Abstract
Gastritis is a commonly diagnosed condition in non-human primates used in biomedical research. As in humans, Helicobacter pylori infection may cause gastritis. The following report presents a method of non-invasive detection and a successful treatment protocol for this common pathogen.
Gastritis and gastric ulcerations in non-human primates frequently present as an incidental finding at necropsy and are normally not associated with major clinical signs. 1 In cases of severe gastritis, animals may show anorexia, weight loss, abdominal pain, bloating, vomiting or regurgitation, melena, hematemesis, ascites, dehydration, tachypnea, tachycardia, pale mucous membranes and/or lethargy.1,2 As in humans, gastric disease can be associated with Helicobacter pylori,1,2 and due to a similar anatomy and pathophysiology rhesus macaques may represent a valuable animal model. 3
In vivo diagnostic tests for Helicobacter spp. in non-human primates typically involve gastric endoscopy and biopsy. The biopsy specimens are then submitted for histology, rapid urease testing, polymerase chain reaction or culture.1,2 For humans, non-invasive diagnostic tests such as quantitative and qualitative antibody testing, urea breath test and fecal antigen tests (FATs) are available.4,5 Amongst these tests, FATs are considered to be suitable for children and handicapped individuals since they do not require the patient’s cooperation, and the use of monoclonal FATs is clinically accepted. 5
Here we report a case of an 8-year-old, male rhesus monkey showing weight loss, paleness, persistent alopecia, deteriorated general appearance and a distinct sour foetor ex ore. Apart from severe hypophosphatemia (0.291 mmol/L [0.872–1.453 mmol/L] 6 ), hematology and serum chemistry values (white blood count [WBC], red blood count [RBC], hemoglobin [HGB], hematocrit [HCT], platelet [PLT], mean corpuscular volume [MCV], mean corpuscular haemoglobin [MCH], mean corpuscular hemoglobin concentration [MCHC], albumin [ALB], alkaline phosphatase [ALKP], alanine aminotransferase [ALT], amylase [AMYL], urea nitrogen [BUN], cholesterol [CHOL], creatinine [CREA], globulin [GLOB], glucose [GLU], total bilirubin [TBIL], total protein [TP], calcium [Ca], chloride [Cl], potassium [K], magnesium [Mg], sodium [Na]) were normal.
Based on the clinical symptoms, a case of severe gastritis was suspected. Hence a proton pump inhibitor (esomeprazole [Nexium™10 mg; AstraZeneca, Wedel, Germany], 0.4 mg/kg twice a day) was used for symptomatic treatment, and a monoclonal FAT (RIDASCREEN™ FemtoLab H. Pylori; R-Biopharm AG, Darmstadt, Germany) was performed. The enzyme-linked immunoassay was done in an accredited laboratory (Institute of Medical Microbiology and Hygiene, Eberhard Karls Universität Tübingen, 72076 Tübingen, Germany) according to the manufacturer’s instructions.
7
Upon receipt of a positive test result, the treatment was extended to a 10-day oral clarithromycin-based triple therapy (esomeprazole, amoxicillin and clarithromycin [the latter two from Hexal AG, Holzkirchen, Germany], 0.4, 10 and 10 mg/kg respectively, twice a day). After the 10-day treatment and in the subsequent weeks, the monkey’s average body weight increased by more than 15% (30 weeks before treatment: Weight change before and after triple therapy and results of consecutive fecal antigen tests (FATs).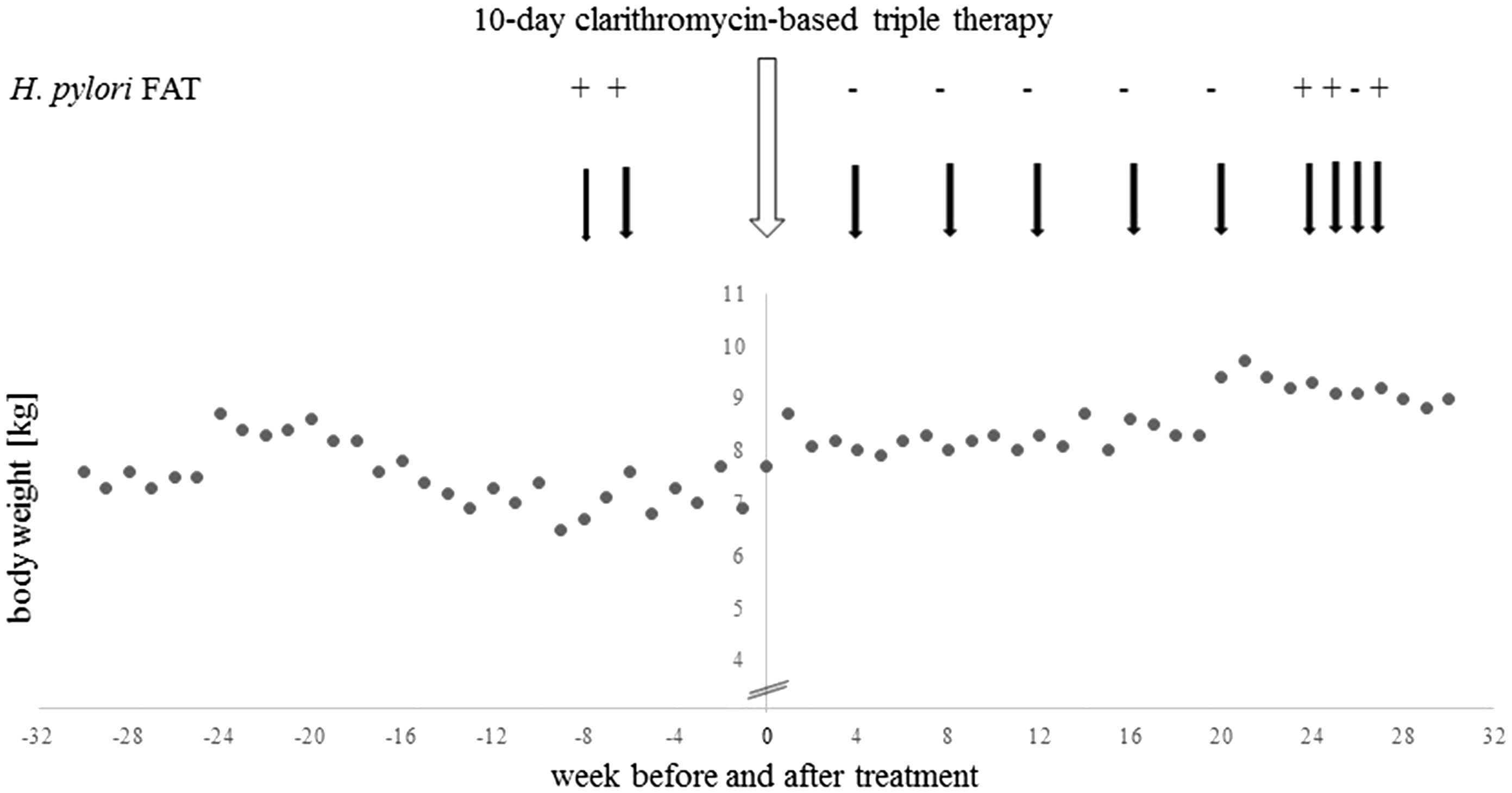
To examine the effectiveness of the treatment, fecal samples were collected and tested at four weeks 5 and again at 8, 12, 16, 20 and 24 weeks after the end of the treatment. Four weeks after the end of the antibiotic treatment, the animal was diagnosed to be negative for H. pylori by FAT and remained negative for the subsequent 20 weeks. At 24 weeks the FAT was again positive and the result confirmed by retesting one and three weeks later (Figure 1). However, the animal’s body weight remained stable and none of the clinical signs observed before treatment developed again. To monitor the status of other animals in the facility, more fecal samples were tested. An animal was considered to be positive for H. pylori if the result was positive twice 5 at a weekly interval, and negative if the result was negative three times at a weekly interval, since intermittent shedding, low amounts of antigen and inhomogeneous dispersal of the antigen in the sample could occur. All animals of the same origin, that arrived together as a group (n = 3), and two out of the five animals which were temporarily in direct or continuously in indirect (alternate group cage sharing) contact with the monkey were tested positive. As none of the monkeys tested positive displayed clinical signs of disease no antibiotic treatment was administered.
The case presented demonstrates that a clarithromycin-based triple therapy, at the dosage and duration used, can control H. pylori infection in a naturally infected rhesus macaque. Clarithromycin was not used in the facility prior to the present treatment, which may explain the efficiency of the drug combination, although the emergence of clarithromycin-resistant strains is of considerable concern in human medicine. 8 Neither of the symptoms observed before treatment nor any other clinical signs of H. pylori-associated gastritis1,2 appeared again. Whether a reinfection or a re-emergence of H. pylori had occurred remained unclear.
Comparison of diagnostic tests for Helicobacter pylori in non-human primates.
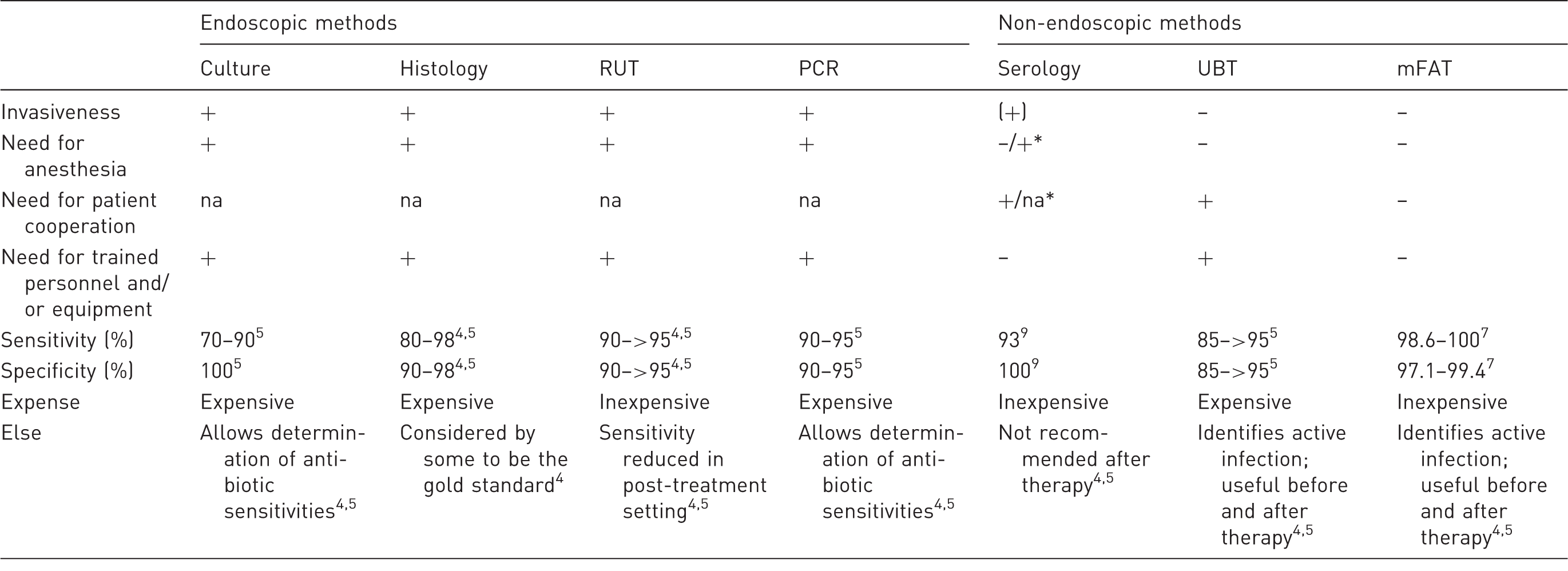
RUT: rapid urease testing; PCR: polymerase chain reaction; UBT: urea breath test; mFAT: monoclonal fecal antigen test; na: not applicable; *in untrained animals.
To apply the monoclonal FAT for routine Helicobacter diagnostic, further studies are required to test sensitivity and specificity as well as potential cross reactions with other new and old world monkey gastric and enterohepatic Helicobacter sp6,10. However, monoclonal FAT presents a promising, non-invasive, inexpensive and simple, though accurate, method for the veterinary clinician to monitor the infection status of H. pylori in primates in captivity.
Footnotes
Acknowledgements
We would like to thank M Venier, U Scheurlen, A Schäfer and the laboratory team for their contribution and support.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
