Abstract
The toxic properties of various nitrosamines in animals and humans are well established. The parenteral or oral administration of the smallest quantities of diethylnitrosamine (DEN) or dimethylnitrosamine (DMN) results in severe liver damage. Most prominent are intense neutrophilic infiltration, extensive centrilobular haemorrhagic necrosis, bile duct proliferation, fibrosis, and bridging necrosis that ends in hepatocarcinogenesis. Due to the robustness of the induced hepatic alterations, the application of DEN in rodents has become an attractive experimental model for studies aimed at understanding the pathogenetic alterations underlying the formation of liver cancer, which represents one of the most common malignancies in humans worldwide. However, several studies have shown that the hepatocarcinogenic effects of nitrosamines might vary with the genetic background of the animals, their sex, their age, and other factors that might impact the outcome of experimentation. We present general guidelines for working with DEN, and a detailed protocol that allows the establishment of highly reproducible liver cancer in mice. The outcome of liver injury after the application of DEN in mice, as estimated by the formation of cirrhosis and cancer, appears to be a suitable animal model for the analysis of some aspects and processes that promote the pathogenesis of hepatocellular carcinoma in humans.
Historic background of the model
The carcinogenic potential of N-nitroso compounds was first brought to general attention in 1937 when it was reported that dimethylnitrosamine (DMN) might be one of the causative agents that induce liver damage in men. 1 This assumption was experimentally confirmed by Barnes and Magee in 1954, who found that a single oral or parental dose (20 to 40 mg/kg body weight) of DMN acts primarily as a liver poison producing severe liver necrosis in rats, mice, rabbits, guinea pigs and dogs. 2 In this landmark report it was further shown that the resulting liver lesion in DMN-poisoned animals is characterized by a sharp line of demarcation between the totally destroyed parenchyma and apparently uninjured liver cell areas. 2 Extension of these studies subsequently revealed that the chronic application of DMN in rats resulted in a very high incidence of malignant hepatic tumours. 3 Although the precise pathomechanism that induced hepatic tumorigenesis was not known at that time, the authors concluded that DMN, by virtue of its chemical and physical properties, may be of fundamental interest for the investigation and analysis of hepatic carcinogenesis. 3 The hepatotoxic and carcinogenic effects of many other N-nitroso compounds including diethylnitrosamine (DEN) were reported several years later.4,5 A large step in understanding the molecular mechanisms that cause the carcinogenic effect of DEN were reported in 1963 in a study demonstrating that the administration of DEN in rats resulted in the ethylation of the N7 atom in the guanines of nucleic acids that were extracted from the liver. 6 Later it was demonstrated that the biotransformation of DEN and DMN to ultimately alkylating metabolites which cause the DNA adduct formation is mediated in a pathway that is dependent on cytochrome P450 enzymes including CYP2E1.7,8 The important role of CYP2E1 for the bioactivation of nitrosamines was ultimately demonstrated in experiments that were carried out in Cyp2e1 null mice. 9 These mice showed a significant lower tumour incidence and multiplicity compared with wild-type animals after treatment with DEN. 9 Due to the carcinogenic properties of nitrosamines, the application of these substances, and in particular DEN, has become highly attractive for inducing liver tumorigenesis in rodents as an experimental model of human hepatocarcinogenesis.
The application of DEN in mice has now become a well-established model for studies aimed at understanding the pathogenetic alterations underlying the formation of liver cancer. Because of its lesser alkylation activity, the repeated application of DMN in rodents for several weeks has become attractive for studying the pathogenesis of hepatic fibrosis rather than just of hepatocarcinoma. When applied in doses of 10 mg/kg body weight for three or four weeks, DMN induces alterations that are compatible with the deterioration of liver functions during the pathogenesis of hepatic fibrosis. 10
Pathogenic mechanisms of liver damage
Various nitrosamines including DEN (Figure 1) are acute hepatotoxins and carcinogens in many animal species.
4
The prolonged oral feeding or parenteral application of DEN in rodents at high doses is extremely effective in inducing hepatic tumours.3,5 Even a single application of DEN at 10–90 mg/kg body weight usually has an irreversible carcinogenic effect in rodents.
11
Structure of nitrosamines. Nitrosamines (more precisely, N-nitroso-dialkylamines) are a class of organic compounds having the general structural formula R1R2-N-NO (left).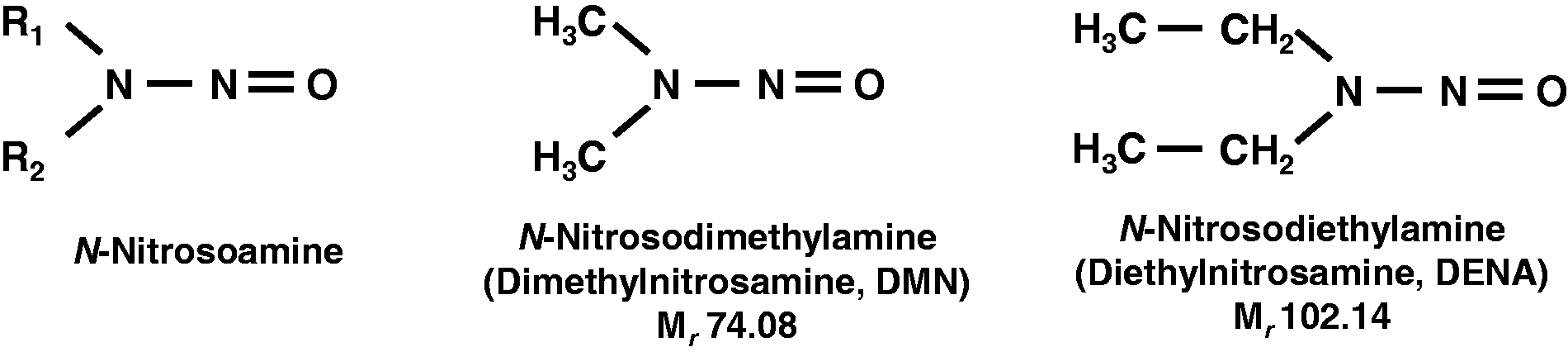
Likewise, DMN acts as a potent hepatotoxin, immunotoxin, mutagen and carcinogen in the rat, mouse, guinea pig, rabbit and dog when given in doses of the order of 25 mg/kg body weight.3,12 This nitrosamine targets the liver primarily, in which it is biotransformed by cytochrome P450-dependent mechanisms that have the highest activity in the centrilobular hepatocytes.
13
The principal metabolizing pathways are rather similar in rodents and humans.
14
The final degradation of DMN leads to the formation of reactive intermediates that are causative in the formation of methylated macromolecules. In particular, the formation of N7-methylguanine and O6-methylguanine in DNA is most critical (Figure 2). This was experimentally demonstrated in 1968 in a study that analysed the degree of methylation of RNA and DNA after the injection of DMN.15,16 Other promutagenic DNA lesions that are generated in high amounts in animals after administration of DMN are, among others, N3-methyladenine and O4-methylthymidine.
17
In the affected DNA molecules, these modified bases give rise either to depurination, yielding apurinic sites that may give rise to guanine to thymine transversions, or result in direct base mispairing. Many aspects of the principal pathways and the toxicity of the potential metabolites of nitrosoamine breakdown have been analysed in detail in cultured rat hepatocytes.
18
Simplified model of the biotransformation of dimethylnitrosamine (DMN). DMN is metabolically activated, mostly in hepatocytes, by cytochrome P450 enzymes including CYP2E1. Reactive metabolites may bind as electrophiles to DNA as is indicated. Please note that the ultimately carcinogenic methyl-cation is only hypothesized and may not freely exist. Typical adducts formed in DNA are N7-methylguanine and 06-methylguanine.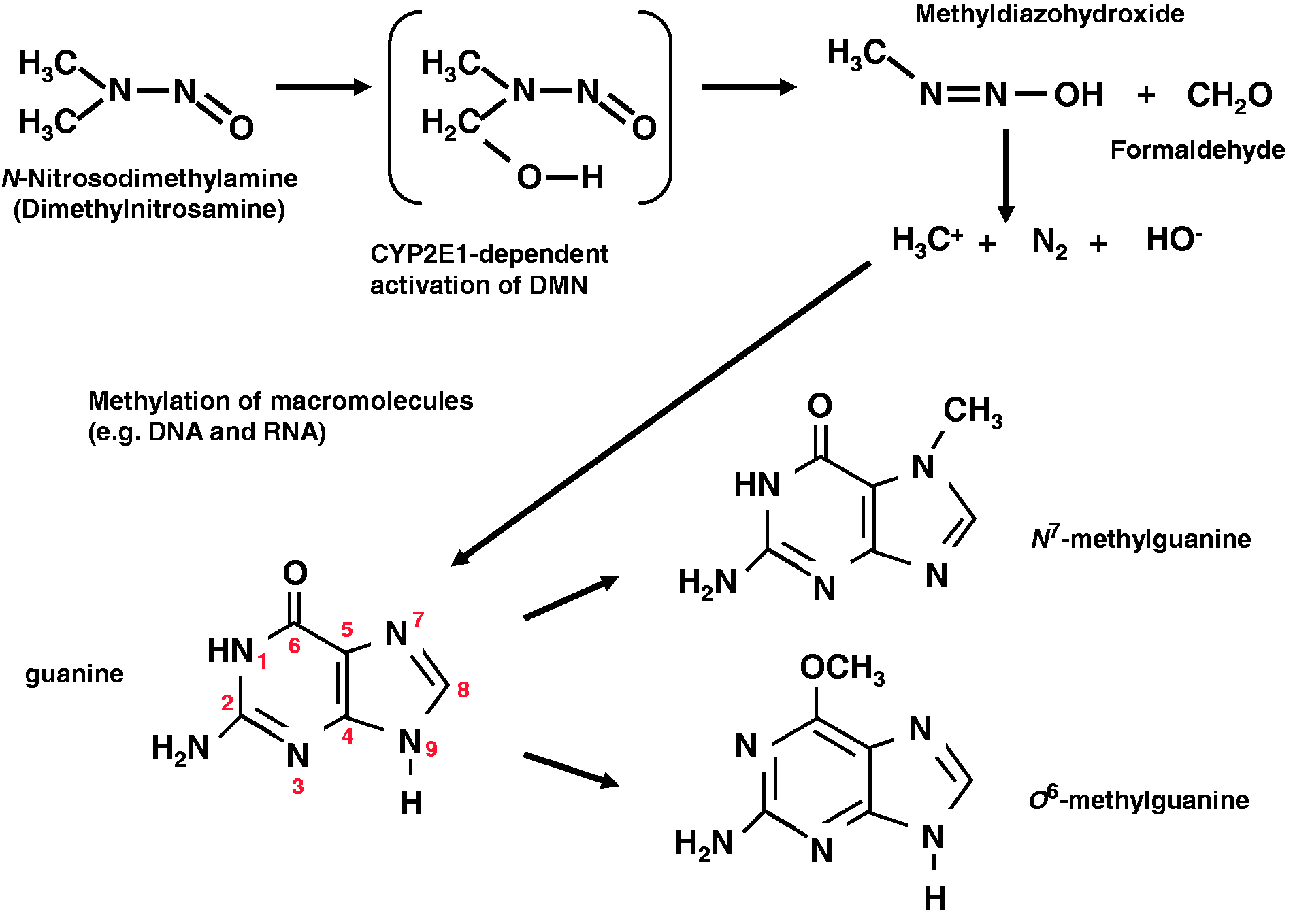
It was also shown that the dealkylation of DMN and DEN to their active mutagenic metabolites can be significantly modulated in both rats and mice by substances such as 3-methylcholanthrene and phenobarbital (PB) that increase hepatic demethylase activity. 19 However, this assumption was somewhat challenged in a later report in which it was shown that the specific alkylation density of the cellular genome after pretreatment with respective compounds is related to drug-mediated changes on the cellular level rather than to alterations in carcinogen metabolism. 20
The hepatotoxicity and carcinogenicity of both DMN and DEN have been comprehensively proven in rats that received these compounds from weaning onwards with drinking water. 21 . The study mentioned indicated clearly that the number of liver neoplasms induced by the treatment was simply proportional to the dose rate and that even quite low dose rates caused a variety of non-neoplastic liver abnormalities such as hyperplastic nodules and shrinkage of hepatocytes. 21 Detailed studies performed with DEN in F344 rats have confirmed that this carcinogen is highly reproducible and will dose-dependently induce liver damage within defined thresholds. 22 Other reports performed on rodents have further revealed that repeated application of DMN for three weeks results in an apparent loss of total proteins in the liver, while there is a significant increase in DNA content.23,24 In addition, oxidative stress caused by DEN is well known for contributing to the pathogenesis in hepatocarcinogenesis.25,26 In some other protocols, DEN is administered in combination with hepatotoxins such as CCl4. The combination of DEN and CCl4 for example was employed in a landmark study in which the contribution of the toll-like receptor 4 (TLR4) signalling the development of hepatic inflammation, fibrosis and a setting of hepatocellular carcinoma (HCC) development was analysed. 27 In this study, HCC was induced by intraperitoneal (IP) injection of DEN (100 mg/kg) at ages 6–14 weeks followed by 6–12 biweekly injections of carbon tetrachloride (CCl4) (0.5 mL/kg IP, dissolved in corn oil) in C3H/HeOuJ and C3H/HeJ mice; while in the treatment scheme of C57BL/6 mice, DEN (25 mg/kg IP) was given at day 15 postpartum followed by 22 weekly injections of CCl4 (0.5 mL/kg IP, dissolved in corn oil). The authors assumed that the additional application of CCl4 increase the endotoxin levels, thereby creating a microenvironment that more resembles the clinical setting in which most human HCC arises.
Experimental procedure
General considerations
Genetic background
It is well accepted that striking inter-strain differences and inherent sensitivities with respect to general liver tumour development exist in commonly used laboratory mice.28–30 The treatment of newborn male C3H/HeJ mice with DEN for example results in the induction of hepatocellular adenomas and carcinomas that are approximately 20- to 50-fold more frequent than that observed in similarly treated male C57BL/6J mice. 31 A detailed description about the relative susceptibility of selected mouse strains to hepatocarcinogenesis can be found elsewhere. 29 Highly susceptible strains are for example C3H and CBA. Intermediate susceptibility is conferred to strains FVB, SM/J, P/J, CE/J, LP and AKR/J, while relatively resistant strains are BALB/c, C57BL/6, 129 and SWR. It is worthwhile mentioning, however, that liver tumours develop in mice of resistant strains, too, but with a delay of about 2–3 months when compared with the sensitive strains.
Age and sex of mice
Detailed studies on hepatocarcinogenesis in mice treated with single low doses of DEN have shown that the livers of infant mice are the most susceptible to the inception of carcinogenesis, while enzymatic competence (i.e. DEN-dealkylating activity) progressively increases with age, reaching approximately the same peak activity in both sexes between the 7th and 15th days of age. 32 At later stages, metabolic activity decreases with age, although more rapidly in female than in male mice, reaching a plateau by the 42nd day of life, 32 which is possibly also explainable by the fact that weanling mice are more susceptible due to a higher hepatic DNA synthesis at this age.
Extensive data in the last decades have shown that the incidence for HCC development is gender-dependent in humans and rodents, with a high prevalence in males. Several studies have also demonstrated that female mice are largely resistant to DEN-driven hepatocarcinogenesis. When mice are repeatedly exposed to DEN for a long time, 100% of the male mice but only 30% of females develop hepatic tumours, and it has been assumed that oestrogens may inhibit hepatocarcinogenesis while androgens stimulate this process.33,34 The observation that female mice are in general less prone to the formation of hepatic tumours is also shown with other tumorigenic compounds such as N-ethyl-N-nitrosourea. 28 A key publication 35 that was extensively considered and discussed 36 has demonstrated that the gender disparity in DEN-driven liver cancer is mostly due to gender-specific differences in interleukin-6 (IL-6) production in Kupffer cells, which in turn are oestrogen-mediated. Accordingly, the gender disparity in DEN-mediated HCC incidence is completely abrogated in IL-6 knockout mice.
In order to avoid unnecessary complications and to reduce the number of experimental mice it is therefore highly recommended that mice of the male gender are used exclusively.
Application routes
There are reports in the literature in which DEN was administered orally to rodents with their drinking water, 37 with their diet, by oral gavage, 38 by exposure via inhalation,39,40 or by intratracheal or intragastric instillation.41,22 However, the most commonly used method is the IP injection of DEN solution42,43 because this administration route allows the proper dosage and guarantees that each animal receives approximately the same amount of nitrosamine.
Duration of treatment
Overview of the use of diethylnitrosamine (DEN) in mouse models.
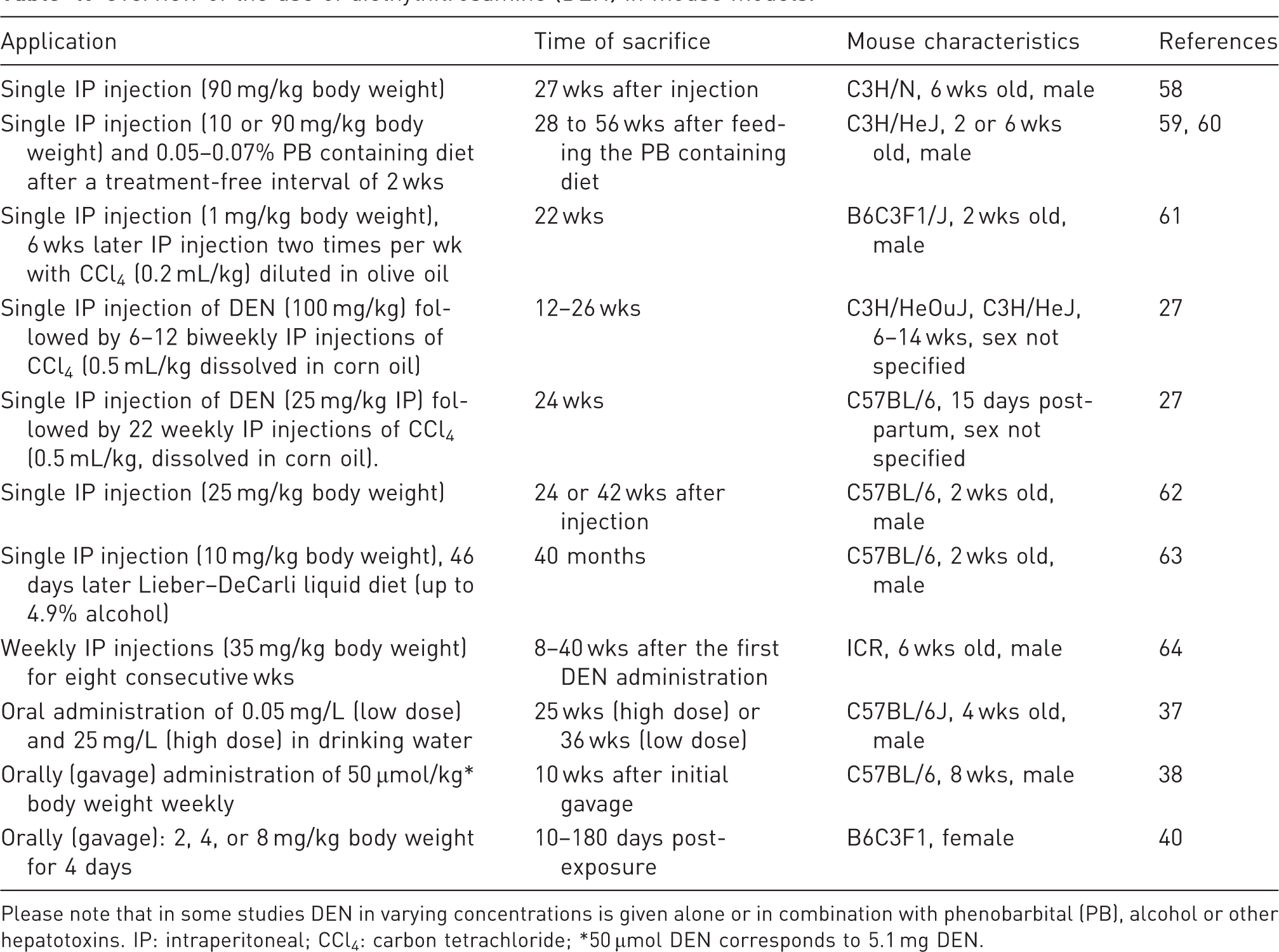
Please note that in some studies DEN in varying concentrations is given alone or in combination with phenobarbital (PB), alcohol or other hepatotoxins. IP: intraperitoneal; CCl4: carbon tetrachloride; *50 µmol DEN corresponds to 5.1 mg DEN.
Initiation–promotion studies and tumour genotype/phenotype
The majority of liver tumours generated by a single dose of DEN in weanling mice (2 weeks of age) are mutated in either the Ha-ras gene (almost exclusively codon 61) or the mutational hot-spot codon in the B-raf gene.45–47 Similarly, tumours generated by a single injection of DEN in 6-week-old mice are mostly Ha-ras or B-raf mutated. By sharp contrast, tumours induced by a single application of DEN to 6-week-old mice followed by long-term treatment with the model tumour promoter PB are 90% or more mutated in Ctnnb1 which encodes the oncoprotein β-catenin.
48
Ha-ras and B-raf mutated tumours are basophilic in the haematoxylin and eosin (H&E) stain and can be identified by positive staining for the phosphorylated form of the extracellular-signal regulated kinase 1/2 (ERK1/2) in the case of Ha-ras mutated tumours or by positive staining for E-cadherin (both Ha-ras and B-raf mutated tumours). For representative examples see Figure 3. By contrast, Ctnnb1-mutated tumours are eosinophilic in nature and stain positive for the marker enzyme glutamine synthetase
49
and several cytochrome P450 isoforms (see review
50
). For a representative example see Figure 3.
Phenotype of mouse liver tumours of differing genotype. Liver sections from tumour-bearing animals were stained immunohistochemically for the following markers: (a) glutamine synthetase (GS), showing positive staining in a Ctnnb1 (encoding β-catenin) mutated tumour; (b) E-cadherin and (c) p-ERK, showing positive staining in a Ha-ras-mutated tumour.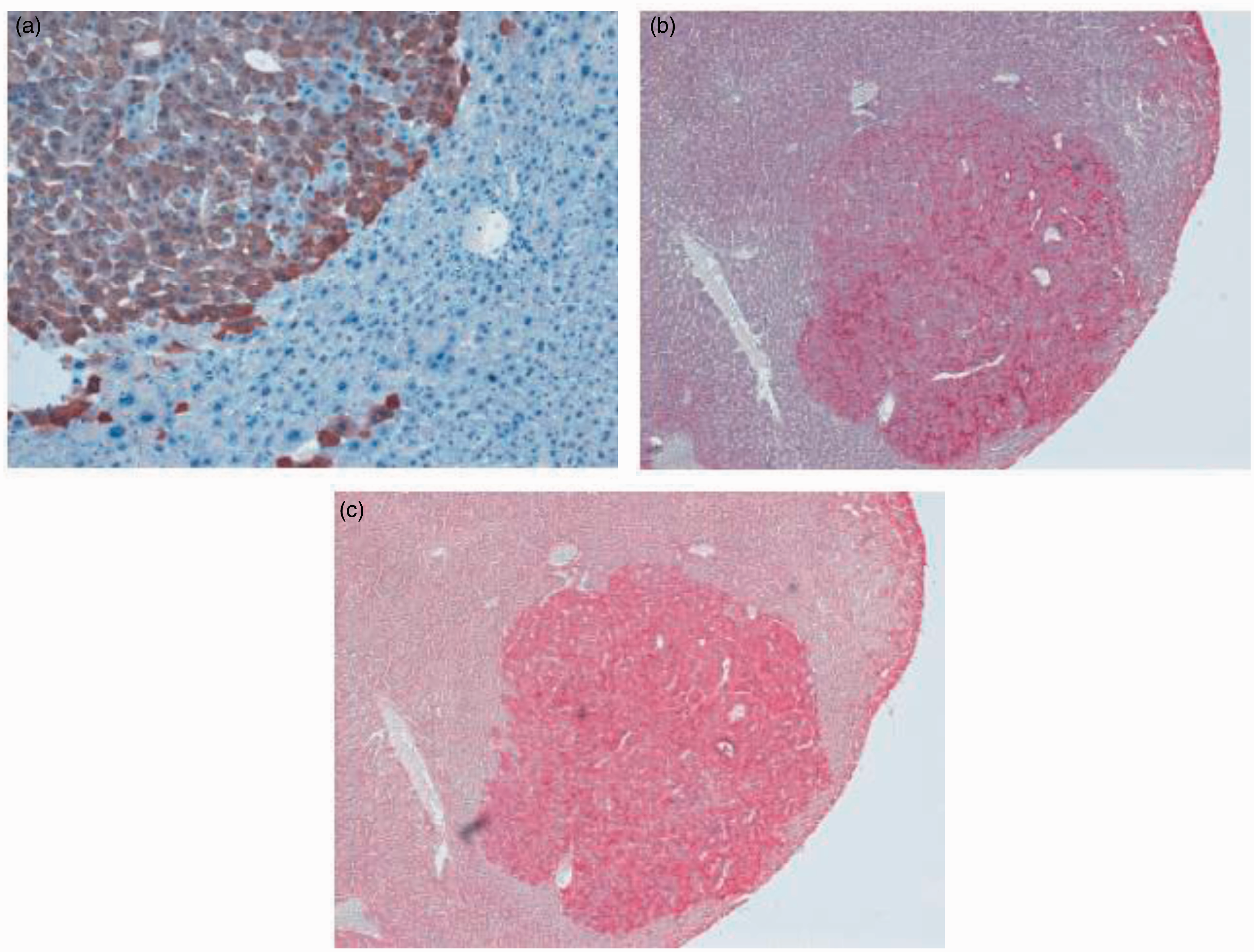
Biometric calculation of requested cohorts
Strain susceptibility to spontaneous and induced liver tumour formation depends on genetic and numerous other factors. 29 In line with this assumption it has been demonstrated that the growth of preneoplastic lesions and the susceptibility to hepatocarcinogenesis between C3H/HeJ (high susceptible strain) and C57BL/6J (low susceptible strain) mice varies by a factor of up to 20-fold, utilizing an identical protocol. 51 Therefore, the required cohorts in an experiment might be highly variable. DEN is highly mutagenic in mice, and our experience has shown that the final number of animals that is necessary to reach an appropriate statistical power depends on many factors. In particular, it is obvious that larger group numbers are required in experiments in which the long-term effects of hepatic carcinogenesis are being investigated. Each experiment needs a detailed biometric analysis in which all factors (such as duration of treatment and expected results) that impact the outcome of an individual experiment need to be considered for the biometric calculation of cohort sizes. If molecular or cellular aspects of hepatocarcinogenesis are being investigated, we recommend starting with pilot experiments in which DEN is administered to only a few animals. Thereafter, the cohort sizes should be adapted when performing the final experiment. Fortunately, the new technology of non-invasive imaging leads to a significant reduction in the number of animals required, even in long-term carcinogenicity studies. 52
Practical implementation
Safety and storage
Based on the high toxicity of these substances, the injection solution containing DEN is always prepared under a fume hood with two tight layers of nitrile gloves, a protective gown, respiratory protection (type P3 mask) and protection goggles in a room with restricted access that is marked as such. Nitrile gloves and syringes that have been used to prepare the solutions are afterwards disposed of in a hazardous waste container. There are also protocols available in which excess quantities of N-nitroso compounds can be destroyed in alkaline solution with an addition of nickel-aluminium alloy. 53 In addition, it is essential that all excrement of animals should be collected on a daily or weekly basis and disposed of as hazardous waste. The animals should be kept litter free and separated from all other animals.
In case of any accidental exposure through skin contamination, inhalation, or needle stick injury, a 3 mL urine sample should be taken of the exposed person and stored at –70 ℃ for an analysis of nitrosamine metabolites. 54 This allows for an estimation of DEN intake and contributes to individual risk analysis and causal attribution for possible future diseases.
Preparations
DEN like many other nitroso compounds is sensitive to light exposure, especially to ultraviolet light, that leads to photodegradation in which dimethylamine, nitrite, nitrate ions, formaldehyde, formic acid, and methylamine are formed.55,56 However, in our experience, DEN is stable at room temperature for more than 14 days in aqueous solution (0.9% NaCl) when stored light-protected. We obtained DEN for our experiments with a purity grade ≥99.0% (product code #73861) from Sigma-Aldrich (Taufkirchen, Germany). The compound was delivered with a detailed safety datasheet that had to be read before working with DEN. Moreover, the safety datasheet that also included information about first aid measures had to be filed, periodically reviewed and placed at a location that was accessible in case of accident.
Procedure
Please note: As outlined above, sex and age are factors that impact on hepatocarcinogenesis in mice. In addition, humoral influences on cancer susceptibility are reported for female mice. To avoid unnecessary variations in experimentation, we recommend using only male mice that are of the same age. Groups of age-matched control mice should be treated in parallel using only the solvent (0.9% NaCl) as an injection solution. Please keep in mind that the handling of mice (i.e. taking them out of their cage, fixation and exposure to a foreign surrounding) is already stressful for them. Therefore, the handler should be well-trained before working with DEN.
During the application of nitrosamines, the animals have to be placed on a heating pad to guarantee an ambient temperature of 37 ℃ (e.g. HP062; Labotect, Göttingen, Germany) to prevent hypothermia. If experienced personnel perform the injection this step can be omitted. All mice that are used in the experiment should be inspected for any signs of distress such as changes in respiration or body temperature, rough hair coat, dull eyes, unusual behaviour, or hunched posture. All mice showing abnormalities should be excluded from the study. Healthy mice should be weighed prior to each injection to accurately calculate the dosage of DEN. The required volume of DEN that must be injected in each animal should be calculated and documented in a precast table. In our laboratory, we have a table listing the doses/injection volumes for a range of mouse weights. We recommend using animals at age 14 days and the application of a single DEN dose of 10 to 25 mg/kg body weight. When using animals at an age of between five and six weeks, we recommend using DEN doses of between 50 and 90 mg/kg body weight (possibly in conjunction with subsequent PB application). As with all injections in animal experimentation, we recommend injecting constant volumes (e.g. 100 µL) for both treatment and control groups. Therefore, it is beneficial if the body weights of the animals do not vary significantly. This further enables the treatment of animals with little delay and helps to minimize stress and unnecessary handling. The solution should be injected in mice using disposable, sterile syringes. To this end, the mouse should be scuffed behind the neck region between the thumb and forefinger. After turning over the hand the animal should rest on the palm against the base of the thumb using a third finger to stabilize the pelvic region. The abdomen should be kept taut, with the head held lower than the rest of the body so that the intra-abdominal organs move towards the diaphragm. The solution should then be injected into the lower side of the abdomen (left or right lower quadrant of the abdomen, preferably the left lower quadrant) using a sterile 27 gauge needle (e.g. 27 G Microlance; Becton Dickinson, Franklin Lakes, NJ, USA). To further avoid injury to the bladder, intestine or other intra-abdominal organs, aspiration is recommended before injection. After finishing the injections, all used syringes and needles should be disposed of in a container for hazardous waste. Control mice should be injected with an identical volume of vehicle (0.9% NaCl) in the same manner as given above. Mice should be re-inspected one hour after injection for abnormalities and thereafter at regular intervals. If repeated injections are necessary, the procedure should be iterated in the same manner up to the schedule end of the experiment. All injections and abnormalities during the injections should be documented punctiliously. At the end of the experiment, mice are sacrificed, e.g. by cervical dislocation, and blood and liver (and other organs) should be collected for subsequent analyses. Excess biological materials and animal cadavers should be disposed of in a container for hazardous waste.
Animal burden/side-effects
Variation in α-fetoprotein (AFP) expression 24 weeks after single diethylnitrosamine (DEN) application.
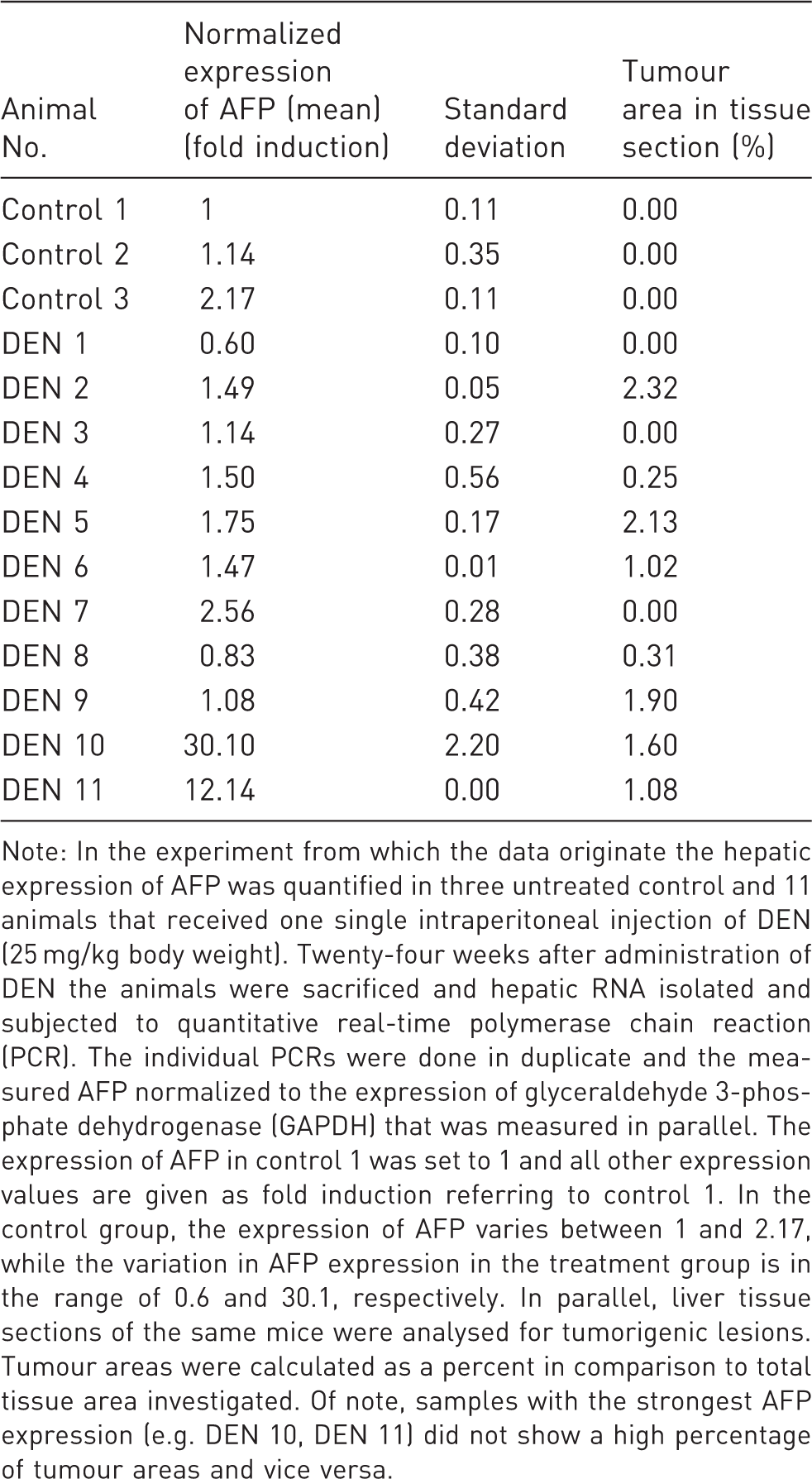
Note: In the experiment from which the data originate the hepatic expression of AFP was quantified in three untreated control and 11 animals that received one single intraperitoneal injection of DEN (25 mg/kg body weight). Twenty-four weeks after administration of DEN the animals were sacrificed and hepatic RNA isolated and subjected to quantitative real-time polymerase chain reaction (PCR). The individual PCRs were done in duplicate and the measured AFP normalized to the expression of glyceraldehyde 3-phosphate dehydrogenase (GAPDH) that was measured in parallel. The expression of AFP in control 1 was set to 1 and all other expression values are given as fold induction referring to control 1. In the control group, the expression of AFP varies between 1 and 2.17, while the variation in AFP expression in the treatment group is in the range of 0.6 and 30.1, respectively. In parallel, liver tissue sections of the same mice were analysed for tumorigenic lesions. Tumour areas were calculated as a percent in comparison to total tissue area investigated. Of note, samples with the strongest AFP expression (e.g. DEN 10, DEN 11) did not show a high percentage of tumour areas and vice versa.
During prolonged application of DEN, histological alterations that are characterized by intense neutrophilic infiltration, extensive centrilobular haemorrhagic necrosis, bile duct proliferation, fibrosis, and bridging necrosis are noticeable (Figure 3). The formation of these histological alterations is dose- and time-dependent. The phenotypic alterations that morphologically characterize the developing tumours and allow their stratification into genotype/phenotype classes can be visualized by enzyme and immunohistochemical stainings. In addition, imaging strategies including magnetic resonance imaging (MRI) allow the non-invasive demonstration of the developing tumour populations in the mouse liver (Figure 4). In particular, the animal burden increases with the number and size of intrahepatic tumours formed. In addition, the increasing impairment of the liver combined with the nitrosamine-induced immunosuppression might lead to systemic inflammation and in turn might develop critically during the experiment and result in high mortality rates. Therefore, the animal burden after application of DEN should be classified as being from moderate to severe. When marked physiological or behavioural signs of animal distress (humane endpoints) are noticed during the experimentation, the respective animal should be euthanized to relieve unnecessary pain or distress. These humane endpoints should be settled prior to the start of the planned experiment.
Demonstration of mouse liver tumours by magnetic resonance imaging (MRI). Mouse liver tumours were induced by a single intraperitoneal injection of diethylnitrosamine (DEN) into mice at two weeks of age. Tumours are indicated by white arrows. The smallest tumour detectable by MRI had a diameter of ∼1 mm. K: kidney. For further details see Ref. 52.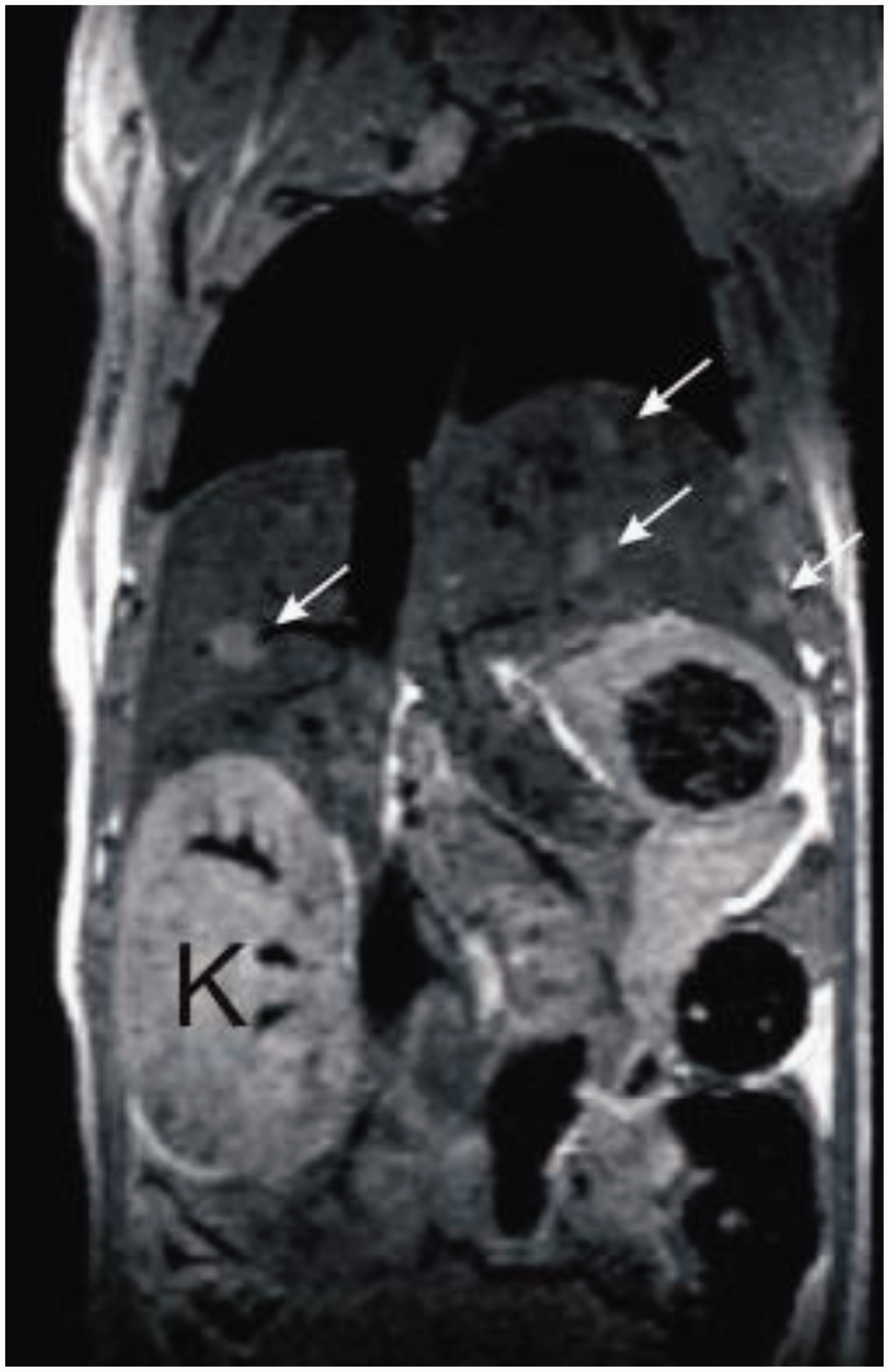
Classification of severity of procedure
According to Article 15 of the EU Directive 2010/63 (http://eur-lex.europa.eu/LexUriServ/LexUriServ.do?uri=OJ:L:2010:276:0033:0079:en:PDF) the estimated degree of pain, suffering, distress or lasting harm of the animals subjected to DEN application should be classified with a scoring system that includes ‘non-recovery’, ‘mild’, ‘moderate’ or ‘severe’.
Details about the classification criteria that underlie this assessment have been established by the Expert Working Group on severity classification of scientific procedures performed on animals. These can be found at: http://ec.europa.eu/environment/chemicals/lab_animals/pdf/report_ewg.pdf.
The IP application of DEN twice a week with no major impairment of liver function is classified as a moderate procedure according to Article 15 of the above-mentioned EU Directive 2010/63. Animals can develop significant weight loss during and after the procedure. Therefore, body weight has to be carefully monitored (daily) and to refine the procedure, extra food (jelly packs or food pellets mixed with water are to be placed on the cage bottom level. In case of ≥20% of weight loss for longer than 48 h compared with the weight at the beginning of the experiment the mouse should be euthanized. In case of tumour genesis humane endpoints should be implemented to restrict pain, suffering, distress or lasting harm to the animals.
Concluding remarks
The sequence of pathophysiological alterations in chemically-induced models of hepatic cancerogenesis has a high similarity with that seen in HCC in humans. In comparison to other mouse models of hepatocarcinogenesis (xenograft models, genetically-modified mice), these chemical models are favourable when HCC development on a natural genetic background is being studied. The carcinogenic capacity of DEN and other N-nitroso compounds are mainly found in the capability of these compounds to be activated by cytochrome P450-dependent pathways into reactive metabolites that alkylate DNA. Nitrosamines evolve their effects in a dose-dependent and cumulative manner. While a single small dose of DEN may not lead to the formation of neoplasms in experimental models, the administration of higher doses or the prolonged application of nitrosamines induces hepatocarcinogenesis after a period of latency. Although the sequence of events following DEN application is highly reproducible, the pathogenesis is also influenced by the sex, age and strain of the mice. Based on our experience and on a multitude of independent reports, we recommend the use of male mice at young age (approximately 2 weeks old) and a singular IP injection of DEN at 10 mg/kg body weight to induce hepatic tumour formation. The majority of tumours generated by this protocol will be basophilic in nature and genotypically either Ha-ras or B-raf mutated. Alternatively, animals at the age of 5–6 weeks can be treated with a single dose of 50–90 mg/kg body weight. If desired it is advisable to administer PB or a similarly acting drug chronically as a tumour promoter. This treatment regimen will produce tumours which are mostly Ctnnb1-mutated. These treatment regimens will form solid tumours in 50–100 days at a high frequency. The body weight of the animals should be regularly measured and documented. Reliable readout systems are the visualization of hepatic tumours by MRI and the demonstration of elevated quantities of AFP in serum. However, the extent of tumour formation should always be histologically proven and documented.
Ethical statement
All experiments were approved by the official State Animal Care and Use Committee (LANUV, Recklinghausen, Germany).
Footnotes
Funding
This work was supported by grants of the German Research Foundation (SFB/TRR57 P04, P13, Q2, and Q3).
