Abstract
A new application of a device enabling the long-term enteral administration of drugs or nutritional supplementation was developed for implementing in research entailing the use of macaques (Macaca fascicularis). After implanting a subcutaneous port, a surgically-placed gastrostomy (SPG) was completed to afford access to the gastric lumen and enable the administration of substances. In this study, the device was left in place for a period ranging between two and 12 months in macaques (n= 16). In five cases, the SPG was used successfully for 8–12 months, until the experimental endpoint was reached. In six cases, the SPG had to be removed earlier due to local infection at the implant site, which promptly regressed after the SPG was removed and antibiotic treatment was administered. One SPG-implanted macaque was euthanized for reasons unrelated to the SPG or the xenotransplantation procedure. In four cases, the SPG was implanted without any complications but has yet to be used to administer substances to the animals. From an ethical standpoint, the SPG device described here minimizes the forced handling of macaques otherwise needed for the oral administration of viscous or unpalatable substances by gavage. The device thus represents an effective refinement that fully complies with the tenet of the ‘3 Rs’ that should be considered by primate centres exposing non-human primates to the long-term daily administration of substances by oral gavage.
In experimental research entailing the use of animals, the tenet of the ‘3 Rs’ 1 is now diligently applied by the scientific community and incorporated in an increasing number of legal frameworks. The European Parliament has recently issued a Directive on the protection of animals used for scientific purposes that aims to harmonize experimental standards across the European Union (Directive no. 2010/63/EU). This document principally underlines the importance of refinements, i.e. the use of methods to ensure that all possible discomfort and pain for the animals is avoided or reduced to a minimum, and that their wellbeing is enhanced by improving their care, treatment and living conditions, taking their lifetime experience into consideration. On the basis of the above assumption, refining an experiment means not only reducing any harm to the animals but also improving their living conditions throughout the experimental procedures, wherever possible. 2
Animals unable to take necessary nutrients orally as a consequence of Parkinson's disease (PD) or similar debilitating neurological disorders require alternative forms of nutritional support to prevent or correct calorie and nutrient deficiencies. When it comes to drugs, enteral administration is associated with a markedly greater interindividual variability in drug bioavailability by comparison with parenteral administration, but it has the advantage of reducing the risk of anaphylactic shock. Using positive reinforcement training (PRT), primates can be trained to accept oral administrations of aqueous solutions from a syringe. The volume of the solution is generally limited and it is often necessary to make the solution more palatable. 3 Alternatively, the enteral administration of substances to non-human primates can be based on the use of nasogastric/orogastric tubes, but these approaches require repeatedly restraining the animals manually. 4 Enteral feeding can also be achieved by means of a gastrostomy or jejunostomy, i.e. a tube inserted permanently in the stomach or jejunum via an open surgical procedure and passed subcutaneously and to the outside through the skin. As an alternative, a percutaneous endoscopic gastrostomy tube can be inserted in the stomach. 5 In all such cases, the tube exit site must be covered with a jacket and the tube secured in a pocket for easy access, minimizing the risk of self-inflicted injury. Some discomfort is generally experienced by the animals involved, however. The surgical implantation of a vascular access port in primates has also been reported as a means for the long-term dosing of drugs, 6,7 and a similar approach has been proposed for enteral nutrition in humans too. 8
Important advances in non-human primate models often enable a rapid progression to clinical practice, making such models the most appropriate for translational research. In this setting, cell transplantation has great potential as a solution for the treatment of terminal organ failures, such as those responsible for type 1 diabetes and PD. 9–11 It has been demonstrated in preclinical studies, however, that the prolonged administration of pharmacological immunosuppression is often needed for long-term graft survival.
Our group recently developed a neural xenotransplantation model in which neural precursors obtained from porcine fetuses 12 are transplanted into immunosuppressed non-human primates with pharmacologically induced PD. 13 The model involves repeated handling of the animals every day, resulting in their unavoidable discomfort. 4
To optimize the animals’ handling and minimize the drawbacks of the technique described, we developed a new refinement, identifying a novel approach to the animals, based on the use of a surgically-placed gastrostomy (SPG) that enables long-term drug administration and/or nutritional supplementation with a reduced impact on animal welfare.
Materials and methods
In this experimental model, PD was first induced in primates with a specific compound, the neurotoxin 1-methyl 4-phenyl 1,2,3,6-tetrahydropyridine (MPTP) 0.25 mg/kg (Sigma-Aldrich St Louis, MO, USA), 14 then the animals underwent transplantation with porcine neurons. They were subsequently treated and monitored for a lengthy period of time after surgery to see whether the transplanted neural precursors matured in the primates’ brain and improved their global motor activity. This model involved prolonged oral treatments for the animals, consisting of the twice-daily administration of immunosuppressive, antimicrobial and symptomatic drugs, in addition to nutritional supplementation where necessary.
Animals
Sixteen 5–9-year-old, purpose-bred female cynomolgus monkeys (Macaca fascicularis) weighing between 2.6 and 4.4 kg were implanted with SPG. All the primates used by CORIT were purpose-bred and supplied by an authorized international company (Siconbrec Ltd, Makati City, Philippines). For each primate, details were recorded relating to the country of origin and supplier, quarantine dates, virological and bacteriological screening and any treatment administered during the conditioning period, in accordance with the Federation of European Laboratory Animal Science Associations (FELASA) recommendations. 15 Only primates in good health and negative on testing for tuberculosis and undesirable viruses (Ebola, Marburg, Herpes B, Simian T-cell lymphotropic virus, Simian immunodeficiency virus, Simian retrovirus, Hepatitis B) were accepted at the CORIT Primatology Centre. 16
On arrival, the primates (body weight 2–4 kg) were housed for the first few weeks in two galvanized wire gang-cages (120 × 320 × 200 cm high) designed specifically to contain up to 10 animals each in clean and secure conditions. A nylon net cage (200 × 400 × 250 cm high) connected by a tunnel to the wire gang-cages, was made available inside the facility as a playroom (various toys were provided for environmental enrichment), where the primates could play, jump and eat for 2–4 h/day, all together or in small groups. Depending on the type of project conducted at our centre (renal or neuron transplantation), a few days prior to surgery the animals were paired and moved to stainless-steel squeeze-back metabolic cages (69 × 104 × 90 cm high) to facilitate observation of their behaviour and the measurement of their food and fluid intake, and urine output. All the cages containing the paired animals were located in the same room to enable visual, auditory and olfactory contact among the different pairs of animals. After positioning the SPG device, the primates were housed for 6–8 days in individual cages to protect the suture. Then the animals were periodically or permanently moved to gang-cages (120 × 320 × 200 cm high) connected by a tunnel to a nylon net cage (200 × 400 × 250 cm high) where they could stay together with other primates because the animals fitted with SPG devices did not require special measures.
At the same time, measures were taken to accustom the primates to being handled by the technicians and to the procedure of dosing through the SPG. Indeed, primates were accustomed to being handled having become used to the husbandry procedures. In particular, they were handled regularly to accustom them to contact with humans and enable their regular clinical examination. In our experience, this approach made subsequent handling and restraint procedures less stressful for the animals.
The Parkinson model
Twelve of the animals with SPGs were induced with PD and underwent a transplantation procedure.
The experiments were conducted in accordance with the Italian Animals Act (Law No. 116 of 27 January 1992) and authorized by a specific Decree of the Italian Ministry of Health. The research project was also approved by our Institutional Animal Care and Use Committee.
Xenotransplantation
Following the onset of a stable lesion, striatal neurons from engineered pig embryos were unilaterally injected into two sites of the left putamen using stereotaxic-guided intervention. Starting two days prior to transplantation, the non-human primates were exposed to a clinically applicable immunosuppressive regimen consisting of cyclosporin A (Neoral; Novartis Pharma AG, Basel, Switzerland) administered orally twice a day, adjusted individually to maintain a 16 h blood trough concentration of 300–600 ng/mL, and mycophenolate sodium (Myfortics; Novartis Pharma AG) administered orally twice a day, adjusted individually to maintain a 16 h blood trough concentration of mycophenolic acid between 3 and 6 µg/mL. 17 In addition, ondansetron (Zofran®, GlaxoSmithKline, Verona, Italy; 0.3 mg/kg daily subcutaneously) was administered as an anti-emetic drug for 15 days after surgery, and lansoprazole (Lansoprazolo Sandoz®; Sandoz SpA, Origgio, Varese, Italy; 1 mg/kg twice a day orally) for gastric protection was administered throughout the postoperative period.
The clinical course of the PD was monitored by implementing a previously reported clinical scoring system, used daily.
18
In particular, four parameters were scored (from 0 to 3) based on the animal's posture and degree of tremor, akinesia and dystonia. Video-recording sessions were also performed every two weeks. In addition, positron emission tomography (PET) studies with 18F-
Application of the SPG system at the CORIT facilities
For each of the 16 animals receiving SPGs, the table shows the weight (before and after SPG implantation) or current weight, the duration of the postoperative follow-up, the number of oral gavage sessions avoided and the total number of port injections performed
*Before implanting the SPG system and after its removal (or current weight)
†Based on the xenotransplantation study design
‡Due to local infection
§Ongoing experiment
**Prior to use of SPG
††Requirement for a viscous high-calorie diet
‡‡Animal acutely unwell due to MPTP treatment; SPG not yet used
n.a.: not applicable; SPG: surgically-placed gastrostomy; MPTP: 1-methyl 4-phenyl 1,2,3,6-tetrahydropyridine
The SPG device
The device
An X-Port* with attachable 8 Fr Groshong® catheter (BARD Access Systems, Rome, Italy) was adopted. The SPG device was originally a totally implantable vascular device; it consisted of two main components, i.e. an injection port with a self-sealing silicone septum (base 22.6 mm × 28.2 mm, internal volume 0.6 mL, 2000 punctures maximum with a 22 gauge needle) and a radio-opaque silicon catheter (50 cm long, internal diameter 1.5 mm, volume 0.6 mL). In particular, a Groshong valve, positioned at the end of the catheter, helped to ensure against gastric juice reflux into the port/catheter system (Figure 1). All materials were biocompatible. Only non-coring needles were used (straight type, 22 gauge, 2.5 cm long, BARD Access Systems) to puncture the silicone septum of the port, minimizing the risk of damaging it.
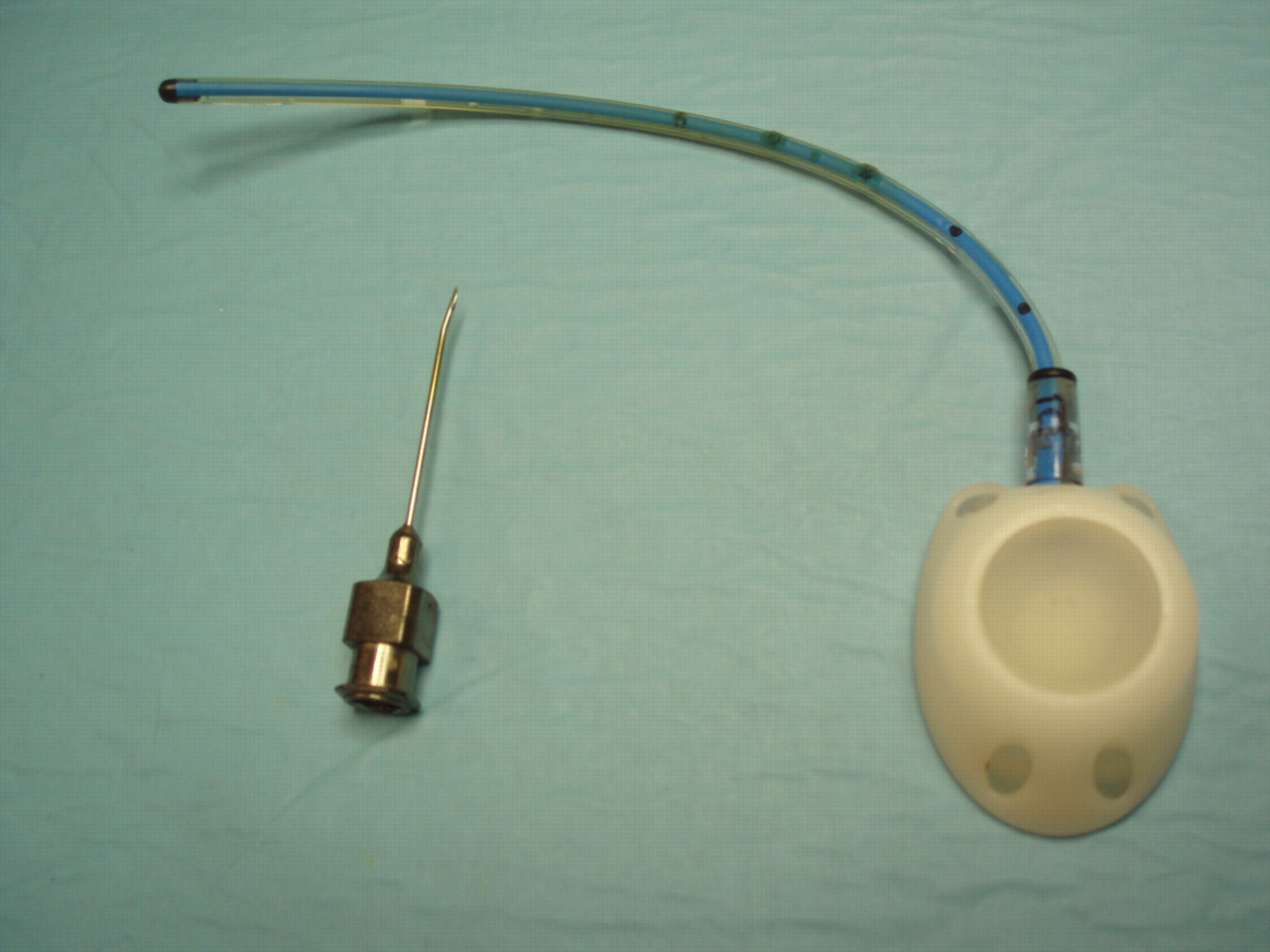
The injection port connected to the catheter, and a non-coring needle
Positioning of the device
Anaesthesia
The SPG was only placed in 16 animals scheduled for PD studies. All animals were premedicated in the squeeze cage with ketamine 10 mg/kg and buprenorphine 0.01 mg/kg (Temgesic®; Schering-Plough SpA, Milan, Italy) mixed in the same syringe and injected intramuscularly. An intravenous catheter was then placed in the saphenous vein and anaesthesia was induced intravenously with midazolam 0.3 mg/kg (Ipnovel®; Roche, Milan, Italy) and propofol (Propovet®; Esteve SpA, Milan, Italy) to effect (mean ± standard deviation [SD] 2.69 ± 1.29 mg/kg). After tracheal intubation, the animal was connected to the anaesthetic machine and a surgical anaesthesia plan was maintained with isoflurane (Isoflo®; Abbott, Latina, Italy) (mean end tidal isoflurane% ± SD 0.93 ± 0.2) in oxygen (100%) through a non-rebreathing system (Jackson Rees T-piece with an adjustable pressure limiting [APL] valve [GPC Medical Ltd, Vikaspuri, New Delhi, India]). Physiological parameters (heart rate, electrocardiogram, respiratory rate, temperature, non-invasive blood pressure, and inspired and expired gases) were monitored constantly throughout the procedure by a multi-parameter monitor (Compact Critical Care Monitor, Datex-Ohmeda Srl, Milan, Italy). A local anaesthetic drug, bupivacaine 1 mg/kg (Bupisolver with Adrenaline®; Solver Pharma Srl, Naples, Italy) was injected intradermally beside the skin incision before the last suture. At the end of the surgical procedure, isoflurane administration was interrupted and the endotracheal tube was removed when signs of awakening were detectable (increased respiratory rate, swallowing or slight palpebral reflex). Once extubated, the animal was placed in an incubator until fully awake to ensure a warm and calm recovery. In the postoperative period, buprenorphine 0.01 mg/kg was given intramuscularly every 8 h for two days. All anaesthesia procedures were uneventful and the animals recovered completely within 30–40 min.
Surgery
An oral antibiotic treatment was administered before surgery (amoxicillin and clavulanic acid, 45 mg/kg/daily and 6 mg/kg/daily, respectively: Augmentin®; GlaxoSmithKline) and repeated daily for five postoperative days. The surgical procedure needed to insert the device was completed by a clinical team of fully-trained surgeons with specific expertise. The procedure lasted approximately 30 min. Following a midline substernal laparotomy (about 3 cm long), a subcutaneous pouch was fashioned on the upper left side of the abdomen, then in the left anterolateral site of the rib cage where the port was to be placed. The anterior wall of the stomach was identified and exteriorized. Using the Seldinger technique, the catheter was inserted in the gastric lumen through its anterior face, between the gastric body and the antrum, equidistant and 7–8 cm from the lesser and greater gastric curvatures. Then the catheter was anchored to the gastric wall with a purse-string suture (resorbable Vicryl 3/0) and passed through the left muscle layers of the anterior abdominal wall, 1–2 cm from the costal arch and about 2 cm from the midline incision. Pexy between the stomach and the abdominal wall around the catheter exit site was performed with four resorbable Vicryl 3/0 stitches. The catheter was connected to the port, paying attention to keep it straight, with no twisting. The port was then inserted subcutaneously in the thoracic subcutaneous pouch and anchored to the external fascia of the rib cage (resorbable Vicryl 3/0), enabling a stable attachment and good usability when the port needle was used (Figure 2). 8 The port was tested and the midline incision was sutured in a double layer (single suture, resorbable Vicryl 2/0). Using this technique, the gastropexy made it possible to extract the catheter, if necessary, simply by traction, without any risk of gastric leakage. Indeed, the small gastric hole closed rapidly, thanks to abdominal muscle elasticity.
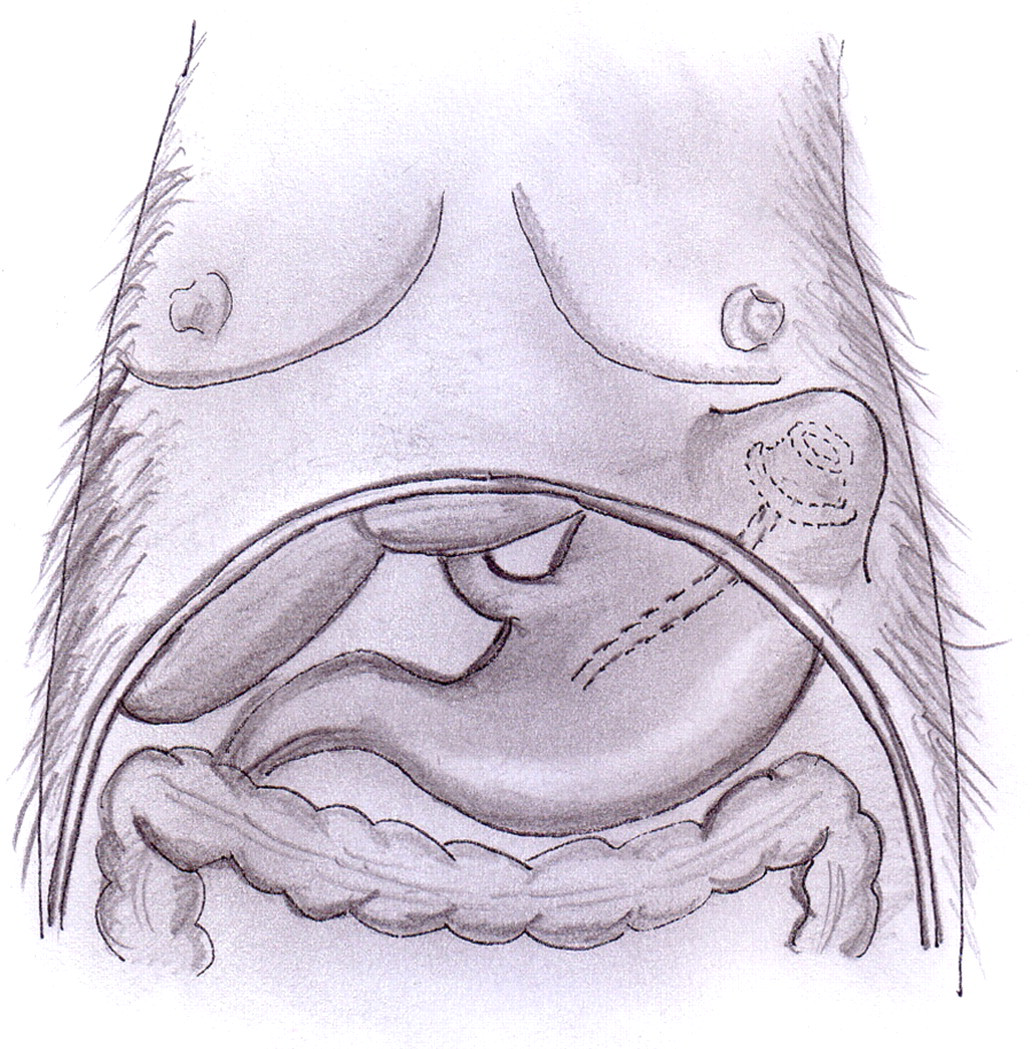
Positioning of the device
All the animals tolerated the subcutaneous implantation well (particularly as regards its anterior position), rapidly resuming their normal diet and regaining their preoperative body weight within 1–2 days.
Removal
In six of the 16 animals, the SPG had to be removed before the experimental endpoint was reached due to local infections. In these cases, a brief surgical procedure, preceded by the administration of ketamine 10 mg/kg intramuscularly, was performed at the port site via a small incision (about 2 cm long), then the port and catheter were extracted by simple traction. Abundant subcutaneous flushing with a mixture of 2 mL iodopovidon solution and 8 mL 0.9% saline solution was performed through the cutaneous incision to favour a speedy healing process. Finally, a cutaneous suture was performed (single suture, resorbable Vicryl 2/0) and an analgesic and antibiotic treatment, identical to the one adopted previously, was administered. The procedure took approximately 15–20 min.
Clinical use of the device
The sutures were removed from day 8 onwards. As early as 48 h following its implantation, the SPG was tested once and then used according to the experimental protocol. Using a sterile technique under strict aseptic conditions, well-trained animal carers performed the procedure twice a day (at 07:30 and at 19:00 h) to administer enteral treatments. When nutritional supplementation was needed in addition to the animal's voluntary food intake, a peptide-based liquid enteral formula was used. In some cases, a higher-energy solution was needed; the high-calorie solution used to satisfy the nutritional needs in these cases was much more viscous and proved incompatible with the diameter of the catheter. These animals were consequently given nutritional supplementation by oral gavage, together with the drugs, and the SPG was temporarily abandoned.
Concerning the handling of the animals, the squeeze-back mechanism was used, i.e. the primates were brought to the front of the cage as gently as possible to keep stress responses to a minimum, while another animal technician disinfected the skin locally with an iodine solution. A non-coring needle was then inserted in the port. Sterile water for injection (5 mL) was administered first to check catheter patency. Then the drugs were injected (i.e. immunosuppressive agents, antimicrobials, gastric protection drugs, cortisone, multivitamin solutions and symptomatic drugs or nutritional supplementation, as necessary). After each use, the access port was flushed with sterile water for injection (20 mL) and the skin was disinfected (with iodopovidon solution) after withdrawing the needle.
Results
To date, 16 non-human primates have undergone SPG implantation at the CORIT facilities.
No surgical complications occurred during the placement of the SPG devices and no obstruction of the system occurred in any of the animals throughout the observation period. None of the animals showed frank skin redness at the needle insertion site, while a slight swelling around the port site was occasionally detectable. When this happened (animals no. 6015B-2065EA), the use of the SPG was temporarily discontinued as a precautionary measure (usually for 3–4 days) and oral antibiotics (amoxicillin and clavulanic acid, 45 mg/kg daily and 6 mg/kg daily, respectively: Augmentin®; GlaxoSmithKline) were administered (for up to 1 week). As shown in Table 1, the device was left in place for a period ranging from 2 to 12 months. Five primates (nos 6805B, 6492E, S6087BK, 7022D and 4495I) received drugs and nutritional supplements as necessary, twice a day, until the experiment came to an end. One of these animals (no. 4495I) could only be treated occasionally via the SPG because its need for a high-calorie diet made it necessary to resort to oral gavage instead of the SPG. No infections at the subcutaneous port were detected in these animals. In four cases (nos 7774A, 7708A, 7568C and 6015B), the port had to be removed after 41, 192, 215 and 233 injections, respectively, due to local, subcutaneous infections with no associated fever or pain on palpation, which promptly regressed after the SPG was removed. These four animals were subsequently given conventional oral treatment (by gavage), which enabled the smooth continuation of the experiments. In two other cases (nos 2065EA and 7919A), a local infection occurred close to the subcutaneous port before it was used and it proved necessary to remove the SPG. One non-human primate (no. B32FE) implanted with an SPG was euthanized for reasons unrelated to the SPG or the xenotransplantation procedure. Finally, the SPG was implanted in four other primates and has yet to be used because the animals have yet to undergo their main surgical procedure.
At necropsy, neither irritation in the stomach mucosa nor lesions in the abdominal organs were detected in SPG recipients, nor were there any signs of fibrosis or encapsulation on serosa where the catheter emerged from the stomach or on the abdominal wall.
Table 1 shows the number of oral gavages scheduled (based on the duration of the experiment) compared with the number of oral gavages ultimately performed. A more than 50% reduction in the number of oral gavage sessions was achieved in six of the eight animals that completed the experiments. These data convincingly demonstrate the validity of our approach.
Discussion
Primate studies represent a fundamental step in the development of novel therapeutic strategies. 13 In this setting, drug efficacy, safety and toxicity studies often imply long-term, repeated oral exposure to the test substance. It is well known that primates can be trained with the aid of PRT techniques 19 and ultimately become willing to come to the front of their cage to accept the administration of small volumes of aqueous solutions. 3 Such an approach is difficult to adopt, however, when viscous preparations, large quantities or unpalatable flavours need to be administered (as is often the case with immunosuppressive drugs). In such cases, oral gavage is almost invariably the only route available for enteral administration. As reported by others, 4,20,21 orogastric intubation is an inherently stressful procedure for primates because, in our experience at least, they are unable to adapt to it. PRT, which is otherwise a very useful method for training animals to cooperate during experimental procedures, does not appear to be applicable to the gavage technique. The problem deserves special attention from an ethical standpoint: the question is which of the available treatment options is the least stressful for the animals involved, while complying in every respect with the experimental protocol. In this context, a more acceptable and less stressful route of administration would greatly improve the general health and welfare of experimental animals.
We have developed a neuron xenotransplantation model that requires the long-term administration of immunosuppressive drugs and often of nutritional supplementation too. The use of the SPG device developed at our centre has proved very helpful and was well tolerated by the animals. For a start, it facilitated the administration of drugs, ensuring the smooth course of the experiment, as well as enabling the administration of nutritional supplements and thereby preventing the substantial weight loss that can occur in animals with Parkinsonism, despite the intensive nursing provided. Indeed, our approach required only gentle pressure on the squeeze-back mechanism to bring the animals to the front of the cage in order to access their SPG. With time, almost all the animals showed signs of becoming used to this procedure, sometimes coming spontaneously to the front of the cage and waiting for the treatment to begin. Secondly, and more importantly, the use of SPG made it possible to optimize our approach to the experimental animals, improving our handling methods and reducing the handling times.
To establish whether a new technique, such as using an SPG for the repeated administration of drugs and nutritional substances in non-human primates, is a genuine refinement of experimental procedures, it is mandatory to undertake an accurate harm/benefit assessment. 22 In our study, the benefits were clearly numerous in terms of improving animal care during experimental procedures. First of all, animal handling and the related induced stress were considerably reduced, since forced handling and gavage were minimized and a more gentle approach could be used. The gentler handling involved was gradually accepted and the animals became used to the procedure and began to cooperate. Indeed, the injection through the port is equivalent to a subcutaneous injection, consisting of a rapidly implemented procedure (less than a minute) that requires no sedation, a measure sometimes needed for oral treatments by gavage in aggressive or nervous animals. In addition, because the animals were not afraid of the procedure, the relaxed approach taken by the trained personnel ensured their less hurried, quieter and gentler handling.
As far as harming the animals is concerned, placing the SPG naturally requires an additional surgical procedure that is not strictly related to the experiment. As already pointed out, however, in the hands of experienced clinical surgeons, the procedure was smooth and rapid, and the animals quickly recovered. When the SPG had to be removed, a new brief surgical procedure was needed at the port site, and both the port and the catheter were removed by simple traction, causing no complications. An additional risk may lie in the potential for local or general infections, but our experience demonstrates that such a risk can be minimized with perioperative antibiotics and careful attention to hygiene to prevent any contamination of the subcutaneous port. While no general infections occurred, there were nonetheless cases of local infection at the injection site (as occurs in the clinical use of such devices in humans), which occurred in 37% (6 out of 16) of our animals. There may be several explanations for this finding: first, the animals were housed under conventional microbial conditions; second, the SPG was used twice a day for several months; and last but not least, all the transplanted animals in the study were pharmacologically immunosuppressed. As for the source of the infection, this is likely to have been at the needle site, since the stomach is normally a sterile cavity in relation to agents responsible for skin infections, and the distal end of the catheter is protected by a Groshong valve that prevents any reflux into the catheter.
The involvement of several animal carers to guarantee regular and safe treatments twice a day, seven days a week, for several months, may at first glance look like an additional risk factor. Conversely, the exclusive involvement of senior personnel specifically trained to deal with the SPG in accordance with standard internal operating procedures does not appear to represent an additional risk factor – at our centre at least.
A further factor that may need to be considered is the need for housing the animals separately while performing the SPG procedure. In general, our transplanted primates were housed in groups, but we isolated them twice a day to perform oral gavage or administer substances through the SPG device, so no additional restrictions in terms of animal housing were adopted for the SPG primates by comparison with animals undergoing oral gavage.
Finally, several PD primates temporarily lost weight during the experiment. This situation is entirely compatible with the weight loss that often accompanies PD in humans 23 and it was unrelated to the SPG implant. On the other hand, the experience we gained of nutritional supplementation enabled us to mitigate this effect in the last four animals undergoing neural transplantation (nos S6087BK, 7022D, 4495I and 7919A).
Taken together, the harm/benefit assessment related to the use of SPG suggests that this new device is valid – as suggested by the large number of injections that we were able to perform successfully and without complications, which resulted in a like number of oral gavage sessions being omitted. Indeed, in two animals we were able to perform more than 340 injections, meaning that we could avoid resorting to oral gavage sessions for a whole year for animals receiving a single daily treatment.
In conclusion, we feel that using the SPG device further improves our approach to experimental animals requiring long-term substance administration. Indeed, our data suggest that a subcutaneously implanted port represents a safe and effective device for primates involved in long-term experiments (lasting several months) and requiring daily oral drug administration, as in the case of transplantation studies or drug efficacy and safety studies. From an ethical standpoint, our experience confirms that the SPG minimizes potentially stressful situations relating to long-term animal treatments. 4 Good animal handling skills reduce the stress for the animals 20 and, in this light, the proposed SPG device is an effective refinement that fully complies with the tenet of the 3 Rs that investigators exposing non-human primates to long-term daily drug administration by oral gavage should bear in mind. This study only represents a preliminary report on a potentially useful technique, however. Indeed, there is scope for further refinement, to reduce the risk of infection for instance. The experience gained on the use of SPG over time at our centre enables us to confirm that this technique has great potential for further development. Finally, we strongly believe that sharing the results of our study can constructively contribute to the ongoing scientific debate on the animal welfare issue. Indeed, we feel it is imperative to circulate information on novel refinement tools in animal research, with the ultimate goal of enhancing the ethical profile of research programmes that entail the use of non-human primates.
Footnotes
ACKNOWLEDGEMENTS
The authors would like to thank Dr Alberto Petrocelli, for his graphic contribution. Special thanks also go to Dr Giovanna Verlato, Dr Arben Dedja, Dr Diana Bertini, Andrea Barzon and Massimo Rizza, whose invaluable contribution and technical support made this study possible.
This work was supported by CORIT (Consortium for Research in Organ Transplantation, Padua, Italy), the Italian Ministry of Health, the Veneto Regional Authorities, and the EU Integrated Project Xenome – no. LSHB-CT-2006-037377.
