Abstract
This study aimed to estimate the reduction in the minimum alveolar concentration (MAC) of sevoflurane induced by low and high doses of methadone (5 and 10 mg/kg), tramadol (25 and 50 mg/kg), butorphanol (5 and 10 mg/kg) or morphine (5 and 10 mg/kg) in the rat. A control group received normal saline. Sixty-three adult male Sprague-Dawley rats were anaesthetized with sevoflurane (n = 7 per group). Sevoflurane MAC was then determined before and after intraperitoneal administration of the opioids or saline. The duration of the sevoflurane MAC reduction and basic cardiovascular and respiratory measurements were also recorded. The baseline MAC was 2.5 (0.3) vol%. Methadone, tramadol and morphine reduced the sevoflurane MAC (low dose: 31 ± 10, 38 ± 15 and 30 ± 13% respectively; high dose: 100 ± 0, 83 ± 17 and 77 ± 25%, respectively) in a dose-dependent manner. The low and high doses of butorphanol reduced the sevoflurane MAC to a similar extent (33 ± 7 and 31 ± 4%, low and high doses, respectively). Two rats developed apnoea following administration of high-dose butorphanol and methadone. These anaesthetic-sparing effects are clinically relevant and may reduce the adverse effects associated with higher doses of inhalational anaesthetics.
Keywords
Laboratory animals should be administered perioperative analgesics not only for ethical concerns, but also to minimize the impact of pain on physiological parameters and consequently on research results. Opioids reduce the minimum alveolar concentration (MAC) of inhalational anaesthetics, and thus, may reduce undesirable dose-dependent side-effects, such as cardiorespiratory depression. 1,2
Morphine, buprenorphine, fentanyl and remifentanil have been shown to decrease isoflurane MAC in rats,
3,4
but there are no reports on the effects of methadone, tramadol and butorphanol on the MAC of other inhalational anaesthetics. This disparity may be because the latter anaesthetics are less commonly administered to rats. Methadone, a mu (μ)-receptor agonist, differs from classical opioids such as morphine in that it also acts as a non-competitive antagonist of the N-methyl-
The MAC of an inhalational anaesthetic is the concentration required to prevent gross purposeful movement in 50% of subjects in response to a noxious stimulus. This parameter, which is expressed as a percentage at sea-level barometric pressure (760 mmHg), 20 has become the standard measure of volatile anaesthetic potency. In most animal studies, the MAC is estimated in each individual; therefore, the MAC is the average of the partial pressures that just prevented and just permitted movement in response to a supramaximal noxious stimulus. Opioids typically decrease volatile anaesthetic MAC although there are marked species and drug differences. 3,4,9,21,22 The effects of morphine on the MAC of isoflurane are well established in rats; 3,23,24 however, there is scarce or no information on the effects of tramadol, 19 butorphanol and methadone on inhalational anaesthetic MAC in rats. Furthermore, the effects of any of these opioids on the MAC of sevoflurane have not been determined. Therefore, we aimed to determine the effect of these opioids on the sevoflurane MAC in rats.
Materials and methods
The reduction of the sevoflurane MAC in response to the intraperitoneal administration of methadone, tramadol, butorphanol, morphine or saline was evaluated in rats. The study was approved by the Institutional Animal Care and Use Committee (La Paz University Hospital, Madrid, Spain). All experiments were performed during the light period (starting at 09:00 h).
Animals
We used 63 adult, male Sprague-Dawley rats (institutional breeding colony originally purchased from Charles River Laboratories, Barcelona, Spain) weighing 319 ± 14 g. The animals were housed in groups of six per cage (Makrolon type IV) and kept on a 12:12 h light–dark cycle (lights on between 08:00 and 20:00 h) at 22 ± 2°C and 40–70% relative humidity. Food (A03 maintenance diet for rodents, Scientific Animal Food & Engineering, SAFE, Panlab, Barcelona, Spain) and water were available ad libitum.
Procedure
Rats were placed in a methacrylate anaesthetic induction chamber (25 × 15 × 15 cm) into which sevoflurane (8 vol%, Sevorane, Abbott Laboratories, Madrid, Spain) was delivered in oxygen (3 L/min) (Sevoflurane Vaporizer, Sevorane Dragër Vapor 2000, Lubeck, Germany). Endotracheal intubation was performed using a 14-gauge polyethylene catheter (Terumo Surflo IV Catheter, Terumo Europe NV, Leuven, Belgium) with the animal positioned in sternal recumbency. A flexible, blunt-tip wire guide was inserted into the trachea with the aid of an otoscope, and this was used to guide the endotracheal tube into the trachea. After the correct position of the catheter was ascertained–by determining gas movement through the catheter with gauze threads and exhaled CO2 with a capnograph–a small T-piece breathing system with minimum dead space was connected. Approximately 2–3 min later, inspired sevoflurane concentration was reduced to 3–5 vol% and the oxygen flow was decreased to 0.75–1.0 L/min.
Monitoring
The left carotid artery was catheterized (Venocath-18, Venisystems, Abbott, Sligo, Ireland) via surgical cut-down, and this catheter was used for invasive arterial blood pressure measurement, via a calibrated pressure transducer (Transpac IV, Abbott Laboratories, Abbott Park, IL, USA) and arterial blood sampling. Systolic, diastolic and mean arterial blood pressures, heart and respiratory rates, and arterial oxygen haemoglobin saturation (via pulse oximetry) were continuously measured (RGB Medical Devices, Madrid, Spain). End-tidal CO2 was monitored continuously (Capnomac Ultima, 5330 Agent Monitor, Datex-Ohmeda, Hertfordshire, UK) and recorded. The effects of the drugs and drug dosages on blood pressure and heart and respiratory rates were recorded before (1 min) tail clamping at each step to determine the MAC value. Rectal temperature was monitored and maintained between 37°C and 38°C by means of a water-circulating warming blanket (Heat Therapy Pump, Model TP-220, Gaymar, Orchard Park, NY, USA) and a standard (60 W) heating light.
Determination of the MAC
Intratracheal gas sampling was used to measure anaesthetic gas concentration for the determination of the MAC. This method was described in detail previously. 25 In brief, a fine catheter with a 0.9 mm external diameter was inserted through the endotracheal catheter, with the tip located at the level of the carina. The proximal end of the catheter was attached to a 10 mL gas-tight glass syringe (Hamilton Syringe, 1000 Series Gastight, model 26211-U, Sigma-Aldrich, St Quentin Fallavier, France). Samples were obtained by withdrawing 10 mL of gas over 5 min by using an infusion pump (model 55–2226, Harvard Apparatus, Millis, MA, USA). The fine catheter was withdrawn between samples. Before the first MAC determination, the end-tidal anaesthetic concentration was adjusted to 2.4 vol%, a value close to the MAC previously reported in Wistar rats. 26 After every step change in the end-tidal concentration of sevoflurane, at least 10 min was allowed for equilibration before obtaining gas samples and applying the noxious stimulus. Gas samples were assayed using a side-stream infrared analyser (Capnomac Ultima, 5330 Agent Monitor, Datex-Ohmeda, Hertfordshire, UK).
The MAC was established as described previously. 20,26,27 A noxious stimulus was applied with a long haemostat (8-inch Rochester Dean Hemostatic Forceps, Martin, Tutlingen, Germany) clamped to the first ratchet lock on the tail for 60 s immediately after the gas sample was obtained from the trachea. The initial stimulus point was 6 cm distal to the tail base and subsequent stimuli were administered 1 cm proximal to a previous test site when the response was negative or 1 cm distal to that point if the response was positive. 28 A positive response was considered as gross purposeful movement of the head, extremities and/or body or a clear movement of the spine. A negative response was considered as either a lack of movement or discreet reactions: grimacing, swallowing, chewing or tail flick. When a negative response occurred, the end-tidal sevoflurane concentration was reduced by 0.2 vol% decrements until the negative response became positive. When a positive response occurred, the sevoflurane concentration was increased by 0.2 vol% until the response became negative. The MAC was considered as the concentration mid-way between the highest concentration that permitted movement in response to the stimulus and the lowest concentration that prevented such movement. Determination of the MAC was performed at an altitude of 650 m and MAC values were corrected to 760 mmHg. 29
Experimental design
Animals were randomly assigned to one of nine groups (n = 7 per group; see below). Once the animals were anaesthetized and instrumented, a baseline sevoflurane MAC was determined, and each animal acted as its own control. Since the initial (2.4%) end-tidal concentration was close to the sevoflurane MAC in Wistar rats,
30
usually only a few step changes in sevoflurane concentration were necessary. Each rat was randomly administered one of the test drugs or saline intraperitoneally and the MAC was re-determined. After opioid administration, the end-tidal sevoflurane concentration was reduced based on the expected MAC sparing effect of the drug in rats,
3,19
dogs
11,16,31–33
and cats.
34
The MAC value was re-determined until the baseline value was regained. Sixty minutes were usually required to determine MAC and 30 min was allowed between MAC determinations. MAC reduction time was considered the total time, in minutes, from the administration of the opioid or saline (time zero) until the MAC regained baseline values. Maximum MAC reduction time was considered the time, in minutes, at which the maximum MAC reduction was determined. Rats were euthanized with an overdose of potassium chloride given intravenously while still deeply anaesthetized at the end of the experiment. A schematic of the experimental design is shown in Figure 1.
Experimental design (see Materials and methods for details). MAC: minimum alveolar concentration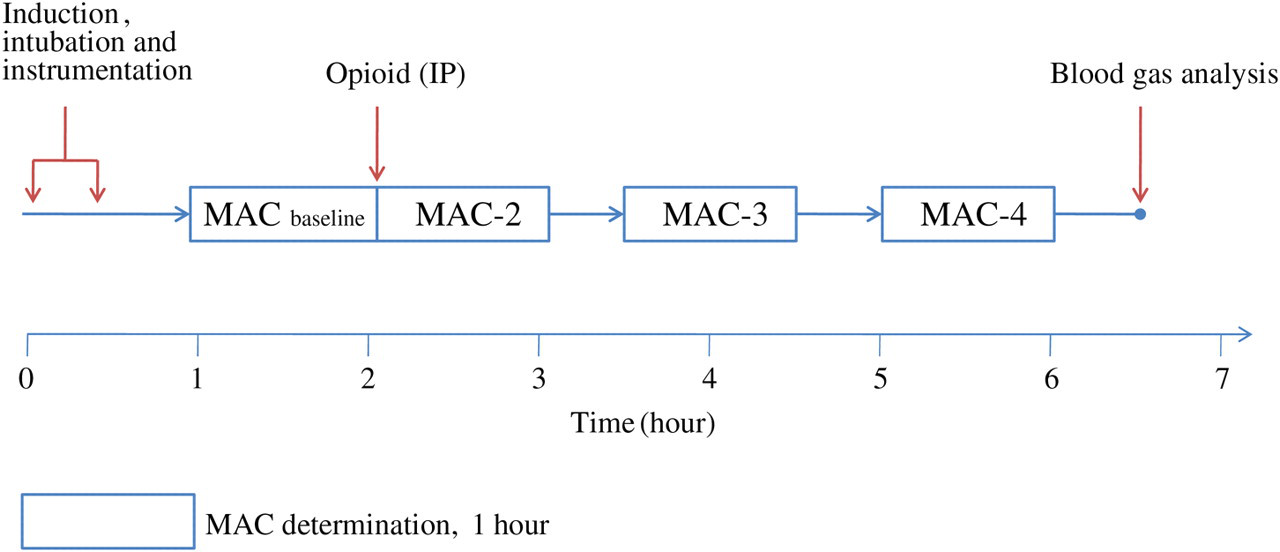
Drug groups
Four opioid drugs were screened in this study: methadone (Metasedin, Esteve, Barcelona, Spain), butorphanol (Torbugesic, Fort Dodge, Madrid, Spain), tramadol (Adolonta, Grünenthal Pharma, Madrid, Spain) and morphine (Morfina, Braun, Barcelona, Spain). Two doses of each opioid, low and high, were selected based on the clinical dose range for the rat, 3,8,35,36 although a pilot study was also conducted in order to select the doses finally employed, taking into consideration the extrapolation of the dose range in other species such as the dog. 37 This study included nine experimental groups; a normal saline-treated control group and eight treatment groups to consider the four drugs at two dose levels: morphine 5 and 10 mg/kg, methadone 5 and 10 mg/kg, tramadol 25 and 50 mg/kg, and butorphanol 5 and 10 mg/kg. Drugs were prepared in 0.9% saline and a total volume of 1 mL was administered intraperitoneally to each rat.
Statistical analysis
A value of seven animals per group was considered adequate based on previous data from similar studies in our laboratory and from calculation of the n value (N query advisor 2.0. Statistical solutions, Saugus, MA, USA). 26 An analysis of variance (ANOVA) for repeated measures was conducted (drug dose × time) by comparing the percentage reduction in the MAC (baseline = 100%). To compare differences between drug treatments at the two defined study times, a one-way ANOVA was employed. The Bonferroni test was used to compare differences among groups. These tests were also used to compare cardiorespiratory data. A 95% confidence interval was established and a P value of <0.05 was set to indicate statistical significance. Results are presented as mean ± standard deviation. The SPSS software version 15.0 (SAS Institute, Cary, NC, USA) for Windows was employed.
Results
Reduction of the MAC of sevoflurane by opioids
The mean baseline MAC determined in all rats (n = 63) was 2.3 ± 0.3 vol% with no differences among groups or doses (P > 0.05). Methadone, tramadol, butorphanol, and morphine reduced the MAC (P < 0.05), although the magnitude of the reduction depended on the drug and dose. A dose-dependent effect on the MAC was observed for all drugs studied, except for butorphanol.
Overall, the decrease in MAC values produced by the low doses of the four studied opioids, but also by the high dose of butorphanol, were similar and no significant differences were determined among them (mean MAC reduction of the 5 groups 33 ± 3%) and significantly less than those obtained with high doses of methadone and tramadol (MAC reduction of 100 ± 0% and 83 ± 17%, respectively). Morphine at a high dose reduced MAC by 77 ± 25% (Table 1, Figure 2).
Maximum reduction in the sevoflurane minimum alveolar concentration (MAC) produced by morphine, methadone, tramadol or butorphanol given at two doses intraperitoneally in the rat (n = 7 per group). Data are expressed as mean ± SD. *Statistically different from saline (P < 0.05). Baseline and postdrug minimum alveolar concentration (MAC) of sevoflurane produced by the opioids morphine, methadone, tramadol or butorphanol given at two doses intraperitoneally in the rat Data are expressed as mean ± SD. The n value is 7 per group (see Materials and methods for details)
†Statistically different from saline (P < 0.05)
‡Statistically different from the low-dose group for the same drug (morphine, methadone, tramadol and butorphanol; P < 0.05)
¶Statistically different from all other groups (P < 0.05)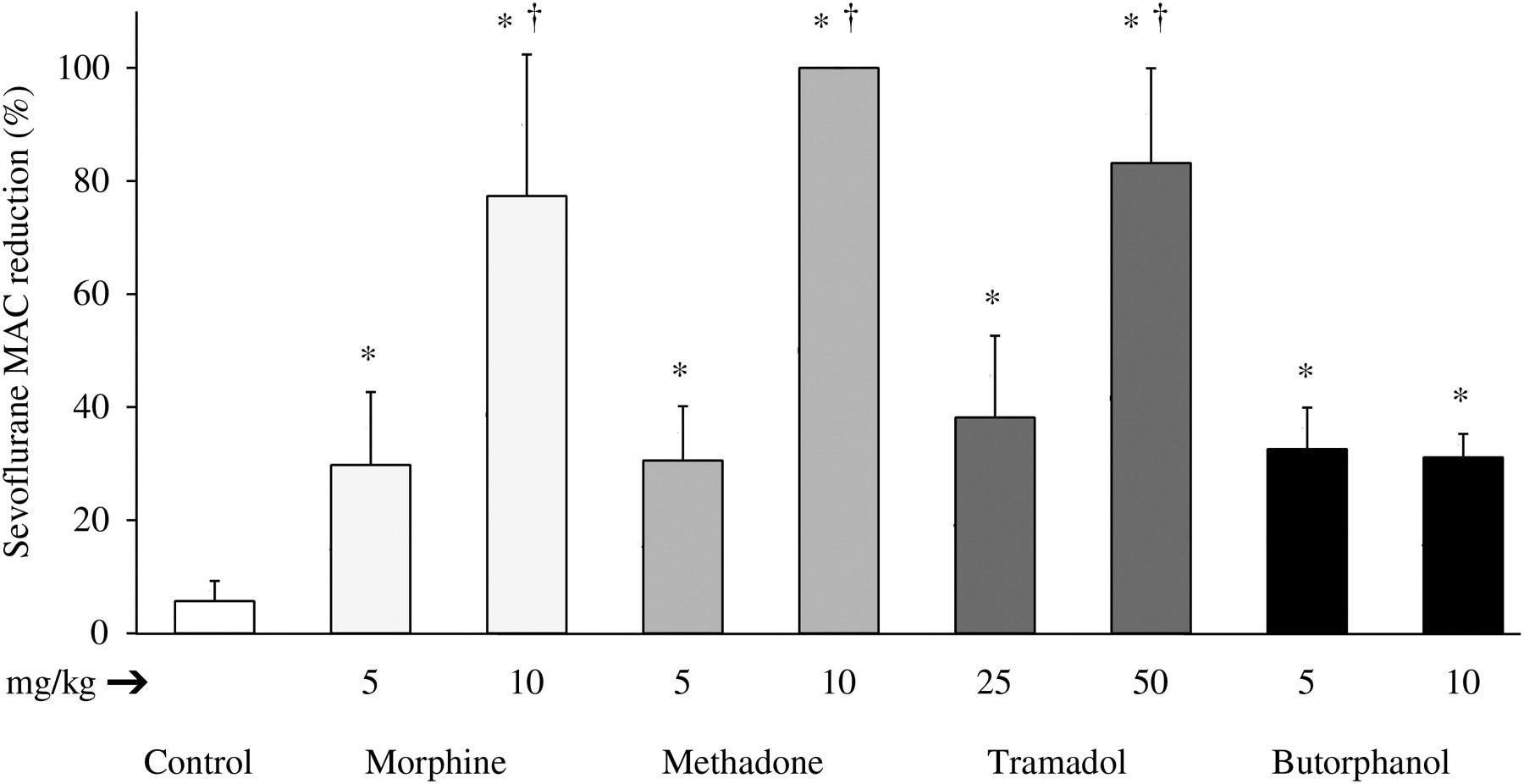
Duration of MAC reduction
The durations of the MAC reduction produced by methadone, butorphanol or morphine at both doses and tramadol at the low dose were not significantly different, whereas tramadol at the high dose produced a longer-lasting MAC reduction (P < 0.05). However, the maximum MAC reduction time was similar for all drugs and doses (Table 1). Overall, each experiment lasted between 4 and 6 h.
Cardiorespiratory effects
Major cardiovascular, ventilatory and oxygenation values before and after (30 min) opioid administration during isoflurane anaesthesia
Discussion
In the present study, methadone, tramadol, morphine and butorphanol significantly reduced the sevoflurane MAC in rats. Morphine (10 mg/kg) reduced the MAC of sevoflurane by 77%, which is greater than the reported decrease of 50% in isoflurane MAC in Wistar rats, 3 and the decrease of 55% in the MAC of halothane when using a morphine dose of 8 mg/kg. 38 A greater morphine dose (20 mg/kg) reduced the halothane MAC by 84%. 38 The sevoflurane MAC reduction of 30% with a morphine dose of 5 mg/kg is comparable with the decrease in the MAC of isoflurane with a similar dose of morphine. 3 Differences among study results may be due to the strains of rats studied 39 and differences in methodology.
The reductions in sevoflurane MAC produced by opioids in the rat cannot be extrapolated directly to other species because clinically relevant differences in the MAC reduction with opioids have been reported among species. For example, morphine (2 mg/kg) decreased the isoflurane MAC by approximately 50% in monkeys and dogs but by only 13% in pigs. 40
Methadone provided a 100% reduction in the MAC, a degree of reduction rarely observed with opioids in rats. Recent reports on the MAC sparing action of intravenous methadone in cats showed a much lower (25%) sevoflurane MAC reduction (0.3 mg/kg) 34 and up to 48% isoflurane MAC reduction in dogs (0.5–1.0 mg/kg) in a dose-dependent manner. 33 The reason for this species-specific difference is unclear and may include non-opioid actions of methadone such as NMDA antagonism 41 or differential effects on opioid receptors, which might facilitate opioid-induced tolerance. 37 It should also be remembered that MAC uses tail clamping as the nociceptive stimulus and this level of anaesthesia (1× MAC) does not provide surgical anaesthesia. 42 Therefore, although a 100% reduction in the MAC was observed, additional doses of inhalant anaesthetics should be administered in order to provide an appropriate level of surgical anaesthesia. The effect of tramadol on MAC reduction in rats may be dose dependent. A low dose of tramadol (10 mg/kg intravenously) decreased the MAC of isoflurane by 13, 19 and in the present study, tramadol at 25 and 50 mg/kg, administered intraperitoneally, decreased the sevoflurane MAC by 38% and 83%, respectively. In dogs, tramadol decreased the MAC of sevoflurane by 26% and 36% at doses of 1.3 and 2.6 mg/kg/h, respectively; however, there was no significant difference in effect between the doses. 16 The MAC reduction with tramadol in dogs is much less than what was determined in the present study; however, it is not possible to say if this indicates a species difference as the tramadol doses used were considerably greater in the rat study.
A dose-dependent effect in terms of MAC reduction was observed with morphine, methadone and tramadol. A similar effect has been previously determined in rats with morphine, 3 fentanyl, 4 remifentanil, 4,30 sufentanil 43 and alfentanil, 38 as well as with the partial agonist buprenorphine, 3 with high doses of these drugs reducing the MAC between 50% and 90%. Despite differences in the dose employed, which may be related with differences in drug potency, a constant infusion rate of the short-acting opioids remifentanil and fentanyl reduced the isoflurane MAC by up to 60% (240 and 60 μg/kg/h, respectively) 4 and sufentanil reduced the MAC of isoflurane by 90% (60 μg/kg/h). 43
Butorphanol produced a relatively small decrease in MAC, with a maximum reduction of 30%; thus, doses greater than 5 mg/kg would provide no further decrease in the MAC of sevoflurane in rats. A ceiling analgesic effect has been described with opioids with agonist–antagonist receptor properties. However, butorphanol exhibits both μ and κ agonist actions depending on animal species and experimental conditions used. 44,45 In the mouse butorphanol may act as a partial agonist at μ receptors to produce its antinociceptive effects, 45 and this may explain its reduced effects on MAC.
Differences in the response to butorphanol have been observed between different species 10,11,46,47 but also within the same species. 9,11 In psittacine birds, butorphanol reduced the effective dose (ED50) of isoflurane by 25%. 48 In cats, a 15–19% MAC reduction of isoflurane was determined at doses ranging from 0.08 to 0.8 mg/kg intramuscularly. 12,49 The effects of butorphanol on MAC are variable within the same species. In dogs, doses of 0.1, 0.2 and 0.4 mg/kg reduced the MAC by 11%, 50 15% 49 and 20%, 11 respectively. However, a lack of MAC reduction has been determined in dogs at butorphanol doses between 0.2 and 0.8 mg/kg intravenously. 9,51 In ponies, a 10% MAC reduction in halothane has been determined with 0.022 and 0.044 mg/kg butorphanol, 52 but only a 3% MAC reduction was determined with 0.05 mg/kg butorphanol in another study. 10 Also the nociceptive stimulus may account for differences in butorphanol antinociceptive efficacy. Butorphanol produced maximum analgesia in the mouse abdominal stretch test 53 and in the warm water tail-withdrawal test, 44,54 and less-than-maximal analgesia was obtained with the radiant-heat tail-flick test in mice, whereas in other studies with assays in which efficacy requirements are high butorphanol is only partially or ineffective in producing analgesia. 45
The MAC method provides reliable and quantifiable data related to anaesthetic potency; nevertheless, the MAC value varies by as much as 20% within a given species but generally by <10% within the same individual animal. 27 However, the reduction in MAC produced by analgesics does not necessarily reflect the actual analgesic potency of the drug 55 since the observed effect on MAC is not simply the result of the antinociceptive effects of the tested drugs, but rather the interactions of the combination between the drug and the inhalational anaesthetic. Nevertheless, the MAC method is regarded as a clinically useful reference because it mimics the intraoperative period. The MAC reduction produced by different drugs, including analgesics such as opioids, allows an estimation of inhalant anaesthetic requirements for surgical procedures and thus an increase in the safety margin derived from reduced anaesthetic doses. 56 Unfortunately, by using the MAC methodology, it is not possible to accurately determine short-lived effects (minutes), because at least one hour is usually necessary to determine the MAC value.
Respiratory depression and apnoea are common side-effects of opioid administration to rats and have been observed after the administration of high doses of buprenorphine, morphine, fentanyl or remifentanil. 3,4 In the present study, the high dose of methadone and butorphanol induced a brief period of apnoea in one rat in each group. This is a limiting factor on the use of these drugs at these doses. Methadone (15 mg/kg) produces respiratory depression in rats resulting in hypoxaemia and respiratory acidosis. 57
No clinically significant hypotension was observed in the present study after drug administration; however, hypotension is a common adverse effect of opioids, 58 especially when high plasma concentrations are quickly achieved, such as following intravenous high dose administration of buprenorphine or remifentanil. 3,4 A transient decrease in blood pressure of 40–45% occurred with morphine (1–10 mg/kg) and a 20–25% decrease with buprenorphine (10–100 μg/kg) after intravenous administration. 3 Hypotension was also observed after intravenous injection of 4.4 mg/kg tramadol in rabbits anaesthetized with isoflurane 18 but not in dogs anaesthetized with sevoflurane (1.3 × MAC), 59 which is consistent with the findings in the present study where no clinically significant hypotension was observed. A potential explanation is that the opioids given in the present study were administered intraperitoneally, whereas previous studies employed the intravenous route. Although intraperitoneally given drugs are rapidly absorbed, the intravenous route results in greater initial plasma concentrations of the opioid and produces an immediate effect. 60
In conclusion, methadone, tramadol, morphine and butorphanol decreased the MAC of sevoflurane in the rat and, with the exception of butorphanol, the decrease in MAC was dose dependent. The anaesthetic sparing effects of opioids should provide an improved safety margin derived from the lower inhalational anaesthetic doses they require. Perioperative opioid use should be considered together with other analgesics such as non-steroidal anti-inflammatory drugs as part of a balanced anaesthesia as is commonly employed in the human or veterinary clinical setting.
Footnotes
ACKNOWLEDGEMENT
This study was supported by Project FIS 08/0422 from Fondo de Investigaciones Sanitarias, Spanish Health Ministry.
