Abstract
Recently, the need for more standardized operation procedures in experimental liver fibrosis research was suggested due to dramatic changes in European animal welfare rules. Here, we present a short series of standard operation procedures (SOPs) summarizing the most relevant and widely accepted experimental models for the induction of liver injury leading to liver fibrosis. The described procedures are based on the long-term experience of the Collaborative Research Centre ‘Organ Fibrosis: From Mechanisms of Injury to Modulation of Disease’ (http://www.sfbtrr57.rwth-aachen.de/), which is supported by the German Research Foundation (SFB/TRR57). These SOPs will help to improve standardization of fibrosis models and to increase the comparability of data between different laboratories with the aim of reducing animal experimentation according to the principle that was proposed in 1959 by Russell and Burch as an ethical framework for conducting scientific experiments with animals, namely the replacement, refinement and reduction (3R) principle. In the first section we focus on the carbon tetrachloride (CCl4) model in mice, which is the toxic model of liver fibrosis induction most commonly used worldwide.
Historic background of the model
Experimental liver fibrosis in mice can be induced by surgical intervention (e.g. bile duct ligation), genetic manipulation of fibrosis-related genes (e.g. Mdr2 knockout mice) or application of hepatotoxins. 1 In particular, the single or repeated administration of carbon tetrachloride (CCl4) has become one of the most commonly used experimental models for inducing toxin-mediated liver fibrosis. However, almost all laboratory use protocols, which might vary with respect to usage of anaesthesia, hepatotoxin preparation, doses, treatment duration, administration routes, or the many other factors that may affect the outcome of experimentation. In the European Union, the laws on the protection of animals used for scientific purposes have recently changed dramatically by the implementation of the new EU Directive 2010/63 that contains 66 restrictive articles. 1 These require that, wherever possible, alternative scientific methods that do not need the use of live animals, should be applied in experimental research. The immediate implementation of the new EU Directive should foster the 3R (replacement, refinement, and reduction) principle that was proposed by William M S Russell and Rex L Burch in 1959 for the more ethical use of animals in testing. 2 To achieve this challenging objective, it is obvious that researchers in the field that use particular models must ensure that they work according to strict general guidelines and accepted standard operating procedures (SOPs). Therefore, we present here a detailed protocol that induces hepatic fibrosis in a highly reproducible manner. In addition, we discuss the historical background to that model and the pathobiochemical mechanisms that contribute to the formation of hepatic insult.
CCl4 is widely used as a solvent for dissolving non-polar compounds, such as fats and oils. The acute toxicity of CCl4 is well established from many animal studies. In particular, studies performed on rats have shown that the lethal dose (LD)50 after acute oral uptake is in the range of 4.7–14.7 mL/kg body weight, depending on nutritional conditions and supplements administered.3,4 Single oral doses of CCl4 in corn oil induce increased liver weight, elevated levels of fat, serum urea, liver enzyme activities, and clear histopathological evidence of liver damage with single cell necrosis; 5 whereas the long-term oral exposure to CCl4 causes marked hepatotoxicity with resulting fibrosis, bile duct proliferation, cirrhosis and even hepatocellular carcinoma (HCC). 6 Toxicity of CCl4 has three or four distinct phases. 7 The first two or three weeks are mainly characterized by necrosis indicated by rising activities of liver specific enzymes and decreasing values of pseudocholinesterase. During the next two or three weeks massive hepatic fat accumulation occurs and serum levels of triglycerides and aspartate aminotransferase (AST) are significantly increased, while hepatic function is reduced. During the third phase, the increase of AST continues, elevated levels of hydroxyproline and triglycerides are found, and overall liver function further decreases. In the final phase, the values of pseudocholinesterase further decrease, and atrophy of the liver is observed. 7 This may be combined with a significant decrease of serum albumin and weight loss which indicates progressive loss of hepatic function during prolonged fibrogenesis.
Pathogenic mechanisms of liver damage
CCl4 is metabolized in the liver by the cytochrome P450 superfamily of monooxygenases (CYP family) to the trichloromethyl radical ( Pathobiochemical sequence of events during carbon tetrachloride (CCl4)-induced liver damage. In the liver, CCl4 is metabolized by cytochrome P450 (CYP) enzymes to a trichloromethyl radical that can be further oxygenated to the trichloromethylperoxy radical. Both radicals are highly reactive and induce complex cellular alterations that result in hepatotoxic damage, inflammation, fibrosis, cirrhosis and hepatocellular carcinoma (HCC).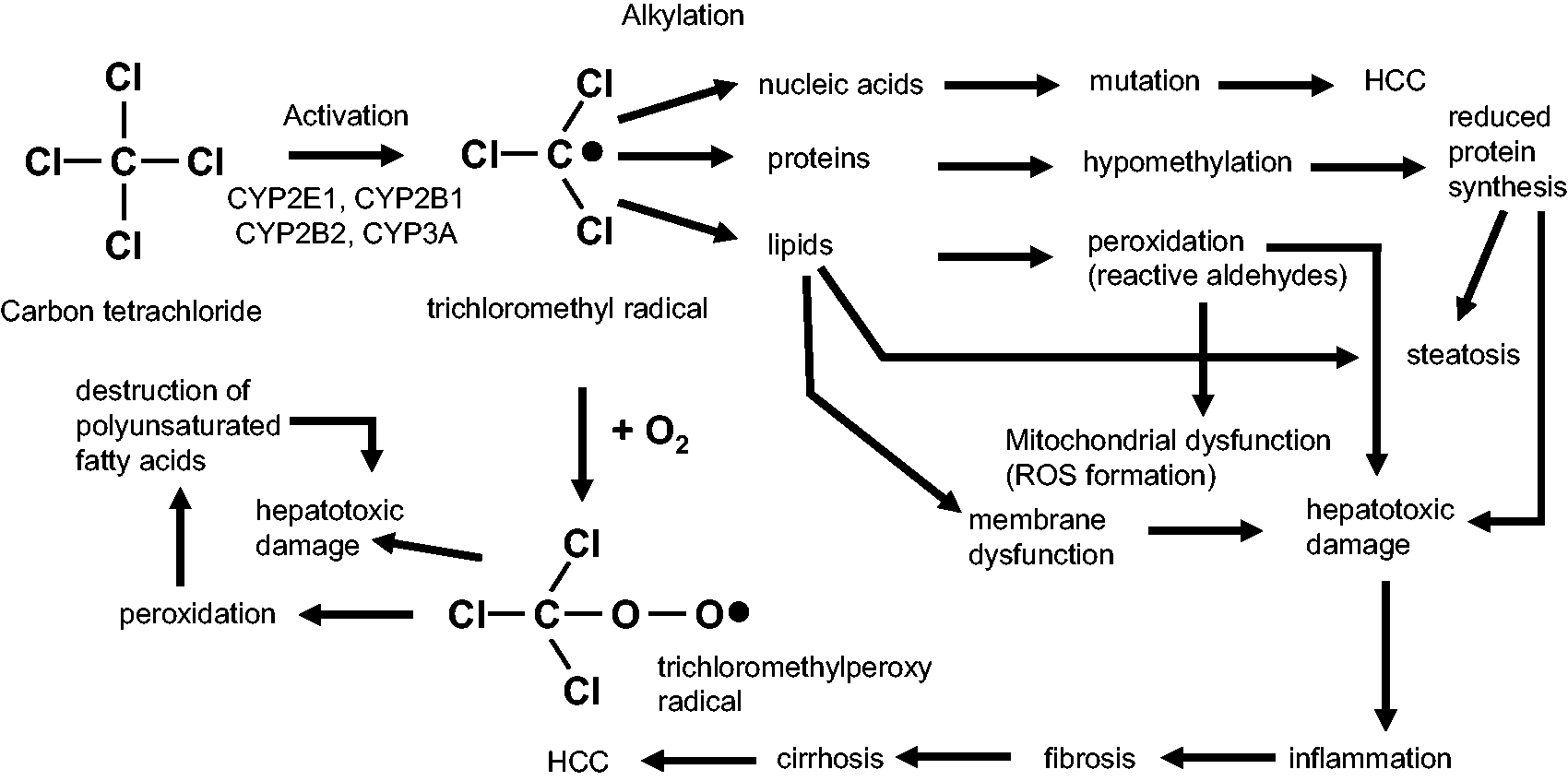
Experimental procedure
General considerations
Genetic background
Susceptibility to carbon tetrachloride (CCl4)-mediated liver fibrosis in different inbreed mouse strains.
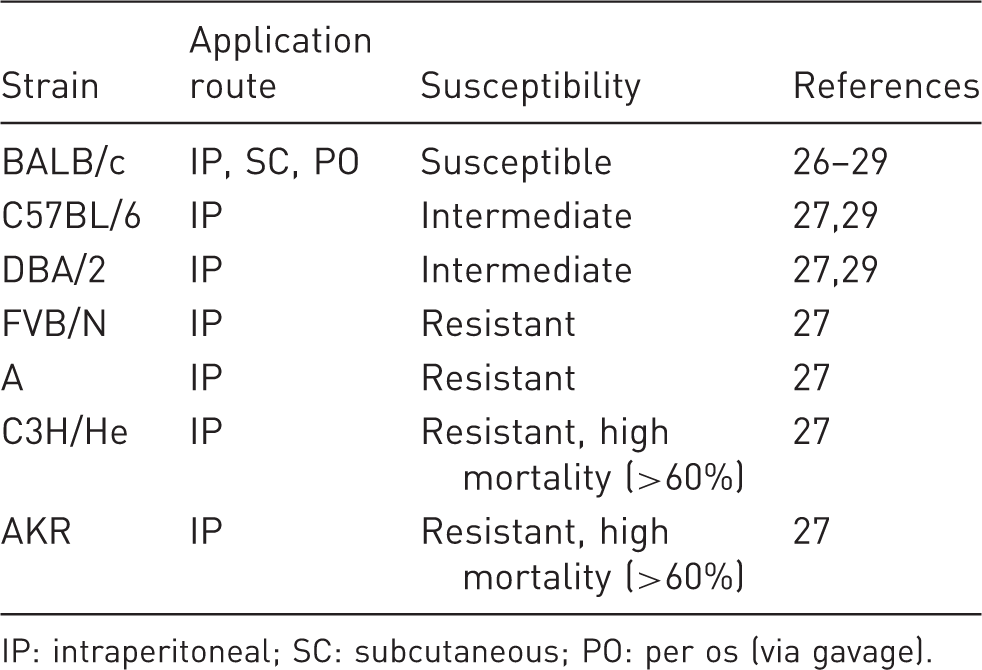
IP: intraperitoneal; SC: subcutaneous; PO: per os (via gavage).
Duration and intervals of treatment
Different responses to CCl4 due to genetic reasons and severity (strength) of fibrogenesis can basically be modulated by application intervals and duration of treatment. In our experiments, intraperitoneal (IP) application of CCl4 in C57BL/6 inbred mice twice a week for six weeks, or alternatively application three times a week for four weeks, results in fulminant deposition of intrahepatic collagen matrix and fibrosis resembling human stage 3 according to Desmet-Scheuer scoring. 10 In highly susceptible strains, four weeks of CCl4 application is usually sufficient for fibrosis induction, while in fibrosis-resistant strains the poor susceptibility can in part be compensated for by prolonged treatment of up to 12 weeks.
Application routes
In principle, CCl4 can be administered through IP injection, inhalation and gavage. The majority of investigators prefer IP application in mice for reasons of excellent reproducibility, good survival rates,
11
ease of performance, and safety. However, for specific aims (such as the induction of portal hypertension) or when the CCl4 intoxication is established within the laboratory in rats, application via inhalation might be more appropriate, but requires a specific extraction hood in the laboratory and expertise (Figure 2). Only a few authors prefer administration of CCl4 via gavage; however, in our opinion, this application route is not to be recommended due to frequent early mortality.
12
Inhalation exposure apparatus. Before exposing mice or rats to a carbon tetrachloride (CCl4)-containing atmosphere, the animals are first stimulated with phenobarbital to induce metabolic liver enzymes and to enhance CCl4-induced liver damage. The animals are then set into an inhalation exposure apparatus and CCl4-enriched air is conducted into the chamber using exposure cycles and conditions outlined under the ‘Practical implementation’ section of inhalation. To avoid accidental exposure and intoxication of research personnel, the procedure should be carried out under an extraction hood. After exposure, the inhalation chamber should be left in the extraction hood for another 10 min before it is removed.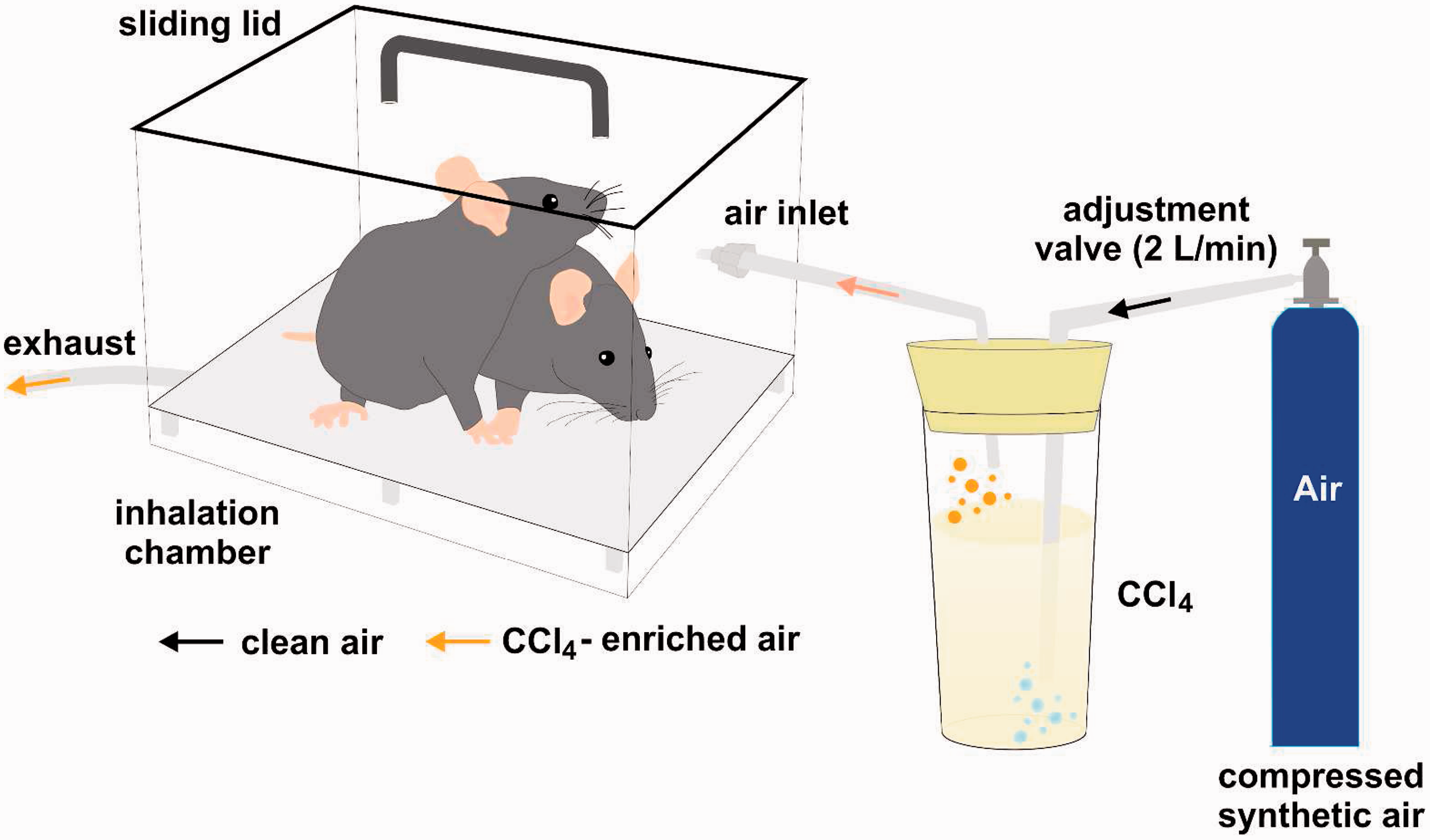
Biometric calculation of requested cohorts (IP treatment and inhalation)
In a typical CCl4 experiment one aims to compare fibrosis susceptibility of wild-type mice with a pharmacologically-treated or genetically-modified strain. Suitable readouts for ongoing hepatic fibrogenesis are, for instance, hepatic determination of hydroxyproline, measurement of alpha-smooth muscle actin (α-SMA) or collagen type 1α1 (Col1) gene expression.
13
Regarding Col1 expression, our data (Figures 3a–3c) typically reveal a variance of approximately 25% in each group.14–16 In addition, we define a difference of 30% in Col1 mRNA expression to be biologically relevant. From these parameters the calculated effect size d is 1.44, as determined with the G Power V.3.1.5 software (that can be downloaded from the Heinrich Heine University, Düsseldorf, Germany, http://www.gpower.hhu.de/en.html).
17
To achieve a statistical power of 80% and a specific α error probability of 0.05 the minimal required sample size in each group is n = 9 mice (see Table 2 and Figure 3d).
Hepatic fibrosis after carbon tetrachloride (CCl4) challenge. Six to eight-week-old male C57BL/6 mice (weighing approximately 18–20 g) received intraperitoneal injections twice weekly of 0.7 mL/kg body weight of CCl4 in equal volume of mineral oil or mineral oil alone for six weeks. Liver specimen from the control group (a) and CCl4 challenged animals (b) were stained with Sirius Red. Increased collagen expression and deposition can be further quantified by (c) by quantitative real-time polymerase chain reaction (PCR). Significant differences (*) with P values < 0.05 and in Student's t-test are indicated. More experimental details are given elsewhere
14
(d). The relationship of total sample size (=animals) and statistical power is depicted. Please note that 18 animals (9 animals/group) are needed under the experimental setting described in our experimentation with a statistical power of 80% (marked as a solid blue line). Higher values of statistical power require more animals per group. Biometric calculation of sample size determined by G Power.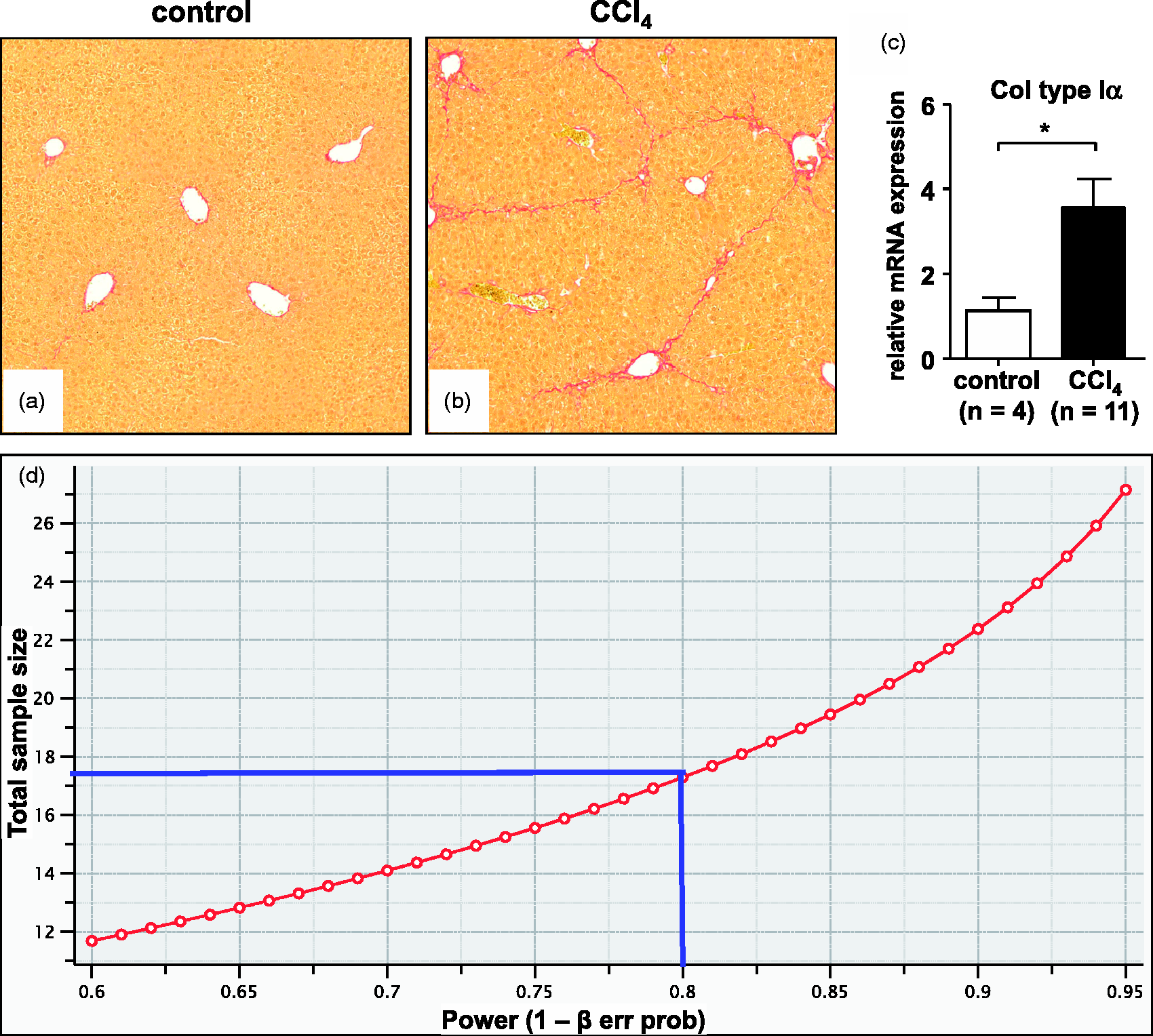

Practical implementation
(a) Intraperitoneal
In general, handling mice (i.e. taking them out of the cage, fixation, and exposition to a foreign surrounding) is already stressful for the animals. CCl4 vapours should be considered to be toxic for humans. Thus prevention of accidental research personnel intoxication is crucial. The level of exposure should be minimized by handling CCl4 in an appropriate location such as a fume hood. Protective clothing including a long sleeved laboratory coat, rubber gloves, safety goggles and a face mask are minimum standards that should avoid exposure over the recommended threshold limits. After application of CCl4, the operators should wash their hands thoroughly. In addition, the novel European animal welfare rules require that only trained laboratory personnel should perform the application of CCl4, ensuring swift and short animal handling times. The actual procedures are as follows:
Mice should be weighed and examined for signs of distress (i.e. changes in respiration, rough hair coat, unusual behaviour, hunched posture). Animals showing these abnormalities should be excluded from the study. CCl4 concentrations usually are 0.5 to 0.7 µL/g body weight diluted in corn oil.18–20 Acute intoxication models with CCl4 usually use higher doses (0.8–2 µL/g body weight) but mice should be sacrificed after 1 to 3 days.21,22 Please note that the injection solutions (CCl4 or CCl4/corn oil) should be at room temperature or close to body temperature. The required volume of CCl4 should be calculated. For practical reasons, we recommend to always inject a constant volume of 50 µL containing a solution of CCl4 (e.g. Sigma-Aldrich, St Louis, MO, USA) in corn oil (e.g. Sigma-Aldrich). Accordingly, the required volume of CCl4 (0.5–0.7 µL/g mouse weight is made up to 50 µL of total volume with corn oil. As an example, one would inject 10 µL CCl4 dissolved in 40 µL of corn oil into a mouse that weighs 20 g to obtain a final dosage of 0.5 µL CCl4/g body weight. 50 µL of CCl4/corn oil solution should be injected in mice using disposable, sterile syringes. To this end the mouse should be scuffed behind the neck region between the thumb and forefinger. After turning over the hand the animal should rest on the palm against the base of the thumb using a third finger to stabilize the pelvic region. CCl4 should be injected into the lower side of the abdomen with the mouse head down to avoid injury to the bladder, diaphragm or intestine using a 27 gauge needle (e.g. 27 G Microlance; Becton Dickinson, Franklin Lakes, NJ, USA). IP injection can lead to local irritation of the skin and CCl4 can cause mild peritoneal inflammation. Animals should be re-inspected one hour after injection for abnormalities and every 24 h thereafter. Termination of experiments and thus CCl4 treatment depends on the specific aim of each study. If immediate proinflammatory alterations are of interest, tissue harvesting after 24 and 48 h is recommended. Established fibrosis can best be investigated after 2–4 weeks of CCl4 treatment, while severe fibrosis (cirrhosis) can be observed after 6–8 weeks of treatment. At the end of the experimentation animals should be sacrificed by cervical dislocation.
Animal burden/side-effects
Usually mice that receive IP injections of CCl4 do not develop severe long-term complications. Although CCl4 intoxication is intended to cause elevated liver enzymes and long-term complications such as portal hypertension, oesophageal bleeding and ascites leading to acute liver injury or liver fibrosis, this condition is usually not associated with pain in humans and thus most likely also not in mice. However, IP CCl4 application is clearly associated with abdominal adhesions and hepatic inflammation and transient spasms.
(b) Inhalation
Inhalative intoxication with CCl4 was first used extensively in rats, and was later transferred to mice. 23 Mice in the inhalation group might be treated with protocols that differ in inhalation time or intervals (commonly twice or three times per week) that either lead to significant fibrosis within four weeks 16 or might progress to liver cirrhosis with low serum sodium levels, ascites and portal hypertension after 11–15 weeks of treatment. Under the most damaging conditions, mortality rates can be as high as 70%, while short-cycle thrice-weekly inhalation usually results in significantly lower death rates (0–10%). Studies using inhalative CCl4 usually aim towards the development of end-stage liver cirrhosis with portal hypertension and are performed for 11–15 weeks. The drawback of this technique is that special equipment and expertise are required to avoid inhalative CCl4 contamination of research personnel because CCl4 vapours are considered to be toxic for humans. Thus, prevention of accidental intoxication is crucial (e.g. usage of latex-free gloves is important), and only trained laboratory personnel should perform the experiments. Every contact must occur, and every potentially contaminated subject must be placed under, the extraction hood.
The actual procedures that we favour in our experimentation are as follows:
First the mice should receive phenobarbital (0.3 g/L in drinking water) for one week before starting the CCl4 inhalation procedure. This treatment should induce liver enzymes and enhance liver damage after CCl4 inhalation. Those animals that are subjected to CCl4 inhalation should be kept to a maximum of six animals per cage. The cage itself and the tube for the gas application should be checked each time before the treatment begins. CCl4 should be filled in a specific evaporation flask. The tube conducting the air should be placed at least 2–3 cm underneath the CCl4 fluid surface in the flask. Compressed air with a flow rate of 2 L/min should be pumped into the flasks containing CCl4 underneath its surface and should bubble out of the fluid CCl4. The tube feeding the CCl4-enriched air should be inserted into the cage. Each treatment should consist of short cycles of CCl4 exposure three times per week (e.g. Monday, Wednesday, and Friday). The conditions should be set to:
1 min of bubbling and 1 min in the gas atmosphere in the first week 1.5 min of bubbling and 1.5 min in the gas atmosphere in the second week 2 min of bubbling and 2 min in the gas atmosphere throughout all the rest of the experiment After treatment, the cage should be left in the extraction hood for another 10 min before it is removed. When a mouse develops ascites, the inhalation should be stopped since ascites is a definitive sign of portal hypertension. The animals should have reached the stage of liver cirrhosis with portal hypertension and at this point the experiments should be conducted (e.g. efficacy analysis of antihypertensive drugs) or the animals should be sacrificed for further analysis (e.g. gene and protein expression in cirrhotic livers).
Mice should then be treated with one cycle/treatment in the initial three weeks, then two cycles/treatment in the fourth week, and three cycles/treatment thereafter. Previously treatment consisted of two or more cycles, and these were separated by 2 min of breathing in ambient air not containing CCl4.
Animal burden/side-effects
Prolonged inhalative CCl4 treatment promotes severe cirrhotic changes in the liver with significant development of ascites 23 and more ascites than IP-treated animals. Therefore, mice subjected to inhalative CCl4 treatment suffer from the complications of liver cirrhosis, show loss in body weight and less food intake, and are more fatigued, but do not experience pain or mutilations.
(c) Gavage
Our consortium does not recommend the regular application of CCl4 by gavage due to high rates of early mortality 11 and additional burdens as specified below. Increased mortality would require higher sample numbers, which is not compatible with the 3R principle. 2 However, if the experimental set-up requires gavage application, investigators should refer to detailed protocols published elsewhere.24,25
Animal burden/side-effects
CCl4 oral feeding causes significant distress to the animal and CCl4 is likely to cause chemically-induced inflammation in the intestinal mucosa. Intubation and application of CCl4/oil into the trachea needs to be avoided, as accidental CCl4 application into the lung is usually instantly fatal for the mouse.
Classification of severity of procedure
According to Article 15 of the EU Directive 2010/63 (http://eur-lex.europa.eu/LexUriServ/LexUriServ.do?uri=OJ:L:2010:276:0033:0079:en:PDF), the estimated degree of pain, suffering, distress or lasting harm of the animals subjected to CCl4 application should be classified following a scoring system that contain procedures to be classified as ‘non-recovery’, ‘mild’, ‘moderate’ or ‘severe’.
Details about the classification criteria that underlie this assessment have been established by the Expert Working Group on severity classification of scientific procedures performed on animals. These classification criteria can be found at: http://ec.europa.eu/environment/chemicals/lab_animals/pdf/report_ewg.pdf.
The IP application twice a week of CCl4 up to four weeks with no major impairment of liver function should be classified as a moderate procedure according to Article 15 of the above-mentioned EU Directive 2010/63.
Prolonged inhalative CCl4 treatment promotes severe cirrhotic changes in the liver with significant development of ascites 23 and more ascites than IP-treated animals. Therefore, mice subjected to inhalative CCl4 treatment suffer from the complications of liver cirrhosis, show loss in body weight and less food intake, and are more fatigued. Because of the high mortality rate the procedure is classified as moderate to severe. In line with the 3R principle, the procedure should undergo a refinement, and humane endpoints (such as lethargy, reduced escape reflexes, reduced food intake, extensive body weight loss >20%, continuous spasm or others) must be implemented with frequent observation points to restrict pain, suffering, distress or lasting harm to the animals. Then the procedure can be reclassified as moderate.
Concluding remarks
Application of CCl4 is a key model in experimental liver research that has been applied worldwide in more than 50,000 studies so far. The model is robust and leads to highly reproducible results. In our opinion, the best treatment route is the application via IP injection. With this SOP we aim to suggest technical and biometric standards facilitating reproducible animal experimentation in line with the new European animal welfare regulations.
Ethical statement
All experiments were approved by the official State animal care and use committee (LANUV, Recklinghausen, Germany).
