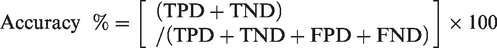Abstract
Pregnancy diagnosis and embryo counting are important end points in reproductive, developmental biology and toxicology studies. The purpose of the present study was to assess the feasibility and efficacy of magnetic resonance imaging (MRI) for early pregnancy diagnosis and embryo counting in the laboratory rat. Female Wistar rats were subjected to whole-body MRI scanning using a 1.5T MRI scanner, employing a isotropic T2-weighted 3D short-tau inversion recovery sequence from day 8 to day 12 post coitum (pc) or without prior mating, under general anaesthesia for pregnancy diagnosis and embryo counting. MRI examination was followed by laparotomy and visual inspection of the uterus to verify MRI findings. By day 8 pc, uterine bulges, characteristic of pregnancy, were depicted as oval-shaped structures of high intensity signal. By day 10 pc, embryonic vesicles were detected at the medial side of the uterine bulges. Pregnancy was diagnosed with 0% false-negative diagnosis and 100% accuracy by day 11 pc, while embryos were counted with 100% accuracy by day 12 pc. In conclusion, MRI proved to be a feasible and reliable non-invasive imaging method of early pregnancy diagnosis and embryo counting in the laboratory rat.
Introduction
The laboratory rat is among the most commonly used animal species in reproductive, developmental biology and toxicology studies. 1 Pregnancy diagnosis and embryo counting are important key end points in such experimental studies. To date, different methods, such as ultrasonography and laparoscopy, have been described for the diagnosis of pregnancy in surviving rat models.2,3 Ultrasonographic examination offers a non-invasive method for diagnosing pregnancy by day 11 post coitum (pc), without, however, providing accurate embryo counting. 2 Laparoscopy, on the other hand, makes accurate pregnancy diagnosis and embryo counting possible by day 7 pc. 3 However, despite the minimally invasive nature of the technique, it results in an inevitable, albeit small, surgical trauma and a short postoperative recovery period.
To this end, the employment of a safe, sensitive and accurate non-invasive method for detecting pregnancy and accurately counting embryos at an early stage of gestation in the laboratory rat would be of great value for researchers. Magnetic resonance imaging (MRI) is a non-invasive imaging method that is considered safe for the human foetus and the development of pregnancy.4,5 MRI has been extensively used in developmental biology and toxicology studies in the rat.6–9 These studies mostly involve MRI scans after day 15 of pregnancy. The aim of the present study was to evaluate the feasibility and efficacy of MRI for early pregnancy diagnosis in the laboratory rat.
Methods
Animals
A total of 78 female Wistar rats (Rattus norvegicus), four months of age, provided from our laboratory’s in-house breeding colony, were used in the study. Colony health status was monitored semi-annually, and rats were found to be free of Mycoplasma spp., adventitious viruses, respiratory and enteric bacteria, as well as ecto- and endoparasites. Rats were housed in polycarbonate cages (Techniplast, Buguggiate, Italy), three rats per cage, in an enriched environment at 20–22°C room temperature on a 12-hour light/12-hour dark cycle. They were provided with nest-building material and were given a commercial pelleted diet (certified rat chow; VIOZOIS S.A., Thessaloniki, Greece) and tap water ad libitum. The breeding facility is licensed by the prefectural veterinary authorities, since it complies with the requirements set by the national legislation on the use of animals for scientific purposes (PD 63/2013) which is in accordance with Directive 2010/63/EU.
Experimental design
Mated rats were randomly allocated to five groups, 13 rats per group, and subjected to whole-body 1.5T MRI scanning under general anaesthesia on days 8, 9, 10, 11 or 12 pc (one group per day) in order to examine their uterus in a blinded fashion for pregnancy and to count the embryos in case of a positive pregnancy diagnosis. Following examination, animals were subjected to direct inspection of their uterus after midline laparotomy by an observer blinded to the MRI results in order to verify the MRI findings on the assumption that visual examination of the uterus provides the most accurate diagnosis of pregnancy as defined by the presence of uterine bulges at this stage of pregnancy. A separate group of control animals (n=13) without prior mating was also subjected to MRI to depict the non-pregnant uterus and also to assess the effect of the procedure on maternal body weight, an index of animal welfare. Dimensions of the uterus and embryos were recorded. Taking into account the relatively low resolution of 1.5T MRI sequences, measurements were made after proper zooming of the figure to achieve the best possible accuracy. Sensitivity of MRI for pregnancy diagnosis was evaluated on the basis of the percentage of false-negative diagnosis, while the accuracy of the method was calculated according to the following formula:
The accuracy of the method regarding embryo counting was calculated according to the following formula:
Accuracy %=100%–(overestimated embryo counts %+underestimated embryo counts %).
Finally, animals subjected to laparotomy were euthanised by exsanguination. The experimental protocol was approved by the Animal Care and Use Committee of our research facility and licensed by the prefectural veterinary authorities, since it complied with the requirements set by the national legislation on the use of animals for scientific purposes (PD 56/2013) which is in accordance with Directive 2010/63/EU.
Mating
Each female rat was caged with a single male rat of verified fertility until successful copulation. The latter was confirmed on the following morning either by detection of a vaginal plug or detection of sperm in a vaginal smear. Smears were examined on an optical microscope (KF2; Carl Zeiss, Hamburg, Germany) at a magnification of 10×. Examination was performed on a daily basis (between 9:00am and 11:00am) until mating confirmation. The day of mating confirmation was defined as day 0 pc. Female rats were kept singly housed after confirmation of successful copulation.
MRI
Rats were subjected to general anaesthesia by intramuscular injection of a xylazine (5 mg/kg)–ketamine (90 mg/kg) mixture to facilitate the examination procedure. They were then secured at dorsal recumbency in an eight-channel phased-array knee coil (SENSE Knee Coil 1.5T/8Ch; Invivo Corp., Gainesville, FL; Figure 1). A MRI scan of the whole body was performed using a 1.5T MRI scanner (Philips Multiva 1.5T MRI system, Class IIa, 2014; Philips Koninklijke, The Netherlands) without intravenous infusion of any contrast agent, employing an isotropic T2-weighted three-dimensional short-tau inversion recovery (3D STIR) sequence (Table 1). Images were analysed using a CDViewer software provided by Philips. Images were reconstructed in coronal and axial planes. A 3D radial plane of maximum intensity projection proved to be helpful in recognising anatomical features of the pregnant uterus.
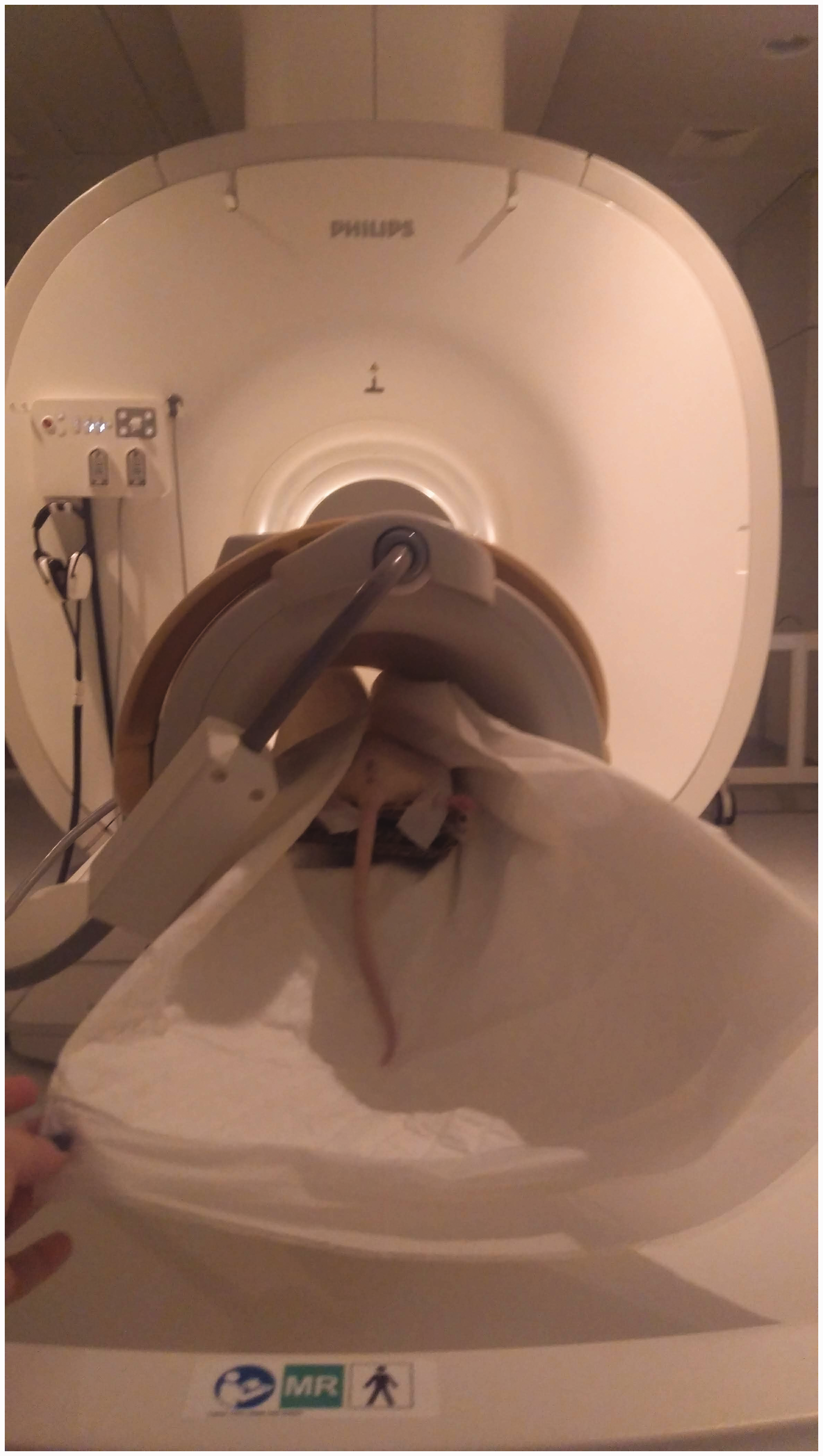
An anaesthetised rat secured at dorsal recumbency in a knee coil before being subjected to magnetic resonance imaging (MRI) for pregnancy diagnosis.
Technical characteristics of 3D STIR sequence when performing MRI scanning for pregnancy diagnosis in rats.

3D STIR: three-dimensional short-tau inversion recovery; ACQ: acquisition; FOV: field of view; MRI: magnetic resonance imaging; NSA: number of signal averages; REC: reconstruction; SAR: specific absorption rate; SED: specific enegy dose; SNR: signal to noise ratio; TE: time to echo; TI: time of inversion; TR: time of repetition; TSE: turbo spin echo.
Statistical analysis
Before the beginning of the study, a sample size calculation was performed with an 80% power and an error set at 0.05 (one sided). According to the estimation, 13 rats per group were required in order to detect a difference of one embryo with one standard deviation between MRI and laparotomy. Data are presented as means±standard deviations.
Results
The normal non-pregnant uterus was not clearly depicted in MRI images. There was no effect on body weight in control animals during a three-day post-examination period (data not shown). By day 8 pc, uterine bulges of pregnant rats were depicted as oval-shaped structures of high signal intensity in a row-of-beads layout (Figure 2). The Y-shaped uterus lay on either side of the midline, with each horn extending from the level of the ovary, caudolaterally to the kidneys, to the uterine body at the level of the urinary bladder. The size of bulges increased with pregnancy development (Table 2). By day 10 pc, embryonic vesicles were detected as oval-shaped structures within the uterine bulges, with a signal of higher intensity (Figure 2). Each embryonic vesicle lay at the medial side of the uterine bulge (Figure 2), and its dimensions increased with pregnancy development (Table 2).
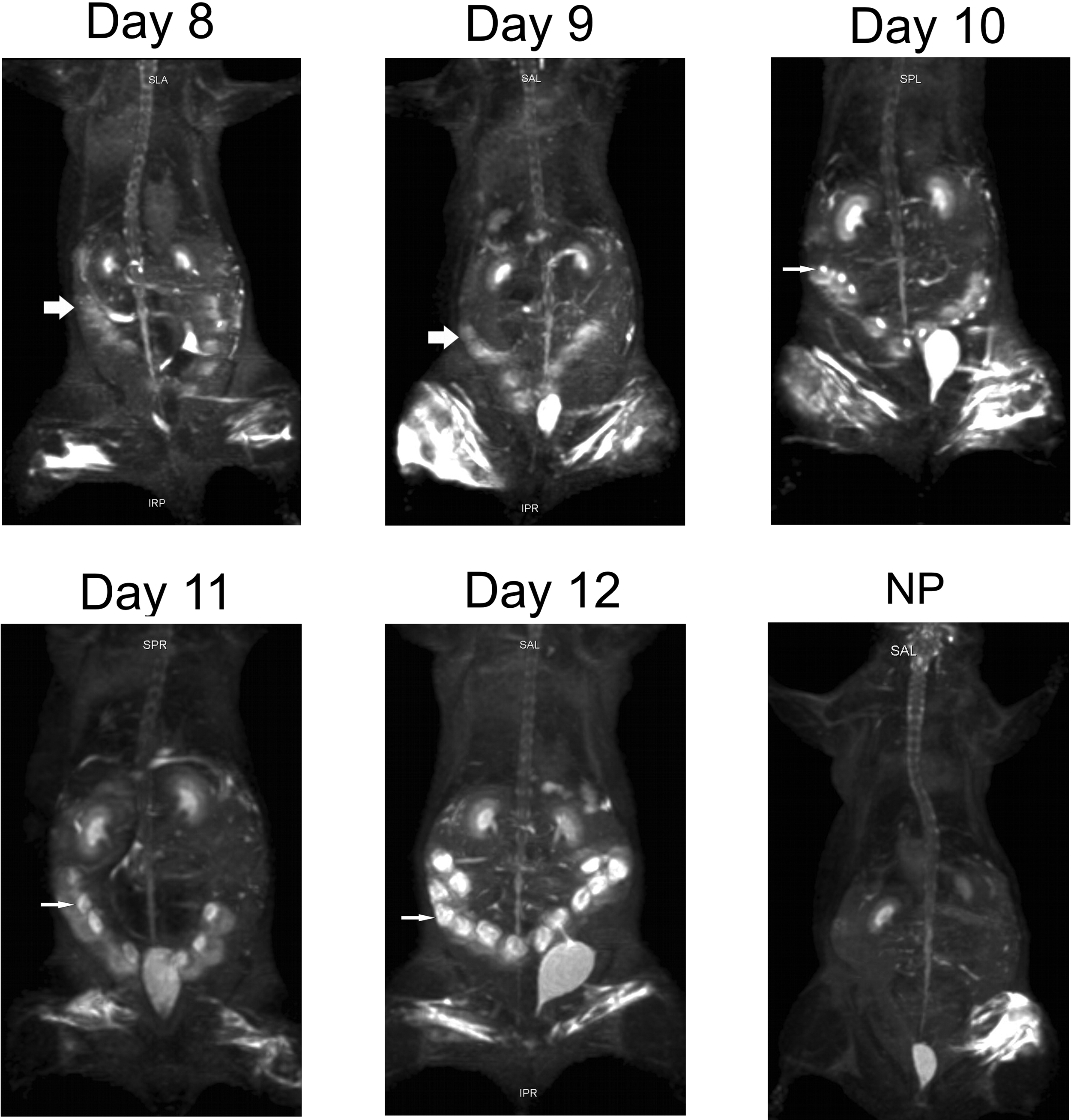
MRI images (coronal plane) of pregnant and non-pregnant (NP) rats. Uterine bulges (thick arrows) were depicted by day 8 pc. Embryonic vesicles (thin arrows) were detectable by day 10 pc. pc: post coitum.
Dimensions (means±standard deviations) of uterine bulges and embryonic vesicles as depicted by MRI during pregnancy in the rat.
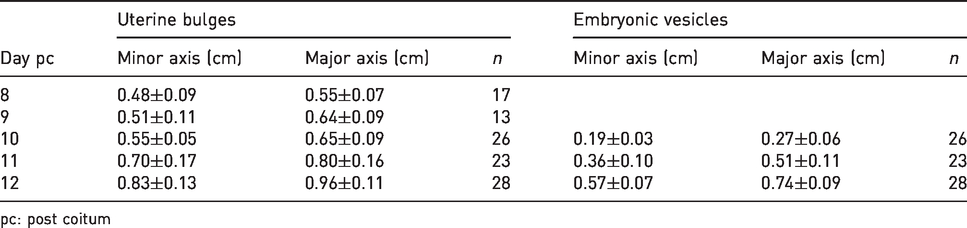
pc: post coitum
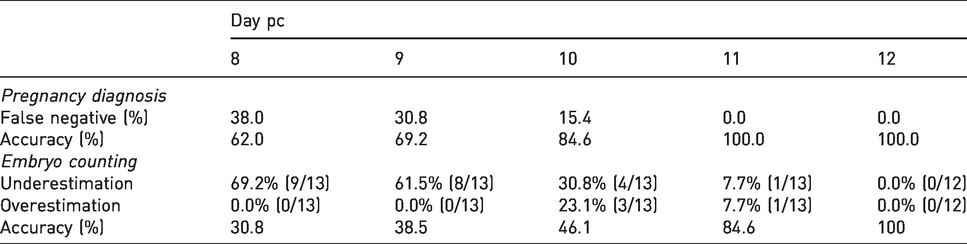
Sensitivity: accuracy of the method
Pregnancy was diagnosed with 0% false-negative diagnosis and 100% accuracy by day 11 pc. Embryos were counted with 100% accuracy by day 12 pc (Table 3).
Sensitivity and accuracy of MRI in pregnancy diagnosis and embryo counting in laboratory rats.
Discussion
The present study assessed the feasibility and efficacy of MRI for early pregnancy diagnosis in the laboratory rat. Pregnancy diagnosis and embryo counting with 100% accuracy were possible by days 11 and 12 pc, respectively, in anaesthetised mated rats subjected to whole-body MRI scanning.
For the purpose of our study, rats were subjected to a fast (3.5-minute) whole-body MRI scan using equipment manufactured for human use established in a university hospital facility. The employed isotropic T2-weighted 3D STIR sequence offered a three-dimensional study of the pregnant uterus that facilitated the detection of anatomical features. The selected MRI sequence effectively nulled fat signal, which led to enhanced conspicuity of tissues embedded in adipose tissue, and provided details of anatomical structures containing a liquid component. 10 The examination was feasible without the need for intravenous infusion of gadolinium-based contrast agents that could adversely affect the embryos. 11
MRI provides unique clinical images of high soft-tissue contrast by using non-ionising radiation. The safety of MRI scanning during pregnancy has been extensively investigated under both clinical and experimental conditions. The assessment of risk from MRI to the foetus is complex, since different factors, such as variable MRI field strength, gradient strength and RF pulse sequences, have to be taken into account. 12 With evolving technical progress in MRI technology, increasingly higher intensities of magnetic fields are applied to human imaging. Short-term exposure of pregnant mice to static magnetic field strengths ranging from 1T to 6.3T13–16 had no adverse effect on embryogenesis, while long-term exposure to 10T caused a decrease in foetal weight at birth and an increase in the number of foetal resorptions. 17 On the other hand, when mice were exposed to all three components of MRI, adverse effects on embryogenesis and foetal growth became evident at even lower magnetic field strengths. A reduction in foetal growth was reported after exposure to 1.5T 18 or 4.7T 19 on days 7, 9 or 12 pc, and an increased percentage of malformations was revealed after exposure for 36 minutes to 1.5T on day 7 pc 20 or for one hour to 0.5T on day 8 pc. 21 Other animal studies, however, reported no effects on embryogenesis, foetal growth or embryo mortality after performing MRI up to 4T.22–26 Therefore, there is inconsistency on the reports regarding the safety of the foetus subjected to MRI.
The American College of Radiology and the Society for Pediatric Radiology recommends no special consideration for use of MRI at any trimester in pregnancy, since there is no conclusively documented evidence for deleterious effects of MRI at 1.5T, such as the one we employed in our study, on the developing foetus. 5 The US Food and Drug Administration and the International Commission on Non-Ionizing Radiation Protection recommend the performance of studies with caution, including a close control of Specific Absorption Rate (SAR) values. In the UK, the Medical Device Agency guidelines require that the SAR value should not exceed 10 W/kg within the foetus. 27 To this end, it is recommended that foetal MRI examination should be undertaken when benefits outweigh potential risks, while care should be taken regarding the timing of exams and SAR limits.5,26,28,29
Uterine bulges were detectable in MRI images by day 8 pc, although with accuracy by day 11 pc. These are characteristic anatomical features of pregnancy in multiparous species and result from the progressive enlargement of the descended into the uterine lumen blastocysts which begin to increase in size on day 6 pc in the rat. 30 Embryos were depicted in MRI images by day 10 pc, while accurate embryo counting was possible by day 12 pc. Laparoscopy has been reported to achieve embryo counting as early as day 7 pc, on the assumption, however, that each uterine bulge detected contains an embryo. 3 Embryos can also be detected ultrasonographically by day 11 pc, albeit without accurate estimation of their number. 2
According to embryological data, rat embryos attain a crown–rump length of 0.2 cm on day 11 pc and 0.45 cm on day 12 pc. 30 These dimensions were similar to the major axis of embryonic vesicles reported in the present study. Images were reviewed using appropriate imaging analysis software at a three-dimensional pattern that facilitated observation and measurement of anatomical structures.
A ketamine–xylazine combination was used for anaesthetising the animals during MRI scanning in order to provide adequate immobilisation of the animals and to reduce motion artefacts. A mixture of ketamine and xylazine is commonly used in pregnant rodents, providing satisfactory anaesthesia, 31 and they are among a range of injectable drugs used for imaging studies. 32 Although the specific anaesthetic regime met the experimental protocol’s requirements, developmental toxicity studies in mice have revealed a reduction of foetal growth. 33 Factors including hypotension, hypoperfusion or hypoxia during the anaesthetic period may contribute to this adverse effect. Ketamine has also been implicated in neurotoxic effects in the developing foetal brain. 34 Other anaesthetic agents, such as isoflurane or tribromoethanol, have been proposed as safer alternative anaesthetic agents with regard to embryo development in survival rodent toxicity models.33,35,36 For procedures lasting more than around 10 minutes, provision of an external form of heat or wrapping of the animal or its extremities with an insulator, such as a plastic wrap, is advised in order to maintain normal core body temperature.32,35
The cost of purchase of MRI equipment is high. Special MRI scanners for rodents have been designed and are available on the market. In the present study, we demonstrated the feasibility of a rat MRI examination using a MRI scanner manufactured for human use, suggesting that a collaboration between laboratories could waive the cost of purchase provided that all necessary hygiene considerations have been properly addressed.
The use of MRI, a safe non-invasive imaging method suitable for longitudinal studies of pregnancy in the rat, contributes to the promotion of Reduction and Refinement, two of the 3Rs principles regarding the use of animals for scientific purposes. In conclusion, in this study, MRI scanning provided a sensitive and reliable imaging method for early pregnancy diagnosis and embryo counting in the laboratory rat that could be used in developmental and reproduction toxicology studies.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship and/or publication of this article.