Abstract
To assess pain in mouse models of bone fractures, currently applied assessment batteries use combinations of clinical signs with spontaneous behaviours and model-specific behaviours, including walking and weight-bearing behaviour. Rearing behaviour – an upright position on the hindlimbs – has a motivational and an ambulatory component. Thus, rearing behaviour might have the potential to be an indicator for model-specific pain in mouse fracture models. To date, the assessment of rearing behaviour in bone fracture models using mice is only scarcely described. In this study, we aimed to determine whether the duration of rearing behaviour is affected by osteotomy of the femur in male and female C57BL/6N mice with external fixation (rigid vs. flexible) and could be an additional sign for model-specific pain, such as the presence of limping. Rearing duration was significantly decreased after osteotomy in male and female mice at 24 h, 48 h and 72 h, but was not affected by anaesthesia/analgesia alone. In male mice, the relative rearing duration increased over 72 h (both fixations) and at 10 days in the rigid fixation group but remained significantly lower in the flexible fixation group. In contrast, in female mice, no increase in rearing duration was observed within 72 h and at 10 days post-osteotomy, independent of the fixation. We did not identify any association between relative rearing time and presence or absence of limping. In summary, our results do not provide sufficient evidence that altered rearing behaviour might be an indicative sign for pain in this model.
Introduction
Reliable clinical and behavioural assessment of laboratory rodents, especially mice, is an ongoing challenge but is crucial to determine the individual’s wellbeing and to prevent pain and distress in animal experiments.1 –3 Pain and distress negatively impact physiological processes and their side effects may result in potentially biased results and a lack of reproducibility.1,4 –9 However, as a prey species, mice mask pain-associated behaviours.3,6,10 Moreover, many methods used for clinical and behavioural assessment of pain are influenced by confounding factors such as anaesthesia and analgesia applied during preceding procedures, handling methods or environmental context.3,6
Bone fractures are a challenging clinical problem and advances in bone fracture research including the use of animal models have contributed to a more in-depth understanding of molecular and cellular mechanisms driving bone regeneration and the risk factors underlying healing complications. 11 To assess pain in mouse models of bone fractures, current assessment batteries use combinations of clinical signs, spontaneous behaviours and model-specific indicators, including walking and weight-bearing behavior. 12 Methods using von Frey filaments 13 and cold or hot plates 2 are routinely used to assess changes in nociception in fracture models. However, these tests are focusing only on nociceptive changes and measure response reflexes after a noxious mechanical or thermal stimulus rather than pain as a subjective experience.2,3,10,14,15 Weight-bearing performance and movement of the fractured limb can give additional insights on potential skeletal pain and mechanical restrictions. In addition, automated gait analysis systems provide precise data on the walking behaviour but are often unsuitable for routine monitoring after surgery as they require sophisticated equipment and extended training beforehand. In contrast, observational scoring of the presence of limping (equivalent to lameness) might not be as precise as automatic gait analysis systems although it can be used as a basic tool to assign numerical values to walking behaviours.11,16,17
Rearing behaviour is a natural and non-evoked behaviour displayed by many mammals, including mice and rats, and has been assessed in different contexts for over 50 years. 18 Rearing is an explorative behaviour and requires the animal to put weight on its hindlimbs to get into the upright position.18,19 Thus, impaired rearing behaviour has the potential to be a useful sign for pain especially after procedures leading to potential restrictions in the hindlimbs. However, to date, only a few studies describe the examination of rearing behaviour for pain assessment in mouse osteotomy models.20 –24 These studies either focused on long-term pain development or did not use osteotomies with external fixators.20 –24 In this study, we examined whether the duration of rearing behaviour was affected by femoral osteotomy with stable external fixation (rigid vs. flexible), and the association between these changes and the degree of limping observed at 24 h, 48 h, 72 h and 10 days post-interventional.
Animals, material and methods
Animals and husbandry
The data in this study were collected from video material recorded during a previous study. 17 For this present study, only the subgroup of animals receiving tramadol in the drinking water for post-interventional analgesia were assessed. 17 The in vivo study was conducted according to the guidelines of the German Animal Welfare Act, National Animal Welfare Guidelines, and was approved by the local Berlin state authority (Landesamt für Gesundheit und Soziales – LAGeSo; permit number: G0044/20). There is no duplication of results published in the previous study. 17 C57BL/6N mice (n = 20 male and n = 20 female) were purchased from either the Experimental Medicine Research Facilities (Charité-Universitätsmedizin Berlin, Berlin, Germany) or Charles River Laboratories (Sulzfeld, Germany). Mice underwent a first intervention (anaesthesia/analgesia) at 10 weeks of age and the second intervention (anaesthesia/analgesia and osteotomy) at 12 weeks of age. Upon arrival, the mice were randomly divided into groups of two per cage and housed in individually ventilated Eurostandard Type II clear-transparent plastic cages (IVC unit). All home cages contained wooden chips (SAFE FS 14, Safe Bedding, Rosenberg, Germany), Envirodri (Shepherd Specialty Papers, USA) and shredded paper as bedding and nesting material. The cages were enriched with a mouse double swing (Datesand Group, Bredbury, UK) and a clear handling tube (Datesand Group, Bredbury, UK), which were removed after osteotomy to reduce the risk of injury. Five days after osteotomy, two single swings (Datesand Group, Bredbury, UK) per cage were reinstalled. Cages were changed weekly by the experimenters only. All experimenters performing analyses were female and animals were tunnel handled throughout the whole experiment. The mice were housed in individually ventilated cages in a semi-barrier facility with limited personal access and a requirement to wear protective clothing when entering the barrier. Regular health monitoring was performed according to the FELASA guidelines (sentinels were partly tested positive for Murine norovirus, Helicobacter spp., Trichomonas spp., and other flagellates).17,25 Housing conditions included a 12/12 h light/dark cycle (lights out from 18:00 h to 06:00 h), a room temperature of 22 ± 2 °C and a humidity of 55 ± 10%. Mice had access to tap water and food (Standard mouse diet, Ssniff Spezialdiäten, Soest, Germany) ad libitum.
Experimental groups, timeline and video recording
Reporting of this study was conducted in compliance with the ARRIVE 2.0 guidelines and the experimental protocol was preregistered (Animal Study Registry; DOI: 10.17590/asr.0000221). Four groups were compared in the study: male mice with rigid external fixator, male mice with flexible external fixator, female mice with rigid external fixator and female mice with flexible fixator (Figure 1(a); females – n = 10 per group; males – n = 9 per group). During the first two weeks upon arrival, mice underwent acclimatization and training (Figure 1(b)). In detail, mice were housed in pairs per cage (IVC; home cage) and allowed to acclimatize to the animal facility. During three training sessions, mice were tunnel handled and transferred to the observation cage for approximately 10 min to familiarize with the handling procedure, observation cage and experimenters. Non-ventilated Eurostandard Type III cages (Tecniplast, Milan, Italy; size specifications: 42.5 cm × 27.6 cm × 15.3 cm; ground area = 1173 cm2), containing the same type of wooden chips as the home cages, were used for animal observation. After the training period, mice underwent a first intervention consisting of isoflurane anaesthesia and the administration of analgesics without a surgical procedure (hereafter referred to as ‘post-anaesthesia’; Figure 1(b)). The same mice underwent an osteotomy on the left femur as well as isoflurane anaesthesia and analgesia administration (hereafter referred to as ‘post-osteotomy’) two weeks after the first intervention (Figure 1(b)).
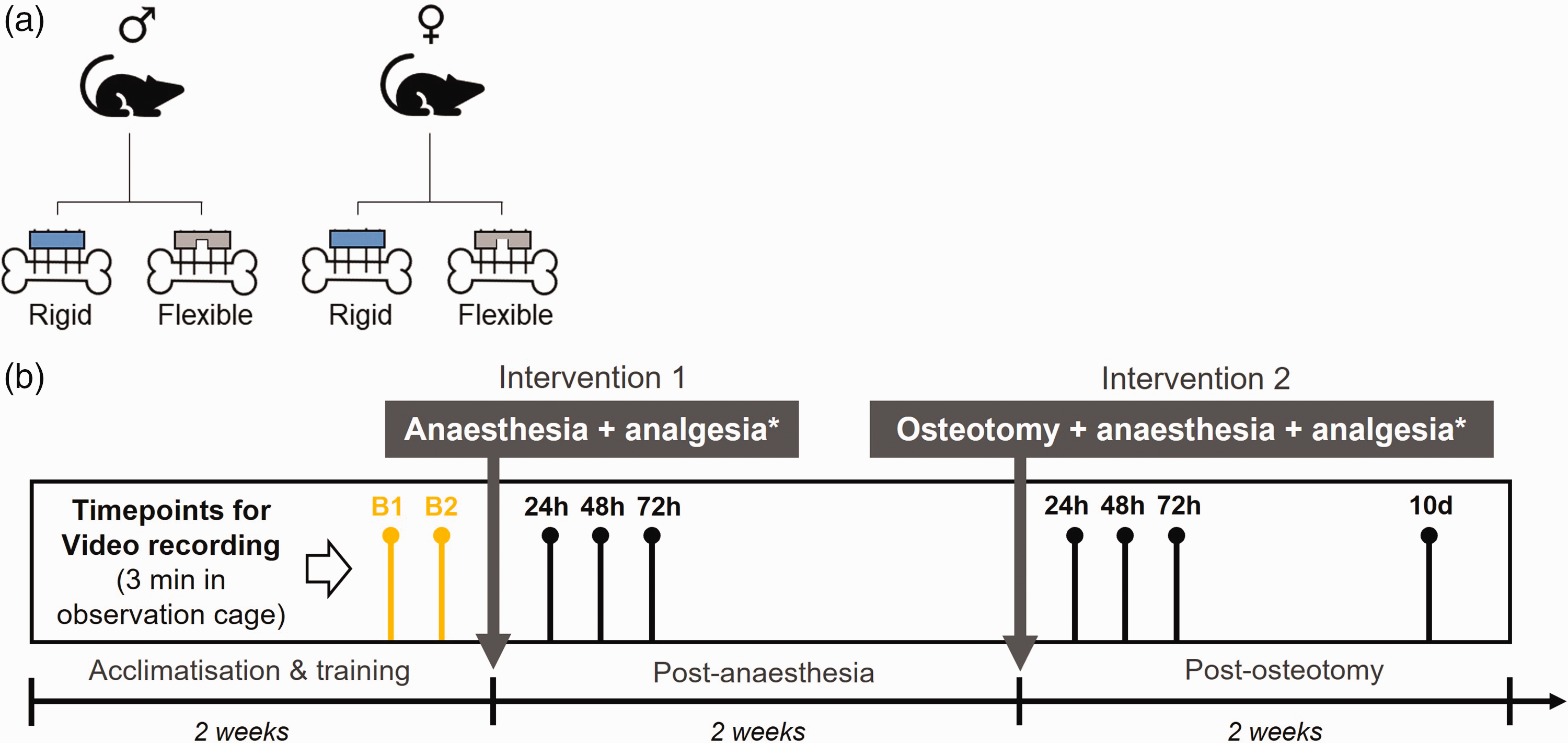
Illustration of group assignment and experimental timeline of interventions and video recordings. Overview on (a) the four different groups, and (b) the experimental timeline. B1 and B2 – signify baseline measurements.
For video recording, mice were individually transferred to observation cages and after approx. 2 min a video was recorded for 3 min before mice were transferred back to the home cage. Two video recordings were performed at the end of the two weeks acclimatization and training period, and before any intervention for baseline measurements at a naïve state (B1 and B2, Figure 1(b)). After the first intervention (post-anaesthesia), videos were taken at 24 h, 48 h and 72 h. Following the second intervention (post-osteotomy), videos were taken at 24 h, 48 h and 72 h and 10 days (Figure 1(b)).
Detailed description of interventions – anaesthesia, analgesia and osteotomy
For both interventions, tramadol was administered via the drinking water (0.1 mg/ml, Tramal Drops; Grünenthal, Stolberg, Germany) for one day before the intervention and for three consecutive days afterwards. Mice additionally received a single subcutaneous (s.c.) injection of buprenorphine (1 mg/kg, Temgesic; RB Pharmaceuticals, Heidelberg, Germany) at the beginning of each intervention. For the first intervention, the mice were anesthetized with isoflurane and then moved onto a heating pad (37°C). Anaesthesia was maintained with isoflurane (CP-Pharma, Burgdorf, Germany; vaporizer settings ranged 2–3%; Draeger Isofluran Vapor 19.3, Draegerwerk AG Co. KGaA, Luebeck, Germany), delivered in 100% oxygen. During anaesthesia, eye ointment, physiological saline (0.5 ml, 0.9% NaCl; B. Braun Melsungen, Melsungen, Germany), clindamycin (45 mg/kg; Ratiopharm, Ulm, Germany) and a single s.c. injection of buprenorphine were administered. Mice were allowed to recover in their home cages under an infrared lamp and were closely monitored. For the second intervention (osteotomy), anaesthesia was induced and maintained as described for the first intervention; eye ointment, physiological saline, clindamycin and buprenorphine were also administered as described above. The left leg was shaved and disinfected with alcohol-based povidone-iodine solution (Braunoderm; B. Braun Melsungen, Melsungen, Germany). A skin incision was made between the knee and hip and the muscles were bluntly separated. After exposing the femur, the external fixator (rigid: 18.1 N/mm; flexible: 3.2 N/mm, both RISystem, Davos, Switzerland) was placed parallel to the femur and fixed with four pins accordingly. An osteotomy gap (approximately 0.5 mm) was created afterwards, using a Gigli wire saw (0.44 mm; RISystem, Davos, Switzerland) between the second and third pin. 16 The muscle and skin were closed with sutures (muscle: 5-0 Vicryl, skin: Ethilon 5-0; both Ethicon, Raritan, USA). After the procedure, the mice were returned to their home cages and recovered under an infrared lamp for at least 30 min. Two different external fixators were used to stabilize the osteotomy – rigid and flexible (Figure 1(a)). Unlike the rigid fixator, which limits movement between the proximal and distal fracture ends, the flexible fixator allows considerable movement (despite stable fixation) which leads to a larger callus formation and delayed healing. It is currently unknown how differences in the external fixator stiffness affect pain behaviour in this model. Therefore, we decided to include both fixation groups in the analysis.
Analysis of video recordings – rearing time and limping score
Each entire video recording – which lasted 3 min – was analysed by either one or two observers for rearing and limping, respectively. Observers were unaware of sex and timepoint (baseline, 24 h, 48 h and 72 h post-anaesthesia) and videos were assigned random numbers unrelated to group allocation or timepoint. After osteotomy, group allocation was known with respect to fixation based on the external fixator; however, observers had no information about the timepoint post-osteotomy (24 h, 48 h, 72 h and 10 days).
Rearing time was measured using a stopwatch. Rearing behaviour was classified as lifting both front paws from the ground and standing in an upright position on one or both hindlimbs. Supported rearing (against the walls) and unsupported rearing were considered equally. The results for the rearing time from both baseline measurements (B1 and B2) were combined as an average baseline measurement (mean). The results of the rearing time analysis are expressed as either absolute rearing (in seconds) or relative rearing time. The relative rearing time was calculated by dividing the average baseline rearing time by the rearing time at the selected timepoints post-anaesthesia or post-osteotomy to control for individual deviations.
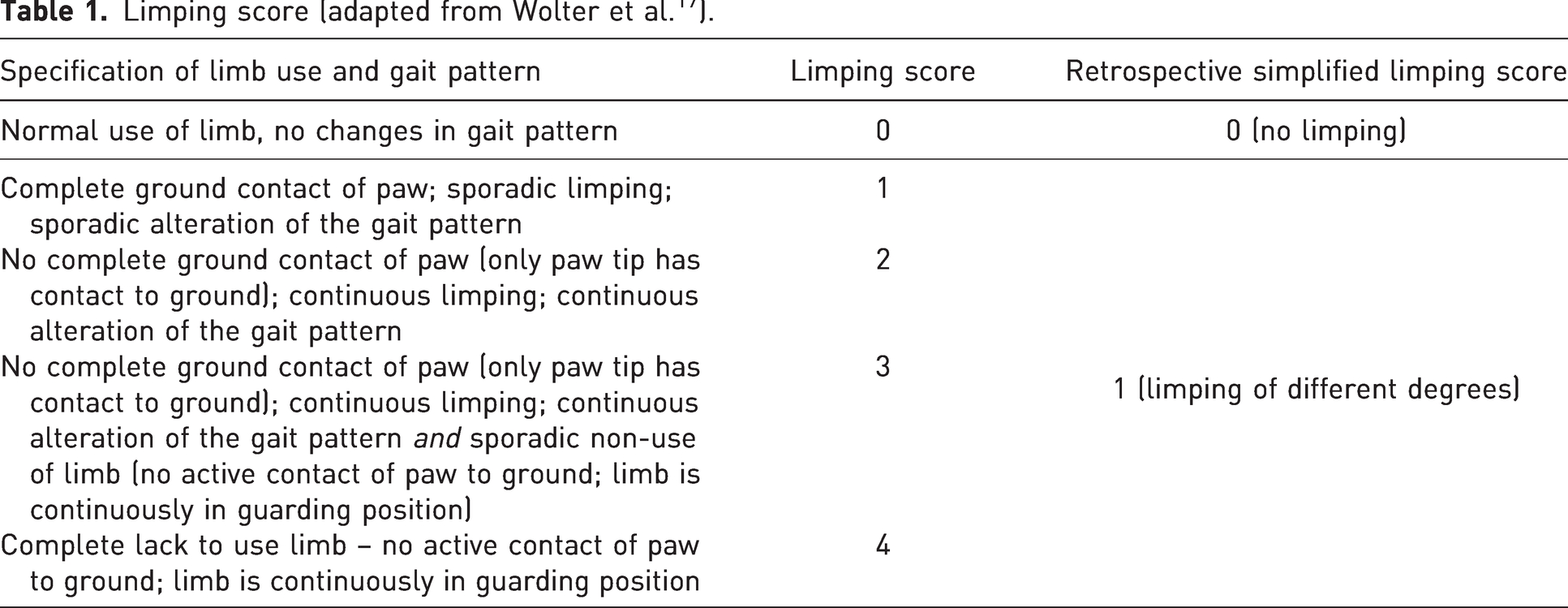
Limping was scored only after intervention 2 (24 h, 48 h, 72 h and 10 days post-osteotomy). For each animal and timepoint, a limping score between 0 and 4 was assigned by two blinded observers (Table 1). For this study’s purpose (association between rearing time and limping), limping scores were summarized as 0 (no limping) or 1 (limping of different degrees) (Table 1).
Limping score (adapted from Wolter et al. 17 ).
Statistical analysis
Sample size was calculated based on preliminary data and the required number of animals was modelled in R (package nparLD). Assuming a 20% difference and a power of ∼80% resulted in n = 10 animals per group. Two male mice (one in each fixation group) were excluded because of health problems unrelated to the study, resulting in a final group size of n = 9 per male group. Rearing data showed a high variability; therefore, values greater than the 75th and 25th percentile plus 1.5 times interquartile range were identified using Tukey’s method in GraphPad Prism and excluded for further analyses (Figure 2(c) – one male; Figure 3(d) – one female). Statistical analysis was performed using GraphPad Prism (V9). Datasets which failed normal distribution testing underwent non-parametric (Wilcoxon, Mann–Whitney, and Friedman) analyses to determine group differences.
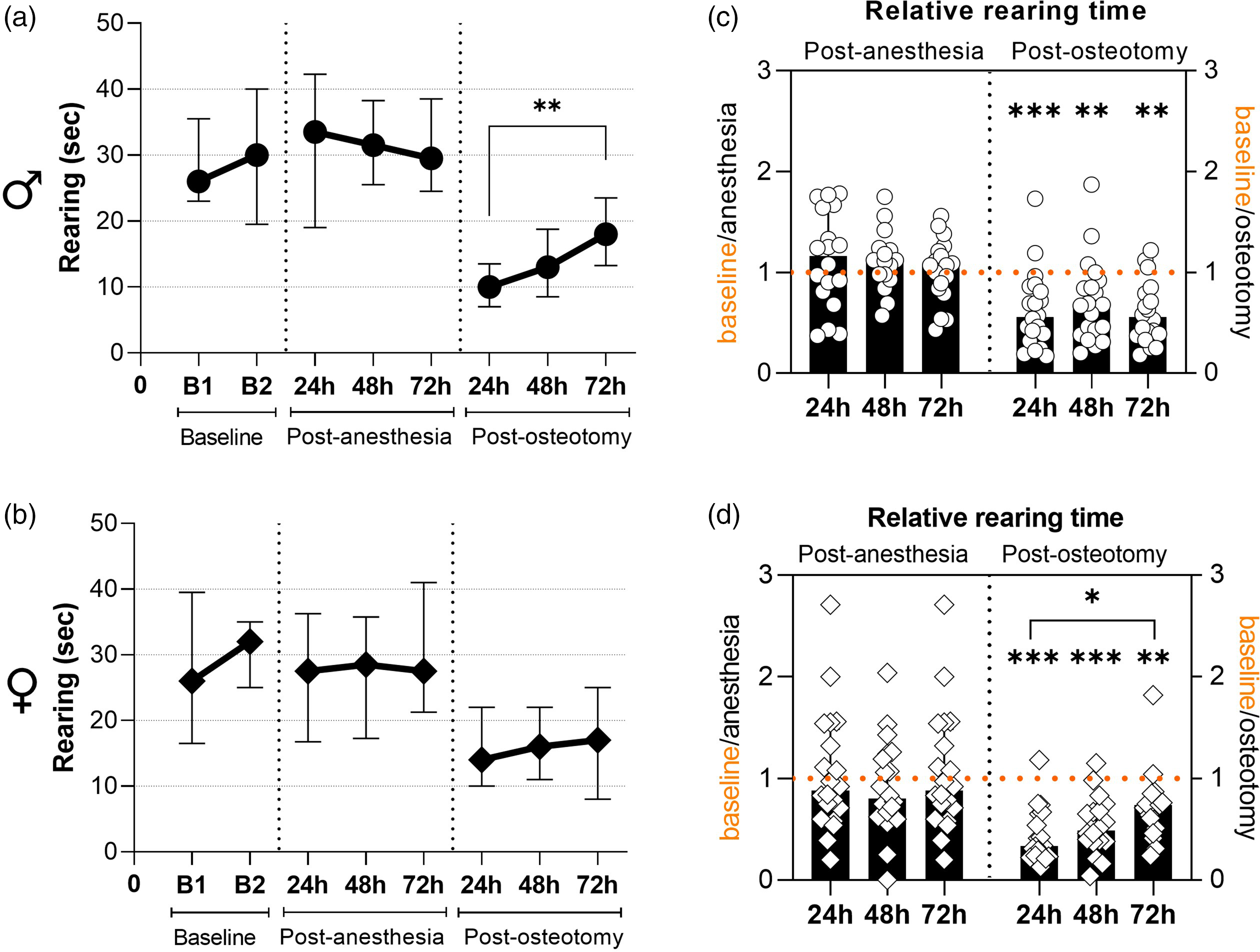
General assessment of absolute and relative rearing time separated by sex (data for both fixations combined). (a) and (b) Absolute rearing time in seconds at two baseline timepoints and at 24 h, 48 h, 72 h post-anaesthesia and post-osteotomy, respectively. All graphs show median with interquartile range for n = 18 (males; (a)) and n = 20 (females; (b)); Friedman tests with Dunn’s multiple comparisons were performed; **p < 0.01. (c) and (d) Relative rearing time after anaesthesia and osteotomy normalized to the respective baseline values. All graphs show median with interquartile range and individual data points representing data distribution for n = 17–18 (males; (c)) and n = 20 (females; (d)); Friedman tests with Dunn’s multiple comparisons were performed between timepoints and Wilcoxon tests to baseline were performed to determine individual statistical differences to the respective baseline measurements; *p < 0.05, **p < 0.01, ***p < 0.001.
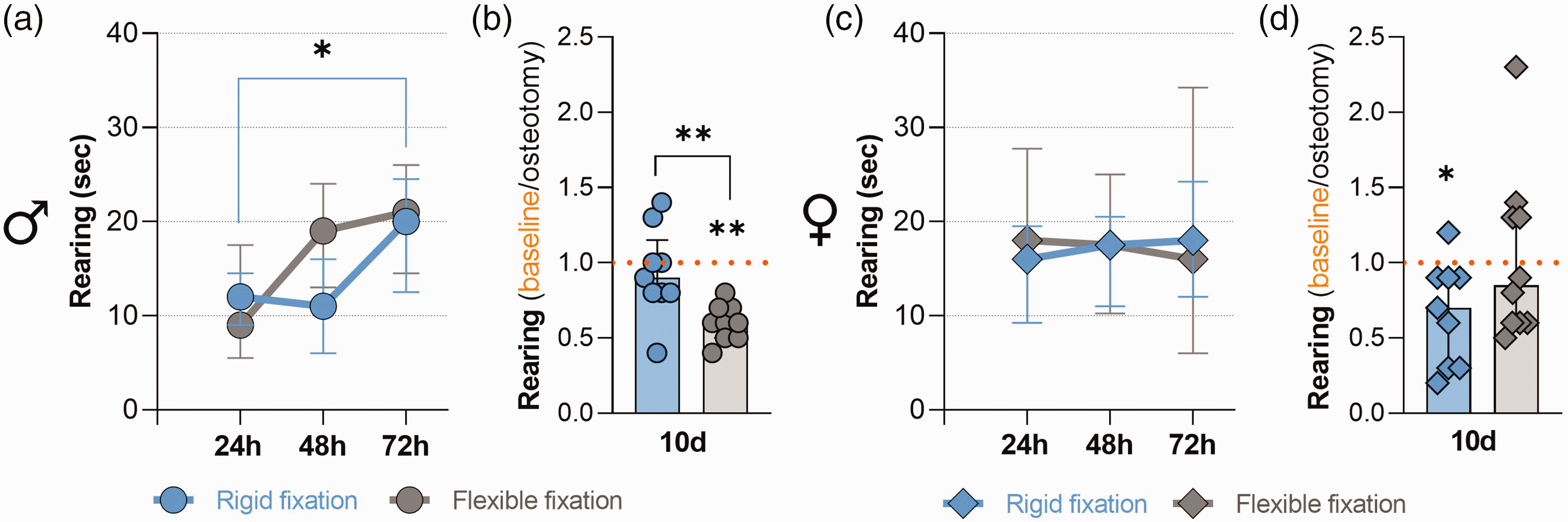
Absolute rearing time separated by fixation stiffness and relative rearing time at 10 days post-osteotomy. (a) and (c) Absolute rearing time in seconds at 24 h, 48 h, 72 h post-anaesthesia and post-osteotomy separated for the two fracture fixations. All graphs show median with interquartile range for n = 9 (males; (a)) and n = 9–10 (females; (c)); Friedman tests with Dunn’s multiple comparisons were performed; *p < 0.05. (b) and (d) Relative rearing time 10 days after osteotomy, normalized to the respective baseline values. All graphs show median with interquartile range and individual data points representing data distribution for n = 9 (males; (b)) and n = 9–10 (females; (d)); Wilcoxon test to baseline was performed to determine individual statistical differences to the respective baseline measurements and Mann–Whitney test was performed between groups; *p < 0.05, **p < 0.01.
Results
General data exploration of rearing behaviour post-anaesthesia and post-osteotomy
Absolute rearing duration in both sexes was similar between the two baseline measurements and between the three different timepoints post-anaesthesia (24 h, 48 h, 72 h) (Figure 2(a) and (b)). To assess deviations from baseline values, a relative rearing time per mouse was calculated for each timepoint post-anaesthesia and post-osteotomy. Individual normalized data analysis (i.e. relative rearing time per mouse) revealed no statistically significant differences in relative rearing duration between baseline and post-anaesthesia (Figure 2(c) and (d); Wilcoxon test with p > 0.05). After osteotomy, absolute rearing duration markedly dropped at 24 h in male and female mice (Figure 2(a) and (b)). In male mice, rearing duration significantly increased between 24 h and 72 h (Figure 2(a); Friedman test p = 0.005; Dunn’s multiple comparison p = 0.008) but not in female mice (Figure 2(b); Friedman test p = 0.37). Individual normalized data analysis supported the longitudinal data analysis by exhibiting significantly reduced values compared with the respective baseline control in both sexes at every timepoint (Figure 2(c) and (d); Wilcoxon test with **p < 0.01 and ***p < 0.001). In male mice, there was no significant difference between normalized values and all timepoints post-osteotomy (Figure 2(c); Friedman test p = 0.4), while a statistical significance was detected in female mice between 24 h and 72 h (Figure 2(d); Friedman test p = 0.014). These findings suggest that despite an increase in absolute rearing time in male mice and in relative rearing time in female mice, baseline values were not re-achieved within 72 h.
Differences in rearing behaviour depending on the fracture fixation
The effects of fixation stiffness on post-osteotomy rearing behaviours were analysed for a 10-day period to determine whether baseline (naïve) values were achieved. Independent of the fixation stiffness, the absolute rearing time in male mice increased over 72 h post-osteotomy, which was only significant between 24 h and 72 h in the rigid fixation group (Figure 3(a); Friedman test p = 0.024; Dunn’s multiple comparison p = 0.04). We found that at 10 days post-osteotomy, relative rearing behaviour of male mice with rigid fixation was was similar compared with the respective baselines (Wilcoxon test with p = 0.69), while remaining significantly reduced in the flexible fixation group (Figure 3(b); Wilcoxon test with p = 0.004). Also, the relative rearing time in the male group was significantly higher in the rigid fixation group compared with the flexible fixation group (Figure 3(b); Mann Whitney test p = 0.005). In contrast, independent of the fixation stiffness, the absolute rearing time in female mice did not change over 72 h post-osteotomy and we found no differences between the two groups in absolute rearing time at 24–72 h post-osteotomy (Figure 3(c)). In addition, we found that at 10 days post-osteotomy, relative rearing was still reduced in female mice with rigid fixation compared with the respective baselines (Wilcoxon test with p = 0.03). In female mice with flexible fixation, no statistical significance was found when compared with the respective baseline values (Figure 3(d); Wilcoxon test with p = 0.7). There was no difference between the rigid and the flexible group (Figure 3(d); Mann Whitney test p = 0.2). To summarize, our data did not indicate differences in absolute rearing time at 24–72 h post-osteotomy when comparing the different fixation groups. However, we found differences in the improvement over 10 days in male mice, which was more pronounced in the group with rigid fixation when compared with the flexible fixation group.
Association of rearing behaviour and limping score
To assess whether the reduction in rearing behaviour after osteotomy was more pronounced in mice with impaired gait (presence of limping), we compared the relative rearing time with the individual limping scores. Animals were scored for limping at 24 h, 48 h and 72 h post-osteotomy. The limping scores were then either categorized as 0 = no limping, or 1 = any degree of limping (Figure 4(a) and (b)). Limping frequency in male mice with rigid fixation was <50% at every timepoint while being >50% at all timepoints in the flexible fixation group, except for 10 days (Figure 4(a) and (b)). We next plotted the relative individual rearing time per category (no limping vs. limping) combining the data of all timepoints (Figure 4(c) and (d)). We found that the relative rearing time was significantly reduced compared with the baseline values independent of fixation and gait (no limping/limping; Figure 4(c) and (d); Wilcoxon test with **p < 0.01 and ***p < 0.001. Mann–Whitney test: rigid fixation p = 0.22; flexible fixation p = 0.7). The following timepoint-resolved descriptive analysis of all datapoints indicated an upward trend towards the initial baseline values in the rigid fixation groups independent of the gait pattern. No upward trend on time-dependent recovery was observed in the flexible fixation group (Figure 4(e) and (f); descriptive analysis without statistical analysis).
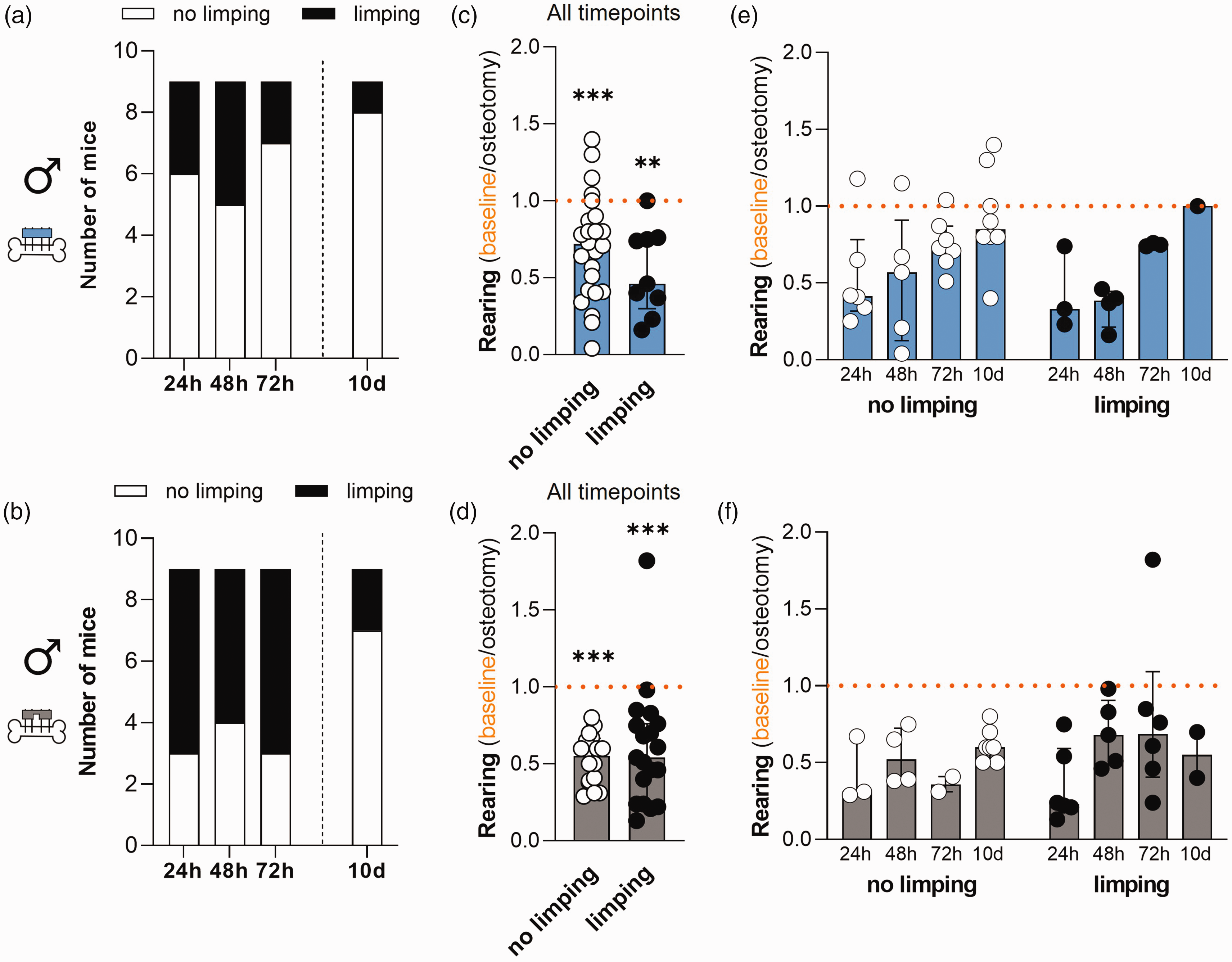
Comparative analysis of relative rearing time and limping score in male mice. (a) and (b) Frequency of limping at 24 h, 48 h, 72 h and 10 days in the (a) rigid fixation and (b) flexible fixation group. (c) and (d) Timepoint combined analysis of relative rearing time in male mice with no limping or limping in (c) the rigid fixation and (d) flexible fixation group. Normalized rearing of animals assigned to limp score category 0 (no limping) or 1 (limping). Wilcoxon test to baseline was performed to determine individual statistical differences to the respective baseline measurements and Mann–Whitney test was performed between groups; **p < 0.01; ***p < 0.001. (e) and (f) Timepoint resolved representation of relative rearing time in male mice with no limping or limping in (e) the rigid fixation and (f) the flexible fixation group. All graphs show median with interquartile range and individual data points representing data distribution. n = 9 (per fixation group; no data exclusion).
In female mice, limping frequency was ≤30% at every timepoint independent of fixation (Figure 5(a) and (b)). We found that the relative rearing time was significantly reduced compared with the baseline values independent of fixation and limping (Figure 5(c) and (d); Wilcoxon test with **p < 0.01). Relative rearing in female mice with flexible fixation was significantly more reduced in mice with altered gait (limping) when compared with mice that showed no limping (Figure 5(c) and (d); timepoint combined analysis; Mann–Whitney test: rigid fixation p = 0.22; flexible fixation p = 0.04). Timepoint-resolved descriptive analysis of all data points exhibited no observable differences between timepoints, gait pattern and fixation (Figure 5(e) and (f); descriptive analysis without statistical analysis). In summary, our detailed analysis did not identify particular association between relative rearing time and gait pattern (presence or absence of limping).
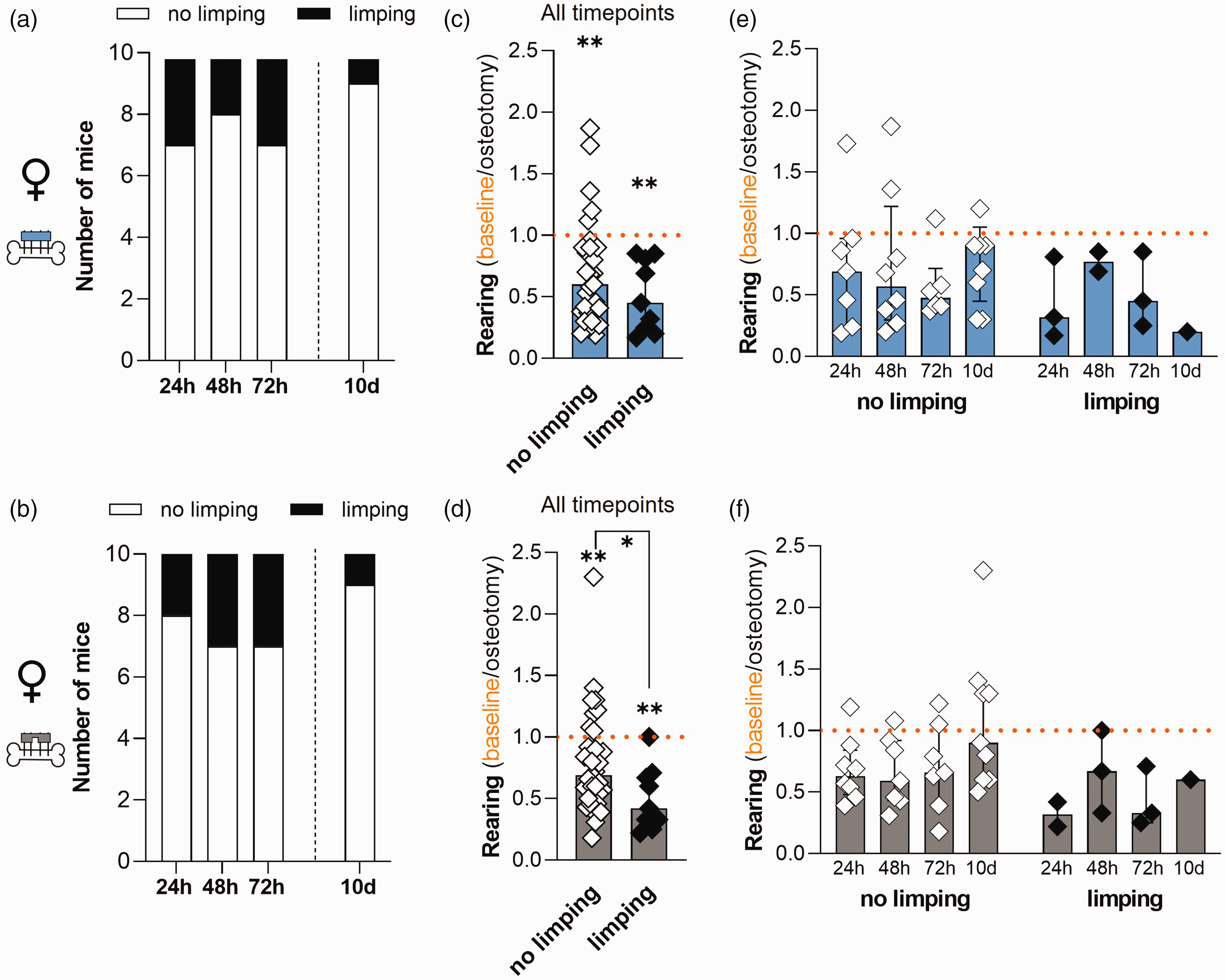
Comparative analysis of relative rearing time and limping score in female mice. (a) and (b) Frequency of limping at 24 h, 48 h, 72 h and 10 days in the (a) rigid fixation and (b) flexible fixation group. (c) and (d) Timepoint combined analysis of relative rearing time in female mice with no limping or limping in (c) the rigid fixation and (d) the flexible fixation group. Normalized rearing of animals assigned to limp score category 0 (no limping) or 1 (limping). Wilcoxon test to baseline was performed to determine individual statistical differences from the respective baseline measurements and Mann–Whitney test was performed between groups; *p < 0.05; **p < 0.01. (e) and (f) Timepoint resolved representation of relative rearing time in female mice with no limping or limping in (e) the rigid fixation and (f) the flexible fixation group representing data distribution. All graphs show median with interquartile range and individual data points. n = 10 (per fixation group; no data exclusion).
Discussion
The analysis of rearing in mice has been used since the 1970s in a wide range of contexts and has also been used as an additional indicator to monitor reduced wellbeing or pain after surgical interventions. Even though rearing behaviour has a high ambulatory component, 18 there are only a few studies reporting the assessment of rearing behaviour after fracture surgery, especially with a focus on the acute phase (72 h post-osteotomy). Hence, this study aimed to evaluate how rearing behaviour changes in mice undergoing osteotomy shortly after the intervention (24 h, 48 h and 72 h) and at 10 days post-osteotomy.
Our data showed a significantly decreased rearing duration at 24 h, 48 h and 72 h post-osteotomy in female and male C57BL/6N mice when combining the data of both fixations (Figure 2). However, in male mice, the relative rearing duration steadily increased over 72 h in both groups (Figure 3). The 10 days evaluation showed an increased rearing duration in the rigid fixation group when normalized to initial values while remaining significantly reduced in the flexible fixation group. In contrast, no increase in rearing duration was observed in female mice over 72 h and at 10 days post-osteotomy. Our observation that rearing behaviour is reduced after surgery is in concordance with several other studies using the analyses of rearing after surgical procedures as an additional indicator of wellbeing after vasectomy and laparotomy.26 –29 Moreover, in our related study involving extensive examination of clinical signs and behavioural changes of these mice, we also observed short-term body weight loss and higher composite scores (>0) shortly after surgery which declined until 72 h. These signs for impaired wellbeing agree with the initial decrease in rearing duration post-osteotomy observed in the current study. 17 We also found that female mice improved slightly faster than males in the clinical, behavioural and model-specific examination over 72 h. 17 However, the rearing behaviour analysis showed that female mice, in general, had a less precipitous drop in absolute rearing duration at 24 h post-osteotomy when compared with the males, but with no improvement over 72 h (Figure 3). It has been previously described that female mice tend to rear more than male mice, 18 while Sturman et al. reported that naïve C57BL/6J female mice reared less than male mice, despite comparable levels of walking activity in response to stress (handling) when monitored in an open field arena. 30 They also reported that rearing behaviour in female mice decreased over the experimental time course of nine days, regardless of whether they were handled or not. 30 Additional studies provide evidence that environmental familiarization affects the rearing behaviour.18,31 Durst et al., for example, reported a high rearing frequency in all groups at baseline which then declined after induction of acute pancreatitis and suggested that the steady decrease of rearing frequency was rather an effect of habituation with a less intrinsic need for exploration than indicative of pain.18,31 In the context of our study and experimental setting, we agree with this suggestion, which is further underlined by the fact that we observed a reduced relative rearing duration in male mice with flexible fixation and female mice with rigid fixation at 10 days but no particluar association with the gait pattern (Figures 3 –5). In addition, gait alteration at 10 days could be likely caused by the ongoing functional restoration of the bone rather than pain, which is also supported by the absence of additional clinical signs at 72 h. 17
The main pain origin is supposed to result from the densely innervated periosteum and depends on the intensity of surrounding soft tissue trauma. 12 In closed mouse fracture models with stable fixation and controlled induction of fracture, the soft tissue trauma can be limited, and pain is expected to peak at two days post-fracture, which is comparable to humans.11,16 After relief of the acute pain during the first days post-fracture, ongoing sub-acute pain is more likely to be associated with the soft tissue trauma while chronicity becomes apparent when the pain persists after complete healing. 12 Several studies focusing on the development of fracture pain have shown more persistent pain behaviours over weeks and some of these studies also monitored rear up behaviour post-interventional.20 –24 For example, a study from Koewler et al. studying fracture-related pain behaviours in closed femur fractures in male C57BL/6J mice identified reduced rearing frequency over 10 days after fracture with no improvement upon specific anti-nerve growth factor (NGF) treatment. 21 Reduced rearing behaviour was also observed by Das and colleagues over 12 weeks when the fractured tibia was stabilized by a cast for three weeks after fracture and compared with a naïve control cohort (no fracture/cast). 20 However, it is important to note that these studies differed in pain management after anaesthesia and/or fracture induction in order to avoid potential interference with pain behaviour and/or to test the potential of specific anti-NGF treatments. This is in contrast to our experimental design, in which mice received a single injection of buprenorphine during surgery and tramadol via the drinking water one day prior and three days post-osteotomy. In a previous study, we initially observed that rearing behaviour occurred rarely, if at all, at 1 h and 6 h after osteotomy, which was comparable to control mice only undergoing anaesthesia and receiving analgesics. 16 Moreover, the data suggested that the reduction in rearing duration was independent of the analgesic applied via the drinking water (tramadol in low and high concentrations vs. buprenorphine) but more reduced in the group receiving higher concentrations of tramadol. 16 Thus, in this current study, we focused on rearing behaviour at 24 h, 48 h and 72 h post-osteotomy, giving the animals time to recover from surgery, while still covering the phase of expected peak pain post-osteotomy. Another study, performed by Magnusdottir et al., assessed pain behaviours in entire male mice and ovariectomized female C57BL/6 mice over six weeks after osteotomy with an external fixator and postoperative analgesia with buprenorphine in a jelly for three days. 22 They observed mechanical and thermal hyperalgesia in all mice post-osteotomy and nociceptive sensitization for up to six weeks. 22 However, their data did not show significant differences in the rearing frequency (measurement started at seven days after surgery), which was monitored using the Laboratory Animal Behaviour Observation Registration and Analysis System for automated home cage monitoring overnight. 22
We also observed that male mice with flexible fixation showed significantly less rearing behaviour than those with rigid fixation at 10 days (Figure 3). Although it is known that more flexible fixations lead to delayed bone regeneration and larger callus formation, data from the above discussed studies indicated comparable nociceptive sensitization in intramedullary pin or external fixator fixations.12,22 However, in our previous study, Catwalk analysis of mice from the flexible group also indicated reduced velocity and relative stride length, suggesting a potential limitation in functional recovery at 10 days which might affect normal walking. 17
It has been reported that rearing behaviour can be affected by numerous factors such as anxiety/fear, injected substances, the light/dark phase, pain, habituation, novelty and familiarization to the environment, the set-up in which rearing is measured, the environment, noise, stress and more.18,30 In addition to these external influences, rearing can also be affected by intrinsic factors such as sex, strain and age.32 –35 Thus, in our study, we tried to reduce and control for as many factors as possible. Therefore, mice of both sexes were trained and were tunnel handled by maximal two experimenters. Moreover, individual baseline measurements were taken, the observations were performed at the same time of the day and the anaesthesia effect was examined on an individual base. Our data showed that rearing behaviour at 24 h, 48 h and 72 h after anaesthesia was not influenced by anaesthesia (Figure 2), which might be an advantage of rearing assessment compared with other methods such as measuring the food and water intake, body weight and the examination of the mouse grimace scale, because these methods can already be altered by anaesthesia and analgesia.16,36,37
However, our study still has limitations. We used only one strain of mice (C57BL/6N) and previous studies reported distinct differences between strains in rearing behaviour.32 –34,38 Moreover, we performed our observations during the light phase, although mice are nocturnal and therefore most active during the night, with previous studies describing differences in rearing behaviour during light and dark phases.23,33,39 In addition, we recorded and analysed the rearing behaviour only during a short timeframe of 3 min to keep the manual analysis feasible. Majuta et al. showed that activity was greatest during the first 30 min after mice were placed in the activity boxes, that is, during the exploratory period, and for the first 3 h following the onset of the dark cycle coinciding with peak night activity. 23 Fahlström et al. also showed that the differences in rearing frequency was most striking in the first 30 min. 35
Because rearing behaviour, comparable to nest building and burrowing, is a normal and spontaneously displayed behaviour, it is relatively easy to assess. 18 However, manual assessment of rearing behaviour is highly time-consuming, while automatic assessments require less time and therefore allow for longer observation periods than manual analyses. Regarding the time-consuming nature of assessing rearing behaviour manually, the number of animals and number of time points is greatly limited and a real-time assessment not possible. Based on these limitations and the associated highly time-consuming analyses, it might be advisable to use automated home cage analysis systems in future studies. The technological progress in automated home-cage monitoring technologies within the last years is promising to make the required equipment more affordable and feasible for routine monitoring of pain or wellbeing in mice.
Our results show that rearing behaviour is reduced after osteotomy with stable external fixation in adult male and female C57BL/6N during the initial phase (72 h) and only partly restored at day 10 post-osteotomy. We found differences between male and female mice and the stability of the external fixation (rigid vs. flexible). In line with previous studies,30,31 the familiarization with the environment might have a more profound effect on this behavioural change than the potential restriction in walking, limiting the significance of rearing as additional indicator for model-specific skeletal pain. Together with the time-consuming nature of the manual assessment of rearing, it seems to be not a practicable tool to be used for routine clinical and behavioural assessment after fracture surgery in mouse models. However, if automated systems are available, limping analysis might be useful as a further pain-indicative behaviour. Finally, further studies are needed to evaluate the value and predictive capacity of changes in rearing behaviour to assess pain in skeletal-impaired mouse models as the current literature on this specific topic is still rare.
Footnotes
Acknowledgements
We like to thank Dr Kristina Ullmann, Bianka Verrett and Sylvia Gundelach for their support during conduct of the animal experiments.
Data availability statement
The authors declare that all data supporting the findings of this study are available within the paper. Further information is made available by the authors upon request.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Charite3R, Charité-Universitätsmedizin Berlin (Project acronyms: RefineMOMo 2.0; RefineLab).
