbiological status were used in this experiment. The rats were housed in 48.3 cm × 26.6 cm × 20.3 cm cages (2–3 per cage depending on size) on Bed-O'Cobs (manufactured by The Andersons Inc., Maumee, OH, USA) Housing conditions were controlled with temperature and humidity maintained at 22 ± 2°C and 50 ± 20%, respectively. Artificial lighting was provided from 07:00 to 19:00 h. The rats were fed a conventional diet of pellets (LabDiet 5010 Rodent Diet, Richmond, IN, USA) and chlorinated tap water ad libitum. An acclimatization period of one week was observed before each experiment.
Anaesthesia
In each rat, anaesthesia was induced with 5% isoflurane (IsoFlo®, Abbot Laboratories, North Chicago, IL, USA) in oxygen (1 L/min), by means of a precision out-of-circuit vaporizer (IsoFluromatic™, Foregger®, Smithtown, NY, USA) within a rodent induction chamber (Harvard Apparatus, Holliston, MA, USA). Following induction, a facemask was positioned tightly around the muzzle and 2.5% isoflurane administered in oxygen (1 L/min). The rat was then positioned in lateral recumbency on a heating pad, and the temperature was recorded via a rectal thermistor (TCA-2DFH; Physitemp Instrument Inc, Clifton, NJ, USA). The rectal temperature was maintained throughout the experiment between 37.0 and 38.0°C. The skin on the left lateral aspect of the base of the tail was clipped and disinfected. A 24-gauge catheter (19 mm; BD Insyte™, Becton Dickinson, Sandy, UT, USA) was preflushed with heparinized 0.9% saline (2 IU/mL) and placed in the left lateral tail vein, using an aseptic technique. Lactated Ringer's solution (Hospira Inc, Lake Forest, IL, USA) was continuously administered via a fluid pump (Compact Infusion Pump; Harvard Apparatus) at 4 mL/kg/h.
The rat was then placed in dorsal recumbency and the ventral aspect of the neck and medial aspect of the right thigh were clipped and prepared using an aseptic technique. The mid-cervical trachea was exposed via surgical cut-down for a tracheotomy and insertion of a tracheal cannula (PE-240 tubing; Harvard Apparatus). Once positioned, the cannula was secured to the distal trachea with a circumferential ligature with silk suture (no. 2; Deknatel, Research Triangle Park, NC, USA). Mechanical ventilation was then provided via the tracheal cannula using a rodent ventilator (7025 Rodent Ventilator; Ugo Basile, Comerio, Italy), which is a piston-driven, time-cycled ventilator creating a non-rebreathing anaesthesia system. During animal instrumentation, anaesthesia was maintained with isoflurane in oxygen at the settings listed above. The inspired-isoflurane concentration was measured via a side-stream infrared gas analyser (S/5™ Compact Anesthesia Monitor; Datex-Ohmeda, Helsinki, Finland). Ventilation was set to maintain an end-tidal CO2 concentration (PETCO2 ) between 35 and 45 mmHg. These values were obtained by using a ventilation rate between 45 and 60 breaths per minute and a tidal volume of 4.0 and 5.5 mL. The PETCO2 was measured continuously with a main-stream capnometer (Capnoguard; Novametrix Medical Systems, Willingford, CT, USA), which was positioned within the expiratory limb of the breathing system, approximately 5 cm from the tracheal cannula's connection to the y-piece. Calibration of the capnometer was performed using calibration gases provided by the manufacturer before every experiment.
Cannulation of the femoral artery was achieved via surgical cut-down over the medial aspect of the right thigh. Once the femoral artery was isolated, a 15 cm long cannula (PE-10; Harvard Apparatus) was attached to a three-way stopcock and prefilled with heparinized saline. The cannula was then inserted into the femoral artery and advanced approximately 3 cm towards the iliac artery and secured with one circumferential ligature of silk suture (no. 2, Deknatel). The three-way stopcock was then attached to an arterial pressure transducer (P23XL; Becton Dickinson, Franklin Lakes, NJ, USA) and blood pressure (BP) was monitored continuously using an AC/DC amplifier (Model P122; Grass Telefactor, West Warwick, RI, USA). The anatomic reference point for the arterial pressure transducer was at the level of the sternum. The arterial pressure transducer was calibrated with a mercury manometer before each experiment. The PETCO2 , pulse rate (PR) (from the arterial trace), and the arterial pressures were continuously recorded using data acquisition hardware (MICRO 1401; CED, Cambridge, UK) and software (Spike2; CED).
Baseline MAC determination
After calibration, the isoflurane vaporizer was set to obtain an expired isoflurane concentration of 1.7% and the MAC of isoflurane was measured using a standard technique. 11,12 A catheter was placed into the tracheal cannula and positioned with the tip at the level of the carina. End-tidal gases were collected into a 20 mL glass syringe via a three-way stopcock attached to the end of the catheter. Samples were collected at the end of each expiration over 10–15 s (7–9 breaths) in approximately 0.75 to 1.0 mL aliquots. The glass syringe was then expelled slowly into the sampling port of an infrared gas analyser (S/5™ Compact Anesthesia Monitor; Datex) to obtain the expired isoflurane concentration. The syringe samples were collected in triplicate to determine a mean isoflurane concentration for any given time point. The gas analyser was calibrated with standard gases (DOT-39 NRC 300/375 M1134; Datex-Ohmeda) before each experiment. The baseline values were defined as all measurements (and calculated values) collected during the pretreatment period.
To determine baseline MAC, a noxious stimulus was applied to the tail with a haemostat (Rochester Pean Forceps 18.4 cm long [straight], Lipshaw, Germany). The level of the tail that was selected as the starting point for stimulation was standardized between rats. The initial compression site was chosen on the most distal site of the tail in which the diameter was nearly uniform before the sharp taper of the tip. Thus, successive compressions were made towards the tail base. Furthermore, each clamp was spaced in 0.5 cm increments cranial to the previous one to avoid the site of the previous compression. The force of the clamp was not measured. However, each clamp was standardized by closing the haemostat to the first ratchet click. While clamped, a continuous to and fro motion was applied for 60 s. Gross purposeful movement was defined as a head lift, kicking of the legs or sustained contraction of the abdominal muscles leading to a change in body position. One, or all, of these responses in association with application of the haemostat were considered as a positive response. A negative response was defined as a muscle twitch or an increase in PR and/or BP alone. If a positive response was elicited, the inspired concentration of isoflurane was increased by 20%. Likewise, for a negative response, the inspired concentration of isoflurane was increased by 20%. A 15 min period was permitted at the new inspired isoflurane concentration before the tail was re-clamped. Our determination of MAC was considered to be the midpoint between the lowest expired concentration of isoflurane that prevented gross purposeful movement and the highest concentration of isoflurane that caused gross purposeful movement in response to the noxious stimulus, and these values were determined in triplicate.
Blood gas analysis
Blood gas samples were acquired 5 min after baseline MAC was determined. Arterial blood samples (0.5 mL) were collected from the arterial cannula in a heparinized syringe and blood gas parameters (pH, PaCO2, and PaO2) were measured immediately using a blood gas analyser (iSTAT; Heska Corp, Loveland, CO, USA). These blood gas values were corrected to the rectal body temperature. After blood sample collection, the arterial cannula was flushed with 0.5 mL of heparinized saline.
Post-treatment MAC determination
Following blood collection, the rat received one of five doses of gabapentin (G1092-25GM; Spectrum Chemical, Phillipsburg, NJ, USA) administered in random sequence by one of the investigators, who was blinded. The dosing groups (mg/kg) were as follows: 0 (G0) (n= 6), 30 (G30) (n= 7), 100 (G100) (n= 6), 300 (G300) (n= 7) and 1000 (G1000) (n= 7). Gabapentin was combined with 0.9% saline to a total volume of 3 mL, with the G0 group receiving only saline. The total volume was injected with a 24-gauge needle into the peritoneal space, approximately 1 cm caudal to the umbilicus. Once the needle was inserted into the peritoneal cavity, negative pressure was applied to the syringe to ensure that the needle was not placed in any visceral organ before injection as aspiration of blood, urine or digestive contents would have indicated an improper needle placement.
After 15 min had elapsed post-injection, a noxious stimulus was applied and if the response was negative, the inspired end-tidal concentration of isoflurane was decreased by approximately 10% every 15 min until a positive response was obtained. If the response was positive, the inspired isoflurane concentration was increased by approximately 10% until a negative response was obtained. To obtain postgabapentin MAC, the noxious stimulus was repeated every 15 min for a total of 2 h after the injection. Based on the results of pilot tests utilizing the same protocol as described and a logarithmic dosing scheme with intraperitoneal gabapentin, the authors observed that any MAC reduction was observed within the first 20 min upon administration, thus in this study post-MAC determinations commenced 15 min postinjection. Approximately 5 min after the postgabapentin MAC had been determined, arterial blood samples were collected for the measurement of blood gas parameters as described above.
BP and PR
Both mean arterial BP and PR were measured over a 30 s period prior to each measurement of the mean end-tidal isoflurane concentration. The mean arterial BP and PR were determined in triplicate, with the baseline and postgabapentin BP values determined as the midpoint between the mean BP at the lowest expired concentration that prevented gross purposeful movement and the highest expired concentration that permitted gross purposeful movement.
Blood gabapentin concentration measurement at end of experiment
After the last post-treatment MAC determination, the inspired isoflurane concentration was increased to 3%. After 10 min, 5 mL of blood was collected from the femoral arterial cannula for immediate arterial blood gas analysis and blood harvest. Collected blood samples were stored at −80°C and later analysed for gabapentin concentrations employing a liquid chromotagraphy–tandem mass spectrometry technique that has been developed for measuring gabapentin concentrations in horse plasma. 13
Data analyses
Data were reported as means ± standard deviations (SD), except where specified otherwise. A Shapiro–Wilk W test was used to test data for normal distribution. A Grubb's test was used to identify any outlier values. No values were identified as being significant outliers. Thus, no data have been excluded from subsequent statistical analysis. To test the hypothesis of a dose-dependent change in MAC following intraperitoneal injection of gabapentin, statistical examination of group data (baseline [=pre-drug] MAC, post-drug MAC and % change in MAC from baseline values) was performed using one-way analysis of variance (ANOVA) followed by pairwise comparisons employing Sidak's multiple testing adjustment method. 14,15 Analyses were performed using the Stata software (Stata Corp, Stata Statistical Software, version 9.0, College Station, TX, USA). A linear regression analysis was performed on the relationship between blood concentrations of gabapentin and the percent change in isoflurane MAC values from baseline values using GraphPad Prism software (Prism 5; GraphPad, La Jolla, CA, USA). A P < 0.05 was considered as indicating statistically significant differences between group data. To test the effect of gabapentin dose administered on arterial BP, PR, PaCO2 PaO2, pH and lactate values, a two-way repeat measures ANOVA (JMP Statistical Software; SAS, Cary, NC, USA) was performed using factors including dose of gabapentin and time postadministration. Significant differences (P < 0.05) were then confirmed using Bonferroni correction. Gabapentin concentrations in blood were compared using the Kruskal–Wallis test (P < 0.05) with significant differences among each group compared with Dunn's multiple comparison tests (P< 0.05).
Results
The age and weights of the rats used in these experiments were similar across groups (age: 95 ± 8 days [G0], 103 ± 11 days [G30], 100 ± 16 days [G100], 91 ± 7 days [G300] and 92 ± 7 days [G1000]; body weight: 362 ± 9 g [G0], 356 ± 14 g [G30], 358 ± 13 g [G100], 355 ± 16 g [G300] and 357 ± 18 g [G1000]). The average baseline isoflurane MAC value was 1.45 ± 0.17%, which did not differ significantly between treatment groups (1.47 ± 0.23% [G30], 1.46 ± 0.23% [G100], 1.48 ± 0.18% [G300] and 1.42 ± 0.2% [G1000]) (Figure 1). Saline administration did not significantly alter isoflurane MAC in our study (Figure 1); MAC values recorded after saline administration varied by only 3.2 ± 0.09% from baseline values. Independent of the gabapentin dose administered in groups G30 to G1000, the MAC value for isoflurane determined after drug administration was not significantly different from the value determined in the G0 group after saline administration (Figure 1). However, the percent change (i.e. decrease) in isoflurane MAC from corresponding baseline values was significantly different in animals of groups G300 (P= 0.02) and G1000 (P= 0.02) when compared with group G0 (Figure 1), but not when compared with groups G30 (P= 0.09) and G100 (P= 0.09, Figure 1).
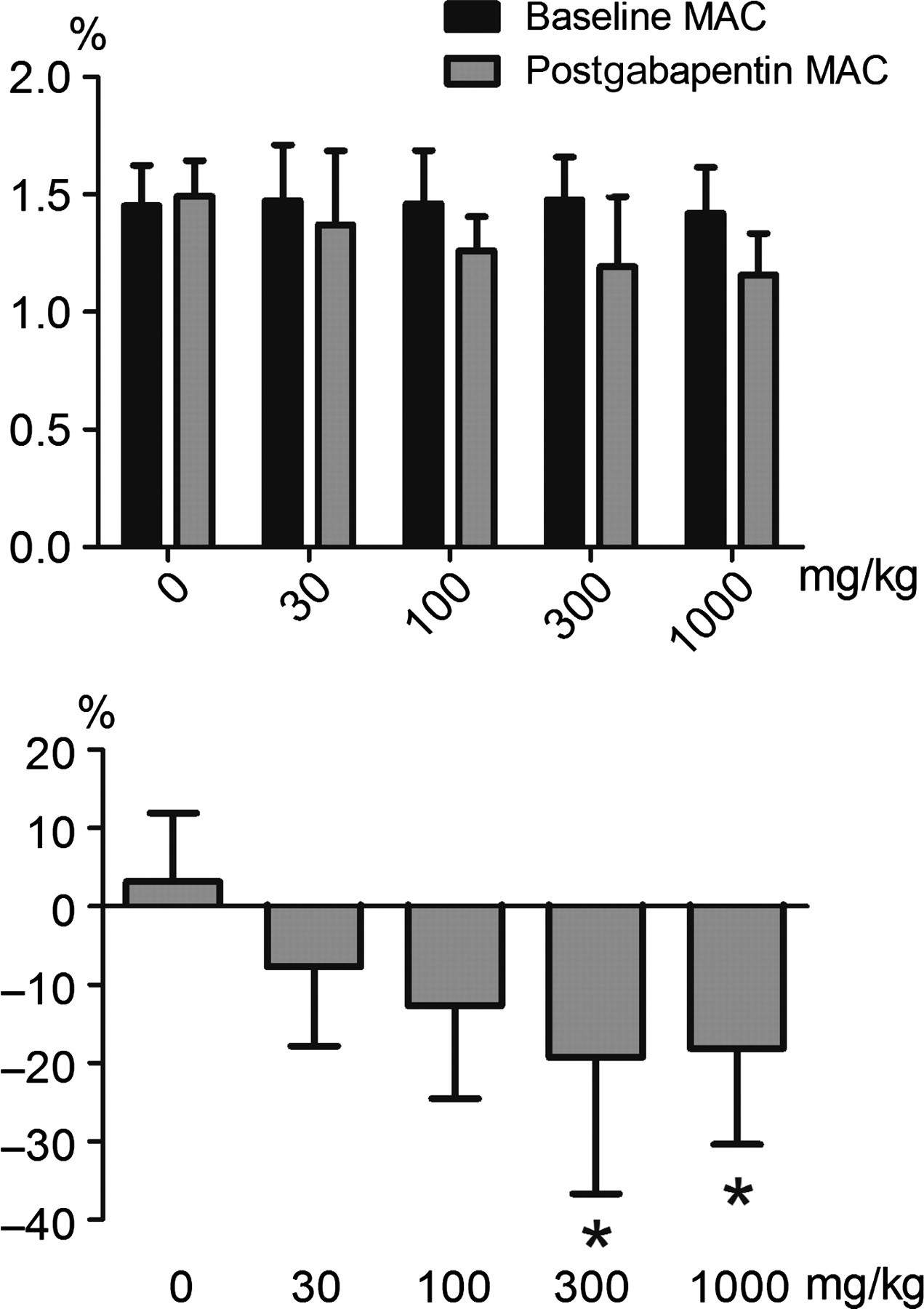
Effects of increasing dose of intraperitoneal gabapentin on isoflurane minimum alveolar concentration (MAC) in adult male rats. The dosing groups (mg/kg) were as follows: 0 (G0) (n= 6), 30 (G30) (n= 7), 100 (G100) (n= 6), 300 (G300) (n= 7) and 1000 (G1000) (n= 7). Upper panel: actual MAC values before and after gabapentin administration. Lower panel: the percent change in MAC from baseline values. Values reported are means ± standard deviations. Lower panel: *P< 0.05, different from the control group (0 mg/kg)
Arterial acid–base and blood gas data, and cardiovascular parameters at baseline and following intraperitoneal saline and gabapentin (30–1000 mg/kg) administration
Data are expressed as means ± standard deviations
*Significantly different from baseline value (P< 0.01)
**Significantly different from baseline value (P< 0.001)
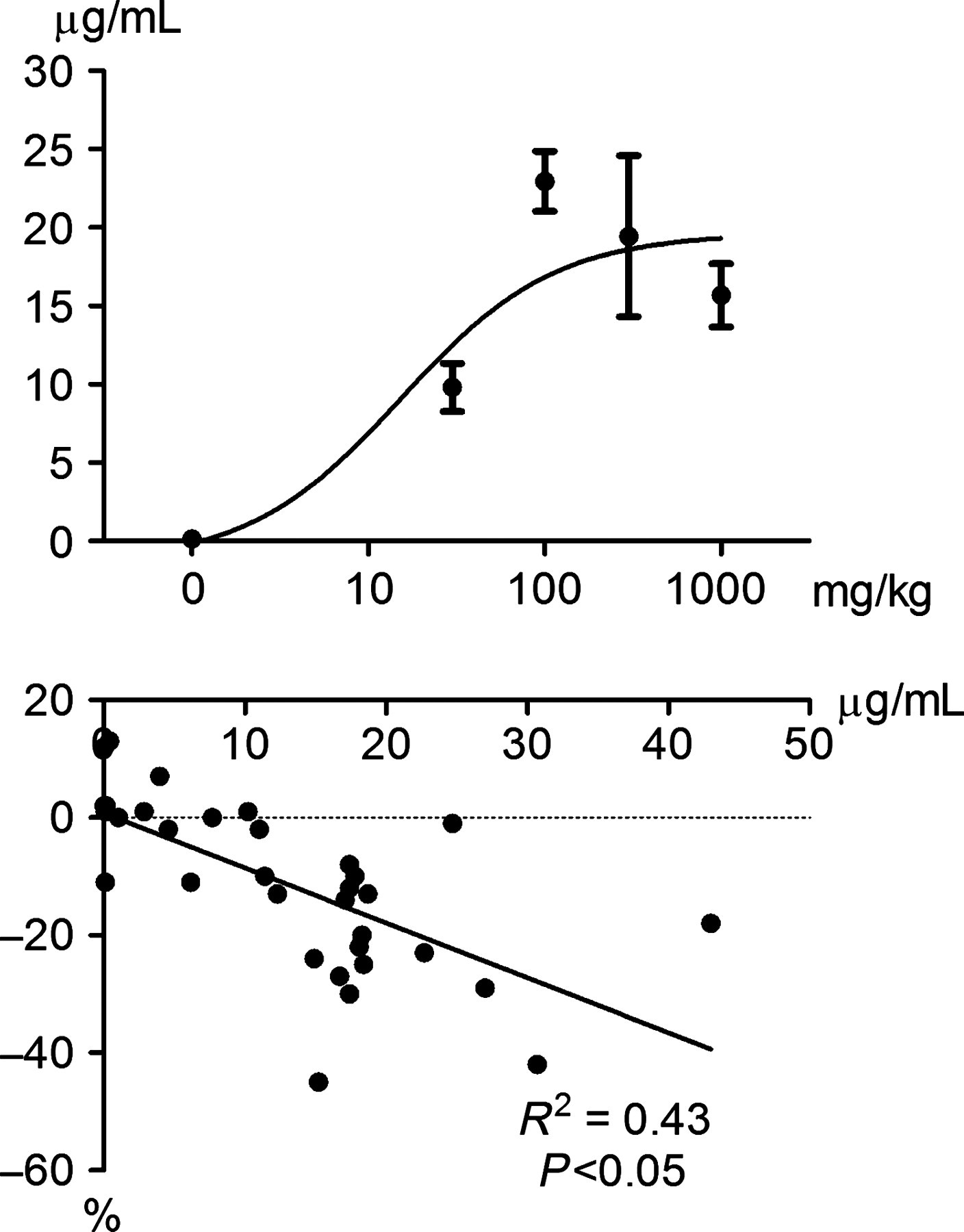
Relationship between increasing doses of intraperitoneal administration of gabapentin and blood concentrations of gabapentin (upper panel) and linear regression analysis of blood concentrations of gabapentin versus the corresponding percent change in MAC of isoflurane (lower panel) in adult male rats. Values reported are means ± standard error. A significant moderate negative correlation was present for the amount of gabapentin in blood and the magnitude of the change in isoflurane MAC from baseline values. MAC: minimum alveolar concentration
Discussion
In this study, we investigated the effect of gabapentin on isoflurane MAC in adult male rats and found that the percent change in MAC from baseline values after 300 (G300) and 1000 (G1000) mg/kg gabapentin was significantly different from the saline control group (G0) and that there was a negative correlation between gabapentin concentrations in the blood and the corresponding magnitude of MAC reduction. However, given the relatively small effect of gabapentin on isoflurane MAC (∼20% reduction from corresponding baseline values) and the lack of a dose-dependency of this effect, the clinical significance of this observation is questionable.
The range of gabapentin concentrations in blood was large for any given dose of gabapentin. This variability was probably due to the route of administration; the intraperitoneal technique has been reported to have a failure rate in the order of 10–20% due to variable uptake from the site of injection. 16 As such, it is not surprising that there was not a clear dose-dependent relationship between intraperitoneal gabapentin and measured blood gabapentin concentrations. This finding may also explain why we did not observe a steady decrease in isoflurane MAC with increasing dose of gabapentin. The lack of a clear dose–effect relationship despite measuring blood gabapentin concentrations within or above the range of plasma concentrations commonly associated with antinociceptive and other pharmacodynamic actions of the drug 17–19 somewhat challenges the interpretation of the MAC reduction as a true pharmacological effect. 20 However, when we performed a linear regression analysis using blood concentrations of gabapentin instead of gabapentin dose, we found a significant moderate negative correlation exists between gabapentin blood concentration and the magnitude of MAC reduction.
In a recent study in cats, 10 the investigators also did not observe a dose-dependent effect of gabapentin on isoflurane MAC nor a relationship between plasma concentration and MAC reduction. In that study, gabapentin was administered intravenously, which one would assume results in less variability in blood concentrations of the compound. While their dose range was much lower than ours, the peak plasma concentrations of gabapentin (20 ± 4 μg/mL [mean ± SD]) achieved with their technique of target-controlled drug infusion were close to the average peak blood concentrations measured in our study (21 ± 14 μg/mL). Only in five animals of our study (3 receiving a dose of 100 mg/kg intraperitoneally and 2 a dose of 300 mg/kg intraperitoneally) the blood gabapentin concentrations were >20 μg/mL. Therefore, it remains speculative at this point whether the authors of the feline study would have observed a similar significant, albeit small, effect on isoflurane MAC if they had extended the dose higher.
Administration of gabapentin had no significant effect on arterial blood gas parameters (PaCO2, PaO2). Changes in acid–base parameters (pH, lactate) were not statistically significant except in the postgabapentin samples for pH in groups G300 and G1000. Both variables remained within the normal physiological range, so these effects are not clinically significant. In a previous study, gabapentin administered at doses of 10, 30, 100 and 300 mg/kg intraperitoneally to Sprague–Dawley rats had no effect on BP or heart rate postadministration. 21
A possible explanation for the lack of any clinically relevant effect of gabapentin on isoflurane MAC might be that gabapentin is primarily an analgesic for persistent inflammatory and/or neuropathic pain associated with hyperalgesia 5 and not against acute noxious stimuli such as a tail clamp as was applied in our experiments. Human studies investigating the reduction of perioperative pain or opioid consumption upon the administration of gabapentin have been inconsistent. For example, a human study evaluating the effect of oral gabapentin alone or in combination with morphine, found it to be ineffective as a sole agent in preventing pain in healthy volunteers subjected to a cold pressor test; only in combination with morphine was gabapentin effective in alleviating acute pain. 18 Another study evaluated the use of gabapentin pre- and postoperatively in patients undergoing total hip arthroplasty in conjunction with a multimodal analgesia regimen and found that it did not decrease subsequent opioid consumption or pain scores. 22 Furthermore, based on clinical investigations in humans it has been concluded that a straightforward dose–response relationship is not apparent from those trials and that in cases of persistent and/or neuropathic pain gabapentin requires a slow titration to effect and hence it can take weeks for analgesia to develop. 23 In addition, a complex pharmacodynamic/pharmacokinetic relationship has been claimed to account for the non-linear relation between dose and effect found in some experimental pain models. 23 Thus, our experimental set-up was likely not suited to expose any antinociceptive potential of gabapentin given that the brief tail stimulus does not simulate a situation similar to persistent inflammatory and/or true neuropathic pain, and the drug was administered in each animal as a single dose only. In addition, this like any MAC study was not designed to test antinociceptive or analgesic properties of a drug per se, but rather the potential of a pharmacological agent to modify the anaesthetic potency of an inhalant agent employing suppression of motor activity in response to a supramaximum noxious stimulus as an observation endpoint.
In conclusion, the MAC-sparing effects of a single dose of intraperitoneal gabapentin are small and neither dose-related nor statistically significant until very large doses are administrated. Future studies will be necessary to further define its role in the perioperative setting, since the drug is potentially useful as an adjunct in multimodal pain therapy of subjects suffering from chronic or neuropathic pain.
Footnotes
ACKNOWLEDGEMENTS
We are particularly grateful to Dr Bernd Driessen, Department of Clinical Studies, The New Bolton Center, School of Veterinary Medicine, University of Pennsylvania for his support during data analysis and his extensive review of the manuscript. We would like to thank Sharon Martinez for her technical assistance in this study and Drs Larry Soma and Cornelius Uboh for the laboratory analysis of our blood samples at the Equine Toxicology and Research Center (West Chester University, West Chester, PA, USA). The funding for this experiment was provided by the Departmental Resource Research Committee, University of Pennsylvania, School of Veterinary Medicine.
