Abstract
The induction of experimental obstructive cholestasis is a reliable model for cholestatic liver diseases in rodents. Bile duct ligation (BDL) in mice provokes typical time-dependent morphological and structural changes in the liver, ranging from liver cell injury and elevated serum enzyme levels after several days, to a severe inflammatory response in the liver after 5–7 days, up to an advanced hepatic fibrosis as soon as three to four weeks after surgical ligation of the common biliary duct. Upon BDL induction, hepatic stellate cells become activated and transdifferentiate into myofibroblasts that produce extracellular matrix proteins such as collagen. In principle, the periportal fibrosis induced by BDL in rat livers is reversible. After the relief of a biliary obstruction, the liver has the capacity to revert to a nearly normal histological architecture and a fully normal biochemical function. When BDL surgery is performed by an experienced scientist, this model has very high reproducibility among all fibrotic models. All these factors corroborate the outstanding value of this model for basic and translational research in biomedicine and hepatology. Nevertheless, this model can result in significant variations when surgery is carried out by untrained personnel or when unconscious modifications are implemented that affect the quality of the intervention. A detailed protocol is provided here for the provision of reliable and reproducible BDL in mice.
Historical background of the model
The induction of experimental obstructive cholestasis in animals is a well-established model. The first experimental investigations of the effects of bile duct obstruction on the liver were performed as far back as the year 1872.1,2 These studies were mainly done in cats and rabbits. The first studies using small cohorts of rats and mice were conducted over 40 years later. 3 Thereafter, the ligations of common bile ducts in rats (that lack gall bladders) and mice (that have gall bladders) have been widely used as experimental models to unravel various aspects of gall bladder function. 4 However, most of these earlier studies in rodents only allowed animals to be kept for short periods, because infections and other surgery-associated complications were frequently observed. As a result, in subsequent studies much more attention was directed towards the maintenance of aseptic conditions during every surgical step. However, all these historical studies have already shown that obstructive cholestasis is an appropriate model for investigating ongoing hepatic insults due to cholestasis. The major alterations include progressive dilation of the bile ducts, increasing focal necrosis centred around the small bile ducts, and other pathological changes that occur in the liver following the first days after complete obstruction of the common bile duct. These phenotypic changes were first reported in a study that was conducted in a large cohort of rats (n = 120) in the middle of the last century. 5 In this pioneering study, a short but precise protocol that allowed bile duct surgery to be performed within 6 min was presented that included information about anaesthetization, opening of the peritoneum, exposure of the bile duct and setting of the double ligatures. Subsequently, a vast literature on details concerning experimental biliary obstruction has emerged. In particular, prolonged bile duct ligation (BDL) was announced as a good experimental model for investigating the dynamic nature of progressive cholestasis that includes inflammation, fibrosis and cirrhosis. 6 Also protocols for partial BDL in mice as a model of acute cholestasis were established. 7 Based on these fundamental reports, we have established an easy, fast and reliable protocol for prolonged BDL in rats and mice in our laboratory that is suitable and reliable for liver fibrosis research. The consistency of this protocol has been confirmed in many studies in which we have investigated variable biochemical and morphological aspects of ongoing hepatic fibrogenesis, profibrogenic signalling pathways, and contribution of diverse immune cell subpopulations for fibrosis progression, and have tested the efficacy of new therapeutic antifibrotic agents, or analysed the function of individual genes or cells in liver biology.8–20 Substantial efforts are currently also being undertaken to improve understanding of the complex inflammatory response during BDL-induced liver cholestasis which consist of three interrelated phenotypes, namely ischemia-reperfusion, leukocytic and angiogenic phenotypes.21,22 In addition, during recent decades strong efforts have been made to develop more sophisticated microsurgical-based methods for inflicting cholestatic liver injury in only selected parts of the liver. 23
Pathogenic mechanisms of liver damage
BDL in rodents has been widely used in research addressing issues of cholestatic liver injury including oxidative stress formation, inflammation, and fibrogenesis. Mechanistically, the prevention of proper bile flow results first in increased concentrations of bile acids in the liver and serum (Figure 1). In response to these alterations, a ductular reaction is induced in which the numbers of biliary epithelial cells and intrahepatic bile ducts increase. In common with other insults of the liver, this reaction is accompanied by infiltration of inflammatory cells such as monocytes, neutrophils, eosinophils, and lymphocytes, while parenchymal cells (i.e. hepatocytes) in areas of proliferated bile ducts become atrophic and the number of extracellular matrices producing α-smooth muscle actin (α-SMA) positive cells increase.
24
Progressive liver damage is further marked by elevated serum activities of aspartate aminotransferase (AST), alanine aminotransferase (ALT), γ-glutamyltranspeptidase (γ-GT), alkaline phosphatase (AP), and lactate dehydrogenase (LDH), and by increased concentrations of total bilirubin, alongside visible jaundice. At the cellular level, specific immunohistochemical markers that indicate the occurrence of the ductular reaction have been well established (cf. Figure 1). Because most of these alterations are also seen in humans suffering from diverse forms of cholestasis, this extrahepatic biliary obstruction model has been recognized as a suitable model for the understanding of congenital and acquired human cholestasis.
25
Pathogenic events induced by surgical bile duct ligation (BDL). After ligating the bile duct, the liver displays a ductular reaction, in which the proliferation of biliary epithelial cells that are positive for cytokeratins (CK) 7, 8, 18 and 19 is increased, whereas hepatocytes that are stained positive for CK 8 and 19 undergo necrosis and apoptosis. Damaged hepatocytes release intracellular transforming growth factor beta (TGF-β) that represents a major profibrogenic agent. During prolonged hepatic damage, α-smooth muscle actin (α-SMA)-positive myofibroblast (MYB)-like cells that originate from quiescent hepatic stellate cells (HSC) increase continuously after BDL. These cells are capable of synthesizing large quantities of extracellular matrix (ECM) proteins including collagens that are deposited in the hepatic parenchyma resulting in fibrosis. The liver is further characterized by inflammation that is triggered by activation of Kupffer cells and infiltration of immune cells that release a multitude of proinflammatory substances. As a consequence of all these alterations, the concentrations of bile acids in the liver and blood and typical serum markers (aspartate aminotransferase [AST], alanine aminotransferase [ALT], bilirubin, alkaline phosphatase [AP], γ-glutamyltranspeptidase [γ-GT]) are elevated. If BDL is prolonged for longer periods, secondary organ damage (e.g. in the kidney) may occur. PDGF: platelet-derived growth factor.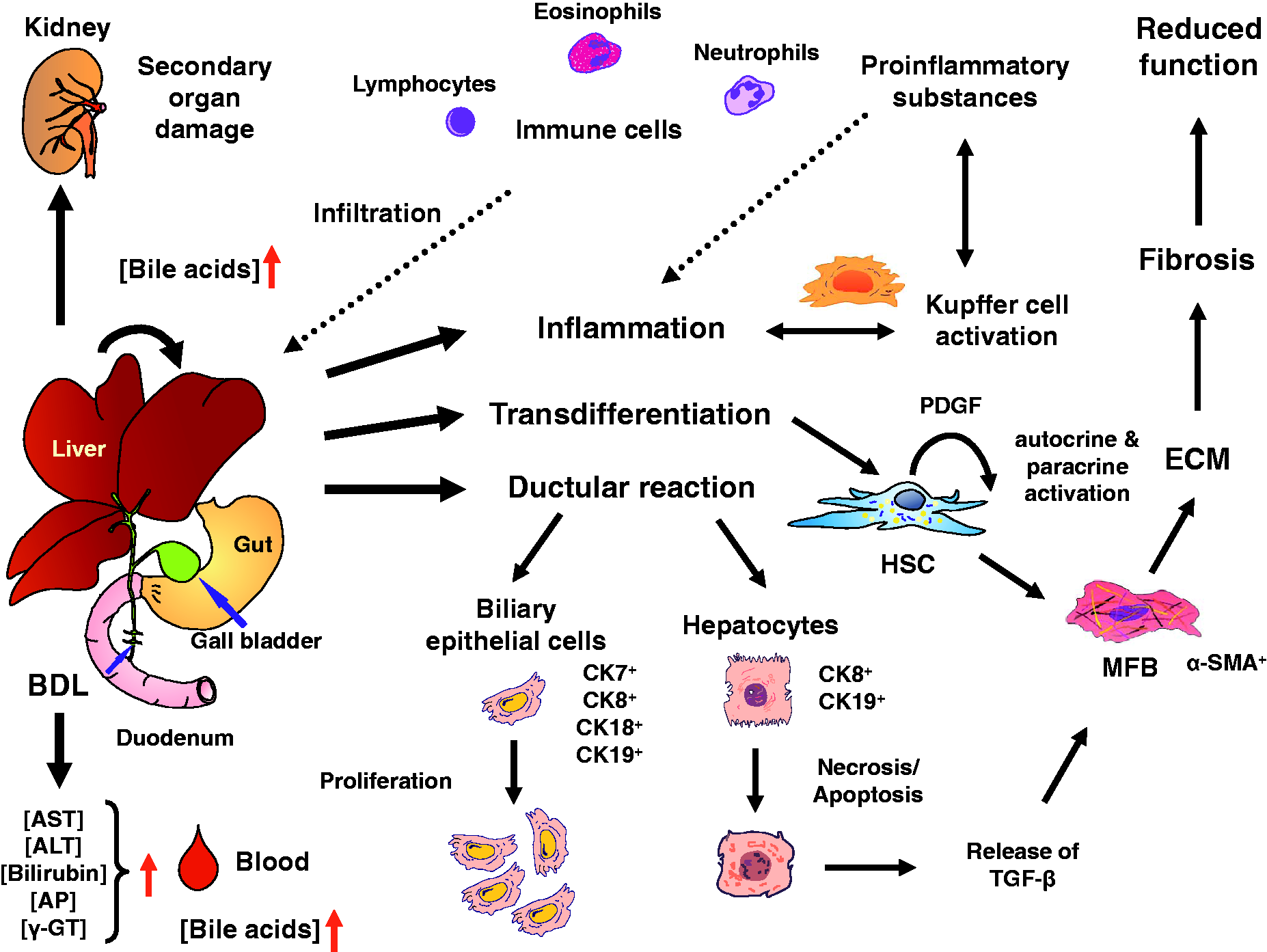
Experimental procedure
General considerations
In principle, BDL is a simple experimental intervention, in which the liver is lifted upwards and the common biliary duct (ductus choledochus) is double-ligated with surgical sutures. This prevents proper flow of the bile that is produced in the liver from the liver to the duodenum (Figure 2).
Schematic depiction of the bile duct surgery. (A) The bile duct is a thin tube that connects the liver and the duodenum. In the liver, intrahepatic bile flow is controlled by a network of fine extensions. (B) For double ligation of the common bile duct, two sutures are each fixed in close proximity by two knots within the region that is indicated.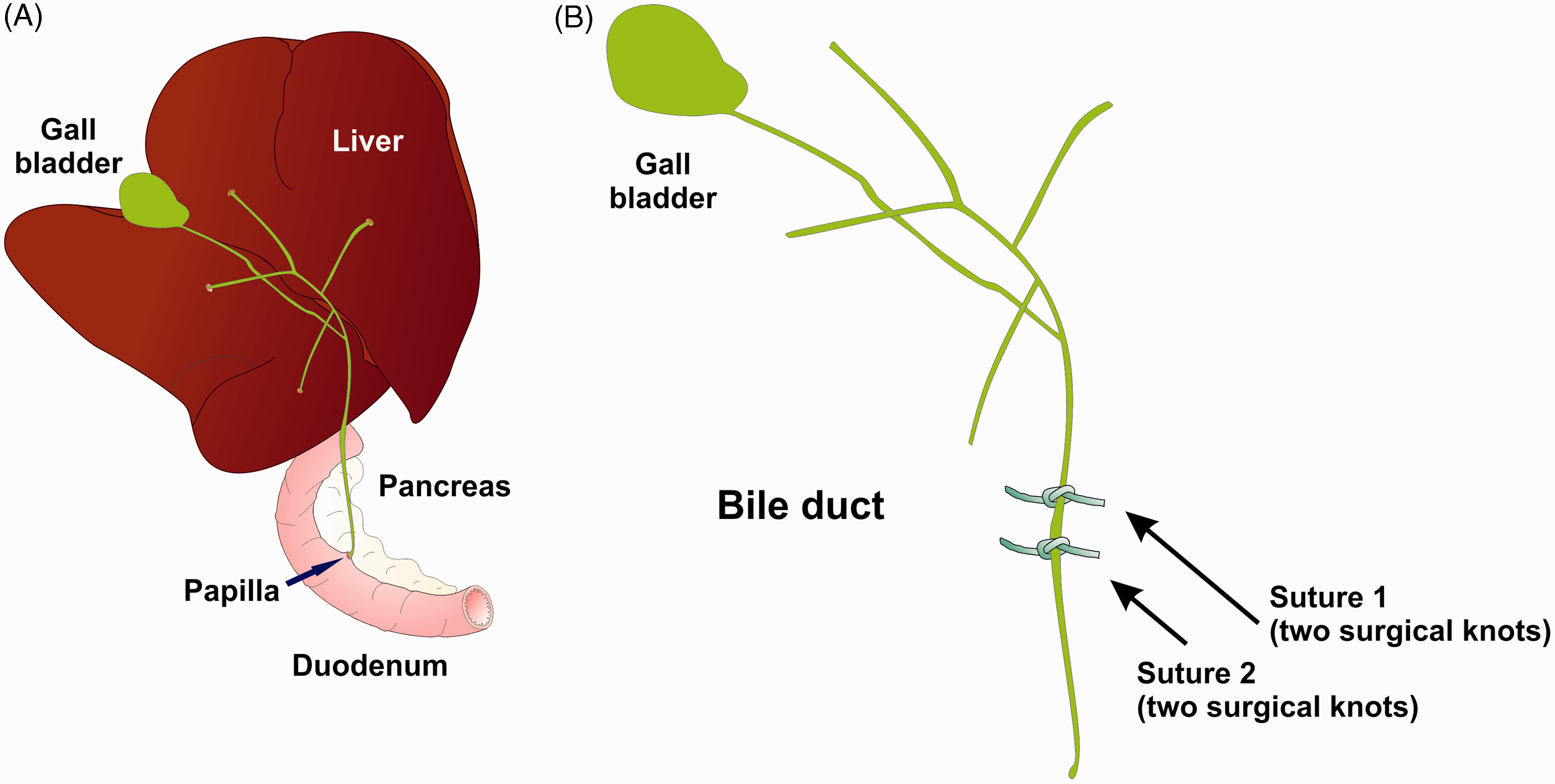
Required laboratory animal facility and legal issues
All animal experimentation that is described below requires strict adherence to national guidelines that may vary within each country as well as to specific institutional guidelines and rules that may vary between different animal facilities or institutional review boards. Furthermore, to allow for high reproducibility between experiments it is essential that the only animals subjected to this surgery are those that are kept permanently under specific-pathogen free (SPF) conditions. In our animal facilities, mice are housed on a 12 h light/dark cycle and have free access to food and water. In cases where animals are received from a commercial supplier or a collaborating laboratory, they are allowed to acclimatize to our conditions for at least one week.
Genetic background and gender
As in most other models, the degree of fibrosis that develops in this model depends on the genetic background of the mice as well as on any transgenic manipulations that interfere with the process of fibrogenesis.26,27 In addition, some studies of the application of therapeutic drugs that modulate bile acid toxicity and cholestasis have reported that the outcome of fibrosis is gender-specific. 28 Potential age- and development-dependent effects on the susceptibility to the sequelae of chronic cholestasis that have been reported in rats must also be considered in mice. 29 Therefore, it should be first clarified which genetic background, sex and age of animals are most appropriate for the planned experimentation. When experiments with large numbers of animals are performed, mice should be randomly assigned to the different groups (e.g. BDL group, sham-operated group, treatment group). When limited numbers of animals are available or intended for BDL experiments, only one sex (preferably male) and a small age range (preferably 8–12 weeks of age) should be used for surgery.
Severity and duration of BDL
The pathogenic events that are induced after performing BDL surgery in rodents is highly complex (cf. Figure 1). Most of the alterations are time-dependent. For example the bile acid composition in liver and serum after BDL increase within the first 6 h after surgery and remain elevated for up to 14 days. 30 Inflammatory gene expression and infiltration of immune cells are observed as soon as five days after BDL, and a suitable time-point in mice for analysing hepatic fibrosis as an endpoint is 21 to 28 days.31,32,17–20
Although it has yet to be proven, other studies in rats have shown that fibrogenic effects of BDL are only transient and may partially reverse spontaneously after three weeks, as demonstrated by declined levels of collagen at later time-points. 33 The same study has also reported on the time-dependent variations in ALT, AST, ALP and γ-GT after BDL. Of course, the severity of liver damage is influenced by the overall quality of the ligation, and there are several studies that require total ligation and others that require partial ligation of the bile duct. The setting of a well-directed, partial BDL for 14 days has been shown to represent a suitable model for acute cholestasis. Compared with total BDL this model results in significantly less necrosis formation and consequent hepatocyte proliferation, lower levels of serum bile acids, and decreases in biliary pressure. 7 The major time-related changes that occur 8 h to six weeks after setting of a total BDL in C57BL/6 mice affect levels of serum markers, transcription of pro-proliferative mediators, hepatocellular and cholangiocellular proliferation, neutrophil and lymphocyte infiltration and accumulation of collagen. 34
Biometrical aspects
Representative sample size calculation for a typical bile duct ligation (BDL) experiment.
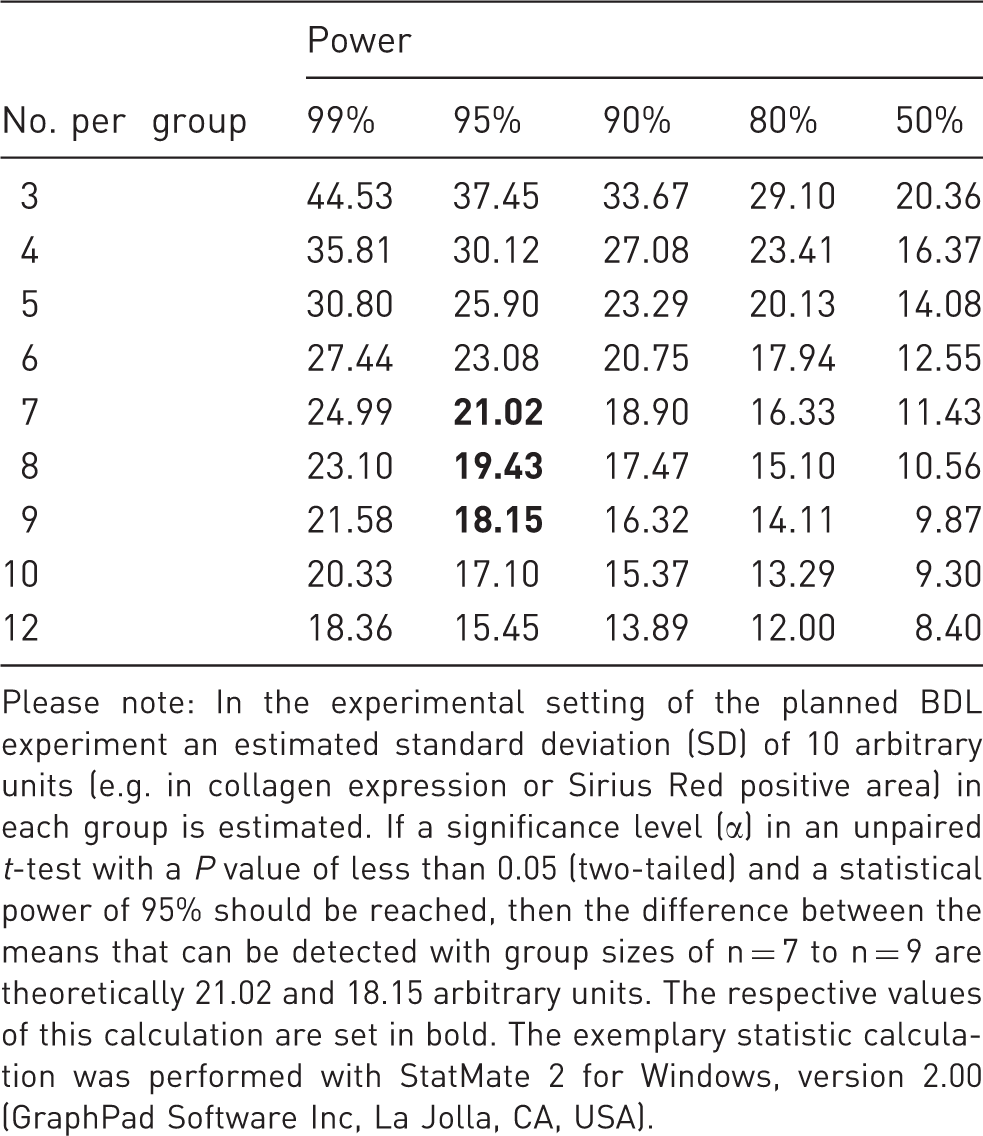
Please note: In the experimental setting of the planned BDL experiment an estimated standard deviation (SD) of 10 arbitrary units (e.g. in collagen expression or Sirius Red positive area) in each group is estimated. If a significance level (α) in an unpaired t-test with a P value of less than 0.05 (two-tailed) and a statistical power of 95% should be reached, then the difference between the means that can be detected with group sizes of n = 7 to n = 9 are theoretically 21.02 and 18.15 arbitrary units. The respective values of this calculation are set in bold. The exemplary statistic calculation was performed with StatMate 2 for Windows, version 2.00 (GraphPad Software Inc, La Jolla, CA, USA).
Reversibility of hepatic fibrosis
Spontaneous resolution from established liver fibrosis with restoration of the normal hepatic architecture has been described in rat livers that were harvested from animals that received twice weekly intraperitoneal injection of carbon tetrachloride and were left untreated thereafter for up to a further four weeks. 38 Similar recovery from biliary fibrosis in animals that have been subjected to BDLs for 21 days and subsequently subjected to biliodigestive anastomosis was reported later.39,40 In mice, a recent study has shown that the reconstruction of bile flow by cholecystojejunostomy allows proper drainage through the constructed anastomosis, resulting in almost complete hepatic tissue repair. 41 All these findings demonstrate that the BDL model is also principally suited for studies intended for investigating the molecular and cellular processes that underlie fibrosis resolution.
Practical implementation
Pre-surgical animal and workplace preparation
To avoid any unexpected complications, all necessary surgical procedures should be done under clean and aseptic conditions. Since it is already known that laboratory routines without any surgical intervention causes stress to animals and have substantial scientific and humane implications for the use of animals in research, 42 operators should avoid any unnecessary handling of animals and thereby cause extension of the surgical process. Therefore, all necessary equipment and reagents should be clearly arranged prior to the beginning of an experiment. In addition, animals that are subjected to BDL surgery should be placed on a controlled heating pad adjusted to a constant temperature of 37℃ throughout the complete treatment (e.g. HP 062; Labotect GmbH, Göttingen, Germany). To avoid eye desiccation, eyes should be covered with appropriate eye and nasal ointment after the onset of deep anaesthesia.
In our laboratory, we use the following pre-surgical work routine for BDL:
All mice that are to be included in the experiment should first be weighted and examined for signs of physical or behavioural abnormalities. Animals showing any kind of irregularities should be excluded from the subsequent surgery. Mice should be anaesthetized by inhalation with 4% isoflurane (2-chloro-2-(difluoromethoxy)-1,1,1-trifluoro-ethane) in 100% oxygen with a flow rate of 4 L/min as inhalational anaesthesia. This anaesthesia not only reduces pain for the animal but also has the beneficial side-effect of acting as a relaxing agent on muscles, thereby simplifying intra-abdominal operations. In a next step, respective mice should be shaved at the abdomen using an appropriate electric fur shaver (e.g. Shaver Favorita II from Aesculap; Braun Melsungen AG, Melsungen, Germany). After the onset of anaesthesia, the content of the isoflurane should be reduced to 1.5–3.0% in 100% oxygen, and the flow rate should be adjusted to 1 L/min. During anaesthesia an ISOFLO vaporiser should be used (Eickemeyer KG, Tuttlingen, Germany) that allows isoflurane concentrations to be properly adjusted. Final preparation of animals should be done by intraperitoneal injection of an appropriate narcotic analgesic that decreases perioperative pain as well as during the recovery period. In our laboratory buprenorphine hydrochloride solution at a concentration of 0.1 mg/kg body weight dissolved in 0.9% NaCl solution is used for this purpose. This semi-synthetic partial opioid agonist is commercially available in many countries (e.g. Essex Pharma GmbH, Munich, Germany). Trade names for buprenorphine are, among others, Temgesic, Buprenex, Subutex, Suboxone and Bultrans. The final solution should be stored in a sterile vial, labelled with the preparation and expiration dates, and stored protected from light in a safe place to which only authorized staff have access (e.g. in poison or narcotics cabinets).
Surgical procedures
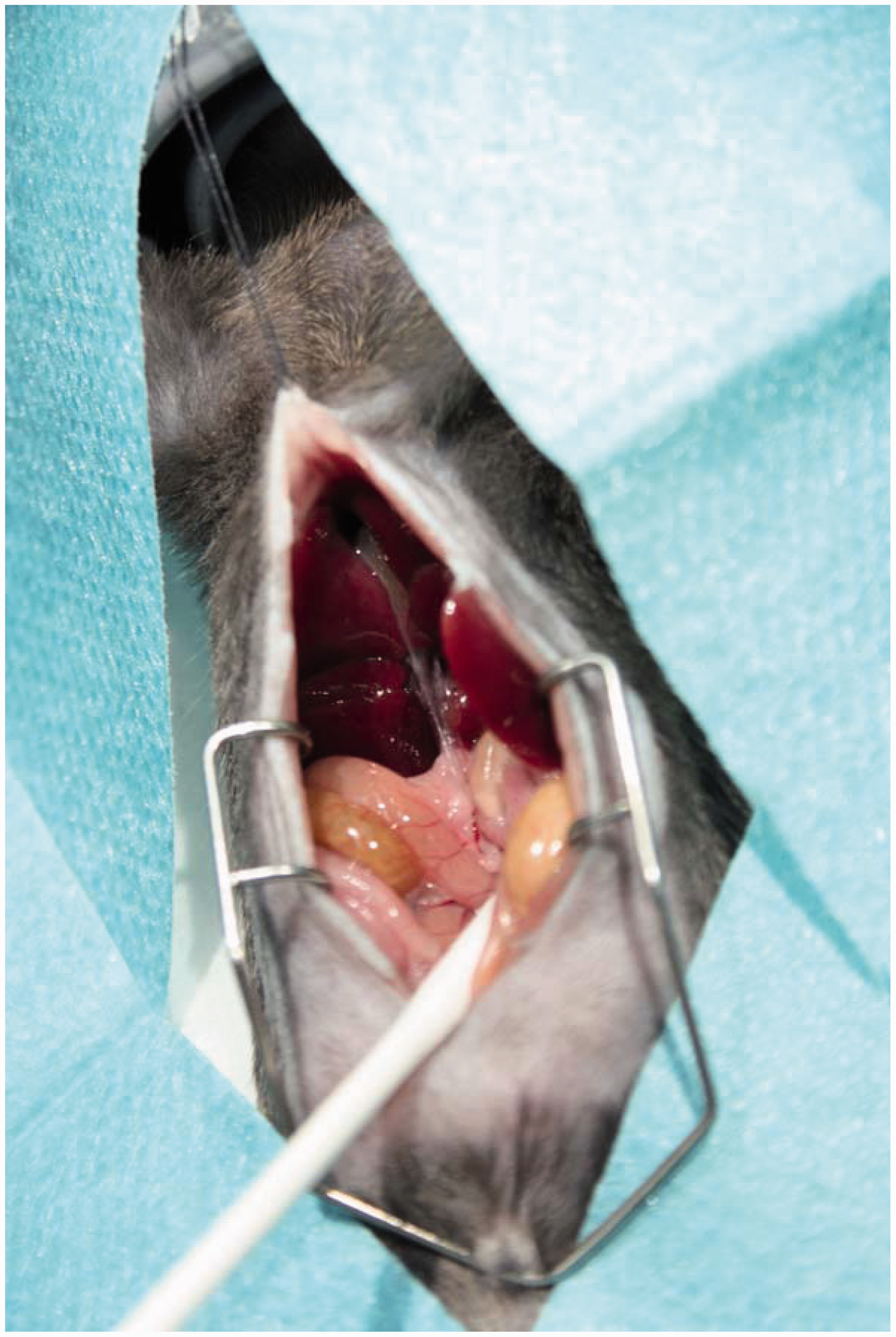
Prior to opening of the abdominal wall, the abdomen is sterilized with a gauze swab that is soaked with an appropriate antiseptic solution. The abdomen of the mouse is opened via a midline laparotomy with an incision of 2 cm in length. For this purpose sterile, razor sharp surgical scissors are routed from the xiphoid process to the region upstream of the urinary bladder. With this incision the cutis and fascia are separated, thereby exposing the underlying peritoneum and the linea alba. Please note that this step risks injury and should therefore be carried out with caution. The surrounding connective tissue that is wrapped around the peritoneum is moved away carefully using the end of the scissors. The peritoneal cavity is opened via a precise cut along the linea alba, and the cavity is enlarged by inserting a holding suture (e.g. 6-0 Mersilk; Ethicon, Johnson and Johnson, New Brunswick, NJ, USA) into the sternum that is subsequently fixed with its two ends on the top of the mask of the vaporiser. To allow unhindered intervention at subsequent steps, a stainless steel Colibri retractor is inserted into the peritoneal cavity to spread the edges of the surgical incision. In our laboratory, Colibri retractors are used with a length of 3 cm and a maximal span of 1.5 cm (Fine Science Tools GmbH, Heidelberg, Germany). Subsequently, the edge of the liver is lifted with a wet cotton swab (use 0.9% NaCl solution), the gut is moved caudally, and the duodenum is pulled down to expose the portal vein, the hepatic artery and the common bile duct (Figure 3). Preparation of the mouse for bile duct ligation (BDL). The abdomen of the mouse is opened via a midline laparotomy. The cavity is enlarged by inserting a holding structure and the operation area is spread by inserting a Colibri retractor. Please note the gleam bile duct that is clearly visible when the ventral side of the liver is lifted and the gut is caudally moved with a humidified cotton swab.
Setting the ligatures
Please note that some laboratories recommend dissecting the bile duct in between the two ligatures. By contrast, we have learnt through experience that potential leaks in one of the knots can result in severe acute peritonitis, ascites, systemic endotoxemia, renal impairment and nephrotoxicity when the bile duct is dissected. Therefore, we recommend leaving the bile duct intact to prevent these serious and unwanted adverse events.
As a first step, the common bile duct is separated carefully from the flanking portal vein and hepatic artery (Figures 4A and 4B). For this purpose, micro-serrated forceps with tips that are 0.5 mm and curved (e.g. type Moria MC31 from Fine Science Tools GmbH) are used. A 5-0 Mersilene suture (e.g. Ethicon, Johnson & Johnson Medical GmbH, Norderstedt, Germany) is then carefully placed around the bile duct (Figure 4C) and tied with two surgical knots above the duodenum. At this step it is essential that the knots are tightly fixed to enable effective obstruction. Another 5-0 suture is then tied in the same manner immediately near the hilum of the liver (Figure 4D). When all knots are fixed, superfluous sutures are removed and the peritoneal cavity is rinsed with a 0.9% NaCl solution. Thereafter, the sternum is lowered and brought into a normal position. The retractor is removed from the cavity and all organs are placed into a physiological position by gently shaking the abdomen. The peritoneum and the cutis plus fascia are then subsequently closed layer by layer and with separate 6-0 Mersilk sutures (Ethicon). Setting the double ligations of the biliary duct. (A, B) For setting the double ligations, the bile duct is first separated from the portal vein and from the hepatic artery using forceps. (C, D) The two 5-0 sutures are pulled through under the bile duct, and each one is fixed with two surgical knots.
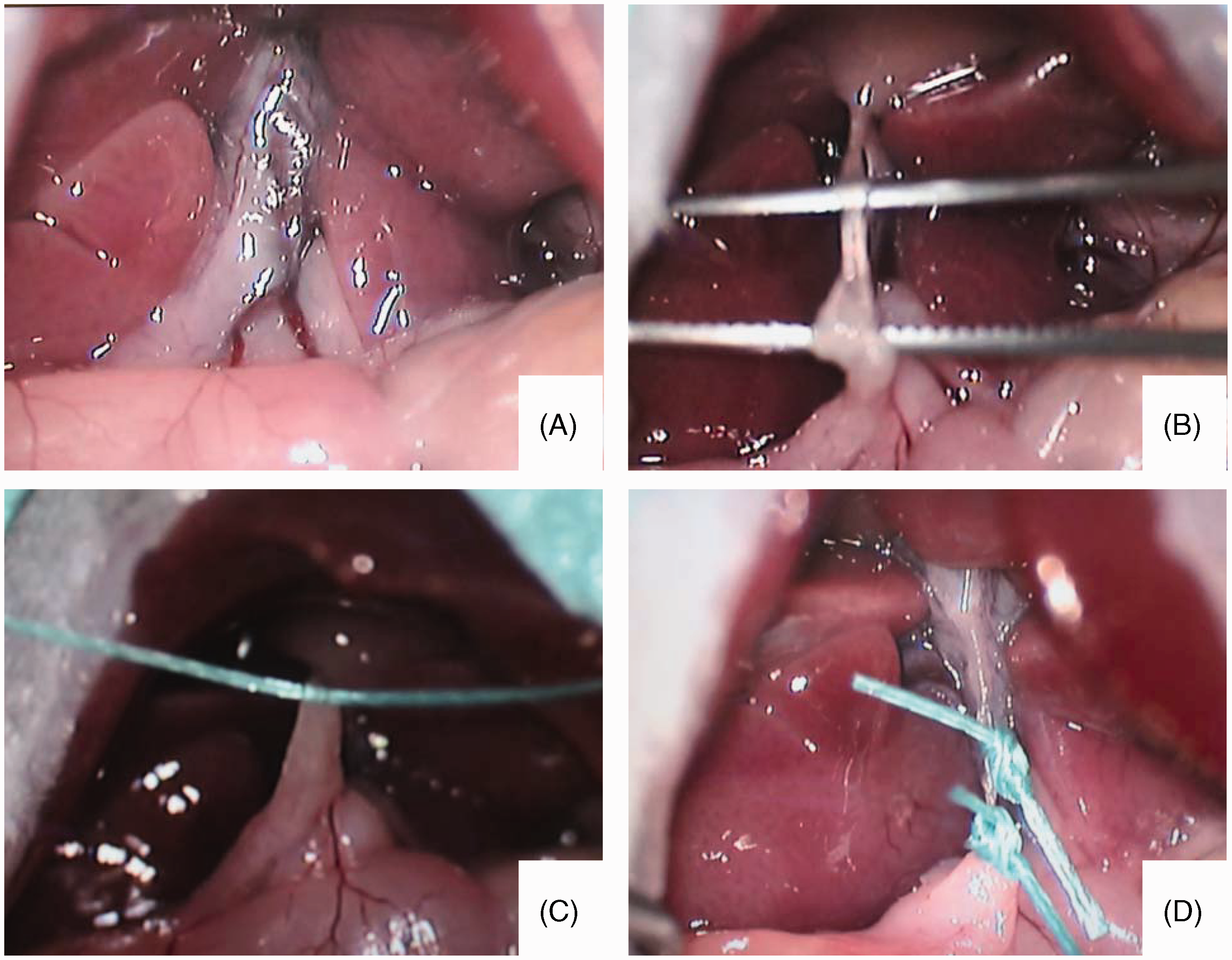
Postoperative animal treatment
After surgery, animals are allowed to recover and are carefully observed until they have recovered fully and behave normally. To guarantee optimal conditions during the recovery phase, it is recommended to warm up cages using an infrared lamp or use an intensive care cage with warmed air. In our laboratory, a standard 100–150 W infrared lamp that can be obtained from any standard household machinery provider (e.g. Petra Electric GmbH, Ense, Germany) is used. The lamp should be placed at a sufficient distance to avoid overheating the animals. When animals are fully awake and recovered, they can be transferred to normal cages with free access to water and food. To detect potential complications as early as possible, animals that underwent BDL or sham surgery should be monitored at regular intervals (e.g. twice daily). In all animals, postoperative treatment with an appropriate analgesia should be done strictly following the local recommendation of the responsible animal care and use committees. In our experimental set-up, analgesic therapy using buprenorphine solution is performed for three days. Successful BDL is recognizable by inspection of the skin and urine that both turn yellow as soon as the second or third day after the surgery.
Controls, readouts and humane endpoints
Studies performed in rats have shown that four weeks after BDL the animals show significant bacterial overgrowth in the ileum that affects nearly half of the cohort. 43 In the same study it has been shown that bacterial translocation to the mesenteric lymph nodes and spleen is also found in 10% of animals that receive only sham laparotomy. These data indicate that intestinal bacterial overgrowth, spontaneous bacteraemia and other infectious complications frequently occur when using this experimental model and that sham-operated animals are necessary controls when applying BDL as a model for studying inflammatory liver insult. This is absolutely mandatory since bacterial components such as lipopolysaccharides alone induce strong inflammatory activity (e.g. acute phase response) and are associated with robust and broadly systemic and central inflammation 44 that may directly or indirectly interfere with the outcome of a specific experiment. It should be further noted that the severity of the bacterial translocation after BDL in mice depends on the genetic background, and is for example significantly higher in C57BL/6 J than in A/J mice.45,46
As mentioned above, sham operations are performed in an appropriate number of animals as a control. Animals that undergo this treatment receive a midline laparotomy as described above. In these animals, the common bile duct is gently touched, mimicking the surgery that is essential for setting the BDL. Similarly, the abdomen of these control animals is closed in layers, and the animals are allowed to recover identically to the animals that have undergone BDL surgery.
Readout systems for ongoing liver damage
Appropriate readout systems for ongoing hepatic damage are increases of ALT, AST, AP, γ-GT and total bilirubin. After prolonged BDL the fibrotic process is histologically indicated by morphological alterations that are visible in haematoxylin and eosin stains (Figure 5A), elevated concentrations of collagen deposits (Figure 5B), and strong α-SMA positivity as a measure of stellate cell activation (Figure 5C), and by increased intrahepatic expression of the proliferation marker Ki67 (Figure 5D). Moreover, the expression of the basic filament protein cytokeratin 7 (CK-7), representing a histological marker of cholestasis and primary biliary cirrhosis, is strongly increased in these livers (Figure 5E).
Immunohistochemical stainings detecting hepatic fibrosis. Representative liver sections of mice that underwent bile duct ligation for 21 days were prepared and stained with (A) haematoxylin and eosin, (B) Sirius Red, or with specific antibodies for (C) α-smooth muscle actin, (D) Ki67, or (E) CK-7. (a–e) The same stains were preformed in liver sections taken from a sham-operated control animal. Scale bars in each panel correspond to 500 µm.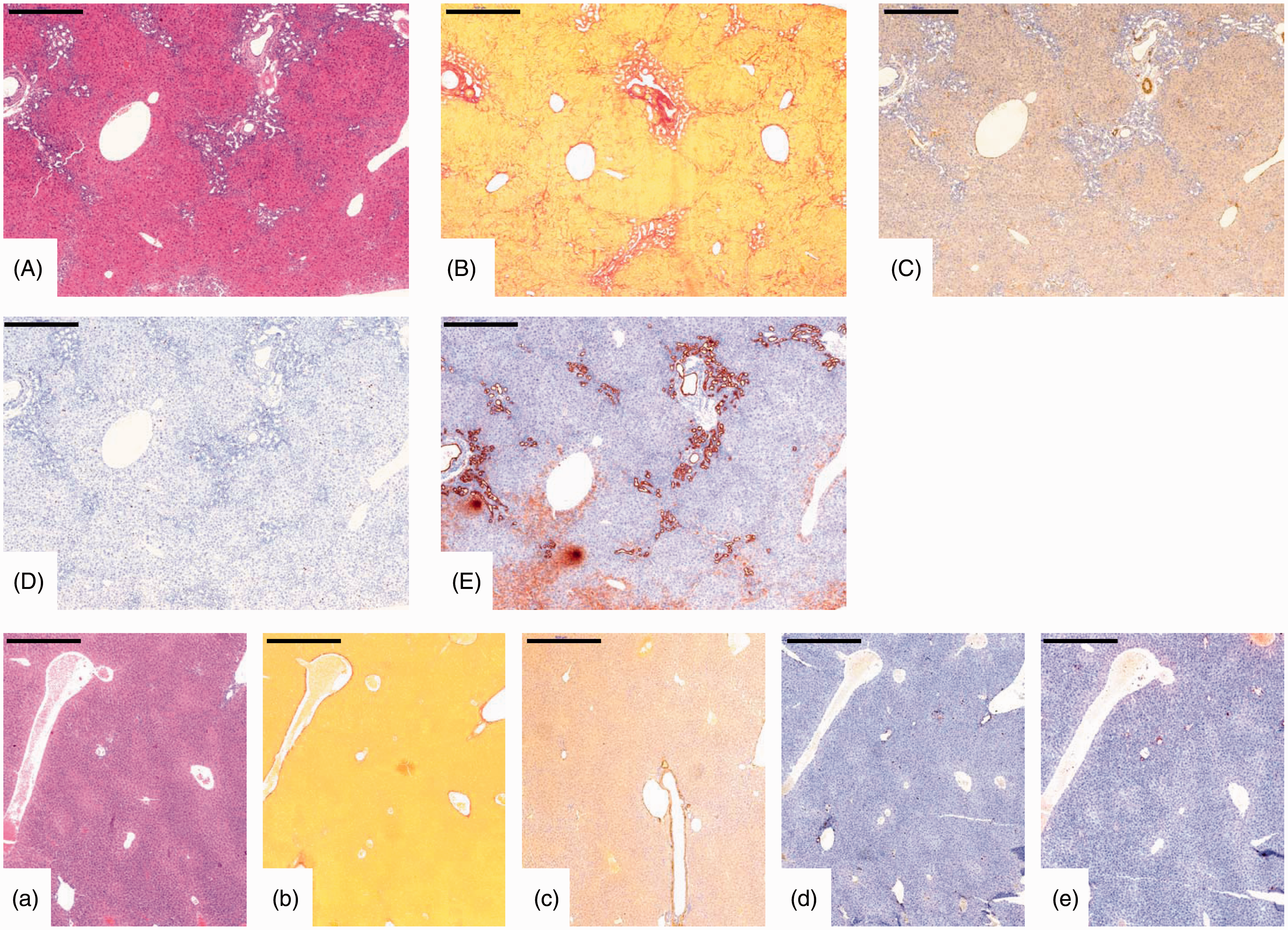
Animal burden/side-effects
The animal burden results mostly from surgery per se. Therefore it is absolutely essential that both the BDL-treated and sham-operated animals receive adequate anaesthetic treatment during the entire surgery and appropriate postoperative management and analgesia. In addition, there is a wealth of literature available showing that rats with prolonged BDL have a lower mean arterial pressure, renal blood flow, glomerular filtration rate, and sodium and water excretion than control rats.47–50 It should be further mentioned that prolonged biliary obstructive jaundice in mice sensitizes animals for endotoxemia as soon as 14 days after the surgery. 51
In our laboratory the animals usually recover within a few minutes after surgery, and postoperative mortality is less than 5%. Although the mortality rate is not significantly influenced by age or sex, we and others have frequently observed a significantly higher value in animals carrying specific genetic alterations. 52 In addition, it is well accepted that genetic background also impacts on the severity of the cholestatic injury.45,46 Moreover, we have observed that the outcome of the inflammatory response or fibrogenesis might be directly affected by genetic alterations which interfere with critical signalling pathways or which are directly associated with the process of ongoing hepatic fibrogenesis.16,17
In our experiments, we have appraised abnormal animal behaviour, excessive debilitating diarrhoea, and bleeding from any orifice or the wound as symptoms of rare complications such as peritonitis, sepsis, internal bleeding, or even of overwhelming pain and distress. If any of these complications should arise, this should be considered to be a humane endpoint for terminating the experiment. In our view, the documentation and scoring of observed adverse effects or clinical signs of animal discomfort that have been proposed elsewhere 53 might be helpful when the described BDL procedure is undertaken for the first time.
Classification of severity of procedure
According to Article 15 of the EU Directive 2010/63 (http://eur-lex.europa.eu/LexUriServ/LexUriServ.do?uri=OJ:L:2010:276:0033:0079:en:PDF) the estimated degree of pain, suffering, distress or lasting harm of animals subjected to BDL surgery must be classified according to the following scoring system: ‘non-recovery’, ‘mild’, ‘moderate’ or ‘severe’.
Details of the classification criteria that underlie this assessment have been established by the Expert working group on severity classification of scientific procedures performed on animals. These criteria of classification can be found at: http://ec.europa.eu/environment/chemicals/lab_animals/pdf/report_ewg.pdf.
The BDL model of up to four weeks with no major impairment of liver function is classified as a moderate procedure according to Article 15 of the above-mentioned EU Directive 2010/63.
Concluding remarks
Prolonged bile duct obstruction in mice is a highly reproducible experimental model that allows the induction of progressive liver damage in mice with low overall mortality rates. The surgery is technically rather simple and quick to perform. After surgery, development of phenotypic alterations follows a predictable kinetic. Therefore, this model is used worldwide by many scientists for studying fundamental aspects of hepatic inflammation, fibrosis, and cirrhosis. Based on our experience, this model is strongly recommended when aspects of acute cholestasis, hepatic inflammation or ongoing hepatic fibrogenesis related to cholestatic diseases are being investigated.
Footnotes
Acknowledgments
The authors would like to thank Mareike Schulz and colleagues for their kind help in preparing photographs of the BDL surgery.
Funding
This work was supported by grants of the German Research Foundation (SFB/TRR57, P9, P13, Q1, and Q3).
