Abstract
The use of animals is essential in biomedical research. The laboratory environment where the animals are housed has a major impact on them throughout their lives and influences the outcome of animal experiments. Therefore, there has been an increased effort in the refinement of laboratory housing conditions which is explicitly reflected in international regulations and recommendations. Since housing conditions affect behaviour and brain function as well as well-being, the validation of an animal model or paradigm to study the brain and central nervous system disorders is not complete without an evaluation of its implication on animal welfare. Here we discuss several aspects of animal welfare, comparing groups of six rats living in the PhenoWorld (PhW), a recently developed and validated paradigm for studying rodent behaviour, with standard-housed animals (in cages of six rats or pair-housed). In this study we present new data on home-cage behaviour showing that PhW animals have a clearer circadian pattern of sleep and social interaction. We conclude that, by promoting good basic health and functioning, together with the performance of natural behaviours, and maintaining animals’ control over some of their environment but still keeping some physical and social challenges, the PhW stimulates positive affective states and higher motivation in rats, which might contribute to an increased welfare for animals living in the PhW.
Animal models play an important role in biomedical research, one of which is the investigation of psychiatric disorders. The laboratory environment and the housing conditions where animals are kept have a major impact on them throughout their lives and will influence the outcome of animal experiments.1,2 Laboratory rodents are usually housed in standard conditions where the limited spatial, structural and social environments can impose constraints on their behaviour and brain development. These contribute to poor ethological measurements and poor welfare, resulting in altered brain functions (see examples3–5) which are critical when studying the brain and diseases of the central nervous system.
In Europe, guidelines for the accommodation and care of laboratory animals are established in the European Directive 2010/63/EU (http://eur-lex.europa.eu/legal-content/EN/TXT/?uri=celex:32010L0063) and in the Commission Recommendation 2007/526/EC (http://eur-lex.europa.eu/legal-content/EN/TXT/?uri=CELEX:32007H0526), which indicate that ‘Animals should be socially housed wherever possible and provided with an adequately complex environment within the animal enclosure to enable them to carry out a range of normal behaviours’. With the development of laboratory animal science and the implementation of the 3 Rs (replacement, reduction and refinement) principles of Russell and Burch 6 in the European guidelines, the use of standard environmental enrichment, also called environmental refinement, is becoming increasingly common for both scientific and ethical reasons.7–10 For rodents, this environmental refinement consists not only of providing them with social partners,11,12 but also of introducing nesting material and/or other objects (e.g. tubes) inside the animal cage to stimulate them to perform species-specific behaviours which has arguably contributed to the increase of laboratory animal welfare.1,8,13,14 The effects of using refined housing on behaviour and brain functions should be specifically addressed on a case-by-case basis when developing new animal models, but evaluation of its welfare implications contributes to a better validation of laboratory rodent models and is good laboratory practice. 15
We have recently developed a new paradigm, the PhenoWorld (PhW), where a group of six rats is provided with a socially and physically enriched housing environment, characterized by a large space, access to running wheels and automated door systems to access food and water, as well as a behavioural testing system. 16 Based on Fraser’s view that ‘for actions to be widely accepted as achieving high animal welfare, in addition to being based on good animal welfare science, they will need to make a reasonable fit to the major value positions about what constitutes a good life for animals’; 17 we aimed to address animal welfare taking into account the three conception approaches described by Fraser, combining basic health and functioning, natural living and affective states of the animals to draw conclusions about their welfare in this study. In our previous publication, where the PhW was validated as a good paradigm for studying mood disorders, we have observed that control animals housed in the PhW presented reduced depressive-like behaviour, higher hedonism and reduced anxiety when compared with animals housed in standard conditions (in groups of six or pair-housed), 16 which are all indicators of a more positive affective state of animals housed in the PhW. We have also observed that the animals in the PhW are able to perform a series of species-specific behaviours, 16 which are not allowed in standard cages due to space restrictions and which can also contribute to the welfare of the animals. Herein we present data on the evaluation of sleep and social behaviours, in a focal sampling analysis of undisturbed animals in their home cages, to draw conclusions about rat welfare while housed in the PhW (in groups of six animals) compared with animals housed in standard housing conditions (in groups of six or pair-housed).
Materials and methods
Animals and housing conditions
Wistar Han IGS (Crl:WI(Han)) specific pathogen free (SPF) male rats (n = 6 per group, total of 18 animals), aged 7–8 weeks of age, were purchased from Charles River Laboratories (Saint-Germain-sur-l'Arbresle, France). The animals were kept in a quarantine room for one week and then transferred to a conventional housing room. The animals were then individually identified and randomly assigned to groups of six animals each and housed in three different conditions: group PhW were housed in the PhW (TSE Systems GmbH, Bad Homburg, Germany); group STD6 were housed in a standard filter-topped transparent cage (610 × 435 × 215 mm, 2065 cm2 floor area) (ref. 2000P; Tecniplast, Buguggiate, Italy); and group STD2 were pair-housed in a standard filter-topped transparent type III cage (425 × 266 × 185 mm, 800 cm2 floor area) (ref. 1291H; Tecniplast). The PhW group had an acclimatization period from living in a standard cage (quarantine conditions) to living in the automated system of the PhW as described in our first description of this system. 16 Briefly, the PhW set-up consists of an area of 1 m2 with a 50 cm height central cage, using corncob bedding on the floor. It is connected to a box with four running wheels by means of two open access tubes, and to two dinking/feeding boxes, each one accessible by means of an automatic AnimalGate (AG). All areas are covered by either perforated Plexiglas or stainless steel grids. In order to automatically register the data per animal, each AG and each running wheel is equipped with an RFID antenna/reader that recognizes each individual animal.
All animals were maintained under standard laboratory conditions: artificial 12 h light/dark cycle (lights on from 08:00 h to 20:00 h), with an ambient temperature of 21 ± 1℃ and a relative humidity of 50–60%; with corncob bedding (Scobis Due; Mucedola SRL, Settimo Milanese, Milan, Italy) and sterile cardboard tubes as housing refinement (autoclaved paper rolls). The rats were given a standard diet (4RF21; Mucedola SRL) and water ad libitum. Health monitoring was performed in compliance with the Federation of European Laboratory Animal Science Associations (FELASA) guidelines 18 according to the recommended SPF health status of sentinel animals maintained within the same room. The PhW central cage contained cardboard tubes with a base of a standard type III cage enabling the animals to climb or jump. During the weekly ‘cage change’ in the PhW system, which consisted of cleaning all areas used by the animals (central cage, running wheel box and drink/feed boxes) and changing the bedding of the central cage, the animals were moved to a standard cage for six animals (ref. 2000P; Tecniplast) for a short period of time.
Animals were handled from the beginning of the study by the same experimenter who performed all behavioural tests and who also changed their cages, including adding new cardboard tubes, once a week.
All experiments were performed according to the European Directive 2010/63/EU and the Portuguese regulations and laws on the protection of animals used for scientific purposes of the Ministry for Agriculture, Ocean, Environment and Spatial Planning, which authorized this study project (authorization 9458 of 2011-05-06). The present study was also evaluated and approved by the University of Minho ethics committee (process code SECVS 097/2013).
Animal individual identification
The skin in the middle dorsal area of the animal was the site of placement for the RFID transponder. This area was previously anaesthetized by a subcutaneous injection of 250–300 µL (100 µL per 100 g body weight) of 0.5% lidocaine (from 2% lidocaine solution; Braun Melsungen AG, Berlin, Germany) using a 25 G (25 mm long) needle. An RFID transponder, 12 mm long × 2.12 mm diameter, 0.09 g weight, covered with BioGlass 8625, and inserted in a 2.6 mm × 32 mm needle, was then subcutaneously injected with the help of a transponder injector (injector and yellow-labelled transponder, ISO FDX-B Standard/manufacturer code 972; Planet Id GmbH, Essen, Germany).
Focal sampling home-cage behaviour
In order to analyse animal behaviour and welfare in the different housing conditions, we used a focal sampling analysis of home-cage behaviour. 19 The animals tails were pen-marked the day before so that on the day of home-cage behaviour recording, they could be videotaped undisturbed in their home cages. The videos were then analysed for sleep duration and for social interaction duration (total time each animal spent sleeping or performing social interaction activities). Each rat was observed continuously for 8 min per session and for two sessions a day, one in the light phase (between 08:30 h and 11:30 h) and another in the dark phase (between 20:30 h and 23:30 h) of the light–dark cycle. The videos were repeated over two consecutive weekends (two videos per housing condition and per phase of the light–dark cycle) and the data presented per animal is the mean value of the two videos. In all the video sessions, and for each of the three housing conditions (PhW, STD6 or STD2), the room where the animals were housed was not disturbed by any researcher or animal caretaker to ensure that there was no interference arising from the presence of any person in the room.
All the videos were scored by the same observer. A second observer scored part of the videos to control for a possible bias. The patterns of behaviour analysed and counted as social interaction were social investigation (sniffing or being sniffed by cage partners); allogrooming or social grooming; aggressive behaviour; play behaviour including pouncing (soliciting behaviour: attempt to nose rub or rubbing of the neck by the play partner); and pinning (lying dorsally on the floor with the partner animal standing over it).19,20
Statistics
Data from the focal sampling analysis of sleep and social interaction activities were analysed by the Kruskal–Wallis non-parametric test, followed by the Dunn–Bonferroni pairwise comparisons method. Outliers exclusions were based on the Z score criteria Z > |3|. In all cases, statistical significance was set at P ≤ 0.05. Statistical analysis was performed using IBM SPSS Statistics, version 22.0 (IBM Corp, Armonk, NY, USA).
Results
Sleeping duration
Focal sampling analysis of the time animals spent sleeping in the light and in the dark phases of the light–dark cycle was significantly affected by their housing conditions, as shown by the Kruskal–Wallis non-parametric test (light phase: χ2(2) = 21.884, P < 0.001; dark phase: χ2(2) = 6.015, P = 0.049). Animals in the PhW were sleeping most of the time in the light phase of the light/dark cycle, and the Dunn–Bonferroni pairwise comparisons test confirmed that the median values of sleep duration obtained for the PhW group were significantly higher than those obtained for the standard-housed animals (STD6: P < 0.001; STD2: P = 0.005) (Figure 1 – light period). During the dark phase of the light/dark cycle, where the animals were supposed to be active and not asleep, the animals in the PhW group presented the lowest sleeping time (only one animal slept for a short time) which was significantly different from STD2 animals (P = 0.045) (Figure 1 – dark period).
Total time animals spent sleeping during the light and dark phases of the light/dark cycle, evaluated by home-cage behaviour focal sampling. Box plots present the minimum (whiskers below the box), the lower quartile, the median (horizontal line crossing the box), the upper quartile and the maximum (whiskers above the box) values for each group. Analysis was performed by Kruskal–Wallis non-parametric test, followed by Dunn–Bonferroni pairwise comparisons method. Significant effects of housing are shown as *P ≤ 0.05 and **P ≤ 0.001.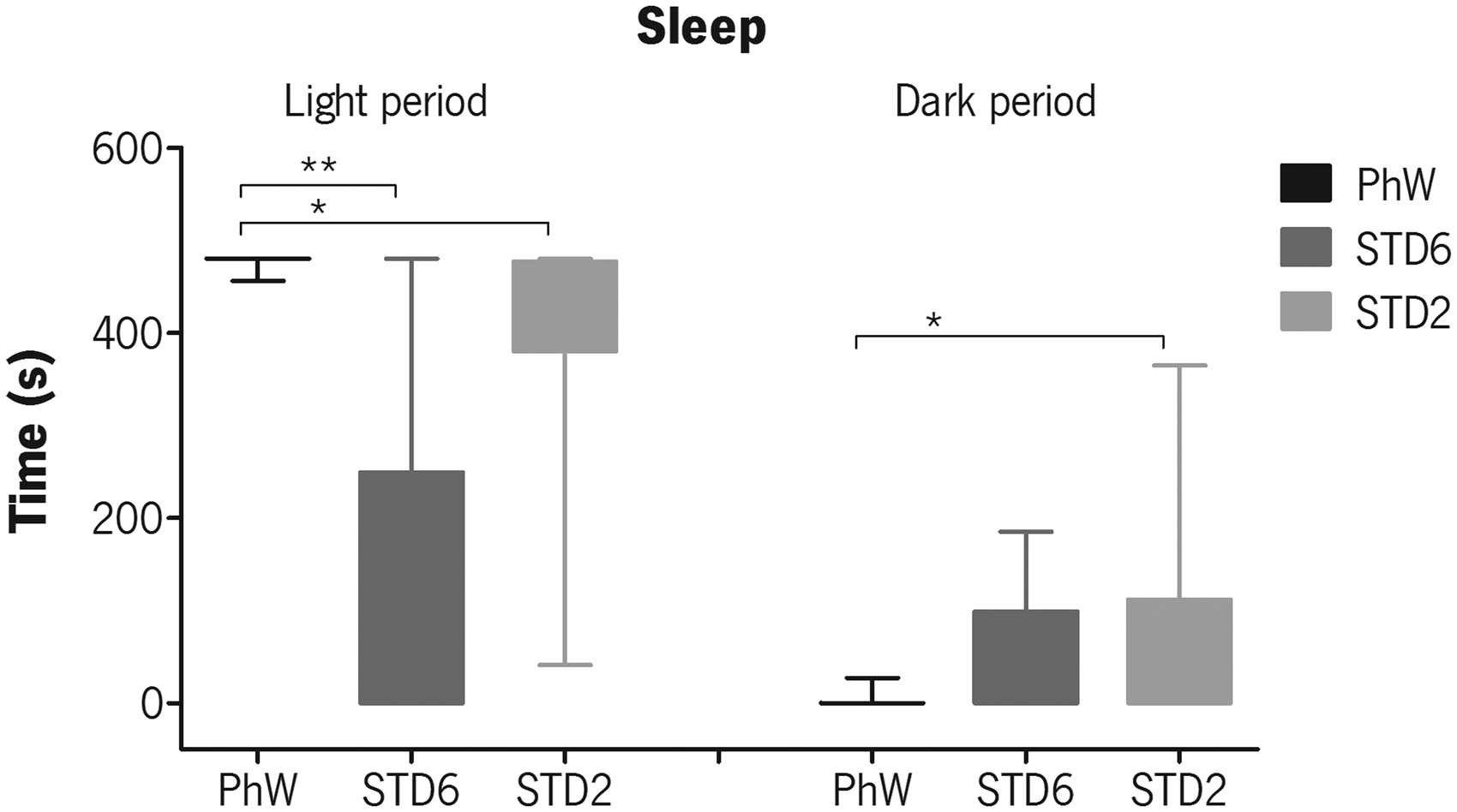
Social interaction duration
Social interaction activities for all housing conditions were observed by focal sampling, mostly during the rats’ active period (dark phase of the light/dark cycle). Kruskal–Wallis non-parametric test confirmed the main effect of housing on these activities (χ2(2) = 12.831, P = 0.002). During this active period, PhW animals dedicated more time to social interaction activities than standard-housed animals, but the Dunn–Bonferroni pairwise comparisons test showed this difference was only significant when comparing PhW with STD2 animals (P = 0.001) (Figure 2 – dark period). Even though the time spent in social interaction activities was reduced in the light phase of the light/dark cycle, where the animals spent most of their time sleeping, we observed that some animals in the standard housing were still engaged in such activities, and a significant effect of housing on activities during this time was shown by statistical analysis by the Kruskal–Wallis test (χ2(2) = 12.358, P = 0.002), with pairwise comparisons showing that STD6 spent significantly more time in social activities than PhW (P = 0.002) and STD2 (P = 0.023) animals in the light phase of the light/dark cycle (Figure 2 – light period).
Total time animals spent in social interactions during the light and dark phases of the light/dark cycle, evaluated by home-cage behaviour focal sampling. Box plots present the minimum (whiskers below the box), the lower quartile, the median (horizontal line crossing the box), the upper quartile and the maximum (whiskers above the box) values for each group. Analysis was performed by Kruskal–Wallis non-parametric test, followed by Dunn–Bonferroni pairwise comparisons method. Significant effects of housing are shown as *P ≤ 0.05 and **P ≤ 0.001.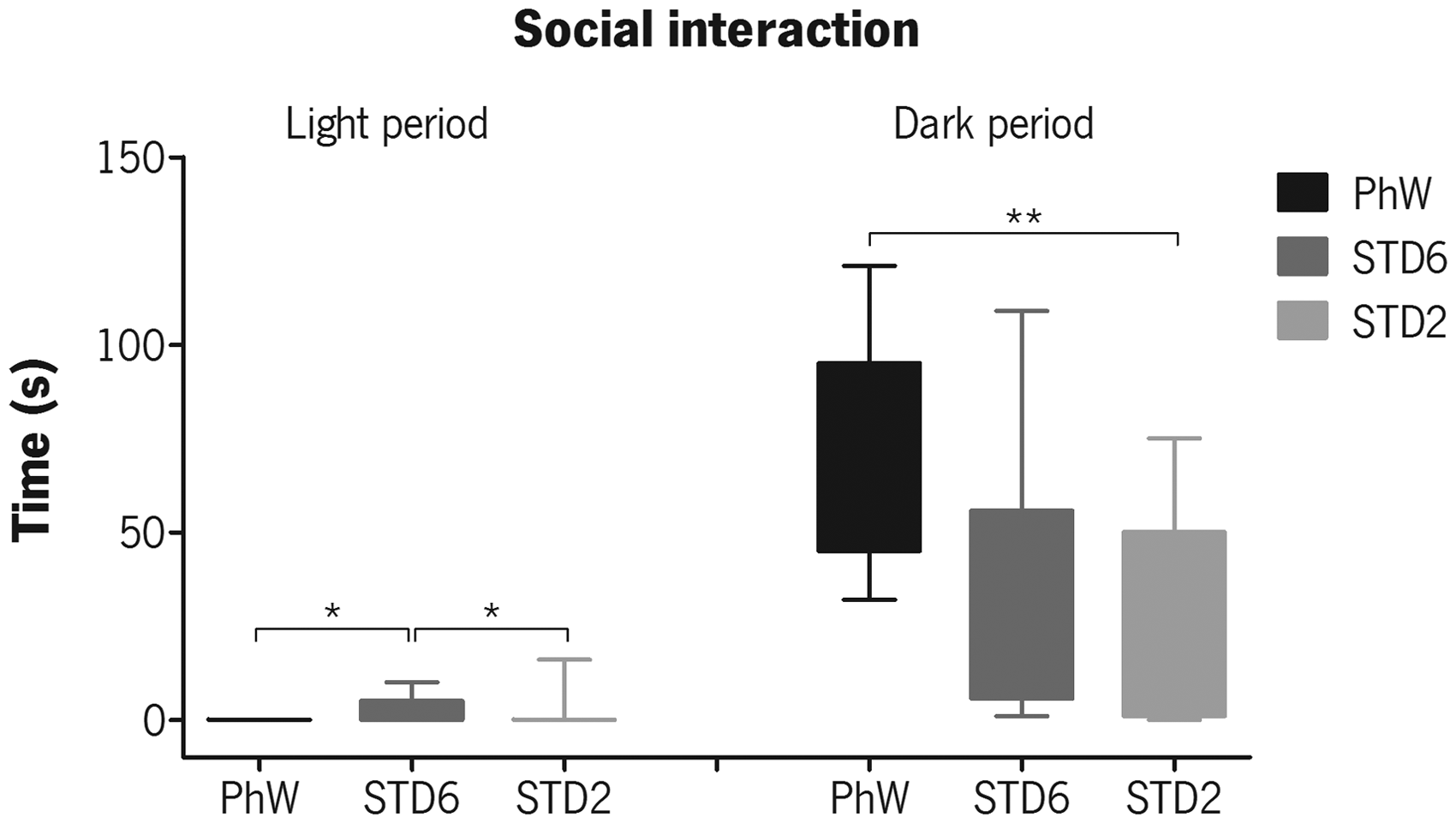
Discussion
Several criteria have been used to assess animal welfare but to understand animal welfare will involve both value-based and science-based evidences together with some degree of subjectivity.7,17 We chose to evaluate animal welfare in light of the three interconnected concepts as proposed by Fraser. 17 Therefore aspects of basic health and functioning, natural living and affective states of the animals housed in the PhW compared with animals housed in standard cages either in groups of six or pair-housed rats (STD6 or STD2) were analysed.
Basic health and functioning
In our study SPF animals kept in conventional laboratory conditions were used and these animals were observed regularly. None of the animals showed signs of injury, pain or discomfort in any of the housing conditions addressed. When analysing home-cage behaviour during the active period of the light/dark cycle by scan sampling, we have previously shown that animals living in the PhW presented an increased number of locomotion events, followed by animals living in STD6, with STD2 animals presenting the lowest number of such events. 16
In the present study we addressed home-cage behaviour by focal sampling sleep and social behaviours. We have shown that animals living in the PhW have a clearer pattern of resting/activity according to the light/dark cycle. PhW animals mostly slept during the light period and were awake and active during the dark period, whereas for STD6 and STD 2 animals this pattern of behaviour was not so evident. Even though STD6 animals were similarly housed in a group of six, the structure they had available was poorer and consisted of a much smaller area per animal. This physical condition could promote social interactions, and certainly did as compared with the pair-housed animals. However this also seemed to create a disturbing element in their behaviour, such as in the resting time (as shown by the STD6 animals spending less time sleeping in the light phase and spending more time sleeping in the dark phase of the light/dark cycle). These findings are in agreement with our previous work where we have shown that plasma corticosterone levels of PhW animals were the lowest at 08:00 h when the lights came on, and the highest at 20:00 h when the lights went off, whereas STD6 animals were the ones with a smaller difference between plasma corticosterone levels at 08:00 h and at 20:00 h. 16 Using these data we can speculate that there is a certain degree of disturbance in the circadian rhythm of animals housed in a group of six but with space limitations, such as those of the STD6 group. Whereas conditions in the PhW promote good circadian rhythmicity of the sleep/wake cycles which is known to affect mood, 21 including the establishment of social interactions during the active period of the light/dark cycle.
All the data discussed so far indicates that PhW can contribute to higher standards of basic health and functioning of the animals, which might contribute to an increased welfare1,3,9 and respecting, as for standard conditions, at least one of the five freedoms defined in the first concept of animal welfare22: the freedom from discomfort (providing comfortable resting areas).
Natural living
We have previously reported that animals in the PhW show basic natural behaviours including exploring, hopping, climbing, running in open areas and tubes between the central cage and the running wheel cage, digging and choosing partners for social interaction, including play. All these characteristics are the behaviours of wild rats but can still be observed in their laboratory counterparts after several generations of breeding in a laboratory environment. 23 The use of environmental refinement objects, such as tubes, in the standard cages also allows for some degree of exploration and climbing and have been shown to contribute to the fulfilment of some of the behavioural needs of rats in the laboratory, contributing to the freedom to express normal behaviour;1,3,8,9,22 however, hopping, digging, climbing and running behaviours are limited in standard cages due to space restrictions. In the PhW, not only are refinement objects provided in the central cage, there is an increased space area and height, as well as free access to running wheels which have all been shown to be beneficial to rats. 24 Combined with social partners, all these structural enrichments available in the PhW provide opportunities for rats to perform more species-specific behaviours that may not be possible in STD6 and STD2 housing conditions, which should improve their welfare.
Freedom from hunger and thirst 22 is also expected in all the housing conditions tested. However in the PhW, even though there is access to food and water ad libitum, the access to the two drink/feed boxes is made through AG, an automated door system that allows only one animal to cross at any one time. We have observed and reported that all animals in the PhW managed to access the drink/feed boxes and to consume amounts of food and water that are comparable with standard-housed animals. 16 However these animals need an adaptation period to learn how to use the AG system which presents an increased challenge for accessing food and water compared with the free access to food and water presented in standard housing. We believe this challenge constitutes a positive stimulus to PhW animals. The sensory and cognitive capacities of animals are known to be a result of evolution in the face of a variable environment, and adapting to a uniform stable artificial environment, such as that of a standard laboratory cage, may be difficult for rats.9,25 Animal welfare concept based on allostasis considers that the capacity to change or adapt to challenges is crucial to good physical and mental health and is good animal welfare. 26 The PhW drink/feed system contributes to such challenges in a controlled laboratory housing set-up, where the animals can still exert a certain amount of control over their lives which is also important for the animal welfare. 7
The ability to perform species-specific behaviours, the presence of social and physical challenges and the ability to have some control over their environment are all present in a PhW paradigm. These contribute to a naturalistic living in a laboratory setting and to the freedom to express normal behaviour, 22 which should improve animal welfare.9,17,25
Affective states
The ‘affective states’ we refer to here are defined by Fraser as emotions and other feelings that are experienced as pleasant or unpleasant rather than hedonistically neutral. 17 We used the gold standard test in the validation of the PhW, which provided insight into the affective states of control rats living in this paradigm when compared with standard housing. Previously, we have shown that control animals living in the PhW present reduced anxiety in the light/dark test, reduced immobility in the forced swimming test (indicating higher motivation to escape the water or reduced depressive-like behaviour) and increased motivation for a pleasurable sweet solution in a sucrose or saccharine preference test (or reduced anhedonia). 16 All these data, as well as the spontaneous motivation to run in the wheels, are indicative of the positive affective state of the animals and adds to the previously discussed contribution to an increased animal welfare in the PhW.
Measurements of spontaneously occurring social behaviour can provide an indication of the affective state of the animals, for example approach and avoidance behaviours may be used to assess the general valence (pleasantness/unpleasantness) of a stimulus to an animal. 27 Rats are known to be a species that need social contact to have a good welfare. 11 In the present study we have verified that even though all the animals in all the housing conditions performed social interaction activities during their active periods, animals housed in the PhW performed these activities for longer periods. This indicates that, even though having a big central cage (with about four times more area per animal than provided in a standard cage) and a running wheel cage for their entertainment as well as extra space in the drinking/feeding boxes, rats living in the PhW are proactive when engaging in social interactions that could be easily avoided. We have previously seen from scan sampling that play behaviour is one of the most frequent social interactions performed by animals in the PhW. 16 Taken together, these findings regarding social interaction also point to a positive affective state, contributing to an increased welfare for animals living in the PhW compared with standard-housed animals.
Conclusions
Our data provide evidence for increased animal welfare of rats as a consequence of living in the PhW paradigm, which promotes good basic health and functioning, natural living including control over some their environment, meeting some physical and social challenges, and positive affective states reflected by the reduced behavioural despair and anxiety, higher hedonistic behaviour, increased sleeping time during the resting period of the day and increased social interactions during the active period of the night. If we could ‘ask the rats’ which housing condition they would prefer in a preference test where the animals were allowed to choose between the PhW and a standard cage, we predict they would choose the PhW.
The physical and social conditions in which animals live in the laboratory have implications for animal welfare and consequently for the experiment outcome, which have to be accounted for.4,12 The PhW provides a laboratory setting that resembles more closely to the real world where many events have to be dealt with. Even though it is less standardized than regular standard housing, it is richer as a study paradigm with a more naturalistic housing set-up. We have previously validated the PhW as a good paradigm for studying mood disorders, 16 but we believe it can be useful in many other fields, for example in future studies of complex emotional and cognitive disorders, metabolism disorders, as well as for rodent studies of non-diseased animals.
According to our data, even though it might be expensive and complex to implement, it would be good for laboratory animal welfare to have the PhW as standard housing for rats.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research received unding from Fundação Calouste Gulbenkian (FCG_P-139977).
