Abstract
Welfare problems in laboratory mice can be a consequence of an ongoing experiment, or a characteristic of a particular genetic line, but in some cases, such as breeding animals, they are most likely to be a result of the design and management of the home cage. Assessment of the home cage environment is commonly performed using resource-based measures, like access to nesting material. However, animal-based measures (related to the health status and behaviour of the animals) can be used to assess the current welfare of animals regardless of the inputs applied (i.e. the resources or management). The aim of this study was to design a protocol for assessing the welfare of laboratory mice using only animal-based measures. The protocol, to be used as a benchmarking tool, assesses mouse welfare in the home cage and does not contain parameters related to experimental situations. It is based on parameters corresponding to the 12 welfare criteria established by the Welfare Quality® project. Selection of animal-based measures was performed by scanning existing published, web-based and informal protocols, and by choosing parameters that matched these criteria, were feasible in practice and, if possible, were already validated indicators of mouse welfare. The parameters should identify possible animal welfare problems and enable assessment directly in an animal room during cage cleaning procedures, without the need for extra equipment. Thermal comfort behaviours and positive emotional states are areas where more research is needed to find valid, reliable and feasible animal-based measures.
The welfare of laboratory rodents has long been a focus of attention. Refinement work within the 3R (replacement, refinement and reduction) concept is used ‘to improve scientific procedures and husbandry which minimize actual or potential pain, suffering, distress or lasting harm and/or to improve animal welfare in situations where the use of animals is unavoidable’. 1 Many of the indicators of poor welfare identified for laboratory mice, such as stereotypic behaviours, barbering of whiskers or fur, and aggression between group-housed males,2–4 occur irrespective of the type of research. They occur in the home cage where the animal spends the majority of its lifetime, and ‘refinement’ applies to the lifetime experience of the animal. 1 Furthermore, factors such as the progression of disease within an animal model or the effects of experimental procedures may themselves lead to deteriorated welfare in the home cage. If so, the cage environment needs to be adapted to meet the specific needs of these animals (e.g. easier access to food and water, softer bedding) in order to reduce the risk of poor welfare. Assessment protocols that aim to give an overview of the welfare of laboratory rodents have been presented in recent years, but to our knowledge, with one exception, 5 they are related to experimental procedures 6 or specific animal models, 7 rather than to a general assessment of the welfare in the home cage. There is as yet no generally accepted protocol that can be used for benchmarking welfare outcomes in an animal facility. This paper is part of a larger project and the protocol presented here has been tested in practice at three animal facilities in Sweden. 8
Appropriate resources and management, such as suitable bedding, nesting material and husbandry routines are preventive measures, or inputs, which reduce the risk of poor animal welfare. However, no matter how good or well thought out this input is, animals of different strains, sexes and ages will react differently to the input, based on genetic background and prior experiences, leading to different welfare outcomes for different individuals.9,10 In addition, even apparently similar environments may be managed differently by animal caretakers, further affecting the animals' experience of a particular situation. 9 This means that when assessing animal welfare it is important to measure the outcomes on the animals themselves, using animal-based measures. 11 Animal-based measures can be applied regardless of housing and management routines and are sensitive to variations in management and to environment–animal interactions. 9 A way of identifying the strengths and weaknesses of husbandry systems in terms of how they impinge on the animals is by using animal-based measures. 12 Therefore, the protocol presented here uses only animal-based measures to assess animal welfare.
When developing this protocol, the intention was to enable identification of possible welfare problems and to benchmark these as animal welfare outcomes for comparisons over time. By assessing welfare at regular intervals, this protocol can be used as an ongoing management tool to document changes in the welfare of animals at a facility. Benchmarking is a way of evaluating current performance, relative to others or oneself, and of identifying areas for improvement. 13 The protocol could be used in all types of laboratory rodent facilities and so be part of the official inspection of the facilities by the regulator or competent authority, even if it may not be necessary to check all measures in order to comply with legislation. The protocol could also be adapted for use in the assessment of the actual severity of experimental procedures. 14 It is further proposed that the work behind establishing this protocol could even be useful for targeting future research areas by leading to the identification of areas where more research is needed to find valid (and feasible) animal-based measures of assessing the welfare of laboratory mice.
The idea behind the protocol
Overview of welfare principles, criteria and corresponding parameters chosen for assessing welfare in mice.
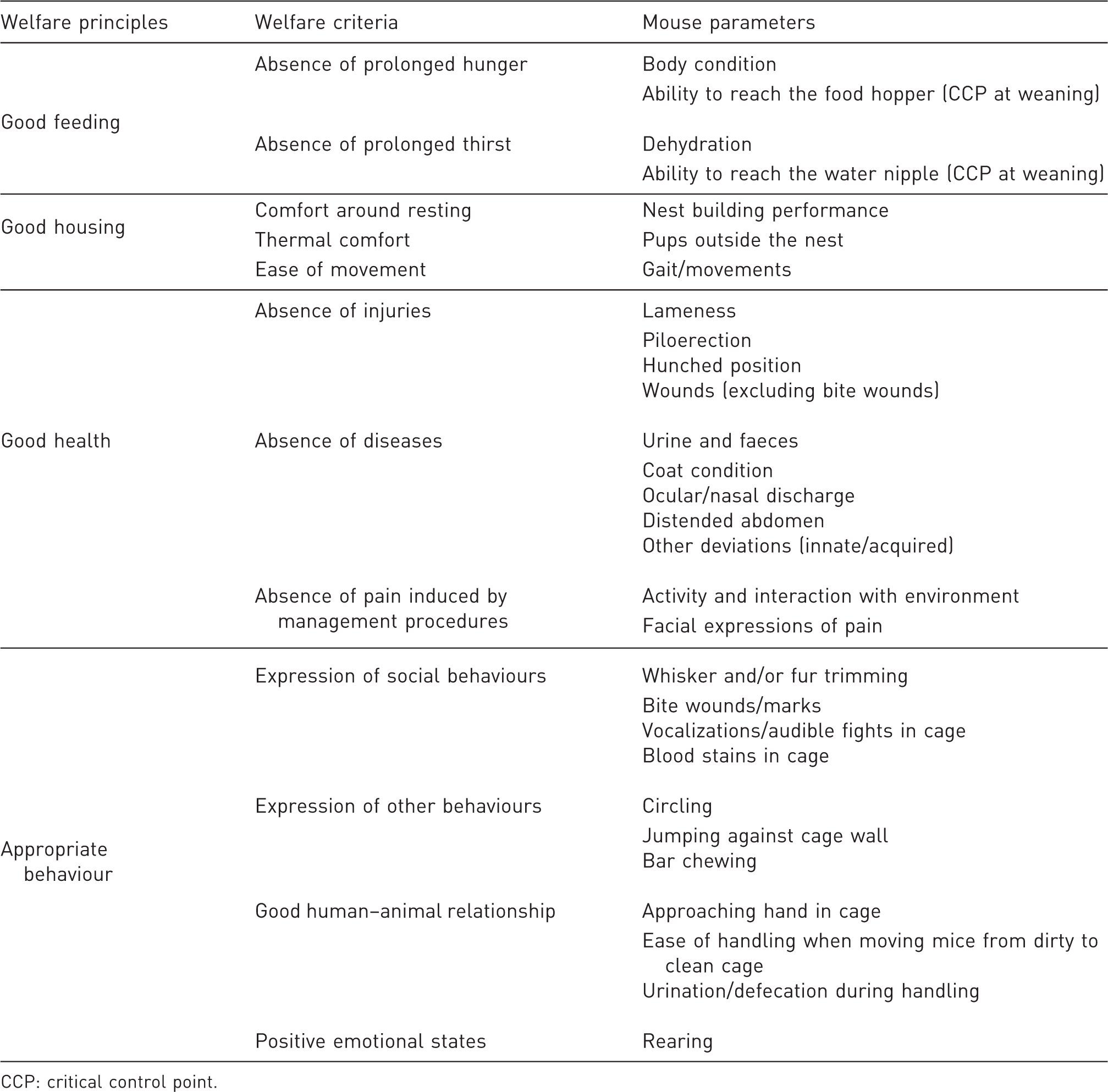
CCP: critical control point.
The protocols that are already available for assessing laboratory animal welfare have not selected their measures based on these 12 welfare criteria, and so it is not clear whether they cover all the different areas of animal welfare. Hawkins et al. 6 mentioned the concept of five domains 15 of potential welfare compromise. These domains resemble the four principles of the Welfare Quality® project; however Hawkins et al. did not propose parameters related to these domains. Some protocols are designed specifically for mice,5,7,16 and some for specific disease models, 7 while another protocol gives more general principles for effective welfare assessment of laboratory animals of all species. 6 There are also protocols available for monitoring the development of pups from birth to weaning,17,18 which is especially relevant in the production of new genetically-modified lines of mice to identify possible deviations from normal development. 18 In addition, several ‘informal’ protocols exist, such as routines for post-operative care, which can be specific for a certain animal model.
Paster et al.
7
used animal-based measures to monitor mice in an abdominal tumour model and define humane endpoints. The protocol by Hawkins et al.
6
emphasized the importance of relevant animal welfare measures (for any species), and proposed that a staff team approach (where different competences are of value) gives the most effective assessment. Evidently, animal-based measures are already being used in laboratory animal science, in the day-to-day assessment of welfare outcomes as a result of an experimental input. But when it comes to the home cage environment and the daily life of the animals outside the experimental situation, it is common to relapse into using resource-based and management-based measures such as cage changing intervals, types of bedding, access to nesting material or nest boxes, and group size when assessing welfare. As an example, in the accreditation of animal facilities, the Association for Assessment and Accreditation of Laboratory Animal Care International (AAALAC International) uses mainly parameters related to resources and management when evaluating the cage environment.
19
But the choice of resources and husbandry routines is often governed by traditions and a combination of scientific and practical considerations.
20
Although these choices may be knowledge-based, they are not systematically assessed for animal-based outcomes (even though animal caretakers perform cage-side animal-based observations as a matter of course), and are not systematically benchmarked over time. Of the published welfare assessment protocols for laboratory animals, the one by Leach et al.5,11,21 is the one most similar to the protocol presented in this paper. These similarities are:
It is an assessment protocol that is intended to be used for benchmarking, and for comparisons between facilities and within facilities. It assesses the welfare effects on the mice of housing and husbandry routines, and not of experimental situations. It uses animal-based measures.
Our protocol is different to the one presented by Leach et al.5,11,21 in that:
Our protocol contains only animal-based measures, while the one by Leach et al. uses many resource and management-based parameters (e.g. resources available in cages, such as nesting material or shelters, and the staff's interaction with the mice). Our protocol is tested for inter-observer reliability (using Fleiss' kappa and Kendall's coefficient of concordance),
8
while the one by Leach et al. is not. The protocol by Leach et al. only assesses welfare at the group level (animal unit) while our protocol assesses both the group and individual animals.
However, the major difference between the two protocols is in the procedures for choosing the parameters. Leach et al. 11 chose parameters through a consultation process with experts who were presented with parameters collected through a literature search. The parameters in our protocol were chosen on the basis of the 12 welfare criteria from the Welfare Quality® project to cover the different dimensions of welfare.
Materials and methods
Selection process for parameters
The parameters selected for each of the 12 welfare criteria are presented in Table 1. The first step of the selection process was to review existing protocols (published, web-based and informal) for animal-based measures. Thereafter these parameters were sorted according to the welfare criteria to identify which criteria might lack existing parameters. In total, 287 unique animal-based measures were listed, of which 224 (78%) were related to disease and injuries (which are two of the 12 welfare criteria). The remaining 63 parameters represented the remaining 10 criteria. For the criteria ‘absence of prolonged thirst’, ‘thermal comfort’, and ‘ease of movement’ only one animal-based measure per criterion was found in existing protocols; ‘saggy skin’ (dehydration), 22 ’ pups outside nest’ 17 and ‘normal gait/movements’ (unpublished clinical score sheet), respectively. For the criteria ‘comfort around resting’ and ‘good human–animal relationship’ only two animal-based measures per criterion were found in the existing protocols; ‘differences in resting posture’ 22 and ‘use of nesting material’ 11 plus ‘response to capture handling’ 11 and ‘response to observer’, 11 respectively.
If there was more than one animal-based measure, the next step was to choose which parameter(s) to use for each criterion. The protocol is intended to be used directly in an animal room in connection with cage cleaning procedures and without any need for extra equipment, which meant that several parameters had to be discarded already at this stage. For example, Hawkins et al. 6 suggested animal-based measures of welfare such as heart rate, blood pressure and level of stress hormones be used if these were available as part of the scientific procedure. However, physiological parameters such as these are not suitable for the protocol in the current paper for the reasons stated above. This exclusion of parameters led to another literature search for alternative parameters for those criteria for which there was no longer a parameter and also for ‘new’ parameters that might have been published after the existing protocols had been designed. In a final step, parameters were selected on the basis of their reliability and whether or not they were feasible. Those with high documented reliability from previous studies were more likely to be chosen.
Several parameters, especially those related to injury, disease, or pain, could have been allocated to several of the welfare criteria. In the Welfare Quality® protocols each parameter can only be used to represent one criterion, 23 since the scores are to be integrated to an overall welfare classification for a farm. Double criteria representation would mean double counting of that score. In this protocol, no overall welfare score is envisaged; however the specificity of the measures is still important in order to identify areas of better or poorer welfare. Also in the future other researchers might be interested in calculating an overall welfare score. For the parameters in question (e.g. ‘piloerection’ and ‘hunched position’ are commonly used signs of severe suffering, no matter what the cause), decisions therefore had to be made as to which criteria they should represent. The parameters needed to be able to identify any problems which might exist. Thereafter further testing, and possibly extra equipment, might be needed to find the reasons behind the problems.
Additional methodological points
Two parameters in the table are listed as ‘critical control points’ (CCP), namely ‘ability to reach the food hopper’ and ability to ‘reach the water nipple’. This concept originates from ‘hazard analysis and CCP’ (HACCP). This is a tool for assessing hazards and for establishing control systems that focus on prevention rather than relying mainly on end-product testing, e.g. for food safety. It is defined as ‘a point, step or procedure at which controls can be applied and is essential for preventing or eliminating a (food) safety hazard or reducing it to an acceptable (critical) level’. 24 For pups, the parameters in question should be tested at a specific time point, namely weaning, when pups must rely on their own capacity to find (reach) food and water. If this fails, the feeding and watering regimes need to be rejected and other methods have to be used. In this case it could mean that food should be placed on the cage floor and an extra-long water nipple should be installed.
In addition to the listed parameters in the protocol, it was decided that there will also be a column for free text to note down comments or observations that do not fit under any of the parameters.
Results
Description of the parameters
The selected parameters are presented in Table 1 according to the 12 welfare criteria. A full explanation of each parameter and its scoring scale, with references if available, is presented in Appendix 1 (see online at http://lan.sagepub.com). The explanation is presented in the form of a set of instructions. Each parameter is explained in a standard format: name, brief description, the different levels of the score, and comments including references to previous studies and especially information on the reliability of the parameter.
Choice of unit of measurement and scales in our protocol
Some parameters have been published with existing validated scales, like body condition score, 7 nest building performance 25 and facial expressions of pain 26 and so these scales were also used in our protocol. For parameters that were taken from existing protocols (although not necessarily validated) we also used the scales from those protocols. Several parameters were given a ‘Yes/No’ scale in our protocol, e.g. ‘fur and whisker trimming’, ‘bite wounds’, ‘distended abdomen’ and ‘ocular/nasal discharge’. This was because, as stated above, the emphasis of this protocol is to identify whether an indicator of poor animal welfare was present or not. The scale for ‘ease of handling’ was converted from an assessment of handlers 5 to an animal-based measure, and then further modified following a pilot study. The parameter ‘approaching hand in cage’ was developed by combining what is proposed for farm animals in the Welfare Quality® protocols and the findings of Hurst and West 27 on mice. The scale (i.e. distance from hand) was adapted to fit a standard sized mouse cage and the time allowed was reduced (from 60 s to 10 s) to be feasible as one parameter in the entire protocol.
The parameters ‘nest building performance’, ‘blood stains in the cage’ and ‘urine and faeces’ can only be measured at cage level, although this would be an individual measure if animals are singly-housed. Stereotypies are measured at cage level, although this measure is based on seeing individual animals performing them. The parameter ‘whiskers and/or fur trimming’ is measured at the individual level, but it is the receiver of the behaviour, not the performer, who is actually identified. The same principle applies to bite wounds. Determining the identity and number of individuals performing stereotyped behaviour, barbering or aggression was considered to be too time-consuming within the scope of this protocol. In addition, we regarded their mere presence in a cage as ‘bad enough’ and therefore did not regard it as necessary to record how many individuals were performing the behaviour. All other measures were recorded at the individual level, since they were relevant for the individual's welfare and since it was possible to score each individual during the cage cleaning procedure.
Discussion
The results of this paper show that it is possible to identify animal-based measures for the assessment of laboratory mouse welfare based on the 12 welfare criteria from the Welfare Quality® protocols. We propose that this protocol could be used to assess the many different dimensions of animal welfare. This protocol is important because it assesses welfare in the home cage, where laboratory mice spend most of their time and where breeding animals spend their whole life. The housing and husbandry routines are therefore very important factors for their welfare. Findings might also be related to experimental procedures, but the intention is to record what is observed, no matter what the cause. Information about the model might then explain the scores the animals received and, although indicating reduced welfare, they might nevertheless be in accordance with what is expected for that animal model. The protocol developed here is feasible to use, because it is based on parameters that can be measured in the animal room, without extra equipment. Most measures can be incorporated into the cage changing procedure and can be performed within 1.5–1.8 min/mouse. 8 Some of the proposed measures such as the parameters ‘ability to reach food hopper/water nipple’ and ‘rearing’ require more time, but it is believed that the information gained is worth the extra time. An assessment protocol needs to be quick, practical and easy to apply before it can be implemented on a large scale. 16 Furthermore, the assessment can be performed by one person. In our study we used two people, to allow discussion regarding the protocol. 8 Once the assessor is familiar with the protocol, we believe that he/she can do it alone during the cage changing routine. In the following sections we discuss the resulting protocol in terms of its validity, the use of animal-based measures, the question of integration of parameters, the need for additional animal-based measures and presenting results to stakeholders.
Validation and benchmarking
In this paper, the process by which the animal-based measures were selected are explained. Whenever possible already validated measures where the inter-observer reliability can be demonstrated were used; thus large parts of the protocol can already be assumed to be valid. The next step in the validation process was to test the whole protocol at animal facilities, to analyse the inter-observer reliability of all measures, evaluate assessment scales, and assess their feasibility. This has been done at three animal facilities in Sweden. 8 These results support our proposal that the protocol can be used for self-monitoring within facilities by benchmarking the findings and tracking them over time. If it is used for self-monitoring, then facilities can choose whether to use the entire protocol at every benchmarking interval or to record some parameters more frequently than others because they might be of extra interest for that particular facility. The results also support the proposal that the protocol could be used to compare facilities with each other, as inter-observer reliability for the scores is generally high. This could potentially be of interest for a large institute or university that might have different animal facilities spread over its campus since local husbandry routines can differ even within the same institute.
Animal-based measures
It is clear from the literature search that research on mouse welfare is not evenly distributed over all the different dimensions of animal welfare. Perhaps not surprisingly, there are many measures related to health but very few, if any, related to comfort in the home cage (e.g. ‘thermal comfort’), ‘ease of movement’ and ‘human–animal interaction’. The reason for mouse welfare research to focus on injuries and diseases could likely be related to the use of different animal models, as well as the relative ease by which these parameters can be measured. Thus, for the criteria ‘absence of injuries’ and ‘absence of diseases’, it was relatively easy to choose parameters that have the best combination of validity, reliability and feasibility. It was nevertheless difficult to choose criteria where there were few or no parameters, and clearly more work is needed to identify and develop animal-based measures related to the home cage environment or handling procedures.
Leach et al. 11 stated that it is important to assess both resource-based and animal-based measures for an effective welfare assessment and to be able to correlate resources with animal welfare outcomes. We have based our protocol on the argument that while these resource-based parameters are important as inputs and as risk factors, they do not need to be assessed on a regular basis. Instead, the emphasis is put on assessing the animals’ response to resources and management, i.e. the animal welfare outcome. This is in keeping with the statement by Whay et al., 12 which states that the animals’ welfare state can be defined by ‘how well they are able to survive and remain fit within the particular constraints of the husbandry system in which they live’. Rather than collecting a large amount of information on resources and management routines (which should have been checked for compliance with legislation and good practice when first established), we suggest that a more time effective assessment alternative is to focus on what might be causing a less than perfect result for a particular animal-based measure. In order to then correctly interpret the results, background information on the strain of mice, age and sex is necessary. In this way the results of the protocol can be used to identify where efforts should be directed in order to improve animal welfare.
Integration of parameters or not?
The measures in the Welfare Quality® protocols can be integrated into an overall assessment of a farm, that can be later communicated as animal welfare information to consumers buying the products. 9 In the case of Welfare Quality® this allows farms to be classified into one of four categories: not classified, acceptable, enhanced and excellent. Such a classification, or at least a threshold of what is acceptable or not, might also be beneficial for the proposed protocol for mice if it is to be used for official inspections. This would involve weighing different parameters and setting thresholds. A similar approach is suggested in the assessment of actual severity of experimental procedures, where the European Commission Expert Working Group on Severity Assessment advocates that everything the animal experiences during a procedure should be considered in the assessment. 14 An equivalent process which rates the welfare parameters used in this protocol could be envisaged. But integrating scores from different parameters is a complicated procedure that requires advanced mathematical models, and ethical decisions have to be made on how to weigh different scores and how to combine different aspects of welfare to give a single value. 23 For the purpose of assessing the welfare of laboratory mice in their home environment, the detailed scores in the parameters are of more interest (for benchmarking) than an overall integrated score. Indeed, details from different individual parameters that might be of interest will not be visible in an overall assessment.
In order to make the protocol user-friendly it is important to consider how to present the results. They could be presented per welfare parameter, per strain, per age group, etc. depending on the interest of that specific animal facility. It should be easy to backtrack from results to the parameters in question to identify reasons behind potential problems revealed in the results. Proper interventions can then be made and effects benchmarked over time. Within the scope of this study no specific effort has been directed towards the presentation of the results, but future work should be performed in order to make the protocol appealing and user-friendly. Further, the development of a management decision tool could be considered to help guide actions resulting from the assessment.
Development of parameters
We failed to find a useful, feasible animal-based measure for thermal comfort in adult mice. At present, thermal comfort is assessed only for pups (pups outside the nest). Behaviours reported to indicate responses to ambient temperatures are: thermotaxis (moving away from stressful temperatures), huddling and nest building in response to cold, and increased surface area exposure or fur licking in response to heat. 28 Gordon et al. 29 found that mice chose lower temperatures for their active period, that there was only a 1 ℃ difference in choice of ambient temperature between singly-housed and group-housed mice, and that young mice (2 months of age) chose lower temperatures but compensated by being more active. Huddling does however seem to occur in a variety of ambient temperatures, e.g. Gordon et al. 29 found that group-housed mice huddled in an ambient temperature of only 1℃ lower than that preferred by singly-housed mice. Thus, unlike most other species, huddling is not a reliable indicator that a mouse is cold. Nest building can also be performed as protection and is regardless of reproductive status or sex. 30 At present, the behaviours mentioned above need further investigation before it is clear which parameters should be incorporated into the protocol.
Since mice are generally housed in ambient temperatures below their thermoneutral zone, there have been fewer studies of whether mice might sometimes be kept in too warm an environment. In individually ventilated cages (IVCs), and especially in cages with high stocking densities, a lactating female risks becoming overheated since she is generating much heat as a by-product of milk production. 31 Lactating female mice kept in IVCs (singly-housed) have been observed to rest outside the nest in a stretched-out position (E Spangenberg, personal observations) thereby increasing surface area exposure. Reliable and feasible parameters of heat stress are therefore also of interest, especially if mice kept in IVCs are to be assessed.
A positive emotional state in animals is one of the 12 welfare criteria and is also an area that is receiving increased attention within animal welfare science. 32 Although it is believed that the traditional welfare parameters indicate an absence of poor welfare, they might not necessarily indicate that welfare is good. Parameters are needed to assess the positive side of animal welfare, but these are not easy to validate. 33 There are many tests for assessing the emotional state in mice, but these generally measure negative states and require specific equipment. We propose using rearing behaviour when a mouse is initially put into its clean cage as an indicator of a positive emotional state. Rearing is considered to be an exploratory behaviour which, like play behaviour, is thought to be associated with a positive emotional state. 34 However, it turned out to be impractical to score rearing in the current set-up of the protocol because as soon as a mouse was put into the new cage, it was intended that the assessor should start to observe the next mouse in the dirty cage. If the mouse did not immediately perform rearing in the new cage, the behaviour would be missed, 8 and to wait for the mouse to perform the behaviour would prolong the observation time per mouse. For the parameter to be included in the protocol the assessor needs to watch the mouse closely when it is released into the new cage. So evaluating this parameter takes some additional time and may be included when a facility is especially interested in positive emotional states or for a particular group of mice or following a specific management modification. A possible alternative behaviour to observe in the home cage would be play behaviour, which is thought to be associated with a positive emotional state. 35 However, play behaviour in mice is performed mainly by juveniles and the interactions are quick and brief. 36 In addition, it would probably need to be observed during the dark period, when mice are active. Hence, at this stage it is not feasible to incorporate this behaviour into the protocol either, but it is a relevant measure of a positive emotional state (and one that can be measured in the home cage) and thus further studies in this area would be beneficial.
In summary, we have created a welfare assessment protocol for laboratory mice using only animal-based measures for evaluating animal welfare outcomes at an animal facility. We have also identified areas, such as thermal comfort behaviours and positive emotional states, where there are very few valid, reliable and feasible measures of mouse welfare and hence where more research is needed.
Footnotes
Acknowledgement
This work was supported by the Swedish Research Council for Environment, Agricultural Sciences and Spatial Planning (grant number 221‐2009‐1664), and was part of the Centre of Excellence in Animal Welfare Science, a Swedish collaborative research platform.
Ethical statement
This manuscript is based on a literature search for parameters to be included in the protocol. Hence, no studies on animals were performed at this stage. It is however mentioned in the manuscript that the assessment protocol was tested at three animal facilities in Sweden. That part was approved by a Swedish regional committee for ethical approval of studies using animals for scientific purposes. Further, the assessment was performed on animals already available at the facilities visited and so was performed in line with the ‘reduction’ aspect of the 3Rs.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
