Abstract
The vaginal epithelium of the adult female laboratory rodent changes from mucous secretion to cornification over the course of the estrous cycle. The morphophysiological changes occur with such regularity, accuracy and precision that the specific stage of the estrous cycle in the rat can be determined by inspection of the vaginal opening and/or exfoliative vaginal cytology. However, in the mouse, post-mortem vaginal histology is often required to determine the estrous cycle stage for ensuring the required level of reliability. Consequently, an excess number of female adult mice are needed to allow for the delivery of sufficient numbers of mice in a desired estrous cycle stage. In this study, we demonstrate that the standard procedure for oocyte superovulation and collection in the laboratory mouse (e.g. injection of equine chorionic gonadotropin followed 48 h later by human chorionic gonadotropin) can also be reliably used to induce changes in the epithelium of 3.5-week-old mouse vaginas in an estrous cycle stage-specific manner (e.g. establishment and replacement of a mucous secreting epithelium with a cornified epithelium; induction of cornification-associated loricrin expression). The superovulation protocol thus allows for the efficient and economic induction of estrous cycle stage-specific characteristics in the Müllerian duct-derived vagina thereby avoiding the necessity of post-mortem identification of the estrous cycle stage. In addition, our study indicates that the laboratory mouse vagina is an excellent organ for studying the sequence of events leading to cornification.
Non-pregnant females usually experience a cyclical sequence of morphological changes in their reproductive organs during their reproductive years. These changes are governed by their internal biological clock and the hypothalamo–hypophyseal–gonadal axis. After puberty, the neuronal centers cyclically drive the development of the late antral follicles and corpora lutea in the ovaries and evoke fluctuations in the synthesis of the steroid hormones estradiol and progesterone. Estradiol levels are high in the late follicular and luteal phases, while progesterone levels peak during the mid-luteal phase of the estrous cycle. Ovarian steroid hormones cause morphological changes in Müllerian duct-derived organs (i.e. fallopian tube, uterus and upper part of the vagina).1–3
In rodents, the female reproductive cycle lasts approximately 4–6 days. Nevertheless, the gross anatomy, histology and cellular composition of the reproductive organs change in such remarkable regularity, accuracy and precision that some of these changes such as vaginal opening and/or the proportion of cell types in the vaginal secretion in rats can be used to identify the specific stage of the estrous cycle (i.e. diestrus, proestrus, estrus and metestrus).4–10
Even under the stress of routine animal facility life (e.g. light, noise, cage cleaning and in-house transport), adult female rats present regular and successive reproductive cycles.7,11–14 In addition, laboratory rats are easy to handle with regard to visual observation of their vaginal opening and robust enough for repeated vaginal swabs. By contrast, reliable estrous cycle monitoring in living mice is technically more difficult and is interference prone. 15 The degree of invasiveness necessary to perform the vaginal swabs can alter the reproductive status and elicit an inflammatory response that can confound the cytological evaluation. 16 Consequently, experimental animal data are preferably collected in female rats when estrous cycle stage-specific morphophysiological conditions are essential considerations in the experimental protocol. However, the increasing use of mouse lines and genetic mouse models makes having a practical and reliable approach for correlating the anatomical and physiological changes which occur during the course of a mouse estrous cycle with other parameters essential.17,18
Combined equine chorion gonadotropin (eCG) and human chorionic gonadotropin (hCG) injections (i.e. superovulation protocols) have been used for over half a century to economically and efficiently induce high numbers of fertilizable oocytes in mice.19–22 In this study, a superovulation protocol that can be reliably used to induce morphological transformations in the vaginal tubes of immature 3.5-week-old mice comparable with those observed in the vaginal tubes of adult female mice during the natural estrous cycle was demonstrated.
Materials and methods
Animals
C57BL/6NCrl mice from an inbred stock were used in this study that was originally obtained from Charles River Laboratories (Cologne, Germany) in 2011 and further propagated at the Medizinisch-Experimentelles Zentrum der Medizinischen Fakultät Leipzig. The mice were bred and maintained in groups (not larger than 5 animals/group) under specific pathogen-free conditions (for information on excluded pathogens and corresponding methods used, see Supplementary Materials [Table S1] found online at http://lan.sagepub.com) in individually-ventilated cages (Tecniplast, Hohenpeißenberg, Germany) at the centralized animal care facility, where humidity, temperature and lights were automatically controlled (12 h light/12 h dark). Breeding pairs (and during weaning) were fed an energy-enriched diet (ssniff M-Z Ereich; ssniff Spezialdiäten, Soest, Germany. For information on composition, see Supplementary Materials [Figure S1] online). At three weeks of age, the offspring were separated by gender and fed a different diet (ssniff R-Z autoklavierbar; ssniff Spezialdiäten. For information on composition, see Supplementary Materials [Figure S1] online). All mice had free access to water. Hygienic status of the mice was monitored twice a year according to recommendations of the Federation of European Laboratory Animal Science Associations (FELASA).
All animal experiments were conducted in accordance with European Directive 2010/63/EU on the protection of animals used for scientific purposes and were performed with permission from the Animal Care and Use Committee (ACUC #T07/13) and the Government of the State of Saxony, Germany.
Immature 3.5-week-old female C57BL/6NCrl mice were used for all experiments in which vaginal epithelial remodeling during eCG/hCG superovulation was studied. In all other experiments, specimens of spontaneously cycling adult female C57BL/6NCrl mice were employed as described below. Mice were euthanized by carbon dioxide exposure.
Tissue samples and animal numbers
Archival formalin-fixed paraffin-embedded ovaries and vaginas of untreated C57BL/6NCrl mice in different phases of the estrous cycle as verified by vaginal histology from previous studies were available at the Institutes of Anatomy and Biochemistry in Leipzig.23,24 This material was used to demonstrate estrous cycle stage-specific changes in hematoxylin and eosin (H&E)-stained sections of ovaries and vaginas of adult cycling C57BL/6NCrl mice. In order to clarify whether gonadotropin stimulation can provoke comparable anatomical changes in the ovaries and vaginas of immature mice, 35 female C57BL/6NCrl mice (3.5 weeks old) were used. The immature mice were randomly assigned to either two control groups (5 mice/group) receiving no stimulation during the experimental period or to five experimental groups (5 mice/group) receiving gonadotropin stimulation according to the protocol below.
Ovarian stimulation
Gonadotropin-induced ovarian stimulation basically followed a widely used protocol that has been proven to be efficient for inducing ovulation and producing viable embryos for more than half a century.19,25–27
The mice in each experimental group were subdivided into three mice which were euthanized 24, 40 and 48 h after intraperitoneal injection of five international units (IU) of eCG/mouse (Sigma Aldrich, Munich, Germany) at 15:30 to 16:00 h, and two mice which were first intraperitoneally injected with 5 IU eCG/mouse and subsequently injected intraperitoneally 48 h later with 5 IU hCG/mouse (Sigma Aldrich). These two mice were euthanized 15 and 48 h post-hCG injection. For comparison, one mouse of each control group was euthanized at each of the time points mentioned in the stimulation protocol.
Vaginal histology
Vaginas and ovaries were surgically excised from the pelvic floor and fixed in 4% formalin for 24 h at various time points after stimulation with gonadotropins. Subsequently, the tissue was rinsed, dehydrated in an ascending graded alcohol series, immersed in xylene and embedded in paraffin wax. The paraffin blocks were cut on a sliding microtome into 7 µm thick sections. The sections were rehydrated in xylene and a descending alcohol series before staining with H&E. The histology of the sections was evaluated under a microscope (Axioplan 2, Zeiss, Jena, Germany), photographed with a ProgRes C3 digital camera (Jenoptik, Jena, Germany) and documented with a digital recording system (ProgRes CapturePro 2.8.8; Jenoptik).
Immunhistochemical detection of loricrin
The structural protein loricrin is involved in the formation of the cornified envelope in the skin.28,29 Loricrin is also present during cornification of rodent vaginal epithelia. 30 The rabbit polyclonal anti-loricrin antiserum (ab85679; abcam, Cambridge, UK) recognizes an epitope within residues 450 to the C-terminus of human loricrin. 31
Deparaffinized and rehydrated tissue sections were microwaved in 100 mM citrate buffer, pH 6 for 5 min and endogenous peroxidase activity was quenched by incubating the sections in a solution containing 3% hydrogen peroxide and 10% methanol for 30 min. Thereafter, tissue sections were blocked in phosphate-buffered saline (PBS) containing 1.5% normal goat serum for 30 min at room temperature and incubated overnight at 4℃ in a humidified chamber with anti-loricrin antiserum diluted 1:200,000 in PBS containing 5% bovine serum albumin. These sections were washed three times with 50 mM Tris HCl, pH 7.6 containing 150 mM NaCl (Tris-buffered saline [TBS]) and incubated with 0.5% biotinylated goat anti-rabbit IgG antibody (B-1000; Biozol, Eching, Germany) diluted in PBS containing 5% bovine serum albumin for 30 min at room temperature. Following three more washes with TBS, binding of the secondary antibody to the anti-loricrin antiserum-labeled tissue sections was visualized with avidin-biotin-horseradish peroxidase complex (Vectastain ABC kit; Biozol) and diaminobenzidine (DAB) solution. The nuclei were stained with Mayer’s hemalaun for 10 s and the sections were mounted in Roti®-Histokitt (Roth, Karlsruhe, Germany). Specificity of loricrin staining was assessed by pre-absorbing the primary antiserum with a 100-fold excess of loricrin peptide (ab95418; abcam), omitting the primary antiserum in the staining procedure and using the dorsal skin epidermis as positive control tissue. 30 Immunostained sections were evaluated, photographed and documented as described above for H&E-stained sections.
Determination of estrous cycle stages
H&E staining of the vaginal epithelium was used to determine the specific stage of the estrous cycle in the spontaneously cycling mice at the time of euthanization. Previously published and widely accepted criteria were employed.6,8,10,23 The criteria for the diestrus stage were the presence of a small, stratified, non-cornified epithelium with numerous proliferating cells in the basal layers. The proestrus stage was characterized by a stratified non-cornified epithelium with one to two extensively mucified superficial layers of cuboidal to cylindrical mucinous cells. The estrus stage was identified by the presence of a tall stratified squamous epithelium with prominent cornification of the superficial layers. Finally, the criteria applied for determining the metestrus stages I and II were the presence of a stratified epithelium with slightly to massively invading and transmigrating leucocytes and incomplete to complete exfoliation of the cornified layers.
Results
Similarity between natural and gonadotropin-treatment-induced ovarian activities
Normal preovulatory follicle development and corpus luteum formation were observed in the ovaries harvested from spontaneously cycling adult mice (Figure 1a and b). In the ovaries of immature mice, sequential treatment with eCG and hCG induced an increase in organ weight, stimulated follicular maturation and brought about multiple ovulations (Figure 1c–e). Corpora lutea were formed at the site of the ovulated follicles. At the end of the combined gonadotropin treatment period, numerous corpora lutea were present. Overall, the coordinated structural changes in the stimulated immature murine ovaries were comparable with those seen in the ovaries of spontaneously cycling adult mice.
Ovaries collected from adult mice at distinct stages of the estrous cycle contain larger antral follicles at the proestrus stage (stars in a) and well-developed corpora lutea at the metestrus I stage (rectangles in b) than those found during the luteal and follicular stages, respectively. Injection of equine chorionic gonadotropin (eCG) alone (c) or followed by human chorionic gonadotropin (hCG) injection (d, e) is a reliable method for inducing terminal follicular maturation (large antral follicles in c, stars), superovulation (freshly ovulated follicles in d, triangles) and corpus luteum formation (organized corpora lutea in e, rectangles) in 3.5-week-old immature female mice (see Materials and methods for details). Large antral follicles and corpora lutea are not present in the ovaries of littermates not injected with gonadotropins (f). Sections are stained with hematoxylin and eosin (H&E). Scale bars as indicated.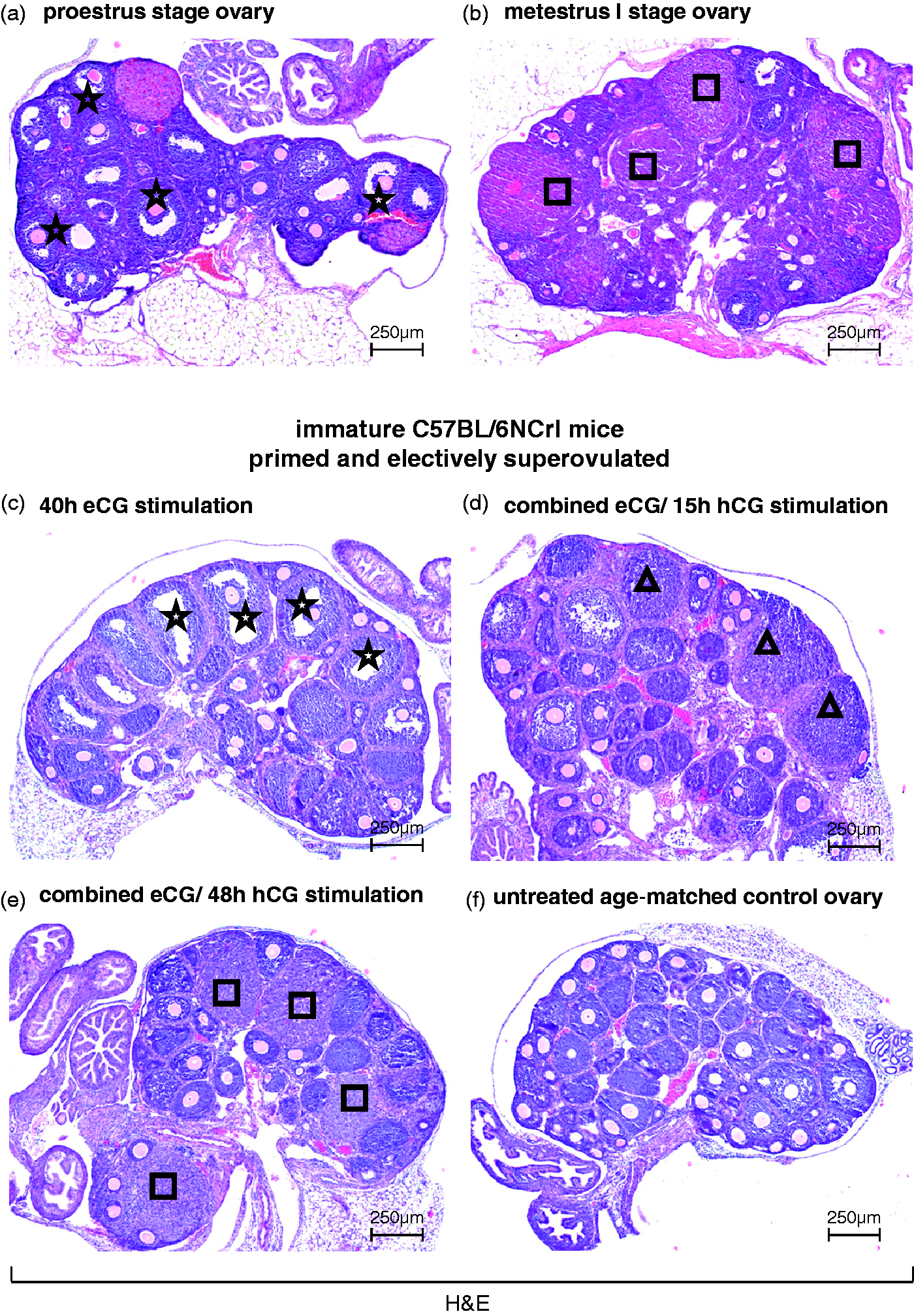
Similarity between estrous cycle and gonadotropin-treatment-induced vaginal changes
The histological changes which occurred in the vaginas of spontaneously cycling adult mice and those caused by the gonadotropin-induced immature murine ovaries were compared.
The vaginal epithelium of the mature female C57BL/6NCrl mice showed all the structural differences expected in the vagina of the spontaneously cycling mouse during all four stages of the estrous cycle. Each mouse examined could be assigned to one of the four stages of the estrous cycle based on the characteristic changes in the vagina (Figure 2a). Vaginas with small stratified non-cornified epithelium and plump polygonal proliferating cells were assigned to the diestrus stage. Those with a stratified non-cornified epithelium with an outer superficial layer of mucified cylindrical cells were referred to as the proestrus stage. Vaginas with a tall stratified squamous epithelium with a broad stratum corneum and a complete loss of mucified cells were assigned to the estrus stage. Finally, vaginas with a slight to massive transepithelial migration of leucocytes and a partial to complete loss of the cornified layers were assigned to the metestrus I and II stages, respectively.
Combined equine chorionic gonadotropin (eCG) and human chorionic gonadotropin (hCG) injections into immature female mice mimic the natural modulatory effects of the estrous cycle on the vaginal epithelium of adult female mice. (a) Characteristic morphologies of the vaginal epithelium are found in spontaneously cycling adult mice: proliferation and mucification from the diestrus stage to the proestrus stage, prominent stratification and cornification at the estrus stage, and desquamation and massive leucocytic infiltration at the metestrus I and II stages. (b) Similar morphological changes in the vaginal epithelium of immature female mice occur after eCG and combined eCG and hCG treatment, respectively: prominent mucification 24 h after eCG injection, beginning cornification within the next 24 h (arrows), prominent cornification 15 h after additional hCG injection. Sections are stained with hematoxylin and eosin (H&E). Scale bars as indicated.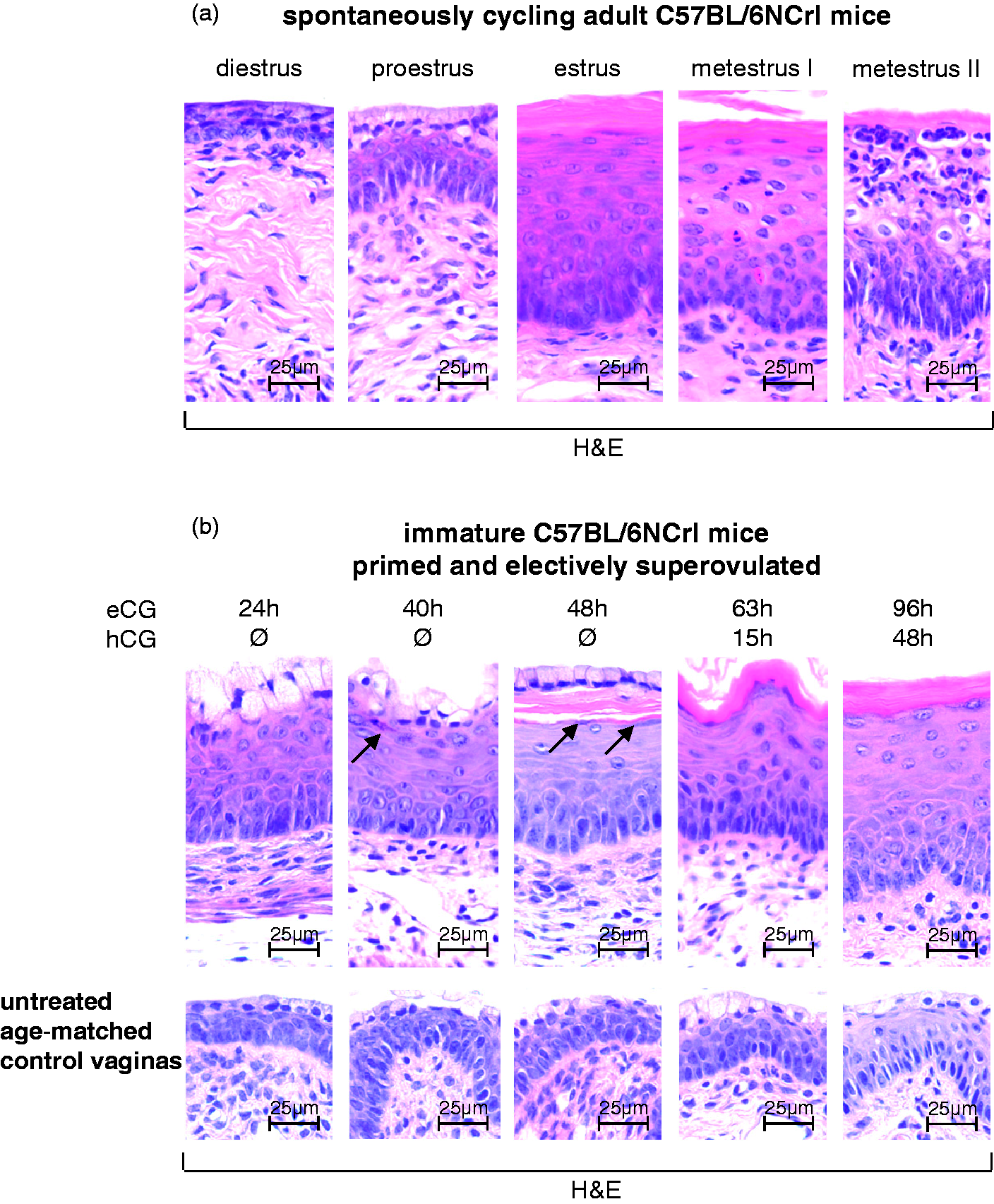
The vaginal epithelia of gonadotropin-treated 3.5-week-old immature C57BL/6NCrl mice showed a similar sequence of remodeling events as described for the vagina of spontaneously cycling mice (Figure 2b, upper row). Injection of eCG caused a gain in size of the vaginal epithelium and an increase in mucification of the superficial epithelial cell layer after 24 h. At that time, none of the ovaries of the animals examined showed preovulatory follicles or corpora lutea (Figure 1). At 40 to 48 h after eCG injection, the epithelial surface was still lined by cells of the mucified type. Beneath these cells, squamous cornifying cells appeared (40 h) and were accompanied by a well-formed cornified layer 48 h after eCG injection. The ovaries of these animals retained many larger follicles that had not yet ruptured. The injection of hCG which followed had further effects on the eCG evoked vaginal differentiation. At 15 h after the hCG injection, the upper portion of the thickened epithelium showed complete and prominent cornification. Remnants of the mucified cell type were no longer seen. In the ovaries, newly forming corpora lutea were present. At 48 h after the hCG injection, the cornification of the upper epithelial layers still persisted. Furthermore, the ovaries were now populated with mature corpora lutea.
Collectively, the exogenous gonadotropin treatment of immature female mice resulted in a sequence of histological changes occurring in their vaginas over the course of the four-day treatment (from mucification to cornification of the epithelium) that was similar to the sequence of vaginal morphologies ascribed to the estrous cycle. The morphological response to gonadotropin-exposure across the vaginas at a particular time point was highly consistent, as was the sequence of morphological events observed over the experimental period.
By contrast, the vaginal histology of age-matched, unstimulated control littermates remained unchanged at the different time points, showing the uniform picture of a small, stratified non-cornified epithelium covered by a single layer of mucoid cells (Figure 2b, lower row).
Similarity between estrous cycle and gonadotropin-treatment-induced differences in vaginal loricrin levels
We next sought to determine whether exogenous gonadotropin treatment of immature female mice was sufficient to cause the de novo expression of specific proteins in vaginas at different stages of the estrous cycle. Loricrin occurs specifically in the granular layer of cornifying epithelia in rodents including the skin, oesophageal and fore-stomach mucosa. 30
First, we determined the expression and localization of loricrin in the cornified envelope of vaginas from spontaneously cycling adult female mice by immunohistochemistry. Loricrin expression was restricted to those stages of the estrous cycle with moderate to prominent vaginal cornification (Figure 3a). The protein was detected in the intimate cell layers underneath the cornified layer. Loricrin staining was absent in the vaginal epithelium of all other stages of the estrous cycle with no cornification (i.e. diestrus, proestrus and metestrus II). There was no loricrin labeling in estrous specimens with cornification when the primary anti-loricrin antiserum was pre-incubated with the loricrin peptide, or after its omission during immunohistochemical staining (as presented online in Supplementary Materials [Figure S2] for a vagina at the estrus stage).
Loricrin expression in the vaginal epithelium of adult and gonadotropin-stimulated immature female mice. (a) Estrous cycle stage-specific expression of loricrin in the cornified vaginal epithelium of adult mice. Loricrin occurs when epithelial cell cornification is present around the estrus stage. (b) Combined equine chorionic gonadotropin (eCG) and human chorionic gonadotropin (hCG) injections can be used to induce the required morphological changes in the vaginal epithelium of immature mice to study cornification-specific gene product expression. Detection of loricrin is by immunohistochemistry (IHC) and nuclear counterstaining by Mayer’s hemalaun. Scale bars as indicated.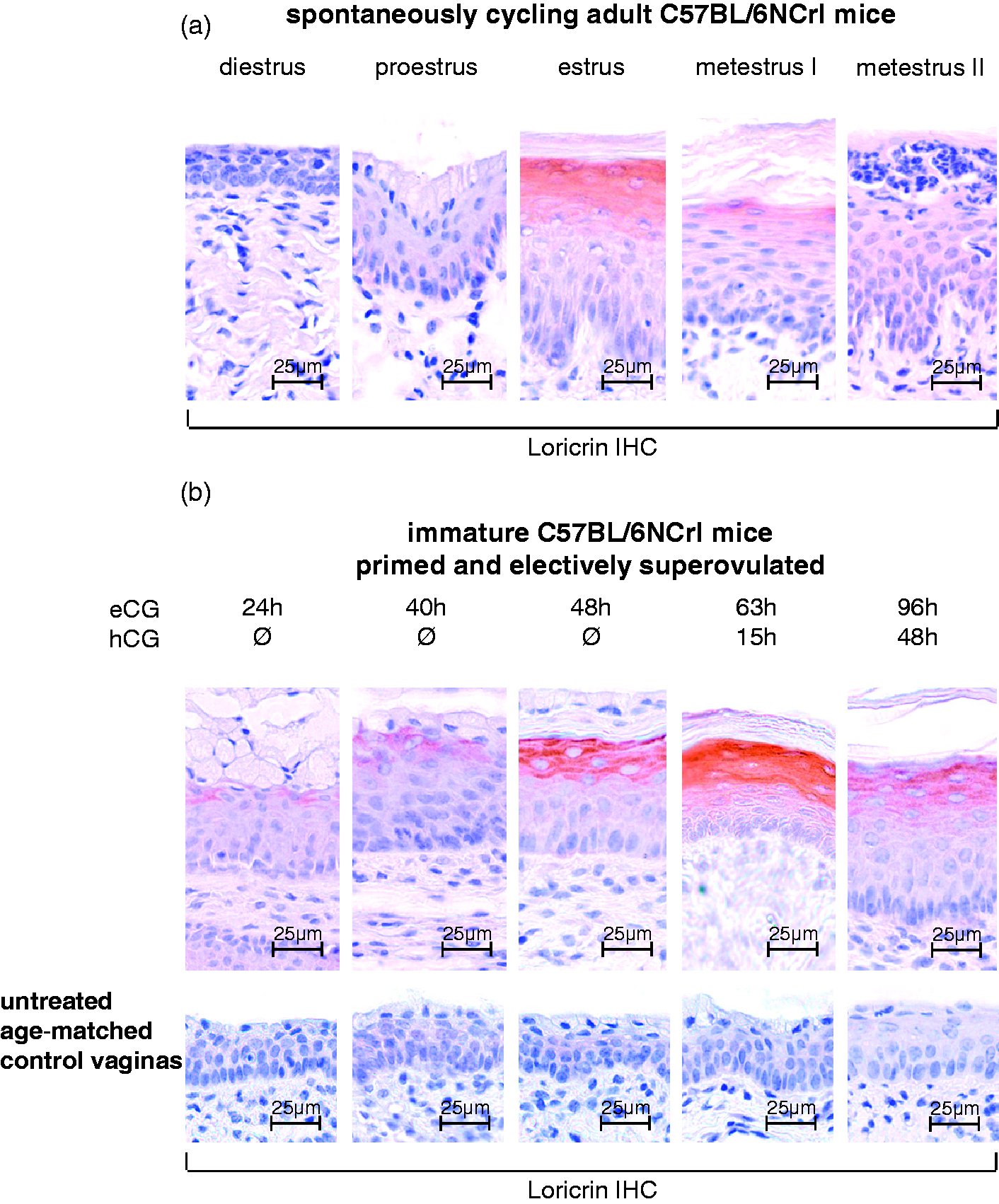
Finally, we examined the expression and localization of loricrin in the vaginas of immature mice after gonadotropin stimulation (Figure 3b, upper row). Injection of eCG caused an increase in epithelial height and intense mucification of the superficial layer after 24 h. Significant cornification of the layers underneath and expression of loricrin were not observed. By contrast, prolonged eCG simulation alone, or in combination with hCG, evoked the above described cornification and strong loricrin staining in the upper parts of the epithelium. Over the entire experimental period, loricrin staining was not present in the vaginas obtained from unstimulated control mice (Figure 3b, lower row). No loricrin labeling resulted in the vaginal specimens with cornification when the primary anti-loricrin antiserum was pre-incubated with the loricrin peptide, or when it was omitted during immunohistochemical staining (as shown online in Supplementary Materials [Figure S2] for a vagina of a combined eCG/ hCG stimulated immature mouse). In conclusion, loricrin expression was induced in the cornified layers of the immature female mouse vagina using our superovulation protocol.
Discussion
Hormonal regulation of the estrous cycle in the female rodent (with particular emphasis on the estrous cycle of rats) has been described in detail. 32 The Müllerian duct-derived organs (i.e. fallopian tube, uterus and upper vagina) undergo cyclical morphological changes under the influence of the ovarian steroid hormones, estrogen and progesterone. In particular, the cyclical shedding of the superficial mucoid and cornified layers in the vaginal epithelium is so remarkable that exfoliative vaginal cytology and histology are used worldwide for the assessment and classification of the estrous cycle stage in rats.6,8,10 Notably, the upper vaginal epithelium of rats has been used to identify xenobiotic estrogens (i.e. non-steroidogenic compounds that mimic the effects of ovarian estrogens).33,34
Unfortunately, mice display estrous cycle irregularities more frequently than rats due to the daily stress of being housed in animal facilities. 13 Furthermore, acceptable exfoliative vaginal cytology is more difficult to obtain in mice than in rats, and vaginal swabs constitute a significant further stress factor for these animals.4,16,35 Therefore, it is widely believed that in mice, unlike rats, the specific stage of the estrous cycle is better assessed by preparation and analysis of suitable vaginal histological sections post-mortem. A recent study by Gal et al. has demonstrated that neither exfoliative vaginal cytology nor gonaodotropin and sex hormone serum levels can adequately predict the estrous cycle stage of a mouse. 15 In their hands, exfoliative vaginal cytology only allowed a rough approximation of the actual estrous cycle stage. The authors have reported a discordance rate between this method and post-mortem vaginal histology of 55%, 77%, 46%, and 31% for diestrus, proestrus, estrus and metestrus, respectively.
Since there is a necessity for determining the specific stage of the estrous cycle in the mouse post-mortem, this entails the use of larger numbers of mice. For example, studies designed to examine the effects of the ovarian estrous cycle on the expression of a particular protein or gene requires many mice. 36 This statement holds true particularly for studies in which a distinct type of vaginal epithelium is required for the successful identification of a gene product associated with a specific step of vaginal epithelial differentiation (as exemplified here for loricrin expression).
In view of these observations, an easy and reliable method is desirable for inducing estrous cycle stage-specific reproductive tract morphologies and gene expression in mice. The application of such a method would allow for a smaller number of animals being euthanized and examined post-mortem, in particular in studies dealing with oestrus cycle stage-specific changes.
We have shown that the injection of immature female mice with eCG alone, or in combination with hCG, promotes sequential morphological changes in the vaginal tube that are comparable with those observed during the estrous cycle of adult female mice. In addition our post-mortem histological examination of the gonadotropin-exposed immature vaginas discloses that the sequential morphological changes under gonadotropin-exposure occur with the regularity and stability required that gonadotropin-exposure could be applied to 3.5-week-old female mice in future studies to locate the animal in an expected oestrus-cycle state at an expected time. This would certainly have the potential of significantly decreasing the number of female mice used in studies in which the effects of stages in the estrous cycle are important starting conditions.
Our study is novel in that previous studies on eCG and hCG stimulations of mice are focused mainly on the timed induction of superovulation in the prepuberal animal.26,37 Therefore, our comparison between exogenous eCG- and hCG-induced and natural estrous cycle-induced changes in the murine vaginal epithelium cannot be directly compared with any previously published studies to the best of our knowledge.
In conclusion, our approach will enable researchers to perform experiments involving estrous cycle controlled structures and functions with reduced animal numbers. Our approach will certainly facilitate the elucidation of gene expression profiles during the series of morphophysiological events that characterize the Müllerian duct-derived organs during the estrous cycle.38–40 In addition, our study indicates that the upper vagina of the laboratory mouse is a convenient model organ for addressing basic and applied questions concerning the process of cornification in a developing stratified squamous epithelium.
The gonadotropin stimulation protocol described herein may need to be modified when a different mouse strain is used, as strain differences have been identified regarding in vitro fertilization and superovulatory protocols.22,37,41
Footnotes
Acknowledgements
We express our profound gratitude to the staff of the transgenic and experimental animal facility for taking care of our mouse strains. We are also grateful to Professor Torsten Schöneberg for helpful comments and suggestions concerning these studies, as well as for critical reading of the manuscript. AR designed the research, assembled the data, and drafted the manuscript. OB interpreted the data, revised the draft and wrote the final version of the manuscript with AR. CM and AS were heavily involved in data acquisition, analysis, interpretation, and presentation. FE, JW and SP helped to tailor the standard stimulation protocol to meet the needs of the study. All authors read and approved the final manuscript version.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: It was jointly financially supported by the Institute of Anatomy and the Institute of Biochemistry, Medical Faculty, University of Leipzig, Germany.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
