Abstract
Mucositis is a common and serious side-effect experienced by cancer patients during treatment with chemotherapeutic agents. Consequently, programmes of research focus on the elucidation of novel therapeutics for alleviation of mucositis symptoms, and these frequently use animal models. However, although these models are assumed to be painful and distressing to the animal, endpoints are difficult to determine. The aim of this study was to evaluate whether a change in burrowing behaviour could provide an indication of disease onset and potentially be applied as a humane endpoint. Baseline burrowing behaviour was measured in healthy animals on three occasions by determining the weight of gravel displaced from a hollow tube. Mucositis was then induced in the same animals by intraperitoneal injection of 5-fluorouracil (150 mg/kg) and burrowing behaviour recorded over three consecutive days. Standard measures of disease progression, including body weight loss and clinical score, were also made. The presence of mucositis was confirmed at necropsy by findings of decreased duodenal and colon lengths, and reduced liver, spleen and thymus weights in comparison with non-treated control animals. Histological score of the jejunum and ileum was also significantly increased. Mucositis onset coincided with a decrease in mean burrowing behaviour which was progressive, however this result did not achieve statistical significance (P = 0.66).We conclude that burrowing may be a useful indicator of inflammation in the mucositis model, although this requires further characterization. Pre-selection of animals into treatment groups based on their prior burrowing performance should be pursued in further studies.
Mucositis is a frequent side-effect arising in 60% of cancer patients treated with chemotherapeutic agents.1,2 This serious condition produces an array of clinical symptoms which may include abdominal bloating, pain, nausea, diarrhoea and weight loss. 3 The condition results from the inhibitory effects of chemotherapy on DNA replication and mucosal cell proliferation. 4 This direct inhibition causes histological changes in the gastrointestinal tract which includes apoptosis, crypt cell hypoproliferation, villous atrophy, collagen breakdown, ulcerated lesions and increased intestinal permeability. 4 The serious nature of these histological features and symptoms renders mucositis as the major dose-limiting factor in cancer treatment regimens. 2 As a result of this, considerable research focus is directed towards the identification of therapeutic targets for symptomatic relief of mucositis symptoms. Much of this research utilizes various rodent models of chemotherapy-induced mucositis.
The rat is a frequently used model for investigation of cancer-therapy-induced mucosal injury and is routinely used for the investigation of new anti-mucotoxic agents. 5 Administration of a chemotherapeutic agent such as 5-fluorouracil (5-FU) or methotrexate, usually via intraperitoneal injection, results in a rapid-onset induction of mucositis identified retrospectively by histological analysis.6,7 For researchers and animal ethics committees a delicate balance has to be struck between achieving the level of inflammation required to investigate disease processes and minimizing animal distress. Given the severity of symptoms reported by human clinical patients, it is assumed, by extrapolation, that mucositis is similarly painful and distressing to rats. Reliable methods of monitoring progression of pain or distress are therefore needed, and humane endpoints need to be set. Pain diagnosis in the rat is challenging because as prey species they tend to mask signs of pain to avoid predator interest. 8 There are therefore few reliable indicators of spontaneous pain that can be used, and many of those indicators that are available are model specific. 9 To date, the most consistent and widely-used indicators of well-being in mucositis models have been body weight change and non-specific clinical scoring. However, these are often retrospective and may not be indicative of the animal’s actual emotional experience.
Spontaneous burrowing behaviour has recently been identified as a promising sign for pain identification in rodents in inflammation-associated models,10–13 complex regional pain syndrome, 14 in prion disease 15 and post-laparotomy; 16 the presumptive basis of the burrowing test being the natural inclination of rodents to burrow, which is a highly conserved behaviour. 17 Studies to date have shown that a decrease in burrowing behaviour correlates well with inflammation or pain onset, and is a sensitive indicator of disease progression. As a low-cost and simple technique it may therefore offer considerable value in the establishment of humane endpoints, as well as in pre-clinical screening of symptom-ameliorating therapeutic agents. However, the majority of studies to date have investigated the behaviour in mice,12–14,16 and in longer-term disease states. 11 The aim of this study was to assess the utility of burrowing behaviour as an indicator of pain or distress in a short-term rat mucositis model, expected to cause an insult characteristic of acute pain.
Materials and methods
Animals and experimental design
The study used 22 male Hsd: Sprague Dawley SD rats (168 ± 14 g) sourced from a barrier-maintained specific pathogen-free (SPF) commercial production facility (University of Adelaide, Laboratory Animal Services, Adelaide, Australia). The source colony was health screened as negative for a range of viral and bacterial rat pathogens including rat coronavirus, parvovirus, Mycoplasma pulmonis and Klebsiella pneumoniae, amongst others. The animals were housed individually in metabolic cages (Tecniplast, Exton, PA, USA) for acclimatization and data collection. Room temperature was maintained at 21–23℃ with a 12 h light/dark cycle. The animals were provided with ad libitum access to potable RO water, and a standard casein-based diet. 18 For in life studies, animals acted as their own controls to minimize the effects of inter-individual variability in data interpretation. Rats were gavaged with saline once daily by the orogastric route for nine days as is standard practice in the disease control group for therapeutic testing studies (between days 0 and 8). On day 6, animals (n = 16) received an intraperitoneal injection of 5-FU at a dose rate of 150 nmg/kg (Mayne Pharma Pty Ltd, Mulgrave, Victoria, Australia) or saline of an equivalent volume (tissue control group, n = 6). Rats were humanely killed at 72 h post-5-FU (or saline) injection by CO2 asphyxiation.
The animal housing and experimental protocols were approved by the Animal Ethics Committee of the University of Adelaide and conducted in accordance with the provisions of the Australian code for the care and use of animals for scientific purposes. 19 This study was conducted as part of a larger programme of work investigating novel therapeutic agents for the treatment of chemotherapy-induced mucositis and hence experimental timeline was influenced by the concurrent study. Provisions of the ARRIVE guidelines were incorporated in study reporting. 20
Daily metabolic data and clinical scoring
Body weight, food and water intake, and faecal and urine output were monitored and measured daily. As a requirement of animal ethical approval, the severity of mucositis was assessed daily, using a disease activity index (DAI), which scored body weight loss, rectal bleeding, stool consistency and general condition of the animal. Each parameter was graded in severity on a scale from 0–3, which was summed to achieve a total score, as described previously. 21
Behavioural testing with burrowing apparatus
The burrow design was adapted from Andrews et al. (shown in Figure 1).
11
Burrows were made in-house and consisted of hollow plastic tubes (200 mm length × 90 mm diameter) sealed at one end. The open end was raised 60 mm above the cage floor to minimize gravel loss when placed in the test cage. Each burrow was filled with 1 kg of gravel (7 mm pea-shingle decorative gravel; Tuscan Path, Hawthorn East, Victoria, Australia). Burrows were located in a test cage which was a standard open-top rat cage (590 mm × 385 mm × 200 mm; Tecniplast, Rydalmere, New South Wales, Australia). These cages had standard recycled paper bedding (Animal Bedding, Fibrecycle, Yatala, Queensland, Australia) as a substrate. Animals had been housed in these cages upon arrival in the facility during acclimatization, and the animals were always placed in their own cage during testing. This was done in order to reduce the effects of novelty during the burrowing test.
The burrowing apparatus.
Rats were habituated to the test cages and burrowing apparatus on two consecutive days prior to experimental treatments. To perform the test, rats were removed from their home metabolic cages and placed into the test cage for one hour. Testing was conducted during the first hour of the animals’ dark cycle. At the end of the test period, the remaining gravel in the burrow was weighed to determine the volume of displaced gravel. To determine the effect of mucositis on burrowing behaviour, rats were assessed for level of baseline burrowing on experimental days −2, 0, and 4 (48 h prior to mucositis induction). Further tests were then performed on experimental days 6–8 following mucositis induction (Figure 2).
Experimental timeline indicating time-points for burrowing data collection.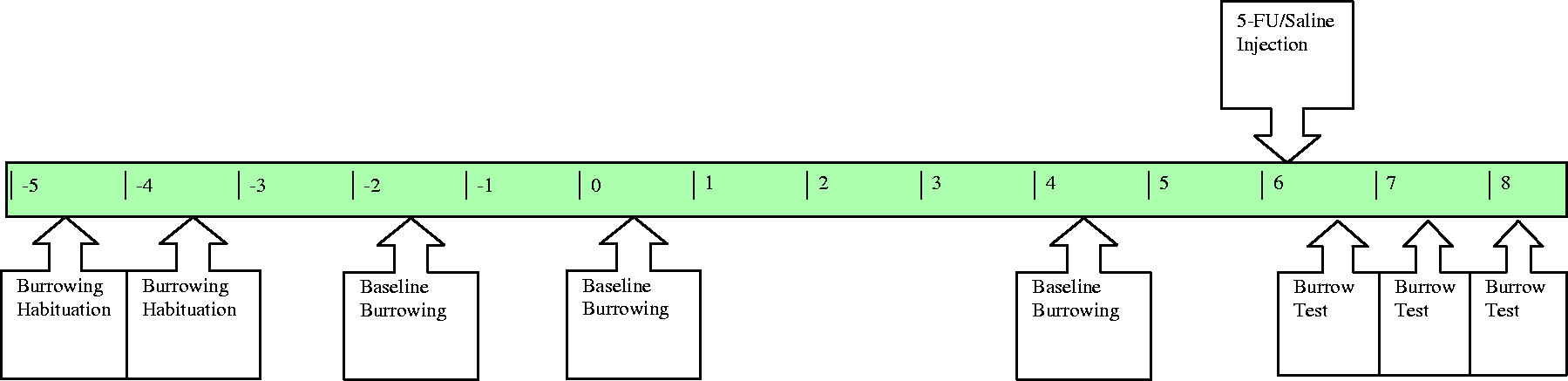
Tissue collection
Following euthanasia, the gastrointestinal tract was removed and placed onto an ice-cold slab. The stomach, duodenum, jejunum, ileum, caecum and colon were weighed with the contents removed. The length of small intestinal tract sections was measured un-stretched. Lengths (2 cm) of the duodenum, jejunum, jejunum–ileum junction, ileum and colon were excised and placed in 10% buffered formalin for histological analysis. The thymus, spleen, heart, lungs, liver, kidneys and adrenal glands were weighed, and samples also taken for histology.
Histological analysis
Tissues samples collected were formalin-fixed for 24 h and then transferred to 70% ethanol, prior to routine histological processing. Transverse tissue sections (4 µm) were embedded in paraffin wax, H&E stained and viewed using a light microscope (Olympus CX-41; Olympus, Tokyo, Japan).
A semi-quantitative scoring system was used to assess intestinal damage of the jejunum and ileum, and assigned an overall damage severity score. Untreated rat intestinal tissue, banked from previous studies, provided a baseline against which to grade all tissues collected in this study. Eight criteria were considered in scoring with each being graded on a scale from zero (normal) to three (maximal damage), that is a maximum attainable score of 24. These criteria included: villus blunting, crypt distortion, reduction in goblet cell number, dilation of lymphatics, thickening of the submucosa, thickening of the muscularis externa, enterocyte disruption and lymphocytic cell infiltration. 6 Tissues were scored in a blinded fashion by one of the authors experienced in histological assessment.
Statistical analysis
Statistical analyses were conducted using XLSTAT (Version 6, 2013). Overall disease severity score was expressed as a median score. Numerical data were expressed as mean ± standard error of the mean. Tissue comparisons between the disease model and healthy animals were made using the student’s t-test. Non-parametric histological scores were compared using a Wilcoxon–Mann/Whitney test. Data recorded daily were analysed using repeated measures analysis of variance (ANOVA) with a post-hoc Tukey’s test. Behavioural data were non-parametric. Due to the limited number of repetitions of each burrowing test transformation of these data was deemed inappropriate and instead the Friedman test was applied with a Dunn’s post test. Differences between means were determined to be significant when P was less than 0.05.
Results
Body weight data and organ weights and lengths
Body weight data are shown in Figure 3. Prior to chemotherapy injection all animals showed a gain in body weight. After injection all animals showed a progressive decrease in body weight which was significantly different at 24, 48 and 72 h in comparison to day 6 (the last day prior to injection).
Change in body weight (a) prior to 5-fluorouracil (5-FU) injection and (b) following 5-FU injection. Data are expressed as mean ± SEM. Significance (P < 0.05) is indicated by a = in comparison with trial day 0 and b = in comparison with day 6 (the last day pre-injection).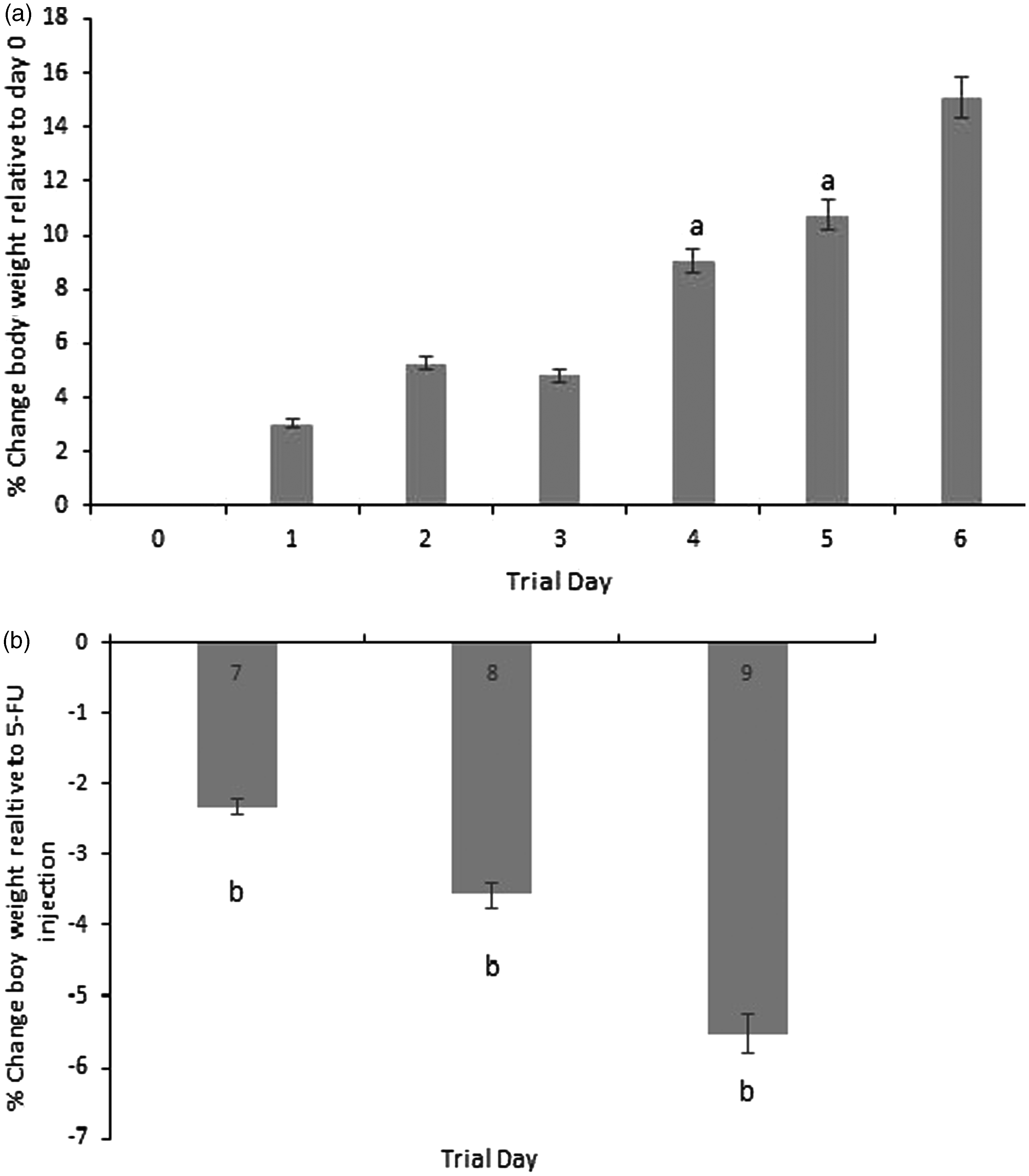
A significantly decreased duodenal (5.3 ± 0.3 cm versus 7.0 ± 0.4 cm; P = 0.006) and colon length (15.2 ± 0.3 cm versus 17.3 ± 0.4 cm; P = 0.002) was observed in chemotherapy-treated animals in comparison to healthy age and sex-matched control animals. A significant decrease in thymus, spleen, liver and jejunoileal weights was also observed in chemotherapy- treated animals (Figure 4). Adrenal gland weights between the two groups were not statistically different.
Weights of visceral and gastrointestinal organs expressed as a percentage of body weight obtained 72 h post 5-fluorouracil (5-FU) injection and compared with age and sex-matched control animals. Data are expressed as mean ± SEM. Significance (P < 0.05) is indicated by a = in comparison with healthy control animals.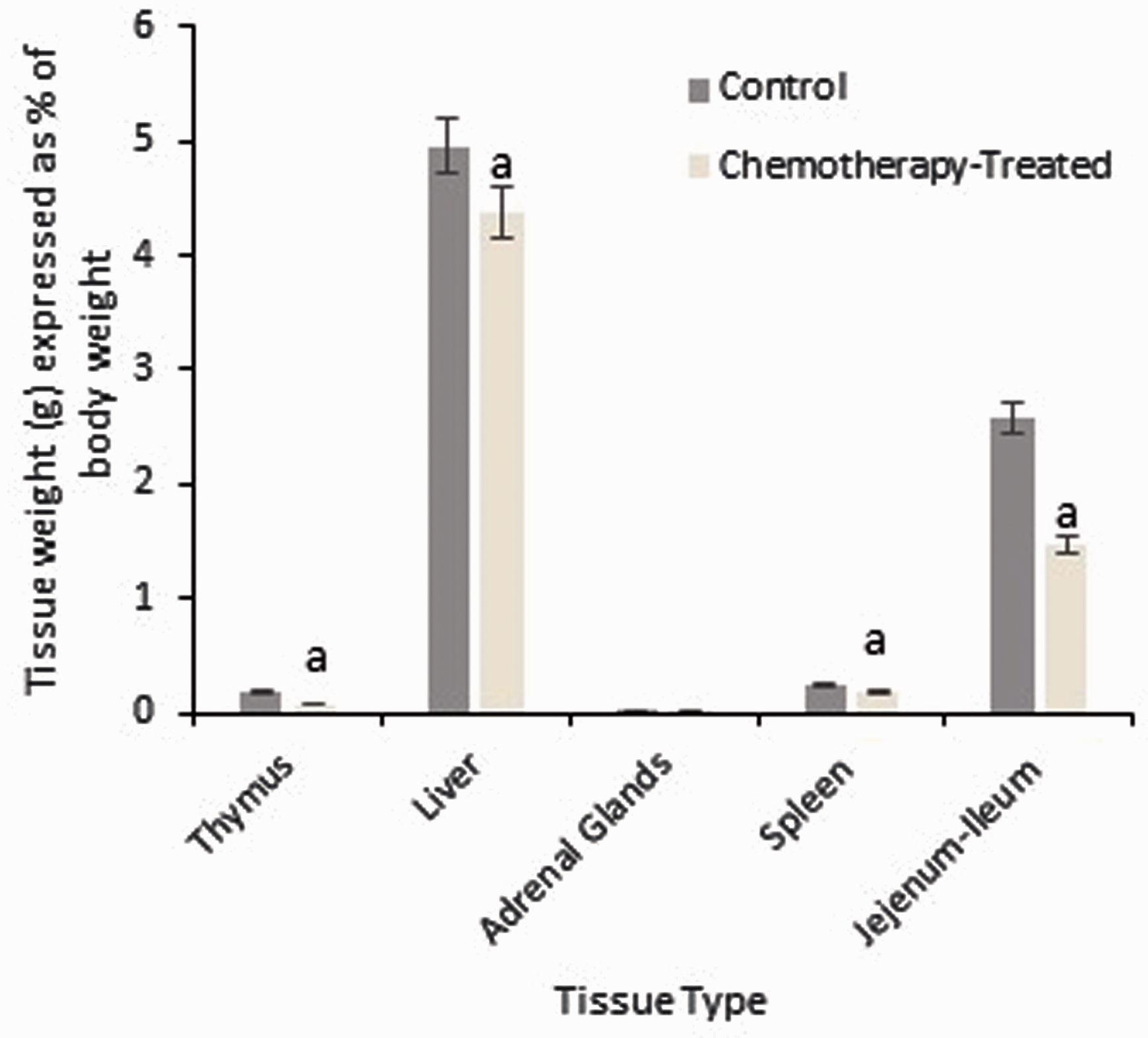
Histological analysis
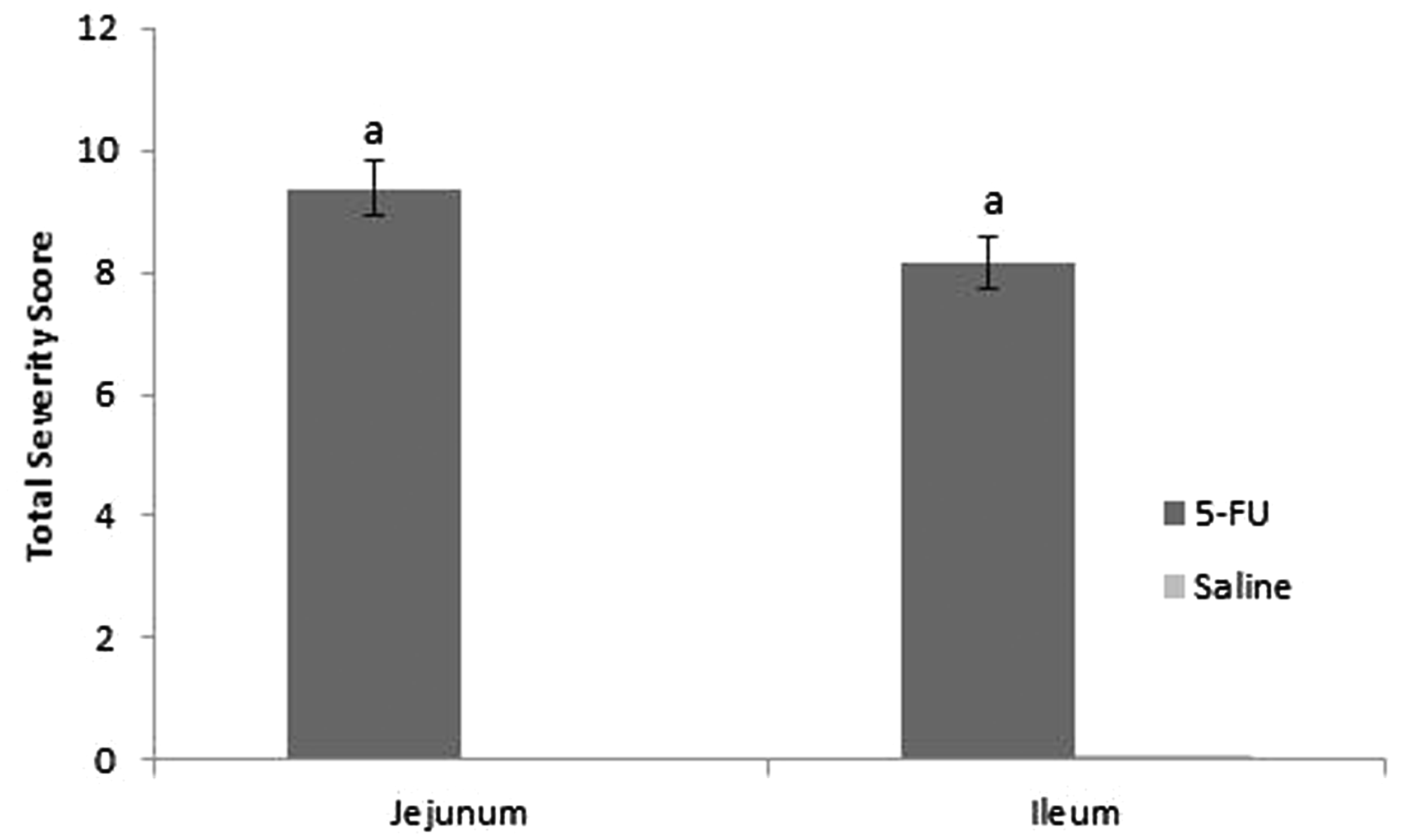
Typical histological features of the proximal jejunum and distal ileum are illustrated in Figure 5. 5-FU injection resulted in tissue damage consisting of villus stunting, crypt cell distortion, reduced goblet cell numbers, polymorphonucleocyte infiltration and thickening of the submucosa and muscularis externa. Histological severity score was significantly increased in the 5-FU treated animals in comparison to the control animals for both tissue types examined (P = 0.0004), providing evidence that the mucositic condition was present (Figure 6).
Photomicrographs (×100) of H&E stained proximal jejunal and distal ileal histological sections in rats 72 h post 5-fluorouracil (5-FU)/saline injection. (a) Jejunal section from saline-injected animal; (b) Jejunal section from 5-FU-injected animal; (c) Ileal section from saline-injected animal; (d) Ileal section from 5-FU-injected animal. Note: Loss of epithelial structure in the crypt and villus, reduced goblet cell numbers and thickening of the submucosa and muscularis externa in chemotherapy-treated animals. Total severity score (out of 24) for the proximal jejunum (10%) and distal ileum (90%) determined through evaluation of eight characteristics. Data are expressed as mean ± SEM. Significance (P < 0.05) is indicated by a = in comparison with healthy control animals.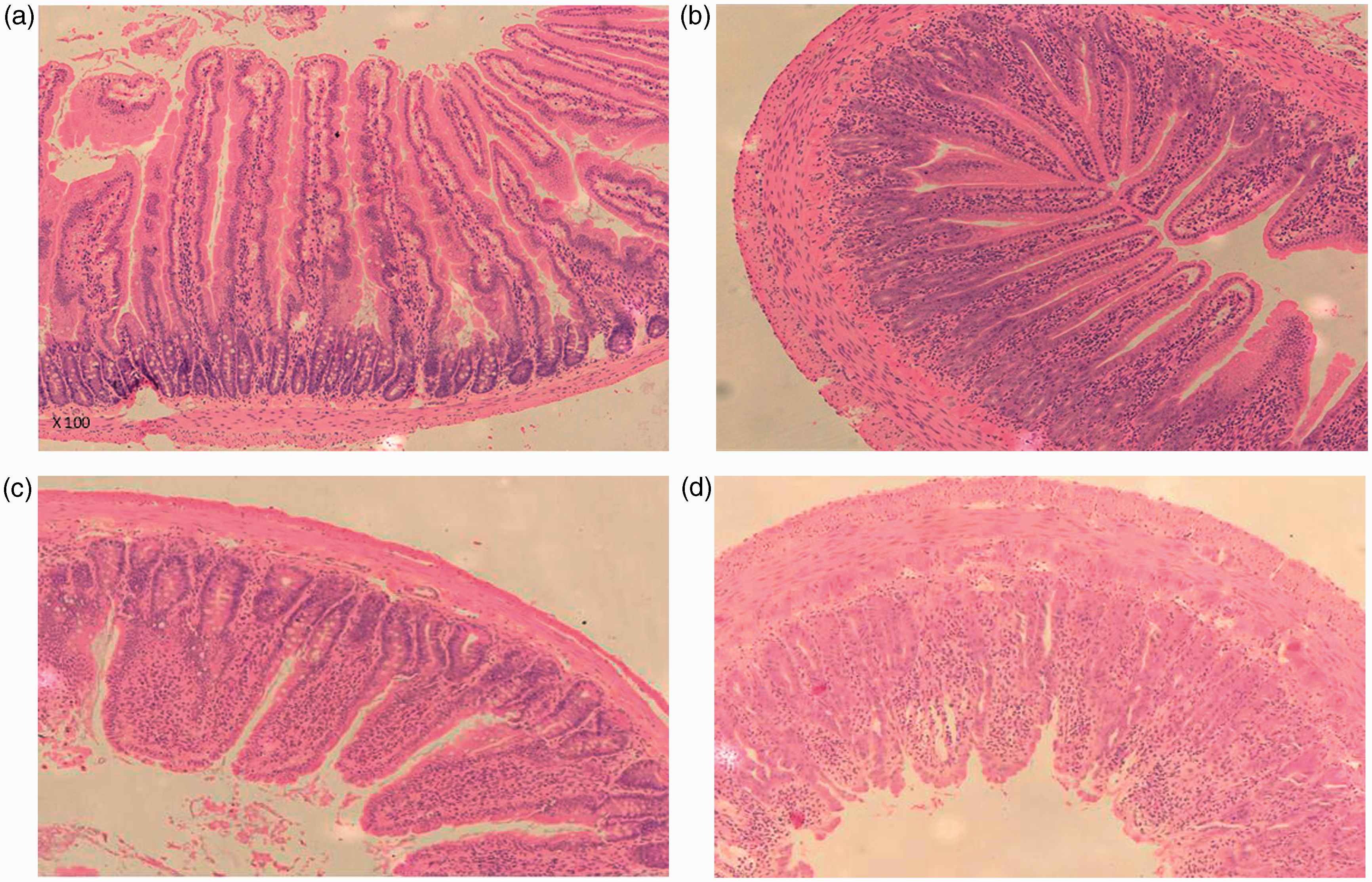
Burrowing behaviour
The burrowing test was simple to perform and animals that had a burrowing tendency were observed to commence this behaviour within the first 10 min of placement in the test cage. Two habituation phases on consecutive days were provided. Out of the 16 animals tested only one animal performed burrowing behaviour on the first exposure to the burrows. On the second day this was reversed with all animals performing burrowing behaviour except one.
Figure 7 shows the volume of gravel displaced over the three baseline periods prior to mucositis induction and in the days following 5-FU injection. While there was a decrease in burrowing behaviour following the injection (which was progressive), no statistical significance was found between means at any of the time-points assessed when analysed using the Friedman statistical test (P = 0.66). An important finding was that the results showed significant intra- and inter-individual variations at all time-points. To illustrate this, the mean for the range of gravel displaced, that is the difference in gravel volume displaced by the rat demonstrating the poorest, versus the most superior burrowing performance, across all time-points was 843 g. In some individual animals burrowing values across the three baseline test days also showed high variability, and in the most extreme case this ranged from displacement values of 10.6 g to 936.6 g. As shown in Figure 7 the mean maximum of gravel displaced at any time-point was at day 4 when 327 g was removed (approximately 33% of total gravel). There was no correlation between histological severity score and burrowing behaviour (R2 = 0.04).
Volume of gravel displaced from test apparatus over a one-hour period. Measurements were taken during baseline testing and after mucositis induction. Data are expressed as mean ± SEM.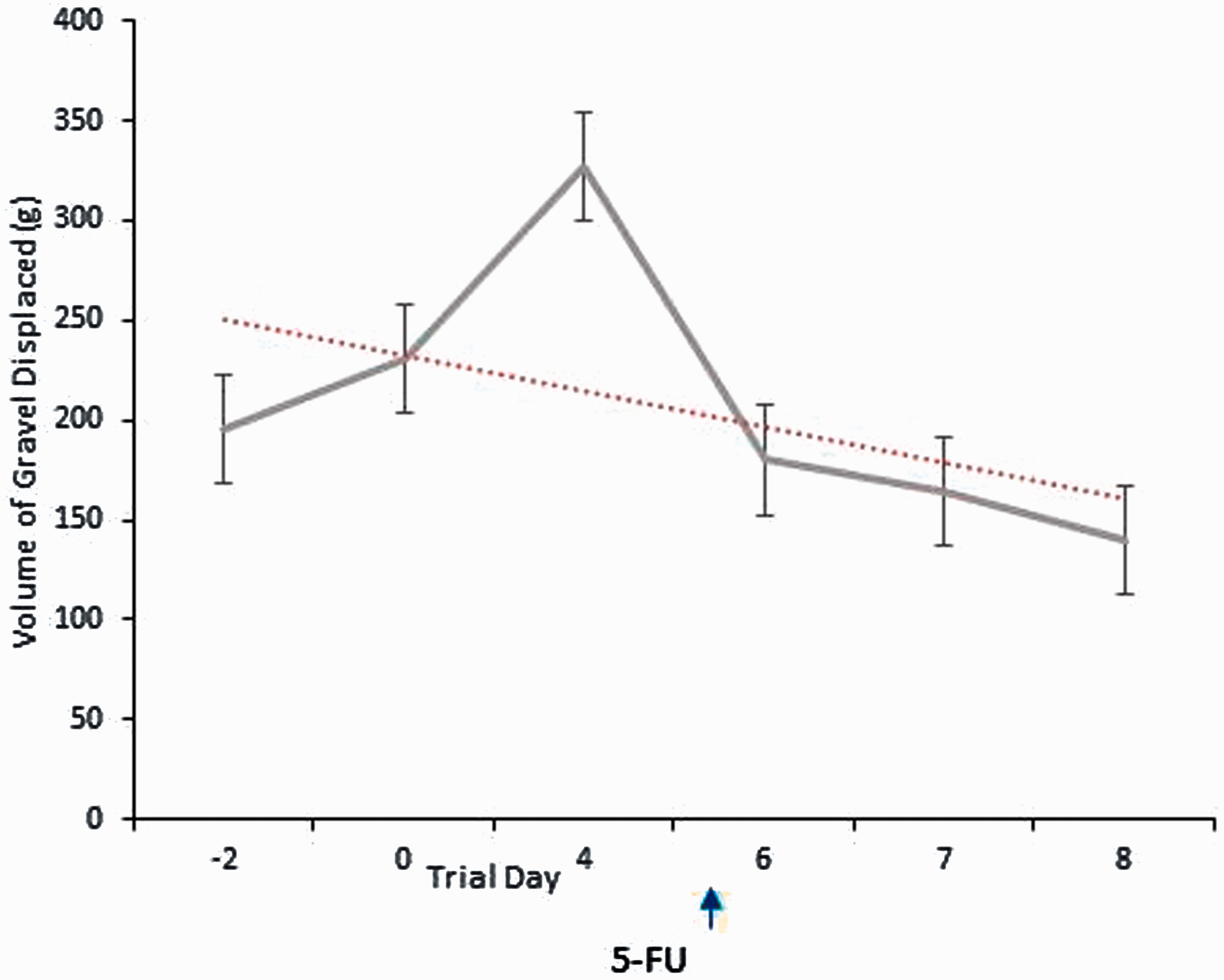
Discussion
The aim of the experiment was to investigate whether burrowing behaviour was a useful assessment tool to evaluate welfare based on its correlation with standard parameters of mucositis disease progression, such as body weight loss. Illness was clearly induced by the injection of 5-FU as evidenced by routinely measured parameters such as body weight loss and DAI. This illness was confirmed as the inflammatory mucositic condition by retrospective examination. The administration of 5-FU significantly decreased duodenal and colon lengths, as well as causing weight reduction of the thymus, spleen, liver and jejunoileum as previously shown.22,23 As predicted, the mucosa was negatively affected histologically as demonstrated by increased histological severity score and observed changes including villus blunting, crypt cell disruption and enterocyte disruption. Burrowing behaviour of individuals showed no correlation with mucosal severity score but this lack of significance may have arisen as a result of a degree of insensitivity in the histological scoring such that there was little inter-individual variation in histology score. Alternatively, due to the affective component in pain perception, there may be a poor correlation between actual intestinal damage and perceived pain, hence burrowing behaviour.
Absolute values for volume of gravel displaced were much reduced in comparison to other studies with a mean maximum volume of gravel displaced of 33% in comparison to approximately 64% in previous rat experiments.10,11 The current study did modify previous experimental designs by using a reduced total volume of gravel (1 kg versus 2.5 kg). However, this was not anticipated to have influenced the propensity to burrow. Instead, this finding of reduced gravel displacement was likely to be as a result of test animal selection (see later). There was a progressive decrease in mean levels of burrowing behaviour following mucositis induction, as evidenced by reduced gravel displacement. However, these values were not statistically different from baseline burrowing behaviour. This could have arisen as a function of insufficient power due to too small a sample size, but given that comparison P values were well outside significance (P = 0.66) this was unlikely. Additionally, Deacon et al. reported that group sizes of 10 were adequate for detecting treatment effects on burrowing, although this was referring to a mouse model. 17 The results of the current study do however demonstrate that burrowing behaviour shows large inter-individual, as well as intra-individual variability. This does not appear to have been reported specifically in previous studies, but rat studies to date,10,11,17 have selected rats to proceed into burrowing studies on the basis of their performance in baseline tests. During such a selection procedure, experimental groups have combined similar numbers of poor and superior burrowing animals across all treatments.
Performance of this pre-selection is more likely to yield a treatment effect on burrowing behaviour, 17 and be appropriate for studies looking into the therapeutic effect of agents. However, in the current study, where the primary focus of burrowing investigation was to allow assessment and implementation of endpoints, this selection was not feasible. Hence, the technique may find limited utility in this context. The significant intra-individual variability could also pose issues for selection of appropriate animals based on baseline values. Andrews et al. 11 conducted baseline tests on two separate occasions, and Rutten et al. 10 on one day, and used these test results to select animals to go into burrowing experiments. However our baseline data shows that individual burrowing values across the three baseline test days could show a large range, hence choice of baseline test day may have considerable influence on the animal’s burrowing behaviour, and its consequent performance in the experimental tests. Previous murine studies have not shown the variability in baseline burrowing values evidenced in the current study.12,15 It is possible that extraneous factors such as facility noise, personnel movement and the move to a novel environment for the burrowing test influenced the animals’ burrowing propensity. However, given that the tests were performed outside of normal working hours in a relatively quiet facility we believe this influence would have been minimal. Given these data it would seem prudent to perform at least three baseline burrowing tests in order to evaluate average and variance of an individual’s burrowing behaviour, prior to experimental group allocation, and before excluding non-burrowing animals. The high level of inter-individual variance shown when animals were not pre-selected also makes it imperative to use animals as their own controls, and to use a number of experimental time-points for assessment of burrowing behaviour in the diseased state.
Our results indicate that a habituation phase to burrowing is critical, and this is likely to improve burrowing propensity and diminish variability between animals. 17 Burrowing tendency is therefore likely to change with experience, and if such a phase is not incorporated into studies, initial novelty of the test set-up may confound results. Other investigators have similarly incorporated such a ‘training’ phase.10,11,17 However, one study demonstrated only a slight tendency for burrowing to increase with experience in rats and mice using burrowed food pellets, but this difference was not of statistical significance. 24 This study highlighted that rats burrowed proportionately less substrate than other species tested compared with their body mass, and therefore study comparisons between rats and mice should be made with care due to these inherent species differences. In our study rats were also housed in a scenario likely to elicit a stress response. Metabolic cage housing consists of grid flooring, an absence of bedding substrate and a smaller living space, all of which have been shown individually to lead to negative behavioural and physiological responses in rodents.25–32 While there have been few studies evaluating the effect of housing condition on burrowing tendency, Bangsgaard Bendtsen et al. did demonstrate that mice increased their burrowing behaviour when subjected to grid-floor housing. 33 As such a consideration of housing scenario may be important in study interpretation.
Previous rat models have investigated burrowing behaviour in peripheral nerve injury models, 11 or inflammation associated pain resulting from the administration of complete Freund’s adjuvant.10,11 These models resulted in an insult that would be expected to impair locomotory behaviour and hence may have had a particular effect on a physical behaviour such as burrowing. However, there is evidence to suggest that burrowing behaviour may be impaired prior to motor impairment possibly due to hippocampal neuropathological changes.13,34–36 Models which would lead to a visceral pain insult, such as that expected as a result of mucositis, have not been investigated in relation to effects on burrowing in the rat. However, burrowing has been shown to decrease following an acute visceral painful event, that of laparotomy, in a mouse model 16 and in a mouse colitis model. 12 Nevertheless, given the previous discussion concerning the difficulties of making species comparisons it may be unwise to extrapolate conclusions from these studies to rats. Sickness behaviour following immune system activation as a result of viral or bacterial infection has also been shown to decrease burrowing performance.13,37,38 Such behaviours are normally evidenced by alterations in motivation towards normal species activities. 39 The malaise-causing effects of intestinal mucositis caused by pro-inflammatory cytokine release, 40 may then also influence burrowing performance in addition to the effects of pain itself. It is possible that the visceral pain insult created as a result of mucositis, especially over the short-time course of the current study, was too subtle to yield any measurable difference in effect on this physical behaviour. Future studies should however consider the evaluation of visceral pain, and its correlation with burrowing behaviour, in rat models, making use of rats selected for their burrowing behaviour.
It is imperative to be able to monitor welfare, and establish and set humane endpoints in animal models such as the mucositis model applied here. Current strategies commonly used in this research area include body weight loss and general clinical condition scoring. However, these parameters suffer from being non-specific to pain and often being retrospective in nature. As such, finding a reliable and sensitive indicator of pain that could be measured simply and inexpensively would be a valuable refinement mechanism. Burrowing, as a non-essential maintenance behaviour, 16 has the potential to provide such a solution, and has shown considerable promise in mouse models of visceral disease. 12 However, based on the current study, we believe that it has limited application at the present time in a rat model for the establishment of humane endpoint criteria. This is due largely to the wide degree of inter- and intra-animal variability which makes interpretation of data difficult. Nevertheless, with further study, it may prove to be useful in applied research investigating analgesic effects on behavioural parameters; or testing novel therapeutics, where pre-selection of rats based on burrowing behaviour would be possible.
Footnotes
Acknowledgements
Thanks are extended to Dr David Butler for his invaluable statistical advice. Prof Gordon Howarth is supported by a South Australian Cancer Research Collaborative Senior Research Fellowship.
