Abstract
Subarachnoid hemorrhage (SAH) from rupture of an intracranial arterial aneurysm is a devastating disease affecting young people, with serious lifelong disability or death as a frequent outcome. Large animal models that exhibit all the cardinal clinical features of human SAH are highly warranted. In this pilot study we aimed to develop a non-craniotomy model of SAH in pigs suitable for acute intervention studies. Six Norwegian Landrace pigs received advanced invasive hemodynamic and intracranial pressure (ICP) monitoring. The subarachnoid space, confirmed by a clear cerebrospinal fluid (CSF) tap, was reached by advancing a needle below the ocular bulb through the superior orbital fissure and into the interpeduncular cistern. SAH was induced by injecting 15 mL of autologous arterial blood into the subarachnoid space. Macro- and microanatomical investigations of the pig brain showed a typical blood distribution consistent with human aneurysmal SAH (aSAH) autopsy data. Immediately after SAH induction ICP sharply increased with a concomitant reduction in cerebral perfusion pressure (CPP). ICP returned to near normal values after 30 min, but increased subsequently during the experimental period. Signs of brain edema were confirmed by light microscopy post-mortem. None of the animals died during the experimental period. This new transorbital injection model of SAH in the pig mimics human aSAH and may be suitable for acute intervention studies. However, the model is technically challenging and needs further validation.
Subarachnoid hemorrhage (SAH) caused by rupture of an intracranial arterial aneurysm is a devastating disease affecting young people, with serious lifelong disability or death as a frequent outcome.
However, pathophysiological mechanisms leading to brain injury after aneurysmal SAH (aSAH) are not well described, and opportunities to study the events occurring immediately after aSAH in patients are very limited. Accordingly, there is a need for clinical relevant animal studies, 1 but a lack of good animal models has limited the development and validation of new treatment modalities. 2
Several in vivo animal models of SAH have previously been described in the literature. In 1928 Bagley et al. published a dog model of blood injection into the cisterna magna and subarachnoid space. 3 Since then a variety of different bleeding techniques have been reported in the dog, primate, cat, rat, rabbit, pig, goat and mouse.2,4,5 Improved imaging and monitoring techniques, combined with reduced costs and easy handling, have increased the popularity of using small animal models.1,2,5 However, large animal models are particularly useful for studies where advanced in vivo neuro- and hemodynamic monitoring equipment as well as repeated blood/cerebrospinal fluid (CSF)/urine/tissue sampling is required. Large animal models are therefore both more relevant and a better option to use in the evaluation of novel treatment strategies. 4
The Norwegian Landrace pig (Sus scrofa) is a large animal with many similarities to human anatomy, physiology, biochemistry and genetics. This makes it an ideal animal for experimental medical research.6,7 In recent years, knowledge concerning pig brain anatomy and neurochemistry has increased considerably. The pig brain is closer to the human brain when it comes to anatomy, growth, development and biochemistry than the brain of smaller animals. Pigs are therefore increasingly being used in neuroscience research.8,9
In this pilot study we aimed to develop and describe a non-craniotomy model of SAH in pigs suitable for acute intervention studies.
Animals, materials and methods
A total of 33 pigs were used in this study. Four of these animals were donated from other researchers who had terminated their experiments for various reasons. Eighteen pigs were used during the first part of the project to pilot various potential techniques for inducing SAH. The remaining 15 pigs were solely allocated to the development of the injection method. In 11 of those 15 animals SAH was considered to be successfully induced. This gives a success rate of 73%.
Data from six Norwegian Landrace pigs weighing 26–38 kg are presented in this final report. Five pigs were assigned for experiments with full hemodynamic and intracranial pressure (ICP) monitoring for 8 h following SAH induction, while one pig was assigned solely for magnetic resonance imaging (MRI) investigations. Two of the pigs were prematurely terminated after 4 h due to technical reasons.
The study protocol was approved by the Norwegian Experimental Animal Board and all experiments were conducted in compliance with institutional animal care guidelines and the National Institutes of Health (NIH) Guide for the care and use of laboratory animals (Department of Health and Human Services [NIH] Publication No. 85–23, revised 1985). The pigs were kept in the animal department for at least two days before the experiments. The conditions in the animal room were strictly controlled at a temperature of 21 ± 1℃, a relative humidity of 55 ± 10%, and a 12:12 h light/dark cycle. The animals were fed on a ‘Format’ diet (Felleskjøpet Agri, Gardermoen, Norway) and had free access to water. After an overnight fast, the pigs were sedated by an intramuscular injection of a mixture containing 15 mg/kg ketamine (Narcetan; Vétoquinol, Ittigen, Switzerland), 1 mg/kg midazolam (Midazolam B Braun; B Braun Melsungen AG, Melsungen, Germany), and 1 mg of atropine (Atropin; Nycomed Pharma, Asker, Norway). The pigs were then transferred to the operating room where they were anesthetized by mask inhalation with 4% isoflurane (Isofluran Baxter; Baxter, Irvine, CA, USA) in 100% oxygen, followed by a lidocaine spray (Xylocain 10 mg/dose; Astra Zeneca AS, Oslo, Norway) in the larynx and subsequently intubated orotracheally. After intubation, an intravenous bolus of 15 µg/kg fentanyl (Fentanyl-Hameln; Hameln Pharmaceuticals, Hameln, Germany) and 10 mg/kg pentobarbital (Pentobarbital NAF, Ås Produksjonslab A/S, Gamleveien, Norway) was given. Anesthesia was maintained with a central venous infusion of 4 mg/kg/h pentobarbital, 0.02 mg/kg/h fentanyl, and 0.3 mg/kg/h midazolam. If there were any clinical signs of discomfort, additional small boluses of fentanyl and midazolam were given. An intravenous bolus of 500 mL of sodium chloride (NaCl, 9 mg/mL) containing 625 mg of glucose was given as a preoperative load. Basal fluid requirement was maintained by intravenous infusion of this NaCl solution at a rate of 3 mL/kg/h. Baseline was defined as the time where all catheters were in place, but before SAH induction. The time when SAH was induced was defined as t = 0 h. From t = 0 h, glucose 500 mg/mL was given at a rate of 0.3 mL/kg/h to keep the glucose levels within the range of 3.0–6.0 mmol/L. All pigs were euthanized by an intravenous overdose of pentobarbital and potassium chloride.
Positioning of catheters
A 20 G vein cannula (Optiva 2; Smiths Medical International Ltd, Rossendale, UK) was inserted into the left ear vein for administration of the fentanyl and pentobarbital boluses right after oral endotracheal intubation. A 14 G central venous catheter (Sekalon T; Becton Dickinson Critical Care Systems, Yishun, Singapore) was introduced into the left external jugular vein for continuous administration of anesthetics and fluids. Two 16 G Sekalon T catheters were introduced into the right and left femoral arteries, respectively, for arterial blood sampling and blood pressure monitoring. In three of the pigs a 5 Fr pulmonary artery thermodilution catheter (Edwards Lifesciences LLC, Irvine, CA, USA) was floated into the pulmonary artery via the right external jugular vein for blood sampling and monitoring purposes. The urinary bladder was drained via cystotomy and insertion of a catheter (Rüsch PTFE AquaFlate; Teleflex Medical Sdn Bhd, Kamunting, Malaysia). Urine output was measured every hour before urine samples were obtained. All wounds were sutured after completion of surgery. A stable, drift-free ICP transducer (Codman MicroSensor Basic Kit; Johnson & Johnson, Raynham, MA, USA) was inserted one centimeter into the brain parenchyma of the right frontal lobe through a burr hole in the skull one centimeter ventrally to the coronal suture, and one centimeter laterally to the interfrontal suture. The burr hole was sealed with bone wax around the ICP catheter to stabilize it and to stop CSF leakage.
Development of the transorbital injection model and induction of SAH
We learned from dissections and computed tomography (CT) studies of the pig brain that the subarachnoid space could be reached by advancing a needle below the ocular bulb through the superior orbital fissure. Using this approach the interpeduncular cistern could be accessed, which allowed SAH induction by injecting autologous arterial blood into the subarachnoid space.
The animals were placed in a left lateral position. Guided by anatomical landmarks on the pig’s cranium a 20 G spinal needle (Spinocan®; B Braun Melsungen AG) was carefully advanced under the right ocular bulb with the needle slightly tilted upwards to reach the bony roof of the orbit. By searching downwards with the needle tip against the orbital bone the superior orbital fissure was located. The needle tip was then gently advanced through the fissure to reach the subarachnoid space in front of the brainstem (the interpeduncular cistern). Correct needle tip position in the subarachnoid space was verified by a clear CSF tap. The colored chlorhexidine method
10
was used to verify CSF (Figure 1). Subsequently, 15 mL of autologous arterial blood was drawn from the femoral artery catheter and steadily injected through the spinal needle over a time period of approximately one minute. The whole procedure was performed under aseptic conditions.
Cerebrospinal fluid tap verified by the chlorhexidine method.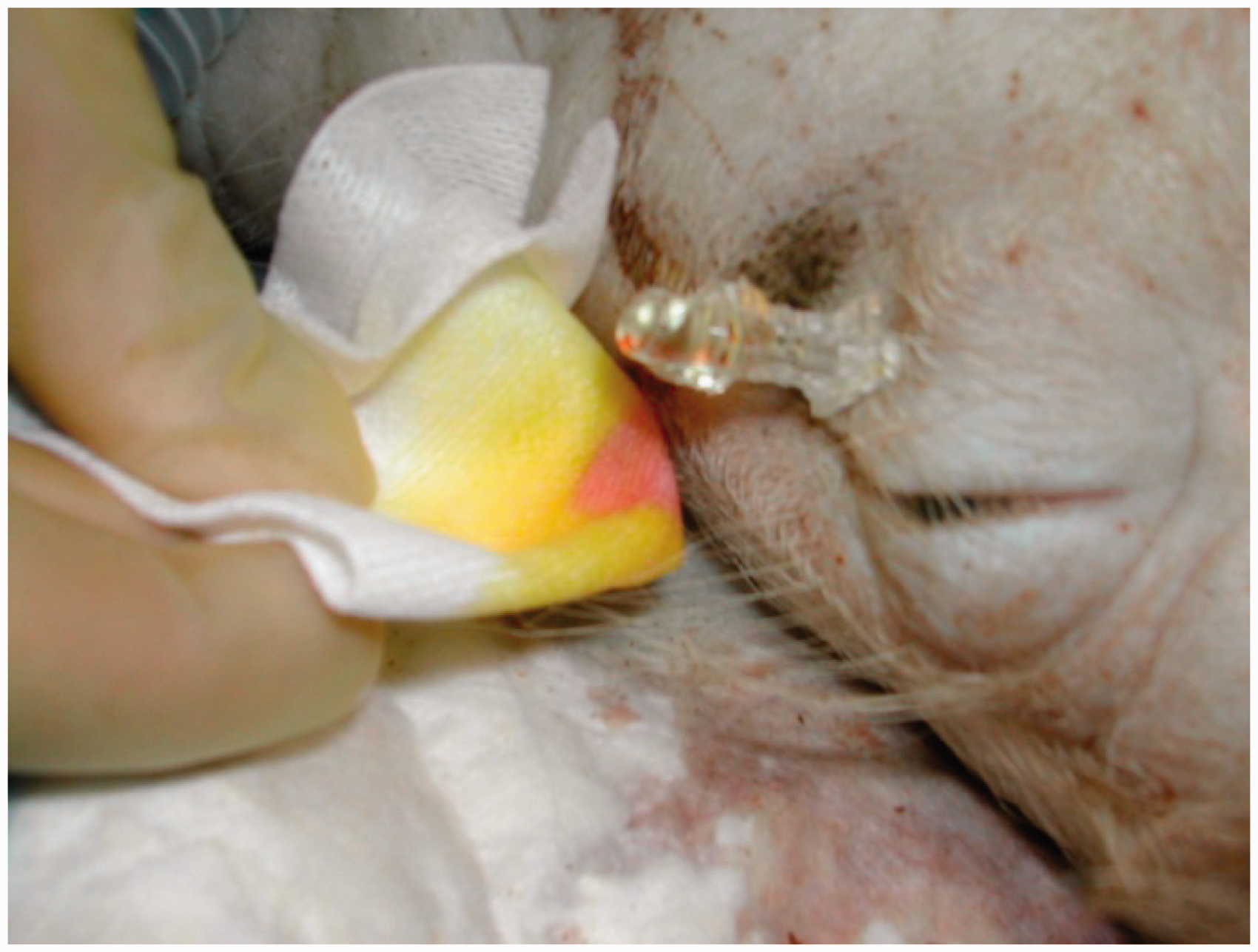
Validation of the SAH model
SAH was verified by macro- and microscopic investigations of the pig brain. In addition, one pig brain was studied by MRI. At the end of each experiment, the skull was opened by craniotomy of the vertex (frontal, parietal and occipital bone) using a Hall® neurairtome (Whittemore Enterprises, Rancho Cucamonga, CA, USA). The brain, cerebellum and brainstem were subsequently dissected free from the dura mater and cranial nerves. Hemorrhage limited by the arachnoid mater was verified macroscopically as the dura mater was incised. Finally, the spinal cord was cut below the brainstem so the cerebrum, cerebellum and brainstem could be removed in one piece and fixated in formaldehyde 20% for further macro- and microscopic examinations.
Histological preparation
Tissue samples were hematoxylin and eosin (H&E) stained and examined by a neuropathologist using a light microscope (Leica DM6000B Research Microscope; Leica Microsystems GmbH, Wetzlar, Germany). Photographs were taken with a digital camera (Leica DFC320; Leica Microsystems GmbH).
Monitoring and measurements
Calibrated transducers (Transpac 3; Abbott Critical Care Systems, Chicago, IL, USA) were used for continuous pressure measurements. Cardiac output (CO) was measured in triplicate, and the results were expressed as the mean value (thermodilution method, 5 mL of normal saline as injection material at <4℃). All measurements were carried out by a single investigator over a period of <4 s at end-expiration (Vigilance Monitor; Edwards Lifesciences LLC).
Hemodynamic measurements were followed by withdrawal of arterial blood samples, which were analyzed immediately at 37℃ for blood gas analysis (ABL 800 FLEX; Radiometer, Copenhagen, Denmark).
The pigs were ventilated using a Servo Ventilator 900 D (Siemens-Elema AB, Solna, Sweden). Positive end-expiratory pressure (PEEP) was set to 5 cmH2O with an inspiration:expiration (I:E) ratio of 1:2. Initial ventilator settings were: minute ventilation of 200 mL/kg/min, respiratory rate of 20/min, and fraction of inspired oxygen (FiO2) 50%. Adjustments were made according to blood gas analysis to achieve a partial pressure of arterial carbon dioxide (PaCO2) within the range of 4.7–6.0 kPa and a partial pressure of arterial oxygen (PaO2) of 12.5–15.0 kPa. Core body temperature was obtained from the pulmonary artery catheter. Temperature was maintained at 38.5 ± 1℃ with the aid of a heating pad or by external cooling as required.
Statistics
All data are presented as mean ± SEM if not otherwise stated. No statistical comparisons were made due to the small number of animals used in the study.
Results
Gross anatomy
SAH after an injection of 15 mL of autologous arterial blood into the subarachnoid space is shown macroscopically in Figure 2a–d. The distribution of blood is evident around the circle of Willis, spreading caudally to the brainstem and pons, into the Sylvian fissure and over both hemispheres and cerebellum. There were no signs of injury to the brain or brainstem parenchyma caused by the spinal needle insertion.
(a) Inferior view of the pig brain after injection of 15 mL of autologous arterial blood into the subarachnoid space. (b) Left lateral view of the pig brain after injection of 15 mL of autologous arterial blood into the subarachnoid space. Cranial (c) and dorsal (d) views of the pig brain showing subarachnoid hemorrhage involving both hemispheres after injection of 15 mL of autologous arterial blood.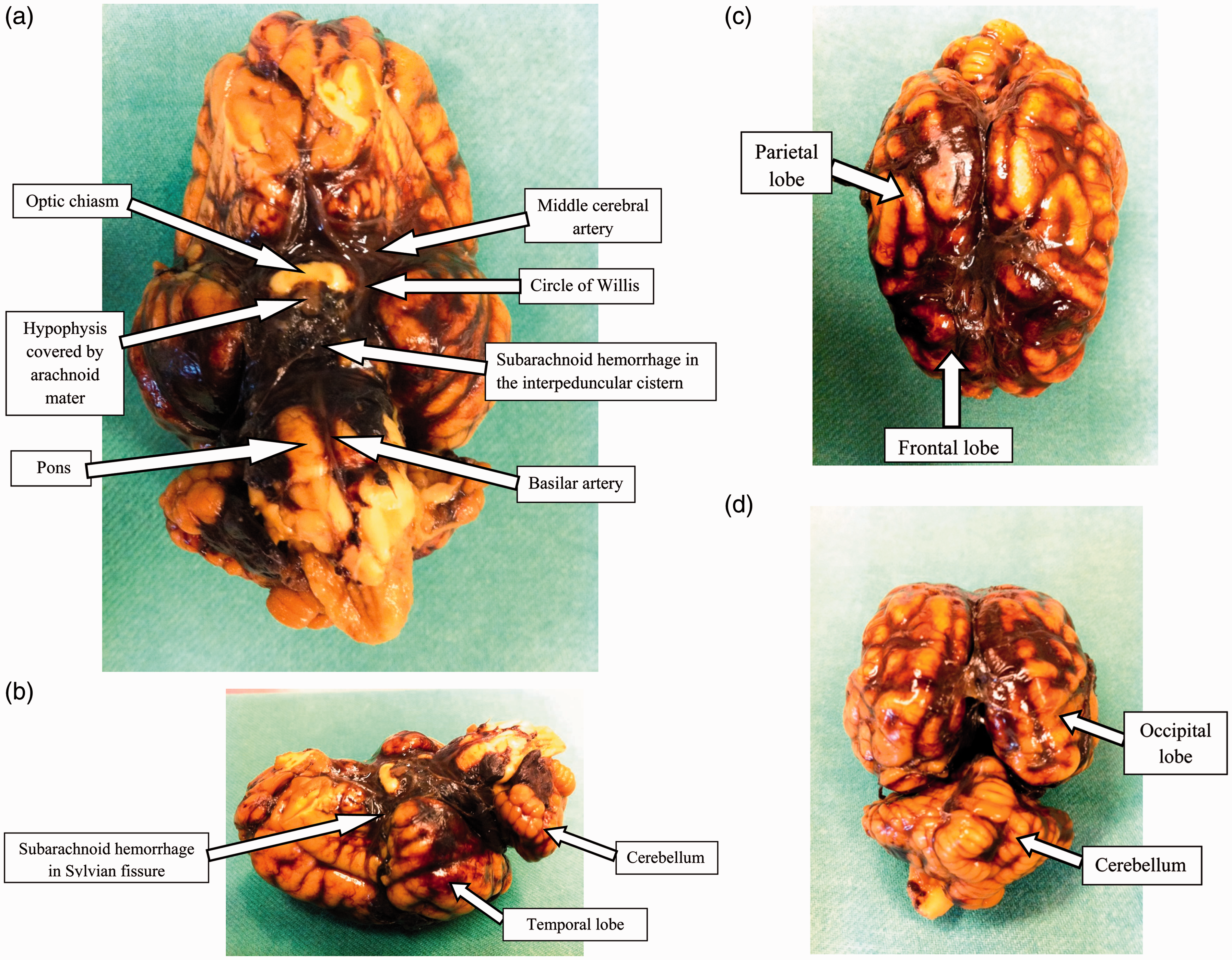
Light microscopy investigations
Figure 3a–c show H&E stained specimens of the left parietal lobe. Extensive hemorrhage consisting of predominantly red blood cells freely distributed in the subarachnoid space between the arachnoid mater and pia mater was observed. In Figure 3a, SAH can be discerned from the intravascular blood in the blood vessels traversing the subarachnoid space. There were also signs of brain edema, as shown in Figure 3c, by vacuolization in the cerebral cortex.
Hematoxylin and eosin stained specimens of the left parietal lobe. (a) Photo showing extravascular blood (predominantly red blood cells) freely distributed in the subarachnoid space. (b) Subarachnoid hemorrhage (SAH) sharply demarcated by the arachnoid mater. (c) SAH demarcated by the arachnoid mater and the pia mater overlying the cerebral cortex. Vacuolization in the cerebral cortex can also be seen as a sign of a developing edema.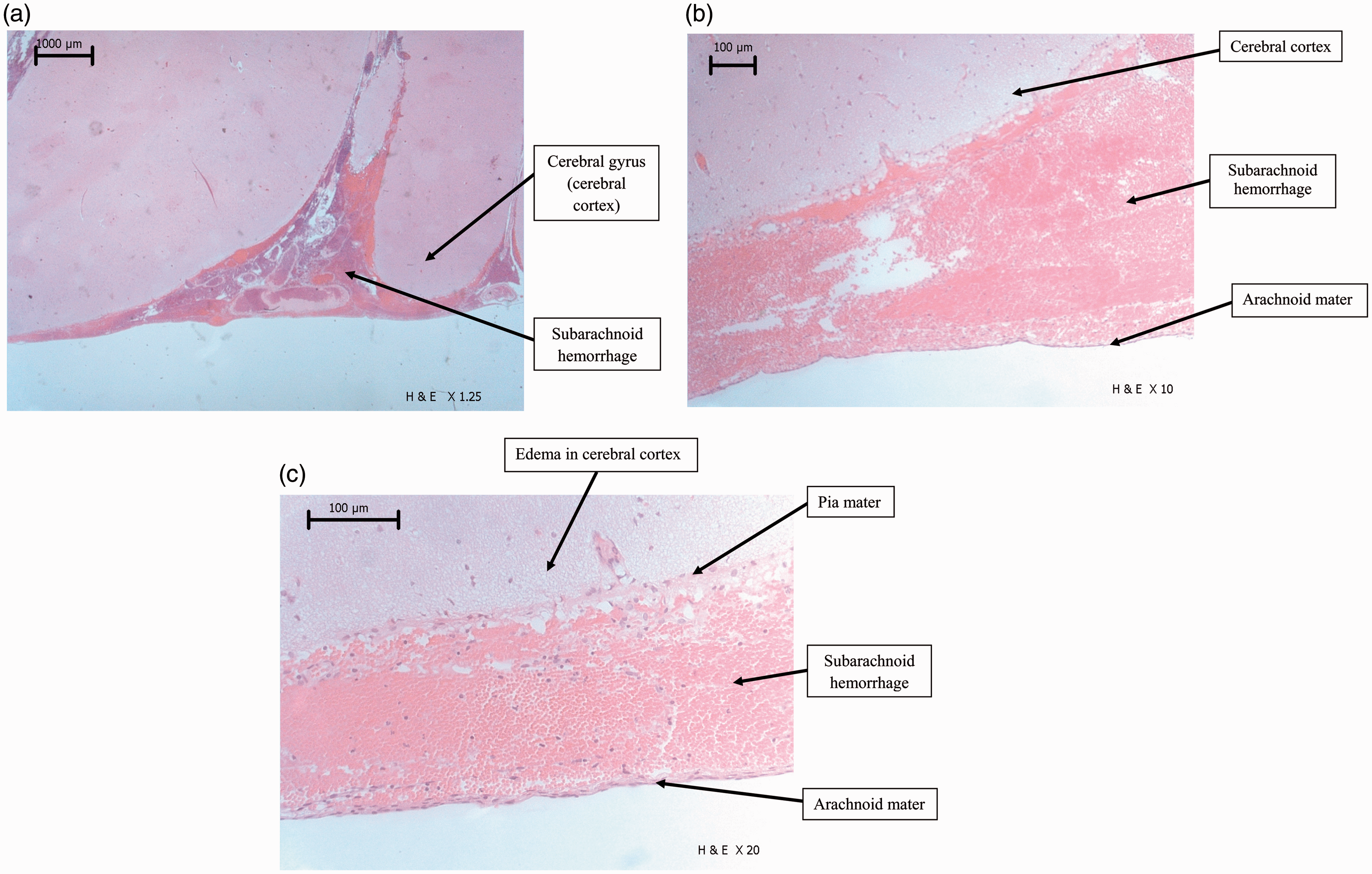
MRI study
The MRI of the pig brain showed massive SAH after injection of autologous blood (Figure 4d–i). However, in these images, some of the hemorrhage could be interpreted as having a subdural location. Hence, a component of subdural bleeding based on the MRI study could not be excluded by our radiologist.
Magnetic resonance imaging (MRI) of the pig brain (fluid-attenuated inversion recovery [FLAIR] images). Three slices are shown at three different time points. (a–c): Baseline images taken before subarachnoid hemorrhage (SAH). (d–f): Images taken 13 min after SAH. (g–i): Images taken 36 min after SAH. In images d–f and g–i blood is seen in the subarachnoid space as white areas around the brain and brainstem and in the ventricles as indicated by straight arrows. Concomitant subdural bleeding over the brain hemispheres, indicated by curved arrows, cannot be excluded.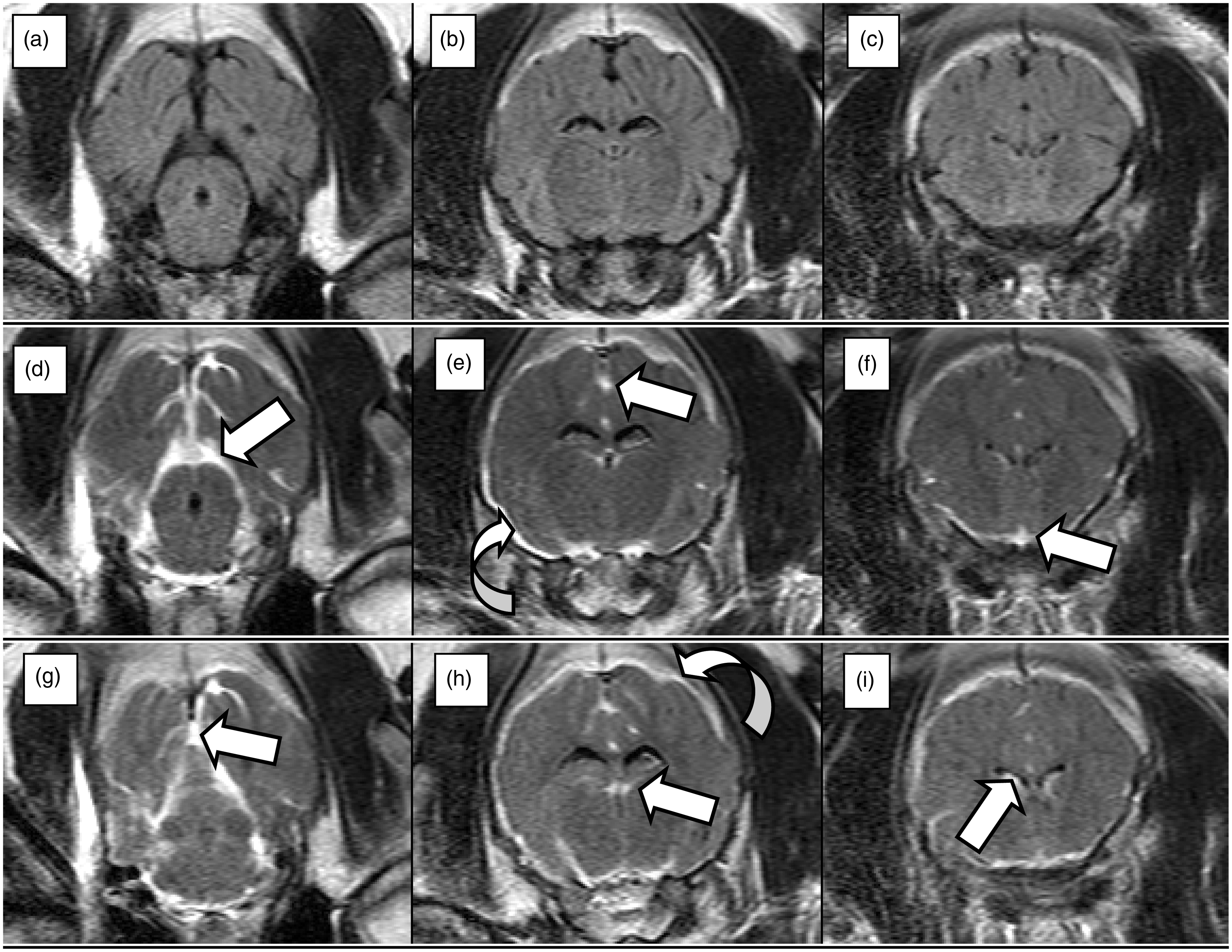
ICP and hemodynamic monitoring
ICP increased and cerebral perfusion pressure (CPP) decreased instantly from the start of blood injection. Despite an increase in mean arterial blood pressure (MAP), CPP was reduced towards zero for a short period of time. The acute increase in ICP and MAP was transient and returned towards baseline values within minutes. Importantly, ICP increased throughout the experimental period while CPP was successively reduced. The latter was due to both increased ICP and reduced MAP (Figure 5a–c). CO was slightly increased following SAH induction (from 3.2 to 4.0 L/min), but gradually decreased towards baseline values up to t = 4 h. Thereafter CO increased slightly towards the end of the experiments, but remained within normal limits throughout the study period. Systemic vascular resistance (SVR) dropped from approximately 2000 dyn × s/cm5 to 1600 dyn × s/cm
5
after SAH induction. SVR continued to decrease during the experimental period, but remained within normal limits. Temperature was kept stable at or slightly below 38.5℃ throughout the study period.
(a) Intracranial pressure (ICP) in pigs with subarachnoid hemorrhage (SAH) (mean ± SEM). (b) Mean arterial blood pressure (MAP) in pigs with SAH (mean ± SEM). (c) Cerebral perfusion pressure (CPP) in pigs with SAH (mean ± SEM).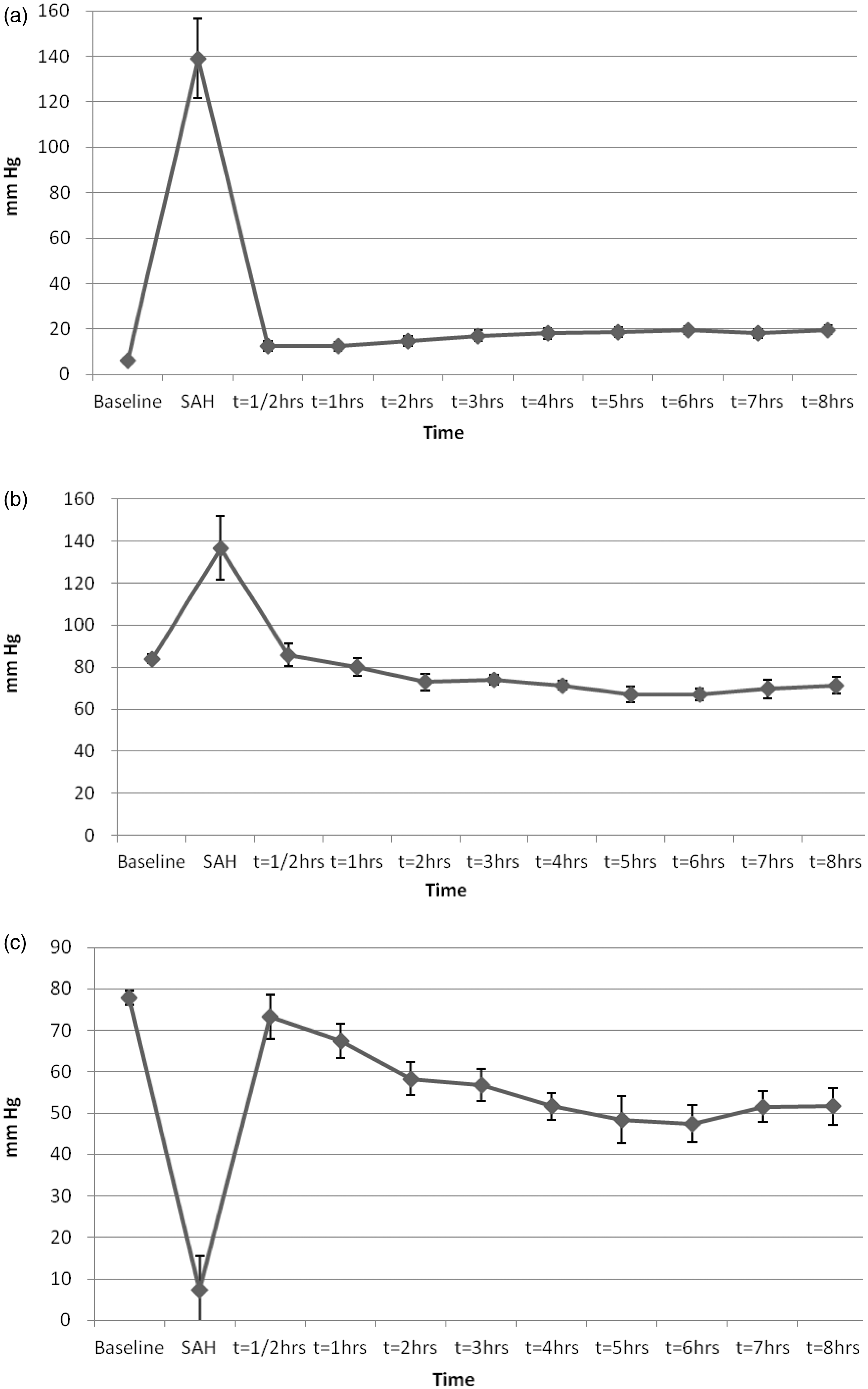
Blood gases, temperature and diuresis
Arterial blood sample data (mean ± SEM).
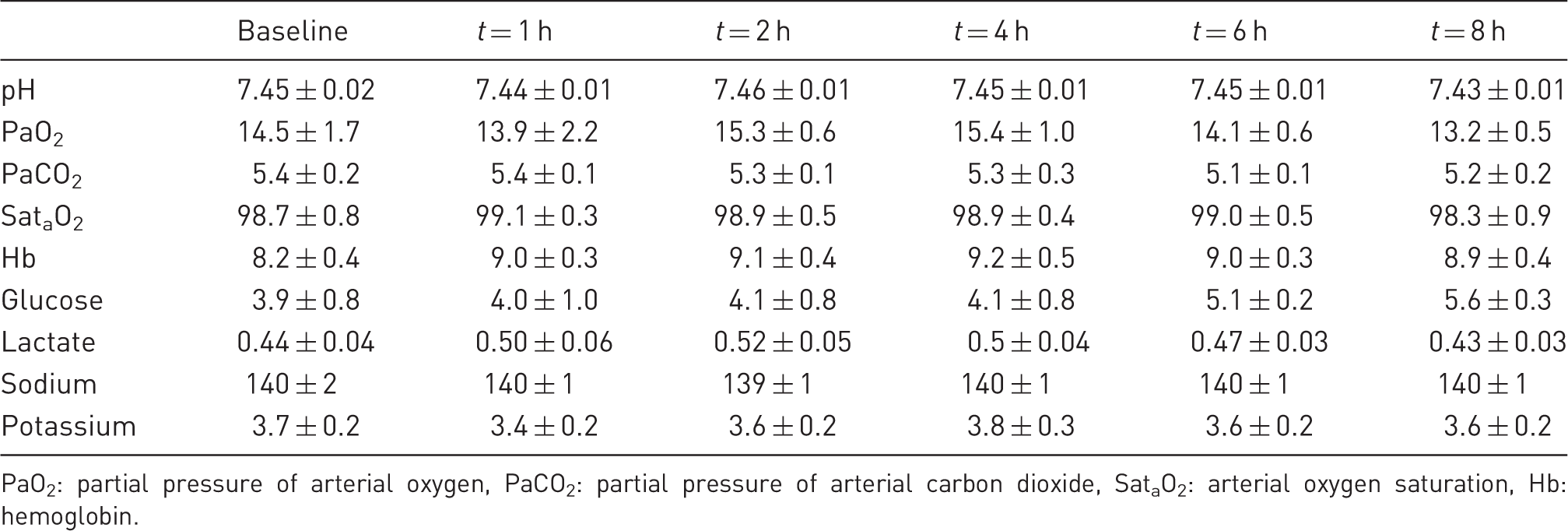
PaO2: partial pressure of arterial oxygen, PaCO2: partial pressure of arterial carbon dioxide, SataO2: arterial oxygen saturation, Hb: hemoglobin.
Discussion
In 1984, Takemae et al. were the first to report a model of cerebral vasospasm in the pig. By performing a C2 laminectomy, a catheter was placed in the prepontine cistern and blood was injected to induce SAH. 11 Later, different porcine models have been developed,12–16 but none of them have gained wide acceptance for use in experimental medicine. 2
Initially, we set out to develop an endovascular puncture model. However, we realized that the pig has a rete mirabile (meaning ‘wonderful net’ in Latin) – a fine-masked capillary plexus interpolated in both the carotid and vertebral artery circulation at the cervicocranial junction – that makes passage of an endovascular catheter impossible.17,18 Accordingly, we decided that developing an injection model was a better alternative.
The subarachnoid distribution of blood was in the region of the circle of Willis, simulating human aSAH caused by rupture of a basilar top or posterior communicating (PCOM) aneurysm. Furthermore, the spread of blood into the Sylvian fissures and over the hemispheres, cerebellum and caudally to the brainstem/pons, closely resembled that of human aSAH (Figure 2a–d). The light microscopy investigations verified hemorrhage confined to the subarachnoid space (Figure 3a–c).
The MRI showed a massive SAH. However, the radiologists could not exclude some concurrent subdural bleeding (Figure 4). When we advanced the 20 G spinal needle into the interpeduncular cistern we made a hole in the arachnoid mater (and dura mater) through which the injected blood could leak into the subdural space. Another explanation for the subdural blood found by MRI was that the pressure of the injected blood might have lacerated the arachnoid membrane and thus created a subdural hematoma (SDH). This is one of the hypotheses for the mechanisms by which ruptured aneurysms can create SDH in humans. 19 Indeed, SAH with concomitant SDH from aneurysm rupture has been reported in humans with an incidence of 0.5–10.3%.19–21
The acute transient ICP elevation and CPP reduction (Figure 5a–c) was in accordance with other experimental SAH models1,22–24 and clinical findings of aneurysm rupture in humans (rebleeding case reports).25–27 A temporary increase in MAP (Figure 5b) accompanying the ICP elevation was also observed and consistent with other models.24,28 After the transient ICP elevation induced by blood injection, ICP remained slightly elevated from baseline values and increased even further throughout the study period (Figure 5a). The edema observed in the cerebral cortex (Figure 3c) may have contributed to the increased ICP. Brain edema is a well-known pathophysiological feature in the course of SAH in both clinical 29 and experimental settings,30–32 and is indicative of ischemia, cell membrane dysfunction, apoptosis and breakdown of the blood–brain barrier. 33 ICP elevation after SAH has also been attributed to the hemorrhage volume, CSF outflow obstruction, diffuse vasoparalysis, and distal cerebral arteriolar vasodilation,25,26,34–37 but we did not specifically target these factors in the present investigation.
MAP decreased throughout the experiment after an initial, but transient increase when SAH was induced. This could be due to vasodilation and a reduction in SVR, which could only partly be explained by the level of anesthesia. An early inflammatory response in the pig causing vasodilation could be another explanation. We could not see any signs of cardiac arrhythmias or ST changes on the electrocardiogram (ECG), or any signs of pump failure that could indicate cardiac dysfunction during these experiments. CO increased shortly after SAH induction and remained higher compared with baseline. Since the heart rate was not significantly raised, the higher CO must have been caused by an increased stroke volume. Rise in sympathetic activity was not specifically measured, but could be a likely explanation behind this observation.
An obvious disadvantage with our model is the lack of a cerebral artery wall puncture, which would mimic aneurysmal rupture in human aSAH. Some studies have indicated that the release of mediators from an artery wall or disturbed perivascular adrenergic innervation might play a role in the pathophysiology causing vasospasm in human aSAH. 38
However, a definite advantage with an injection method is the opportunity to control the volume of blood injected and thus the hemorrhage size and severity of SAH. By contrast, large variations in the hemorrhage volume have been shown in an established endovascular perforation model of the rat. 1 Moreover, unacceptably high mortality rates have been a challenge with all these endovascular models.23,39,40
Another advantage with an injection model is the possibility of running control experiments with injections of artificial CSF instead of blood, making it possible to specifically study the pathophysiological importance of blood components in SAH, controlling for ICP elevation mechanisms. Variations in the injected volume (blood/artificial CSF) would allow us to study different severity grades of SAH. This is of major importance for studying different clinical courses of SAH, which are related to the degree of SAH. 41 Although it might be an opportunity to modify hemorrhage volumes by varying filament/puncture device sizes, 42 the same degree of control and reproducibility would not be possible in vessel puncture methods. 1
Injection of blood into the cisterna magna is a much used SAH model in different animal species.16,43,44 However, human aSAH does not normally occur in the cisterna magna and blood distribution in this model will mainly be in the posterior cranial fossa and spinal canal.16,37,42,45 It has been speculated that this may cause local effects on the brainstem that could create an undue persistent MAP elevation and earlier recovery of cerebral blood flow (CBF), which would make the model unsuitable for acute SAH studies.1,42 Although head-down tilting maneuvers of the animal is performed to make blood spread to the subarachnoid space at the basal parts of the brain, blood distribution in this model does not resemble human aSAH to the same degree as in the injection model we have presented in this paper. Furthermore, great variations of the actual retained blood in the subarachnoid space have been found in a cisterna magna injection method of the rat, probably because blood distributes into the spinal canal. 1
A potential weakness with manual injection models is the operator-dependent variability. Although the injected volume may be fixed, the degree of the acute ICP elevation is determined by the rate of the injected blood. Efforts to control for this include different arterial–subarachnoid space shunt models controlling the bleeding time, 46 computerized pressure-controlled systems, 47 and manual injections regulating the injection speed by aiming at keeping ICP at the same level as MAP during the injection. 28 In most studies, however, the manual injection is performed over a certain period of time – without describing the methodological considerations as to how the specific injection time was chosen. We set up a manual manometric pressure-controlled system, where we were able to measure the injection pressure, aiming to control it at MAP levels. However, using this method, the injection of 15 mL of autologous arterial blood took several minutes and the ICP did not increase to MAP levels. We realized that the diameter and length of the spinal needle caused too much resistance to give sufficient blood flow at the MAP level injection pressure. A needle with a greater diameter could reduce the resistance, but after testing different injection methods we decided to use the method described in the present paper. The injection volume of 15 mL (0.5 mL/kg) of autologous arterial blood was chosen after comparison with other injection models11,48 and after testing the effects of various blood volumes injected.
We wanted to develop an easy and inexpensive technique, without the need for advanced radiological equipment. A disadvantage with a blind introduction of the needle into the subarachnoid space is clearly that it constitutes a potential for damaging brain parenchyma and intracranial vasculature. However, we did not find any signs of parenchymal damage from the needle insertion, except for small hemorrhages in the orbital fat. We cannot totally exclude small intracranial bleedings as traces of blood were seen at the beginning of the CSF tap in some of the animals, but in those cases CSF cleared up instantly and there were no signs of increased ICP until blood was injected. Injury to the hypophysis was difficult to examine because it was stuck in the sella turcica and in most cases ripped off at the pituitary stalk (infundibulum) during brain removal.
The reproducibility of our method with respect to blood distribution and ICP/MAP changes was good. However, the success rate of needle placement in the subarachnoid space/interpeduncular cistern getting a clear CSF tap was only 73%. Hence, in the pigs where we could not find the subarachnoid space a SAH by blood injection could not be performed. We believe that a stereotactic procedure with the aid of MRI or CT techniques might improve the success rate, but this would also increase the costs and complexity of the method. However, we would suggest that further refinement of this model should include some type of imaging techniques to aid the injection procedure.
None of the animals died during the experimental period. This is comparable with other large animal injection models of SAH11,43,49 and is opposed to the high mortality rates in endovascular perforation models.1,23,50 The low mortality rate makes this a suitable method for both acute and chronic experiments. However, one might argue that a higher mortality rate would be desirable when studying the acute phase of SAH to reflect the sudden death rates from aSAH in humans. 51 In future studies, this can possibly be adjusted by considering using a larger injection volume to create a more severe SAH.
In conclusion, this new transorbital injection model of SAH in the pig mimics human aSAH and may be suitable for acute intervention studies. However, the model is technically challenging and needs further validation.
Footnotes
Acknowledgements
We would like to thank the staff at the Surgical Research Laboratory for their kind help and assistance during these experiments. The authors alone are responsible for the content and writing of this paper.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
