Abstract
Oral gavage is commonly used in pre-clinical drug evaluation, but is potentially aversive and may induce behavioral effects independent of compounds under investigation. This study examined the combined effects of repeated oral gavage and disease induction on anxiety-like behavior in the experimental autoimmune encephalomyelitis (EAE) model of multiple sclerosis. The C57BL/6J and NOD/ShiLtJ EAE variants were exposed to sham-EAE induction or untreated control conditions, and either daily oral gavage or home cage conditions. Anxiety-like behavior was subsequently assessed in the elevated plus maze. C57BL/6J mice exhibited increased anxiety-like behavior, relative to NOD/ShiLtJ mice, in response to repeated gavage, whereas sham-EAE induction and repeated gavage were associated with increased anxiety-like behavior in NOD/ShiLtJ mice. Thus, exposure to the induction procedure and repeated gavage differentially altered subsequent anxiety-like behavior in the two EAE variants. Future pre-clinical studies should rely on prior evaluation of parameters of the experimental design using sham-EAE mice. Additionally, less aversive administration routes should be utilized wherever possible to ensure that procedures do not distort effects of the therapeutic under investigation.
Introduction
Multiple sclerosis (MS) is a chronic autoimmune disorder of the central nervous system (CNS) typified by a range of symptoms including visual and motor disturbances, bladder dysfunction, and neuropsychological deficits. 1 In particular, anxiety, depression, and cognitive deficits are prevalent among individuals with MS, with approximately 35% of MS-affected individuals reporting anxiety, compared with 25% of the general population and 43–65% of MS-affected individuals diagnosed with cognitive deficits.2,3 In the last decade, the significance of emotional and cognitive symptoms as primary manifestations of the disease has gained greater appreciation. Concurrently, substantial advances have been achieved in treatment options, particularly in the development of oral drugs, 4 which significantly modify the natural history of the disease. However, the potential benefits of these novel treatments on anxiety and cognitive deficits are still unclear and studies based both on human subjects and MS animal models are limited.
Pre-clinical evaluation of MS candidate therapeutics is most commonly performed using experimental autoimmune encephalomyelitis (EAE), a neuroinflammatory disease induced in rodents and non-human primates by sensitization with a CNS protein or peptide. 5 However, the use of this model presents a number of challenges, 6 specifically with regard to the investigation of psychological symptoms associated with the disease. 7 For example, EAE protocols often require emulsification of the neuropeptide in Freund’s complete adjuvant, combined sometimes with the use of the toxic protein from Bordetella pertussis (PTX), which enhances blood–brain barrier breakdown and causes significant lymphocytosis.5,8 Additionally, oral therapies in experimental mice are commonly administered by gavage, which is potentially anxiogenic. 9 Finally, mouse EAE exists as variants differing in clinical and immunopathological progressions; thus myelin oligodendrocyte glycoprotein (MOG)-induced EAE exhibits a chronic progressive profile in C57BL/6J mice, 10 but a relapsing–remitting profile in NOD/ShiLtJ mice. 11 To improve the use of the EAE model in pre-clinical drug evaluation, the potential confounding interactions between these multiple variables need to be identified and evaluated. Here, we specifically addressed the anxiogenic effects of gavage and disease induction components using vehicle-only (sham-EAE) and control mice of these two mouse strains and the elevated plus maze (EPM) test, a validated test of mouse anxiety-like behavior.12–14
Animals
Our animal facility complies with regulations set by the National Health & Medical Research Council of Australia and experimental procedures were approved by the institutional animal ethics committee. Colonies of NOD/ShiLtJ and C57BL/6J mice were established from breeding pairs purchased from the Animal Resource Centre (Canning Vale, Western Australia) and housed at the La Trobe Animal Training and Research Facility at 23°C, and 12:12 h light:dark cycle, on standard rodent chow (Barastoc Rat and Mouse Feed, Ridley AgriProducts) and tap water ad libitum. As standard practice, water bottles are weighed daily to ensure adequate hydration. The C57BL/6J mouse strain does not appear to have obvious adverse phenotypes. 15 The NOD/ShiLtJ strain, on the other hand, 15 exhibits spontaneous diabetes with higher incidence in females (80–100% by 30 weeks) than males (40–60% at the corresponding time) and breeding is limited to two litters per female. Diabetes is inhibited by Freund’s adjuvant,16,17 so this condition is not a major issue when NOD/ShiLtJ mice are used for EAE. However, all mice will have pre-insulitis by 9 weeks of age. Mice of both strains are used in experimentation from 9–10 weeks and females only are used due to the well-documented differences in disease incidence (3F:1M) and clinical sub-types (relapsing–remitting MS being more common in women) in MS. 18 Mice were checked daily by trained animal technicians. Prior to disease induction, mice were weighed to ascertain that they had reached the expected weight relative to age (NOD/ShiLtJ strain https://www.jax.org/strain/001976; C57BL/6J https://www.jax.org/strain/000664) and after each procedure mice were kept under observation by investigators for 20–30 min.
Materials and methods
Sham-EAE induction and surveillance of animals
Sham-EAE induction was performed by two subcutaneous injections in the inguinal region, 10 each containing 100 µL sterile phosphate buffered saline (PBS; 0.01 M phosphate, 15 mM NaCl, pH 7.4), emulsified in 100 µL of complete Freund’s adjuvant (F5881-6X10ML, Sigma-Aldrich, St. Louis, MO), supplemented with 4 mg/mL of heat inactivated Mycobacterium tuberculosis H37 Ra (231141, Becton Dickinson, Franklin Lakes, NJ). Mice were then immediately injected intraperitoneally (i.p., C57BL/6J strain) or intravenously (i.v., NOD/ShiLtJ strain) with 350 ng of Bordetella pertussis toxin (PTx) (P7208-50UG, Sigma-Aldrich) in PBS. A second PTx injection was administered 2 days later. NOD/ShiLtJ received PTx via the i.v. route, as i.p. delivery is associated with lower disease incidence in this strain. There is no difference in C57BL/6J EAE incidence, whether i.v. or i.p. injected for PTx administration. Following sham-EAE induction, mice were weighed on days 2, 7, and 9.
Oral gavage
Oral gavage was conducted as described previously. 9 Briefly, a 3.8 cm long, 2 mm wide blunt gavage needle (Able Scientific, Canning Vale, WA, Australia) was attached to a sterile 1 mL syringe containing 100 µL sterile PBS. Following mouse restraint, the gavage needle was gently inserted in to the mouth and then stomach of the mouse, PBS quickly dispensed, and the tube then safely removed from the mouse. This was conducted from day 0 to day 8 and no gavage was conducted on day 9 (test day). Non-gavaged/non sham-induced mice were weighed according to the same schedule as the sham-induced mice, as controls to ensure there were no ill effects from the gavage or induction procedures. All mice reached their experimental end point with no adverse effects.
EPM test
The sample size was determined based on previous research using the MOG-induced C57BL/6J EAE variant, 12 where EAE mice showed increased anxiety like behavior in the EPM (% time in open arms; mean ± sd; EAE, 16.14 ± 8.38; control, 30.07 ± 18.41, d = 0.97). For statistical power of 0.8, with alpha set at 0.05, the minimum total sample size required to detect a similar effect is N = 72 for the behavioral experiments, 19 which is within the range of our total sample size of N = 85. The EPM test was conducted during the light cycle of day 9 following induction, for 5 min, in a soundproof room under dim red lighting (40–41 lux). The EPM was constructed of acrylic, consisting of a central platform (5 cm × 5 cm) with four branching arms (30 cm × 5 cm) at right angles to each other, in which one pair of opposite arms were enclosed by gray opaque walls (16 cm high) and the other pair open. The maze was set on legs 36.5 cm above the ground. A 2 mm-high lip was placed around the edges of the open arms to prevent the mice from falling off. Behavior was recorded using a HD webcam connected to a PC, by an investigator blind to treatment conditions. Throughout the study, the same room was used, and the same researcher (PK) conducted the EPM test. The apparatus was cleaned with 70% ethanol and dried before each individual test to remove odor traces. Behavior parameters were analyzed using Ethovision XT (Noldus Information Technology, NLD). Two data points outliers (0.5% of total data set) were removed from analysis; specifically, 2 locomotor activity values were omitted as described. 20 A 2 × 2 × 2 univariate analysis of variance (ANOVA) was then performed using the Statistical Package for the Social Sciences (SPSS, v.22; IBM, Armonk, NY). Following statistically significant interaction or main effects, post-hoc tests using Fisher’s protected least significant difference (LSD) tests were conducted.
Corticosterone assays
At experimental end-point, all mice were killed by CO2 inhalation and blood collected by cardiac puncture. Plasma was isolated by centrifugation at 10,000 g for 10 min at 4°C, aliquoted, and frozen at −20°C. When required, plasma samples were thawed and total corticosterone concentrations measured using the corticosterone ELISA kits (Enzo Life Sciences, Farmingdale, NY) as described. 21 Corticosterone concentrations were measured in duplicate, following dilution of 1:80 in kit buffer and the experiment repeated three times. Plates were read with a Synergy HT absorbance spectrophotometer (BioTek, Winooski, VT) at 405 nm. Upper and lower levels of detection were 20,000 pg/mL and 32 pg/mL, respectively, while the inter and intra assay coefficients of variability were 8.9% and 2.3%, respectively.
Results
Health of the animals over the experimental time course
Monitoring of NOD/ShiLtJ and C57BL/6J sham-EAE induced or control groups. NOD/ShiLtJ and C57BL/6J cohorts were divided into sham-induced or untreated groups. Half of each group received daily gavage with saline, or no further treatment (n = 10–15 per group). On day 9, anxiety-like behavior was evaluated in all mice by the EPM test. The effects of treatments on the health of the animals were monitored over the course of the experiment, by visual checks and weighing at days 0, 2, 7, and 9. The weight change over the time course of the experiment (day 9 − day 0) for each group is shown as the mean ± SEM, where weight at day 0 is set at 100%. All mice had a clinical score of 0 for the duration of the experiment.
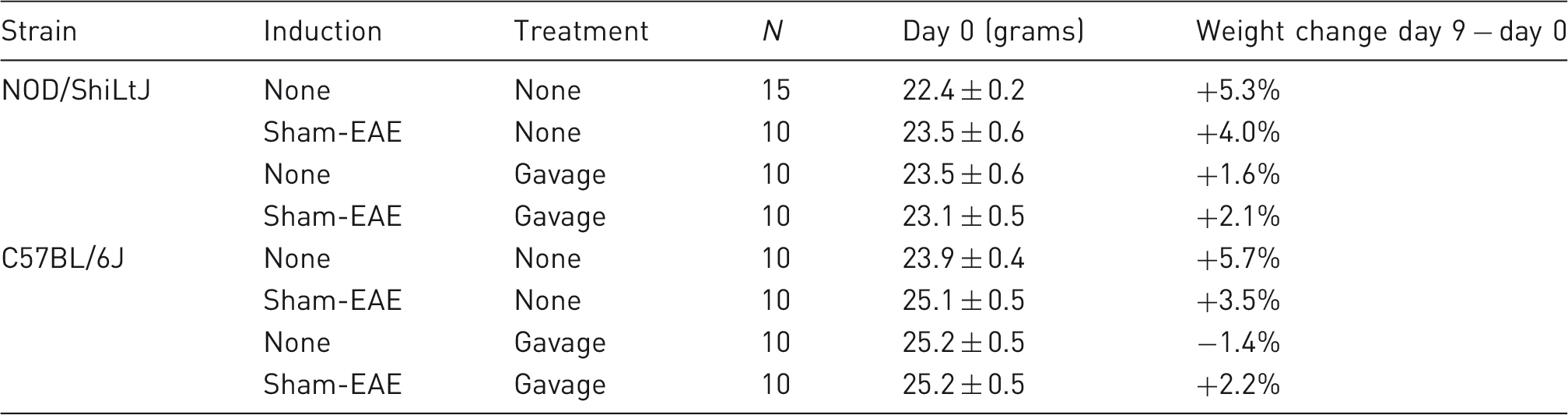
Evaluation of activity
We first identified that exposure to the sham-EAE induction procedure and repeated oral gavage altered locomotor activity in the EPM in a strain dependent manner (strain main effect, F(1,75) = 43.416, p < 0.001; induction main effect, F(1,75) = 4.265, p = 0.042; strain × gavage interaction, F(1,75) = 10.545, p = 0.002; Figure 1(a) and (b), Table 2). Post hoc tests indicated that C57BL/6J mice exposed to control conditions (control and control + gavage groups) and C57BL/6J mice exposed to sham-EAE + gavage conditions showed decreased total distance moved compared with their NOD/ShiLtJ counterparts. On the other hand, among NOD/ShiLtJ mice, those exposed to the sham-EAE induction procedure alone showed decreased locomotor activity compared with control and sham-EAE + gavage treated mice (Figure 1(a)).
Effect of exposure to the sham-EAE induction procedure and repeated oral gavage. Distance moved (cm) in the EPM test in NOD/ShiLtJ (a) and C57BL/6J (b) mice and duration of time spent in open arms (as a percentage of time spent in all arms) in NOD/ShiLtJ (c) and C57BL/6J (d) mice are shown. NOD/ShiLtJ (e) and C57BL/6J (f) plasma corticosterone concentrations following the EPM test are represented. Bars show within-strain significant differences (+ = induction, † = gavage) and * show between-strain differences; **p < 0.01, ***p < 0.001 versus respective NOD/ShiLtJ group; +p < 0.05, ++p < 0.01 versus respective control group; †p < 0.05, ††p < 0.01 versus respective no-gavage group, Fisher’s protected least significant difference test. Data are expressed as mean ± SEM. The main effects and interaction effects of the 3 parameters under investigation, on locomotor activity. The main effects (strain, induction, and gavage) and interaction effects between these parameters are shown in terms of distance moved (cm), in a 2 × 2 × 2 univariate ANOVA. Significant differences (p ≤ 0.05) are found for the strain effect and induction effect while an interaction effect is only found between strain × gavage.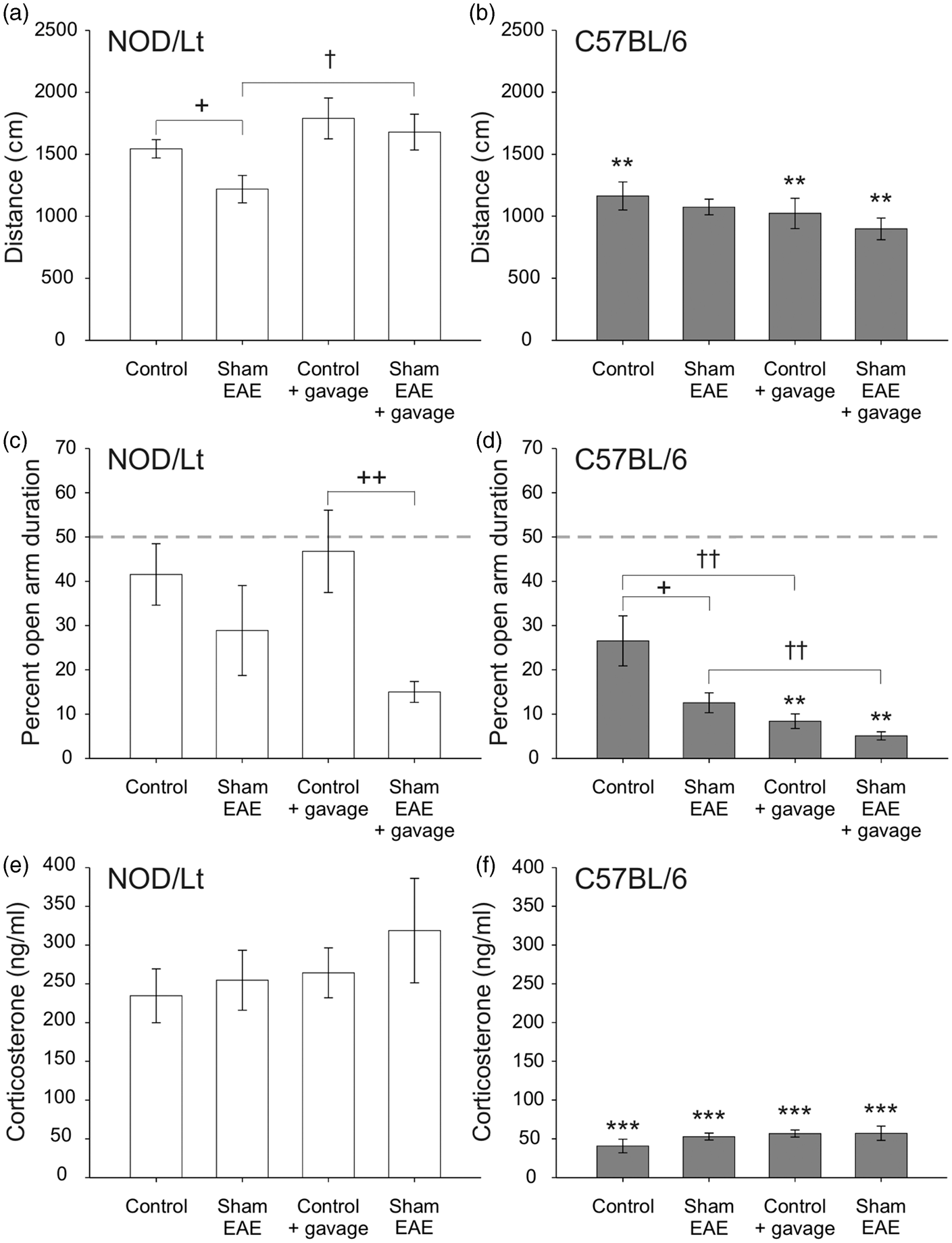
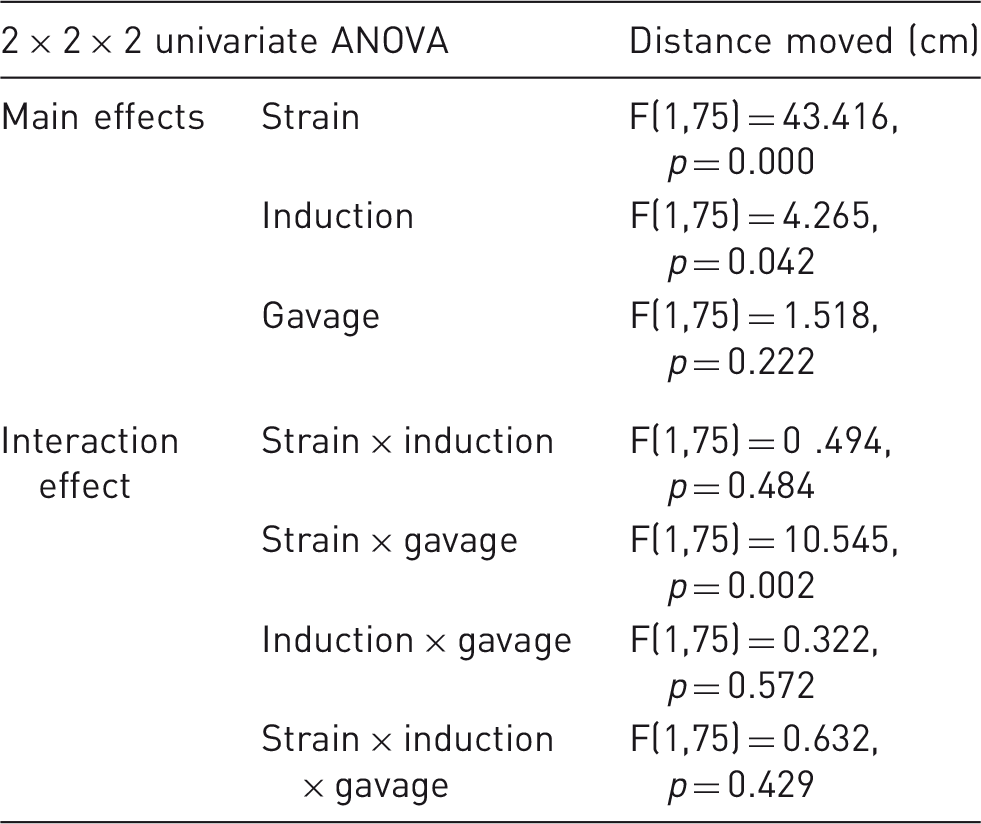
Evaluation of anxiety-like behavior in the EPM
The main effects and interaction effects of the three parameters under investigation on the percentage of total time spent in open arms of the EPM. The main effects (strain, induction, and gavage) and interaction effects between these parameters are shown in terms of percent open arm duration, in a 2 × 2 × 2 univariate ANOVA. Significant differences (p ≤ 0.05) are found for the strain effect and induction effect, while there are no interaction effects with statistical significance.
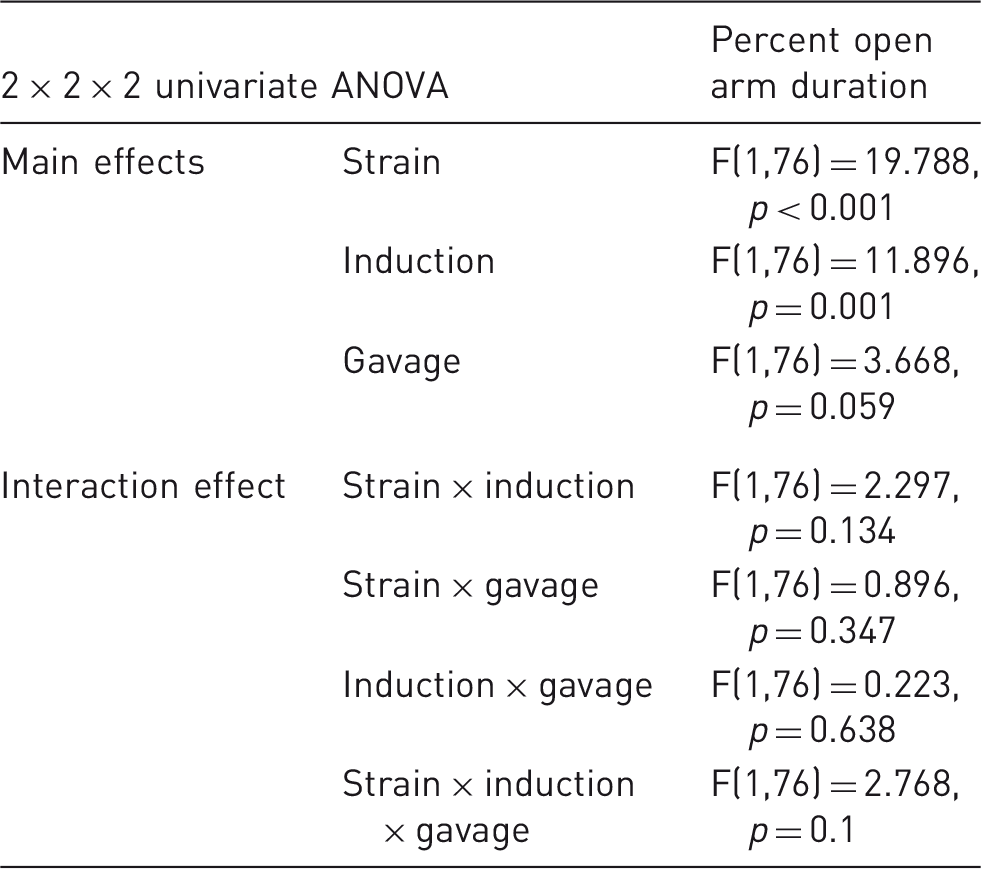
Corticosterone assays
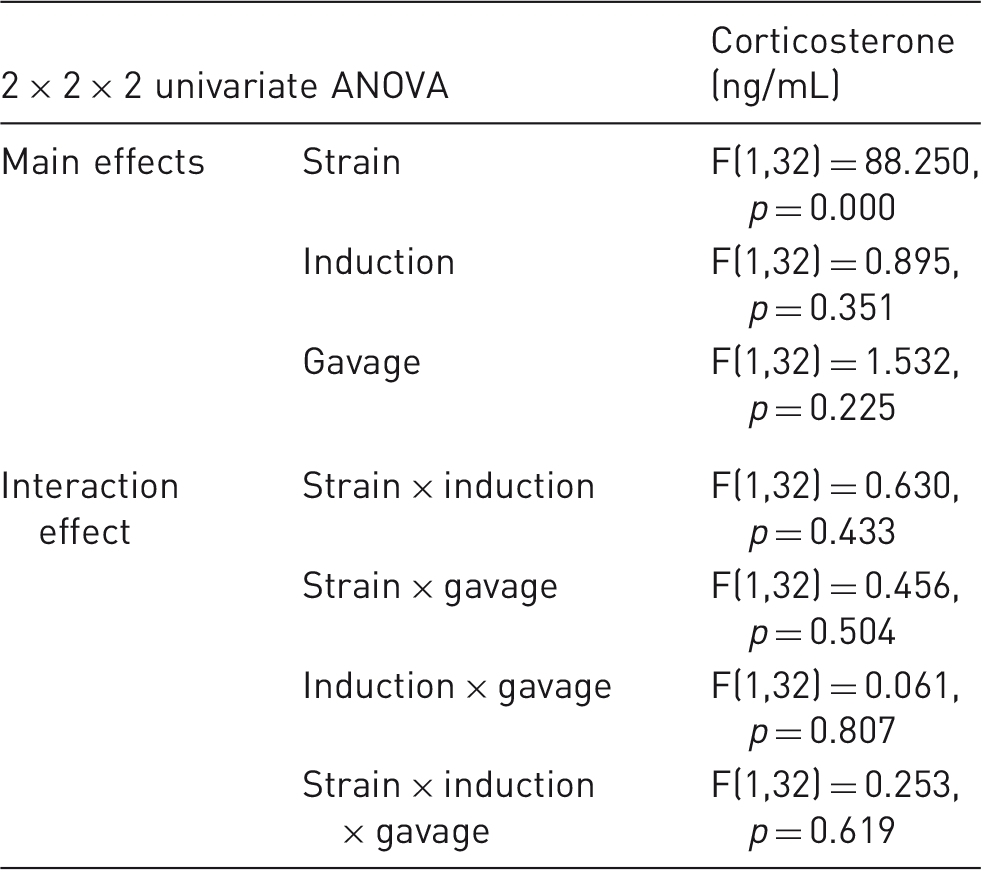
The main effects and interaction effects of the three parameters under investigation on plasma corticosterone concentration. The main effects (strain, induction,and gavage) and interaction effects between these parameters are shown in terms of corticosterone (ng/mL) in a 2 × 2 × 2 univariate ANOVA. Significant differences (p ≤ 0.05) are found for the strain effect only, while there are no interaction effect with statistical significance.
Discussion
Multiple EAE variants have been validated for generation of proof of principle for MS immunopathology. On the other hand, the present study reveals the potential confounding effects of components of disease induction and drug delivery on investigations of neuropsychological aspects of the disease, particularly when these focus on the pre-onset stage. Thus, differential effects of EAE induction and exposure to daily gavage between strains were clearly evident. Sham-EAE was associated with anxiety among NOD/ShiLtJ mice exposed to repeated gavage, but not those exposed to home cage control conditions. In contrast, sham-EAE induction increased anxiety-like behavior in C57BL/6J mice exposed to home cage control conditions, but not those exposed to repeated gavage. It is possible that among C57BL/6J mice, the gavage procedure is sufficiently aversive to produce a floor effect in anxiety-like behavior, thereby decreasing detection of any behavioral effects of the sham-EAE induction procedure. That is, even though there is an induction effect present in the C57BL/6J mice, the gavage effect is masking any of the induction effect when both procedures are performed on the same animal. Exposure to repeated oral gavage increased anxiety-like behavior in control and sham-EAE induced C57BL/6J mice, but was without effect on NOD/ShiLtJ mice. Therefore, it appears that the sham-EAE induction procedure explains most of the variance within and between strains, while gavage strongly affects the C57BL/6J mice (Figure 1(c) and (d)). The reason for this differential strain effect is unclear, given the identical conditions used for housing and handling the two mouse strains. On the other hand, it is also important to consider the potential effect of the different route of PTx delivery between mouse strains. I.p. injection of PTx results in at least 95% disease incidence in the C57BL/6J strain. In NOD/ShiLtJ mice the same route of PTx delivery is associated with only 50-60% disease incidence, whereas 95% disease incidence is achieved via i.v. PTx injection. The i.v. route of PTx injection may result in more severe damage to the vasculature and blood–brain barrier which may not be fully restored by the time point of 9 dpi when the EPM test is performed, thereby exposing the brain environment to blood components. A later time point for behavioral testing is not suitable because of clinical onset and ambulatory difficulties observable from 11–12 dpi in both mouse strains.
Although there were no effects of sham-EAE induction or repeated gavage on plasma corticosterone concentrations, results indicated higher plasma corticosterone concentrations in NOD/ShiLtJ mice compared with C57BL/6J mice. Specifically, regardless of the treatment, NOD/ShiLtJ mice had higher levels of corticosterone compared with C57BL/6J mice. Studies evaluating corticosterone levels between group-housed mouse strains showed variability in baseline corticosterone levels between strains, suggesting that the variance observed in endogenous corticosterone levels in this study are genetically predisposed. 22 The higher corticosterone levels in NOD/Lt mice may provide an explanation for the higher resistance to EAE development because higher corticosterone levels are associated with a dampening effect on EAE development. 23 Consistent with this hypothesis, evidence has been presented for a negative relationship between increasing corticosterone levels and EAE severity. 24 Thus Whitacre et al. showed that in both mice and rats, restraint stress administered before EAE induction resulted in inhibition of EAE, whereas the same treatment administered after EAE initiation had no effect on disease development. They concluded that elevated endogenous glucocorticoid levels exert a profound effect on development of autoimmune-mediated CNS injury. Taken together, the evidence suggests that the corticosterone levels may explain some of the differences in EAE-induction and EAE pathophysiology between these strains.
Taken together, our data show that the C57BL/6J EAE variant will prove more suitable for candidate MS drug evaluation. On the other hand, given the induction effect and relative high corticosterone concentration in NOD/ShiLtJ mice, this EAE variant is perhaps less suitable, despite the attraction of its relapsing-remitting clinical profile. Importantly however, the data illustrate the complexity of interactions between disease induction (namely eliciting of a strong immune response and transient breakdown of the blood–brain barrier) and other parameters of experimental design (daily handling and gavage) inherent in the EAE model and their profound impact on subsequent behavior. Thus, the sham-EAE group represents a valuable experimental paradigm, by allowing prior identification and quantification of such interactions, under conditions that are not masked by clinical disease development. These observations are also pertinent to other experimental autoimmune diseases, for example collagen-induced arthritis, where Freund’s adjuvant mediated activation of the innate immune system is a crucial step in inducing autoimmune disease. 25
We propose that for the improvement of animal welfare, future pre-clinical evaluation of candidate MS drugs should be preceded by detailed evaluation of interactions between EAE variant and variables of the experimental design in the sham-EAE group. Finally, in the last decade, oral drugs for MS, including Fingolimod (Gilenya), dimethyl fumarate (Tecfidera), and teriflunomide (Aubagio), which are water soluble, have been developed. Given the positive impact of the oral route on medical compliance, growing interest in the development of oral MS drugs is anticipated.26,27 Therefore, substitution of gavage by less aversive methods, such as delivery in the drinking water, wherever possible, has translational value.
Footnotes
Acknowledgments
We thank AJ Lawther (School of Psychology and Public Health, La Trobe University, Bundoora, Victoria, Australia) and the La Trobe Animal Research & Training Facility (LARTF) technicians for technical assistance.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Novartis Pharmaceuticals Australia and the School of Psychology and Public Health, La Trobe University, Bundoora, Victoria, Australia. P. Kocovski received an Australian Postgraduate Award (APA) from the Australian Government Department of Education and Training. C.S. D’Souza received a La Trobe University Postgraduate Research Scholarship (LTUPRS).
Supplementary information
Supplementary Table 1 can be found online with this article, http://journals.sagepub.com/doi/full/10.1177/0023677218756156. Further supplementary information can be obtained from: ![]() .
.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
