Abstract
Oral dosing by gavage is often used to test compounds in minipigs. This method is also used for certain nutritional studies that require exact dosing. This procedure may be stressful for the animal and requires the assistance of more than one technician. We investigated whether a gastrostomy tube could be placed and maintained in Göttingen minipigs using a single port laparoscopic technique. As part of another study, laparoscopic gastrostomy tube placement was performed in 12 Göttingen minipigs (32 ± 2 kg) under general anesthesia. The procedure involved single port laparoscopic visualization of the stomach and placement of a locking pigtail catheter into the fundus region of the stomach. The minipigs were followed for three weeks after surgery and macroscopic and microscopic tissue reactions were evaluated at necropsy. All catheters were successfully placed and were easy to use. At necropsy it was evident that the catheter had entered the stomach in the fundus region in 11/12 of the animals. In one animal the catheter had entered the antrum region. None of the animals developed leakage or clinically detectable reactions to the gastrostomy tube. Histopathologically, only discrete changes were observed. Single port laparoscopic tube gastrostomy with a locking pigtail catheter is safe, simple and reliable and is an appropriate alternative to, for example, percutaneous endoscopic gastrostomy, when long-term enteral delivery of pharmacological or nutritional compounds is needed. The use of the gastrostomy tube was easy and, based on subjective assessment, feeding was minimally stressful to the animals.
Oral dosing by gavage is often used in pigs when testing new drug candidates for regulatory toxicology but also when performing certain nutritional studies that require repeated, exact dosing or aspiration.1–3 The placement of a gastric tube in a conscious pig can be technically challenging and stressful for the animal, in particular if repeated frequently. Gavage may cause esophageal trauma, accidental dosing into the lung or pharyngeal injuries if the gastric tube is caught in the pharyngeal diverticulum during placing.2,4,5 Consequently, an alternative to gavage is preferred for long-term studies when frequent or continuous intragastric dosing is required.
Nasoesophageal-, esophagostomy- and gastrostomy-tube feeding are the most commonly used techniques in both companion animals and humans when alternatives to oral administration and gavage are needed.5–8 Due to the anatomical appearance of the pig snout, placing nasoesophageal or nasogastric tubes is not possible. However, permanent orogastric tubes with exits through the cheek are easily placed in newborn piglets but are less suitable for extended use in adult pigs as they are technically more difficult to place in grown pigs and are often regurgitated or chewed by the molar teeth.9–11 Instead, permanent gastrostomy tubes are more suitable and favorable for long-term studies in adult pigs.
There are various ways of placing gastrostomy tubes using minimal invasive surgery.8,12,13 Initially endoscopic techniques were developed to replace the original invasive approach of tube placement. One such technique is the percutaneous endoscopic gastrostomy (PEG) technique, which has gained much popularity since it was introduced in 1980. 14 A commonly used method of PEG tube placement is the pull technique. In this technique, a flexible video endoscope is inserted into the stomach and the site of tube placement is determined. A percutaneous applied suture is pulled from the stomach and out through the mouth. The gastrostomy tube is then mounted to the suture and pulled back into the stomach and out through a small skin incision in the abdominal wall. Although generally safe, PEG tube placement may be associated with potential complications such as a risk of inadvertent bowel or organ injury, misplaced/dislodged tubes, intraperitoneal leakage and hemorrhage.8,12,15 Many of the complications reported after PEG tube placement have been attributed to a lack of direct intraperitoneal visualization during placement of the gastric tube. Radiology-guided techniques and non-endoscopic blinded techniques have also been described with varying success and complication rates in humans and companion animals.6,13
Today laparoscopic gastrostomy (LG) tube placement is a technique commonly used in humans as an alternative to PEG; and has been suggested as a safer method, particularly in children, where more severe complications are seen after PEG than LG.7,8,16 This technique provides a means of ensuring safe and correct placement of the gastrostomy tube under direct visualization of the stomach and other abdominal organs. Long-term gastrostomy tubes are usually placed with the intention of creating a permanent fixation of the stomach to the abdominal wall (gastropexia) and a fistula; which makes replacement of tubes possible when needed. This technique requires gastrostomy tubes and fixation devices; which are able to retract and fixate the stomach to the abdominal wall. Tube gastrostomy with gastropexia and fistula formation is primarily intended to prevent leakage. 12 Even though considered a routine procedure, this technique is slightly more demanding and may cause discomfort in some animals, as has been observed in dogs. 17 The use of a simple gastrostomy tube without gastropexia, and with a low-risk of leakage may simplify the LG procedure and reduce the need for more advanced gastrostomy devices. Even though PEG tube placement has proven feasible in minipigs without major complications 18 the laparoscopic technique could provide a useful alternative or an even better way of placing a gastrostomy tube in pigs especially if laparoscopy for other reasons is part of the planned study protocol or if laparoscopic but not endoscopic equipment is available. The use of a gastrostomy tube instead of repeated gavaging reduces stress and discomfort to the animals and improves animal welfare, which is in compliance with the 3Rs (replacement, reduction, refinement).
We thus investigated whether a single port laparoscopy aided technique could be used to position a percutaneous inserted gastrostomy tube in a Göttingen minipig. The function of the tube was evaluated throughout a three-week intervention period, and correct placement and tissue reaction to the tube were determined at necropsy.
Materials and methods
Animals
The animals were treated in accordance with the Animal Experimentation Act of Denmark, which is in accordance with the Council of Europe Convention ETS 123. The study was approved by the National Animal Experimentation Board.
Twelve male Göttingen minipigs (body weight [BW] 32 ± 2 kg) were obtained from Ellegaard Göttingen minipigs A/S (Dalmose, Denmark) for a nutrition study. The animals were allowed two weeks of acclimatization, during which they were in separate pens and fed restrictedly (Altromin 9023; Brogaarden, Gentofte, Denmark) with free access to water.
Perioperative preparation and anesthesia
Two days before surgery only fluid nutrition (Nutridrink; Nutricia Advanced Medical Nutrition, Allerød, Denmark) was given to the minipigs and straw bedding was removed from their pens. Twelve hours prior to surgery they were restricted from all food. Water was provided ad libitum.
On the day of surgery, the pigs were sedated with 0.06 mL/kg BW of ‘swine mixture’ (125 mg tiletamine [0.75 mg/kg] and 125 mg zolazepam [0.75 mg/kg] [Zoletil 50; ChemVet, Silkeborg, Denmark] + 125 mg xylazine [0.75 mg/kg] [Narcoxyl vet 20 mg/mL; Intervet, Ballerup, Denmark] + 125 mg ketamine [0.75 mg/kg] [Ketaminol vet 100 mg/mL; Intervet] + 25 mg butorphanol [0.15 mg/kg] [Torbugesic vet 10 mg/mL; Scanvet, Fredensborg, Denmark]), as previously described. 19 Anesthesia was induced with propofol (1–2 mg/kg BW intravenously) (Rapinovet vet 10 mg/mL; Schering-Plough Animal Health, Ballerup, Denmark). After intubation and placement of an esophageal tube, the pigs were kept on a balanced total intravenous anesthesia (TIVA) with propofol (5–15 mg/kg BW/h) and fentanyl (10–30 µg/kg BW/h) (Haldid; Janssen-Cilag A/S, Birkerød, Denmark). The animals were ventilated mechanically with 60% oxygen with a tidal volume of 10 mL/kg BW and a respiratory frequency of 10/min, in order to maintain an end tidal CO2 of 45–60 mmHg.
Perioperative antibiotic prophylaxis (20 mg/kg) (Cephazolin Fresenius 1 g; Fresenius Kabi, Bad Homburg, Germany) was administrated intravenously at the time of induction and at the end of procedure. Heart rate, non-invasive mean arterial pressure (MAP), oxygen saturation by pulse oximetry (% SpO2), partial pressure of carbon dioxide (PaCO2), and electrocardiography (ECG) were continuously monitored. Fluid infusion (Ringer Acetate; Fresenius Kabi, Uppsala, Sweden) was given at 10 mL/kg BW/h. If the animals developed bradycardia during the procedure, here defined as a heart rate of below 40 beats per minute, a bolus of atropine (0.005 mg/kg BW) (Atropine DAK 1 mg/mL; Nycomed, Roskilde, Denmark) was given.
Surgical procedure
Prior to the placing of the gastrostomy tube, two central venous catheters (CVC) (Silastic silicone tubes, 0.040 in. internal diameter × 0.085 in. outer diameter; Dow Corning, Seneffe, Belgium) were implanted in the external jugular vein after surgical cut down, as described elsewhere.20,21 The pigs were then placed in left lateral recumbency and the right flank extending from the costal arch to the tuber coxae was shaved and prepared for surgery. Ventrally in the flank a local analgesic abdominal wall block (a mixture of Xylocain 20 mg/mL + Marcain 5 mg/mL; AstraZeneca A/S, Albertslund, Denmark) was administered before the incision. A 2 cm skin incision was made and a 5 mm laparoscopic trocar (Endopath XCEL, Ethicon Endo-Surgery; Johnson & Johnson AB, Birkerød, Denmark) was placed. Intraperitoneal position was confirmed visually before pneumoperitoneum was established with CO2 insufflation (Karl Storz insufflator; Karl Storz Co, Culver City, CA, USA). CO2 flow was 0.6 L/min to reach intra-abdominal pressure at approximately 10 mmHg. The peritoneal cavity was inspected and the stomach was located with a 5 mm, 30° laparoscope (Karl Storz Endoscopy, Holte, Denmark). The entry of the gastrostomy tube through the abdominal wall was determined and after making a local analgesic wall block a small skin incision was made. A 10.2 Fr locking pigtail catheter (Multipurpose Drainage Catheter; Cook Medical Interventional Radiology, UK) was inserted into the abdomen just caudal to the costal arch under laparoscopic guidance. The stomach was manually inflated with a self-inflating rubber bulb connected to the esophageal tube until sufficient gastric tension was achieved. The pigtail catheter was then positioned into the body of the inflated stomach and inserted through the parietal wall of the inflated stomach, in a stabbing motion. The position of the catheter was thoroughly evaluated and the pigtail-tip was locked and secured. The stomach was decompressed and left in a position that prevented tension on the gastrostomy tube. The laparoscope was removed and the gastrostomy tube was anchored to the skin with a Chinese finger trap suture at the base of the catheter. A tension-free position left approximately half of the gastrostomy tube extracorporeal. The catheter was further sutured to the skin along the full length using a continuous suture pattern (Dexon 2-0; Syneture, Tyco Healthcare, Norwalk, CT, USA). The fentanyl infusion was stopped as soon as the pigtail catheter was in place and the propofol was discontinued once the trocar was removed.
Post-surgical management
Immediately after surgery the animals were given 0.4 mg/kg Metacam intramuscularly (Boehringer Ingelheim Danmark A/S, Copenhagen, Denmark). The following three days the pigs were given a daily oral suspension of Metacam (Metacam 1.5 mg/mL; Boehringer Ingelheim Danmark A/S) through the gastrostomy tube. In the case of fever, amoxicillin (Clamoxyl Prolongatum; Orion Pharma Animal Health A/S, Nivå, Denmark) was administered intramuscularly at a dose of 1 mL/10 kg BW. The animals were allowed to eat standard minipig chow diet (Altromin 9023; Brogaarden, Gentofte, Denmark) as soon as they recovered from the anesthesia. Maintenance of the gastrostomy tube was done by daily flushing of the tube with small volumes of tempered water. At the initiation of the nutrition study the animals were randomly allocated to receive either total parenteral nutrition (Kabiven; Fresenius Kabi, Bad Homburg, Germany) through the CVC (n = 6) or liquid enteral nutrition (Fresubin; Fresenius Kabi, Bad Homburg, Germany) through the gastrostomy tube (n = 6). The animals were monitored several times during the day and any changes related to clinical status or behavior suggesting pain or discomfort were noted.
Post-mortem examination
After three weeks, the pigs were euthanized with a lethal dose of pentobarbital (200 mg/kg BW) (Pentobarbital 200 mg/mL; Copenhagen University Pharmacy, Copenhagen, Denmark). The position of the gastrostomy tube was evaluated, and any signs of tube-related tissue reaction were noted. Tissue surrounding the tube was collected for histopathological evaluation. After 48 h in 4% formalin, the tissue samples were anonymized; routinely processed and embedded in paraffin. Samples were cut into 2–4 µm serial cross-sections for routine histology (hematoxylin and eosin (HE) staining). Histological evaluations were performed by two observers.
Statistical analysis
Data are reported as means ± standard deviation (SD). To compare the effect on hemodynamic parameters before and after insufflation, a one-way analysis of variance (ANOVA) was used. Statistical analyses were conducted using Graphpad Prism 5 (Graphpad Software Inc, La Jolla, CA, USA), and a P value <0.05 was considered significant.
Results
In all minipigs, the gastrostomy tube was successfully placed (Figure 1) and the use of the gastrostomy tube was easy. Based on a subjective assessment of the behavior of the minipigs, feeding was only minimally stressful. The duration of the laparoscopic procedure varied between 20 and 30 min in total. In one animal, the catheter perforated both sides of the gastric wall after initial insertion. This pigtail catheter was repositioned immediately and only minimal bleeding was observed. Two days after surgery, one pigtail catheter was occluded by gastric content, and the animal had to be re-anesthetized. The original pigtail catheter was removed and a new catheter was positioned without any problems. The remaining catheters were fully functional throughout the study and no differences in function of the tube were observed between the animals that were given daily nutritional infusions through the tube and the animals fed parenteral nutrition.
Representative images of (a) the gastric tube connected to the swivel line, (b) the entry point of the gastric tube at the skin surface, (c) the course of the pigtail catheter within the peritoneal cavity, and (d) the position of the gastrostomy tube in the fundus region of the stomach.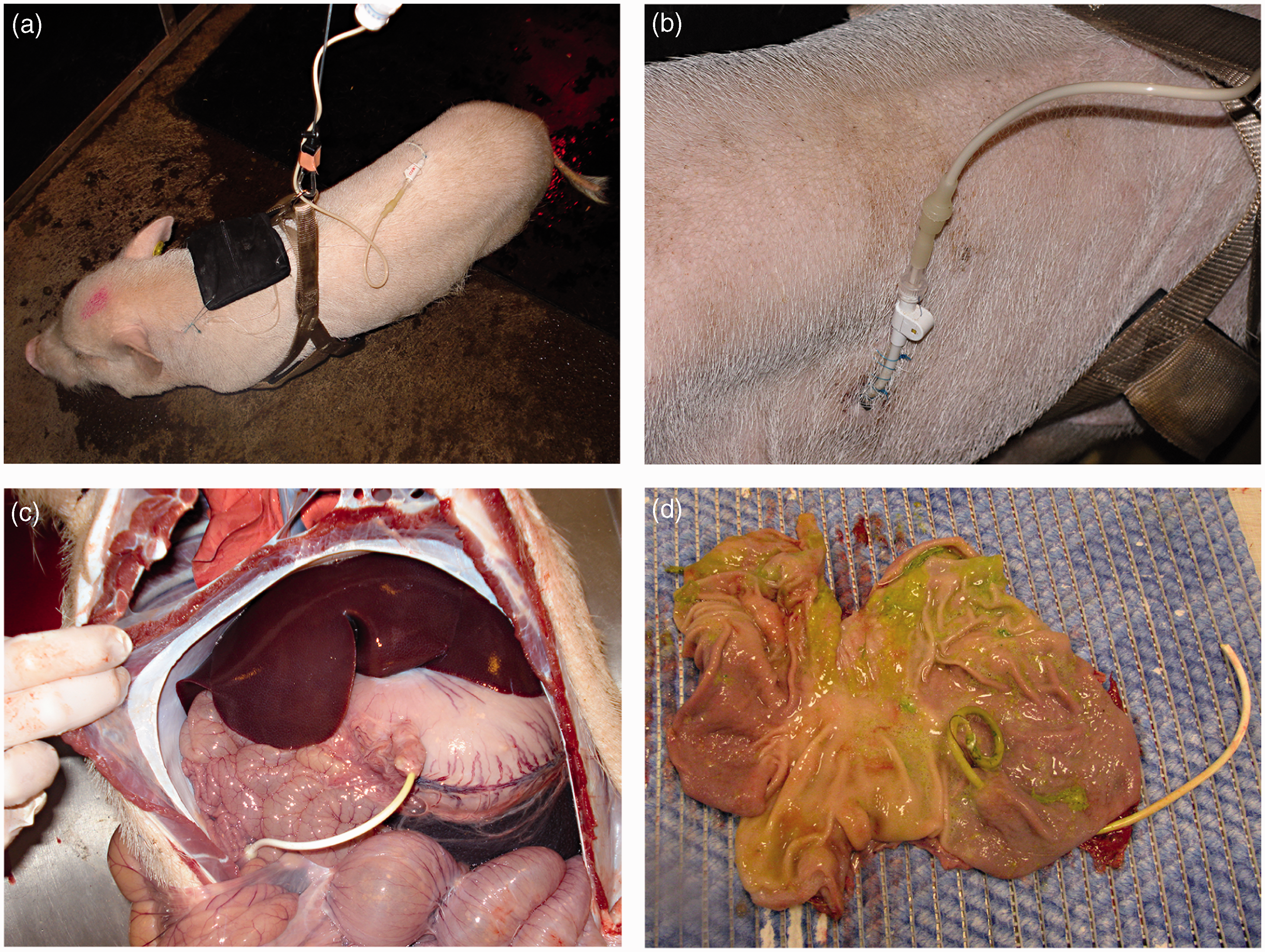
Neither acute intraoperative nor postoperative complications related to the pigtail catheters were observed. None of the animals showed signs of ileus or bowel strangulation at any time. Two pigs developed temporary diarrhea once they were put on liquid enteral tube feeding, and in both cases this was resolved within a day. In the pig where the tube was repositioned the antibiotic and analgesic treatment was restarted after the second procedure. It was not necessary to supplement any of the pigs with additional analgesics other than stated in the protocol. A few hours after surgery one animal developed a fever. Additional antibiotics were given and the pig fully recovered on the following day. All pigs accepted being connected to a swivel line for 10–14 h per day during the nutritional infusions (Figure 1a). The pigtail catheter remained in place and the suturing of the catheter to the skin worked well in keeping the catheter in place (Figure 1b).
Necropsy findings
All gastrostomy tubes were covered with a well-developed peritoneal lining between the stomach and the abdominal wall (Figure 1c). No signs of leakage, fistulation or hemorrhage were observed. In 11 pigs, the pigtail catheter was inserted into the fundus region (Figure 1d) and in one pig into the antrum region of the stomach. In one pig a small abscess (2 × 2 cm) was found in the abdominal muscular layer adjacent to the entry of the tube.
Histology
Histological evaluation revealed minor pathological changes in relation to the tube. Evident implantation sites were characterized by a discrete local chronic-active inflammation intermixed with mild to moderate numbers of neutrophils and eosinophils (Figure 2). A few foreign-body-type multinucleated giant cells containing numerous haphazardly arranged nuclei throughout the cytoplasm were also present. No inflammatory or neoplastic cells were found in the adjacent tissue. The inflammation was primarily seen in the muscular layers and only a few changes could be seen in the mucosa.
Local chronic-active inflammation in the gastric wall at the entry point of the gastrostomy tube (*). Macrophages, neutrophils and eosinophils present near the implantation site. Hematoxylin and eosin (HE) staining. Scale bar = 200 µm in (a) and 50 µm in (b).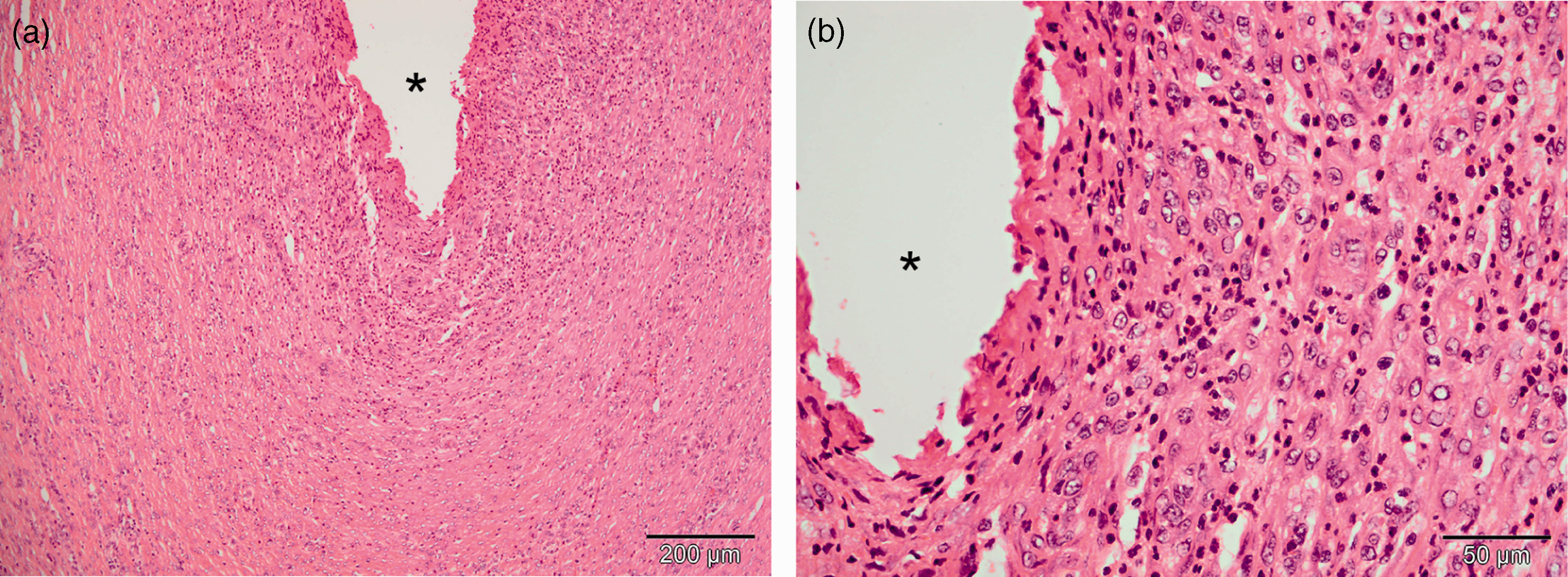
Hemodynamic parameters
Heart rate decreased significantly when the abdomen was inflated (Figure 3a), and did not increase after the abdominal pressure was released. The decreased heart rate did not, however, influence MAP, which was constant through the laparoscopic procedure (Figure 3b).
Hemodynamic parameters. (a) heart rate, (b) mean arterial pressure (MAP), (c) oxygen saturation, and (d) end tidal CO2 before, during and after insufflation of the abdomen. Mean and standard deviation is indicated. *P < 0.05.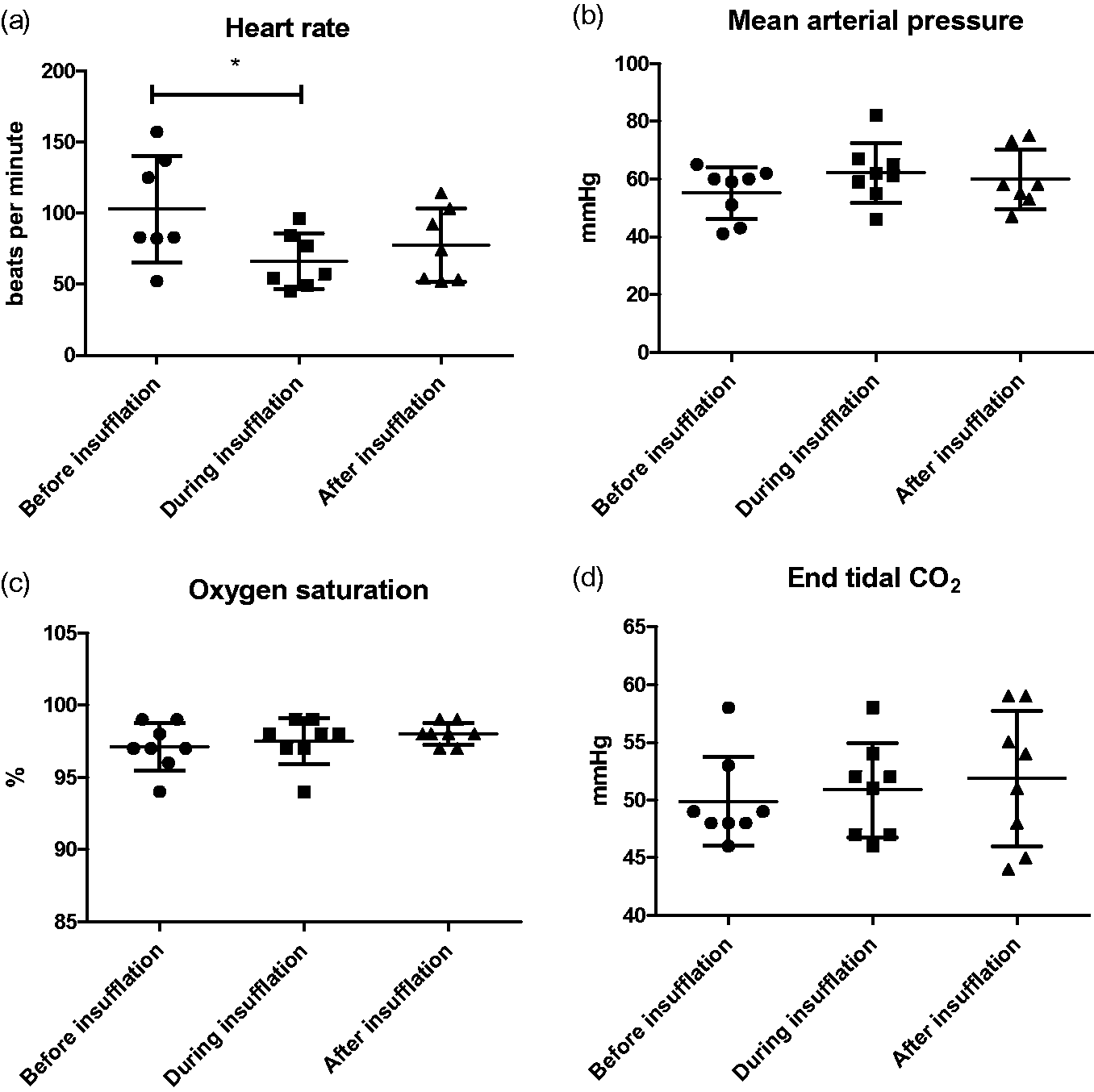
No significant change was noted in oxygen saturation measured before, during or after insufflation of the abdomen (Figure 3c) and this was stable at approximately 97 ± 0.5%. End tidal CO2 did not change significantly in relation to the insufflation (Figure 3d). One pig developed bradycardia after the TIVA was finalized and was given atropine.
Discussion
The described single port laparoscopy aided percutaneous gastrostomy technique is safe, simple and is also reliable for long-term use. The procedure is easy to learn even with minimal laparoscopic experience, and is relatively quick to perform. It is possible to perform PEG in minipigs, but if an endoscopic equipment is not available the laparoscopic approach can be an attractive alternative. However the procedure requires proper anesthetic maintenance with a ventilator where it is possible to set an adequate positive end-expiratory pressure (PEEP), to avoid pulmonary compression during the laparoscopic procedure. The single port laparoscopy without the use of laparoscopic instruments prohibited manipulation of the stomach. However, inflation of small amounts of air into the stomach and direct visualization made it possible to insert the gastrostomy tube without additional instruments. A temporary pulse increase was observed when the stomach was inflated (data not shown), but this was quickly resolved once the tube was in place and the pressure was relieved; however this underlines the necessity for adequate analgesia. The lack of instruments for manipulation, though, makes positioning of the gastrostomy tube slightly less precise. In the present study it was not necessary to convert to open surgery in any of the cases, and the sole case of replacement due to catheter occlusion was uncomplicated. Obstruction is indeed a very common complication, with obstruction incidences as high as 45% in patients with PEG. 21 Consequently, proper maintenance of the tube is important. The cases of temporary diarrhea were likely due to the dramatic change in feed rather than the intragastric administration, and post-tube gastrostomy diarrhea is considered a common complication. 12 In all cases, the tube was completely peritonealized after the three weeks, indicating proper healing following the procedure. No signs of macroscopic pathological reaction to the tube were observed at necropsy, and the histopathological reaction seen in the gastric wall was strictly located around the entry site of the tube. The chronic mild inflammatory reaction found in the gastric wall was comparable to similar foreign body reactions described in pigs. 19
Using this approach we were able to completely abolish the problems with organ perforations that have previously been reported when using the ELD tube applicator technique in pigs, which is a blind percutaneous gastrostomy technique. 3 Further, no signs of post-operative pain were observed in the present study, so the simple analgesic protocol was adequate. Feeding through the tube was possible without animal restraint, although in some studies where continuous intragastric dosing is required it may be necessary to keep the animals on a swivel system. Also, pigs should be housed individually to avoid pen mates biting the tube or causing injury to the skin wounds. In this study, a few pigs scratched the tube against the walls of the pen repeatedly. This could be a behavioral response to tight sutures in the skin. However, no tubes were displaced because of this and no self-inflicted injuries to the skin were observed.
Another important issue to consider is hemodynamic stability. It is always a concern when conducting laparoscopic procedures that the increased intra-abdominal pressure may lead to hemodynamic instability and compromised respiratory function. This can lead to difficult anesthetic maintenance and poor organ perfusion. It has previously been shown that piglets can tolerate an intra-abdominal pressure, using CO2 insufflation, of up to 20 mmHg, without showing any signs of hemodynamic compromise. 22 In the current study, with an intra-abdominal pressure of 10 mmHg, no signs of hemodynamic instability were observed. Hence the cardiovascular consequences of the procedure in this study were clinically insignificant.
We have shown that the LG procedure can be an attractive and perhaps safer alternative to PEG in pigs. In children, it has been argued that the LG procedure should be used instead of PEG, as in general the complications seem to be more severe with PEG than with LG and because conversion to open surgery is more frequently needed for PEG.7,8,16 We conclude that single port laparoscopy aided percutaneous gastrostomy with a locking pigtail catheter and without gastropexia is safe, simple and reliable, and an appropriate alternative to PEG when frequent or continuous intragastric dosing is required in long-term pig studies.
Footnotes
Acknowledgements
We would like to thank the technicians at the Laboratory Animal facility, Frederiksberg Campus, Faculty of Health and Medical Sciences, University of Copenhagen for taking excellent care of the animals. Fresenius Kabi is thanked for kindly donating the nutritional products. Fresenius Kabi did not have any influence on design or conduction of the study.
Declaration of conflicting interests
None.
