Abstract
Poor quality data in preclinical trials can result from inconsistent and unstandardized experimental processes. Unpredictable pre-intervention variability generates unreliable data, biases outcomes and results in needless waste of animals and resources. We applied Define-Measure-Analyse-Improve-Control (DMAIC) quality improvement processes to pilot development of a swine model of trauma, haemorrhagic shock and coagulopathy. The goal was to reduce variability through protocol standardization and error reduction. Six male Sinclair swine were sequentially anesthetized, intubated, mechanically ventilated and instrumented, then subjected to multiple-hit injury, followed by fluid resuscitation monitoring and coagulation testing. Experimental tasks were defined and mapped. Performance measures were task performance times, subject stabilization time and number of task execution errors. Process improvement was assessed by reduced times and errors, and subject stability at target physiological values. Previously-overlooked performance errors and deficiencies were identified. ‘Mistake-proofing’ actions included personnel retraining, revisions of standard operating procedures and use of checklists. The quality improvement pilot trial produced a stable model with reduced protocol deviations. Data quality can be improved and animal waste minimized, if experimental planning incorporates strategies to ensure protocol adherence and reduced operator performance variation and errors. Properly designed pilot trials can be essential components of refinement and reduction strategies in animal-based research.
Introduction
To measure experimental outcomes on animal subjects, invasive instrumentation procedures and other manipulations may be required before the experimental intervention can be tested. However, protocols can be complex, with high potential for deviation and error. Operator training, technical skill sets and competency may vary considerably among staff and learning effects can result in large performance changes over time. The choice of protocol-specific procedures 1 and non-test interventions (e.g. analgesia, anaesthesia)2,3 can profoundly influence both the animal’s baseline physiological status and study outcomes. 4 Systemic flaws in protocol design and execution may not become apparent until after experiments are underway. Trial-and-error changes (‘tweaking’), unplanned deviations to protocols and variation in operator performance can seriously compromise data integrity. Therefore, logistic issues in the pre-intervention phase may have potentially large and unpredictable effects on research outcomes and result in unreliable poor-quality data.
A pilot study consists of a short series of mock runs of the proposed trial, with the objective of informing design and conduct of future primary trials.5–7 Thus, pilot trials represent an important 3Rs refinement and reduction tool (replacement being the third R). Identification and correction of problems in a small-scale pilot greatly reduce risk of experimental failure and subsequent waste of animals. Unfortunately, pilot studies often represent missed opportunities to address problems before a main trial. 8 Often studies are labelled as ‘pilots’ merely because sample sizes are small and inadequately powered.5,8,9 Obtaining effect size data for sample size calculations may be one reason for conducting a pilot. 5 However, a pilot also provides the opportunity to build quality directly into the study design that will result in a more informative (and ethical) trial.10,11 A pilot should have one or more clearly-defined aims: protocol refinement and correction (‘can it work?’), determining primary outcome feasibility (‘does it work?’), determining if the intervention can be made more broadly applicable (‘will it work?’).5–7,12 The process can be made more efficient by targeted quality improvement strategies, such as Define-Measure-Analyse-Improve-Control (DMAIC). DMAIC follows an organized iterative sequence of task mapping, process standardization and problem identification to direct specific operational changes designed to improve performance. 13 Quality improvement methods have been applied successfully to manufacturing, aviation, health care delivery and surgical procedures.14–16 They result in demonstrable improvements in process reliability and quality of end products, and ultimately reduce both costs and waste. 17
We report a pilot study designed specifically to inform development of a swine model of trauma and coagulopathy, used in a subsequent randomized controlled trial. 18 The paper is organized in two parts: (a) the DMAIC template, as specifically applied to this pilot, and (b) the development arc used to determine the finalized protocol. To our knowledge, this is the first study to use DMAIC methods in the context of animal model development. We reasoned that efforts directed to improving ‘upstream’ process quality (namely protocol standardization) would result in more uniform human performance, and consequently reduce ‘downstream’ animal variability.
Methods
Ethical statement
This study was approved in advance by Virginia Commonwealth University Institutional Animal Care and Use Committee (VCU IACUC #AD10001189) and US Department of Defense Animal Care and Use Office (ACURO: USAMRMC Proposal Number 13011003). Animal use and welfare procedures were conducted in accordance with national guidelines (National Research Council. Guide for the Care and Use of Laboratory Animals, Eight Edition. Washington DC: The National Academies Press, 2011, pp. 220) and under the oversight of VCU Division of Animal Resources veterinarians and staff.
Pilot animals
Six male Sinclair swine (Sinclair Bioresources LLC MO; mean delivery weight 25 kg (SD 1.4); 7–8 mo old; health-status certified) were maintained in the institutional holding colony for a minimum of two weeks before surgery. They were housed singly in open-fenced runs with rubber mat floors; ambient conditions were 12:12 L:D and 23℃. Animals were fed commercial swine chow (Teklad Laboratory Mini-swine Diet 7037; Harlan, Madison WI) supplemented with fresh vegetables and water ad lib. Toys and individual free run of the holding room during daily pen cleaning were provided for enrichment. Average weight of pilot animals on the day of surgery was 36.1 kg (SD 3.5).
Animals were habituated to daily gentle handling to minimize stress19,20 and prepare them for pre-surgical sedation. For the first three days, the main handler (PSR) sat down in the pen and allowed the animals to approach. Habituation of the animals to touching was performed with a 1.3 cm x 0.75 m white PVC pipe. The animals were gradually accustomed to allowing the pipe to touch their legs and hindquarters; the pipe was then used as a scratching stick. Animals were positively reinforced with food-treat rewards (the preferred reward was marshmallows). All animals began to solicit scratching and petting within a day, frequently lying down and presenting their belly. The animals were then habituated to having the handler approach them and scratch their hindquarters with gloved hands.
Pilot-study design: Problem formulation
Problem statement and study rationale
The problem statement is a concise and specific description of key issues, needs and potential performance gaps that the study is intended to address (see Supplementary material, Table 1). An in-depth background assessment from comprehensive literature searches and existing data should be performed both to confirm that relevant issues will be addressed and to understand the problem environment.
In this study, a swine polytrauma/shock model was required as a test platform for assessing efficacy of a specified treatment to minimize trauma-induced coagulopathy. 18 Database searches (PUBMED, Web of Science) were conducted at least four times over three months to identify consensus elements of animal coagulopathy models.21,22 However, the team agreed that a pilot was necessary because of substantive methodological differences between studies in experimental methods and ‘coagulopathy’ definitions (such as outcome variables poorly associated with coagulopathy, or failure to demonstrate clinically-relevant changes), thus confounding evaluation and comparison.1,22
Process goals and objectives
Goals are a broad statement of what the project is expected to accomplish in the context of the problem statement. The objectives identify the specific measurable items that must be included to meet study goals.
The goal of this pilot study was to produce a standardized and repeatable animal shock-trauma model (‘can it work?’) resulting in clinically significant and persistent coagulopathy (‘does it work?’). The objectives were to (a) standardize all procedures in the pre-intervention phase; (b) reduce performance variability; (c) reduce animal physiological variability. Operational stability was defined as reduced performance variation, reduced surgical and pre-intervention times and elimination of protocol deviations and procedural errors. Animal stability was defined as individual attainment of pre-specified ‘target’ physiological values.
Experimental design
This pilot study was an observational series of sequentially-performed experiments, each experiment on a single animal. Formal sample size determinations were not performed, because the number of practice sessions required to attain operational stability (and hence the number of animals) could not be determined a priori. However, the pilot sample size of six animals was justified on the basis that six was the maximum number of animals that could be housed at our facility at one time, constituting one experimental ‘block’. Because the surgical technical team was already highly experienced, we anticipated protocol and operational stabilization could be achieved without further use of animals. Animals were randomly assigned to order of surgery by ID number, using a computerized randomization algorithm (available at www.randomization.com).
DMAIC Implementation
DMAIC (Define-Measure-Analyse-Improve-Control) process formalizes identification of tasks to be performed and in what order, identifies checks to ensure tasks are performed correctly and specifies actions to be performed when problems occur.16,23 The overview of the DMAIC process for this study is summarized in Figure 1.
Overview of the Define-Measure-Analyse-Improve-Control (DMAIC) problem-solving process.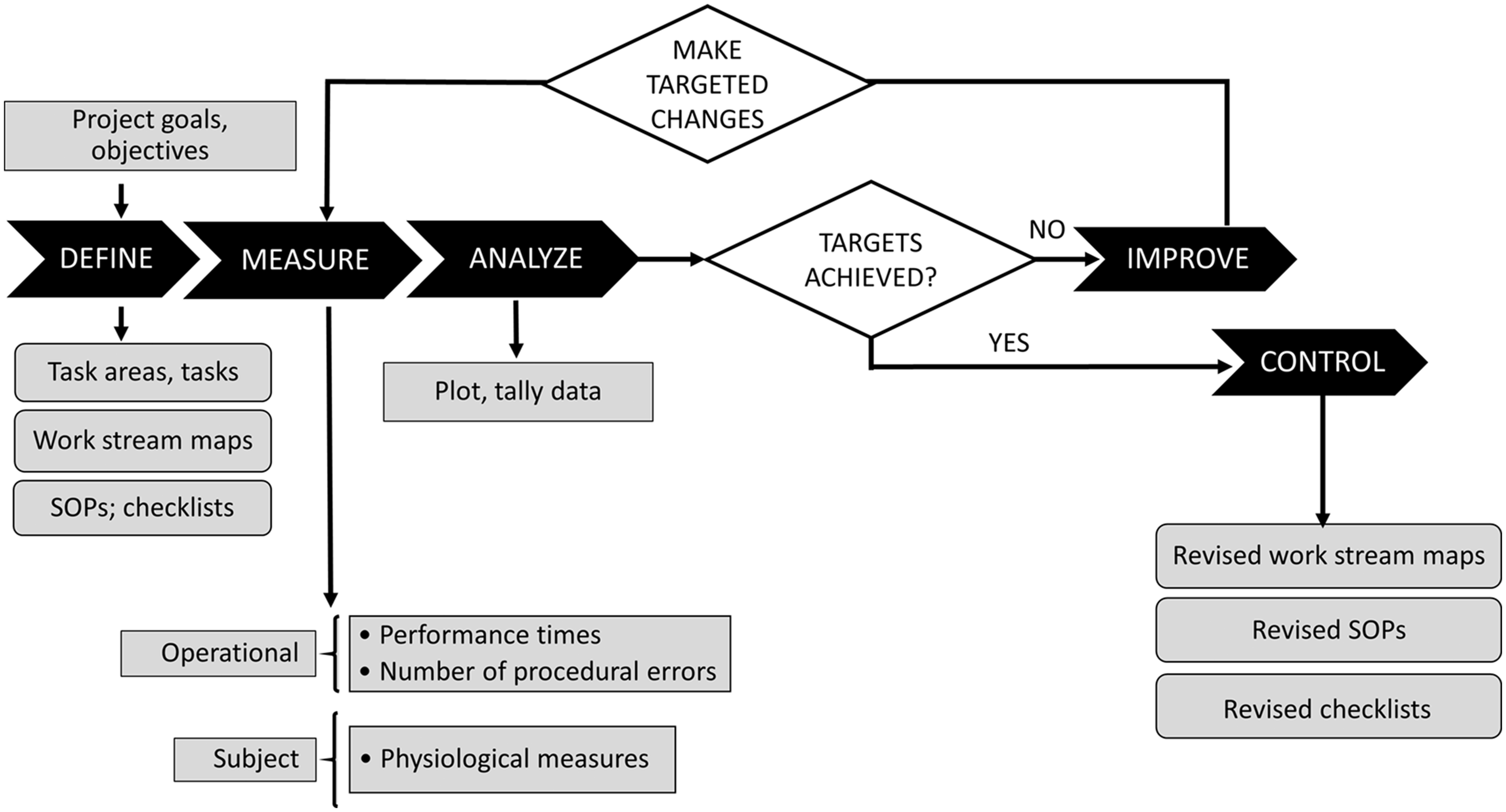
The scope of work is visually captured in a process or work flow map that identifies all required tasks, the order in which they are to be performed, timelines, process boundaries (when to start and stop a process) and deliverables expected from each step. A map makes it easier to understand the current state of the process and identify unnecessary work, problem areas and bottlenecks, and if new tasks are required.24,25 It also streamlines and prioritizes problem solving; for example, targeting upstream rather than downstream problem areas for remediation is often more effective and efficient, because effects of upstream changes will frequently impact processes occurring later in the process chain. 26
The ‘process’ in this study was the entire set of operating procedures for each individual experiment, from the animal’s colony entry to final disposition. Initial planning meetings involved all personnel and focused on consensus identification of tasks, task sub-processes, task sequence, task location, personnel required, data to be obtained, key performance metrics and potential bottlenecks. We used Utstein 27 and ARRIVE 28 guidelines to guide identification and prioritisation of critical tasks: (a) surgical preparation; (b) instrumentation; (c) stabilization; (d) shock-injury induction; (e) resuscitation; (f) post-resuscitation monitoring; (g) euthanasia and tissue harvest. Information was collated in a work stream map. Standard operating procedures (SOPs) for each sub-process were developed and incorporated into laboratory documentation. Checklists 29 for each sub-process were posted on the laboratory walls near the respective work areas.
We assessed two process categories: operational and subject-specific (Figure 1). Operational performance was assessed by time to perform designated tasks, time to subject stabilization and number of critical errors in task execution. Subject stability was assessed by matching of key physiological variables to a priori target values at baseline and at the end of the shock-injury phase (see Supplementary material 2, Study protocol).
Task performance times were defined as the predetermined interval between start and completion of a specific task (see Supplementary material 1, Table 2). We distinguished between ‘set’ task times and ‘targeted’ or ‘untargeted’ performance times. ‘Set’ times were predetermined in the protocol for the standardization of operational phases; these included times for blood sample collection, minimum stabilization time (30 min) and duration of shock-injury (2 hr) and post-resuscitation monitoring (4 hr). ‘Targeted’ performance times were mandated by best-practice requirements. For example, maximum target intubation time was <1 min; gentle but fast intubation is essential to avoid hypoxic complications and damage to the airway. 30 Otherwise, strict time-performance goals for major tasks were not imposed (‘untargeted’). The team was told only to ‘do their best’ and keep in focus the primary objective of achieving a physiologically stable preparation.
Procedural errors (Supplementary material 1, Table 3) were defined as failure to perform a predefined action required for a given task. The target goal was zero errors. Errors were categorized (1 = observed, 0 = not observed) with respect to three task areas: airway management tasks (intubation, oxygenation and ventilation), general procedural tasks and physiological monitoring tasks. Protocol troubleshooting and adjustments were concentrated on these areas.
All performance times were entered on the surgical run sheet for each animal. Errors identified during the experiment were corrected and comments to that effect entered on the surgical run sheet; otherwise deviations were identified after trial completion. Errors were tallied for each trial.
This was a pilot study designed to test methodology of an evolving protocol, not an hypothesis-testing study,31,32 so conventional inferential statistical tests were not appropriate. 6 Performance metrics were assessed by counts and plots − simple descriptive statistics and visual examination of process behaviour charts. 13 Descriptive statistics 6 were calculated in SAS proc means (SAS v.9.4. SAS Inc., Cary NC); summary statistics for angular data 33 were calculated with the SAS macro %circular_moments. 34
The entire team reviewed each experiment to identify problems and brainstorm solutions. Two experienced anaesthesiology clinicians and a veterinarian reviewed and critiqued airway management and ventilation procedures. The coagulation team suggested improvements in sample collection and transfer. Suggested solutions were implemented for the next trial in the series. ‘Mistake-proofing’ actions for the subsequent experiment included documentation of revisions, updating standard operating procedures and checklists, and posting revised checklists and tracking metrics. Explicit reminders and time cues were displayed prominently as colour-coded checklists and on white boards for reference and to minimize reliance on memory.
The pilot study of six experiments was too short for calculation of meaningful process limits (nor was this the objective of the pilot). However, we retrospectively assessed robustness (or ‘sustainability’) of improvement by comparing performance metrics of the six pilot experiments with 14 consecutive experiments in the subsequent primary study (the finalized protocol and results for the completed trial are published elsewhere. 18
Performance was assessed by visual inspection of process charts for task performance times, number of errors and subject stability metrics. Sustainability was assessed by permutation tests on means and standard deviations (measure of process change and variability respectively). 35 Values for the 20 experiments were randomly assigned 1000 times to either the ‘pilot’ (n = 6) or ‘primary trial’ (n = 14) group, the new difference between groups calculated for each random assignment and compared to the original group values. Permutation programmes were written in SAS proc iml and proc sql (SAS v.9.4, SAS Inc., Cary NC). Critical values were estimated from percentiles of the approximate distribution, calculated in SAS proc stdize. (SAS v.9.4. SAS Inc., Cary NC).
Study protocol
Figure 2 shows the finalized task flow map, with approximate times, specific tasks and performance elements for each step. Supplementary material 2, Study protocol describes the decision-making process used to stabilize portions of the protocol where there was no methodological consensus: namely physiological stabilization, iatrogenic injury model and resuscitation.
Work stream map for individual animals. (a) Pre-intervention phase: colony entry and acclimation; pre-surgical preparation; surgical instrumentation. (b) Post-instrumentation phase: stabilization, iatrogenic shock/injury. (c) Experimental phase: resuscitation, post-resuscitation monitoring, euthanasia, tissue harvest.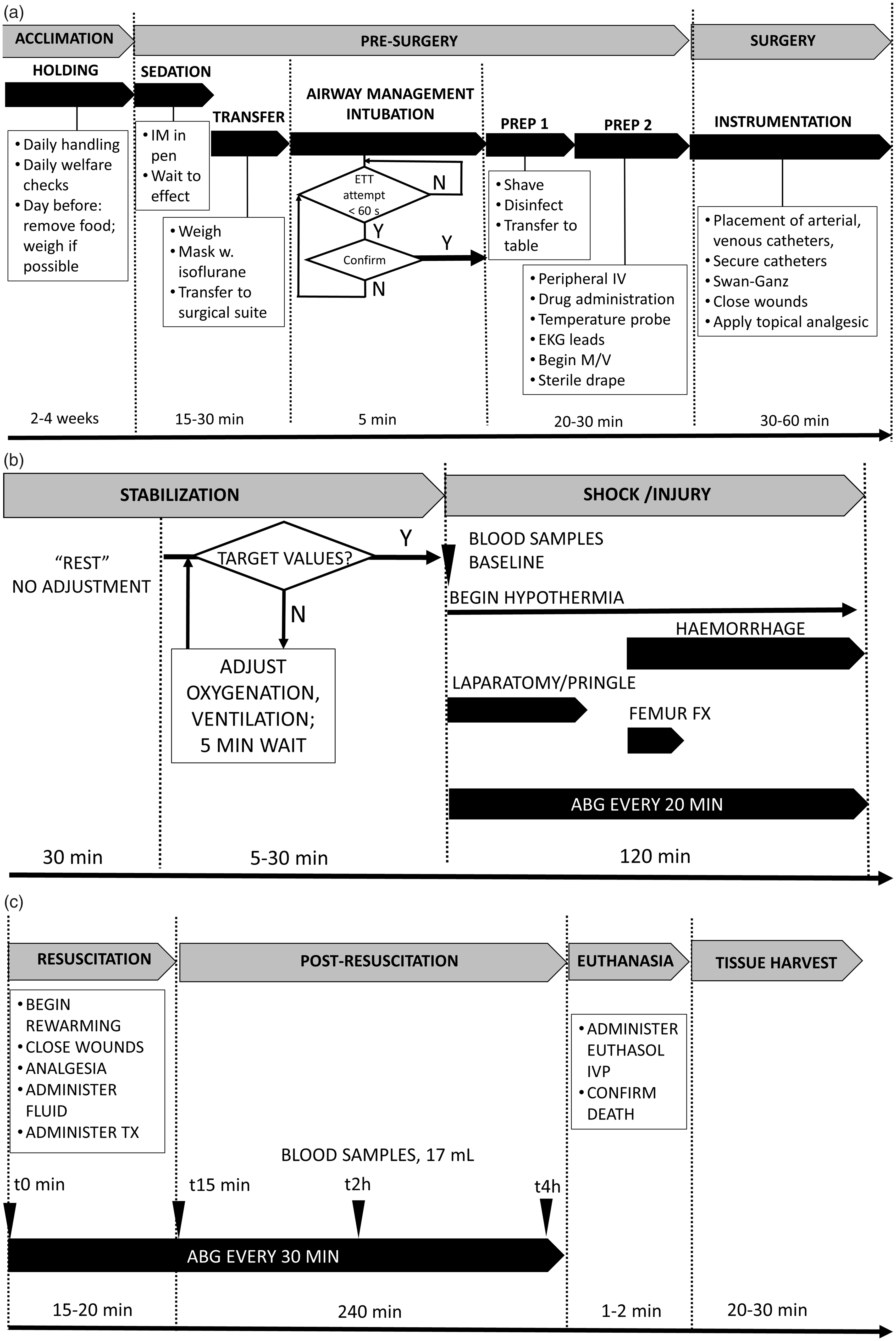
Results
Performance
Times for surgery and catheter placement continued to show modest reductions beyond the end of the pilot series; average reduction in time between the pilot and main study was 23 min (p = 0.03) for total surgery times and 10 min for catheter placement times (p = 0.025) respectively (Figure 3(a) and (b)). In contrast, average time from anaesthesia induction to the first skin incision (Figure 3(c)) showed little change (5 min; p = 0.7). All processes measured showed 5 to 10-fold reductions in variation (p = 0.01) between pilot and primary studies. Fluctuations in ‘incision time’ resulted from differences in times required to thoroughly clean and disinfect surgical sites on each animal (5–25 min; median 10 min), as some animals were cleaner than others.
Sequential performance times for pilot (○) and primary study (•) experiments. (a) Total surgery time; (b) catheter placement time; (c) incision time (Time from anaesthetic induction to first skin incision) (See Supplementary material, Table 1 for complete definitions.)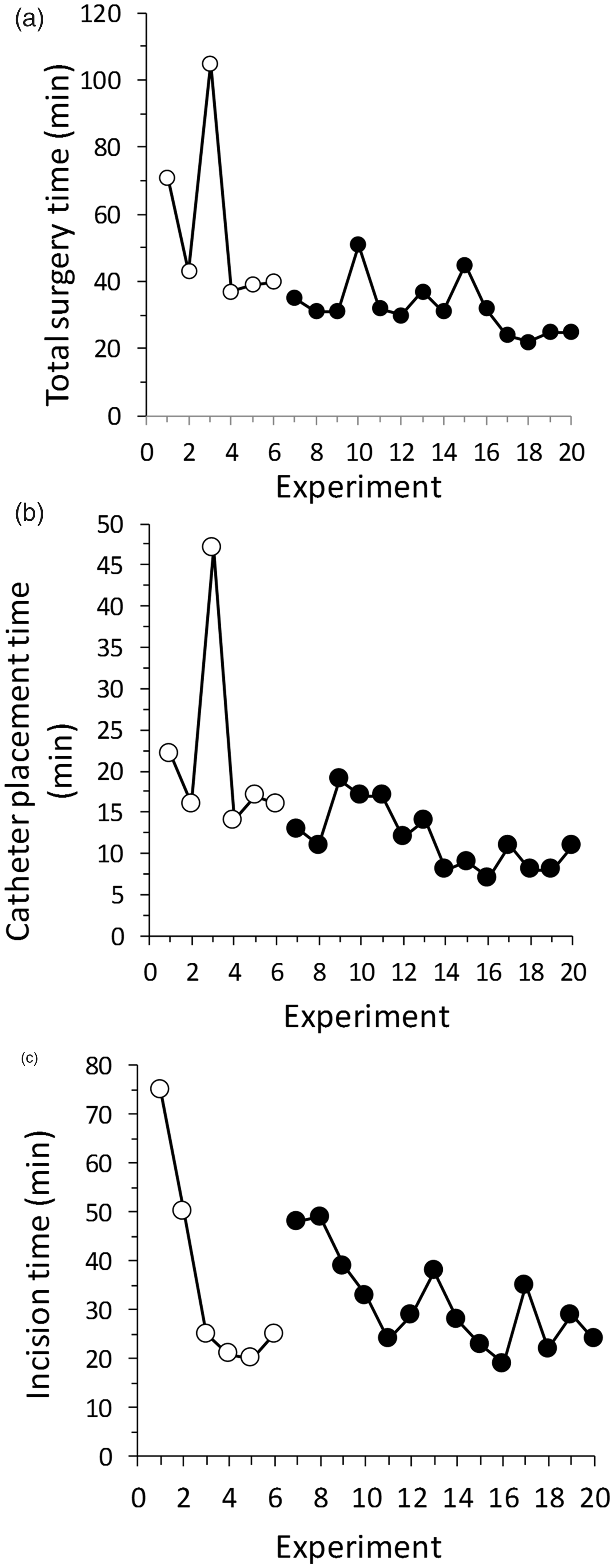
The performance target of zero errors was not achieved during the pilot (Figure 4). Most frequent errors were: missed blood samples at designated time points (3/6 experiments); overly-rapid isoflurane depletion before the end of the experiment (4/6 experiments); single intubation attempts >1 min (5/6 experiments), sPO2 <92% during baseline and stabilization for more than 10 min without documented corrective action (5/6 experiments); omission of two or more scheduled mechanical sigh breaths (6/6 experiments); and four or more documentation omissions (e.g. recording of major events, start/end times; 6/6 experiments).
Number of critical errors during pilot (○) and primary study (•) experiments. (Errors defined in Supplementary material, Table 2.)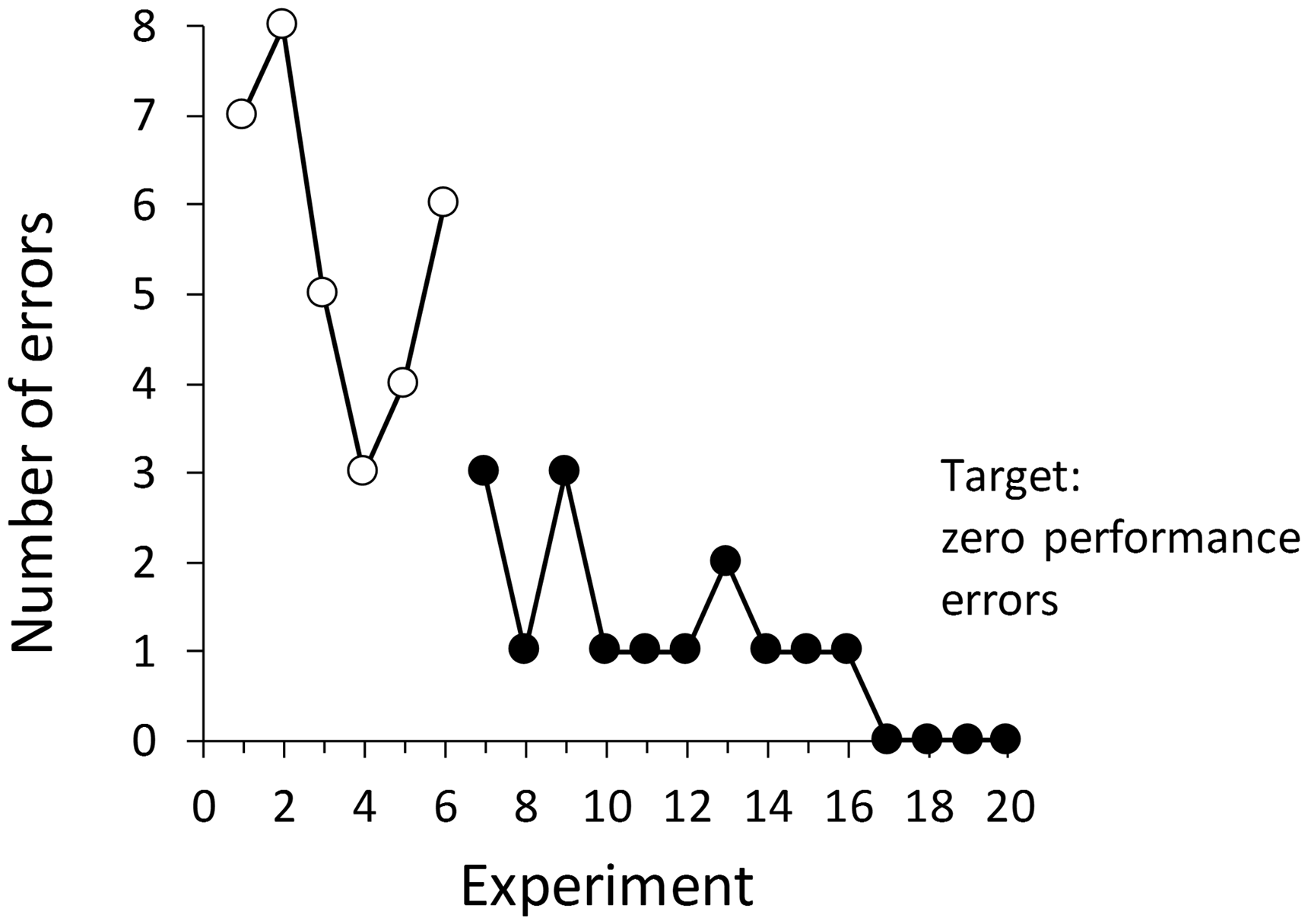
Animal model results are given in see Supplementary material 3, Animal model performance.
Problem correction strategies
The problem of missed blood samples was eliminated by tracking and updating blood sample collection metrics for each experiment, with predetermined sampling times and completion checkoffs displayed on a white board. After consultation with anaesthetists, overly rapid isoflurane consumption was corrected by reduction of oxygen flow rates from historical norms established for previous large animal protocols. Checklists were revised by colour-coding high priority task areas, with prominent display at strategic points around the lab. Error reduction was most effectively accomplished by embedding checklists and action items (e.g. reminders for mechanical sigh breaths) in the surgical electronic run sheets. Action items served as reminders to data scribes, making them less likely to omit crucial information. A dedicated data scribe was essential to ensure process oversight and act as oversight manager without distraction from other tasks.
Of concern were persistent deficiencies in airway management. Unacceptably long intubation times (Figure 5) during the pilot were due in part to insufficient sedation contributing to difficulties in airway visualization and tube placement, (supervised) learning of novice personnel and previously unrecognized skill issues. Remedial actions included increased sedation of animals before intubation, extensive personnel retraining and skill practice after each experiment and personnel replacement. Airway management was designated to two team members with the most consistent and best performance times. The target objective of successful intubation on the first attempt in <1 min was achieved consistently after pilot completion.
Total intubation times during pilot (○) and primary study (•) experiments. Dotted line is the target maximum intubation time of 1 min.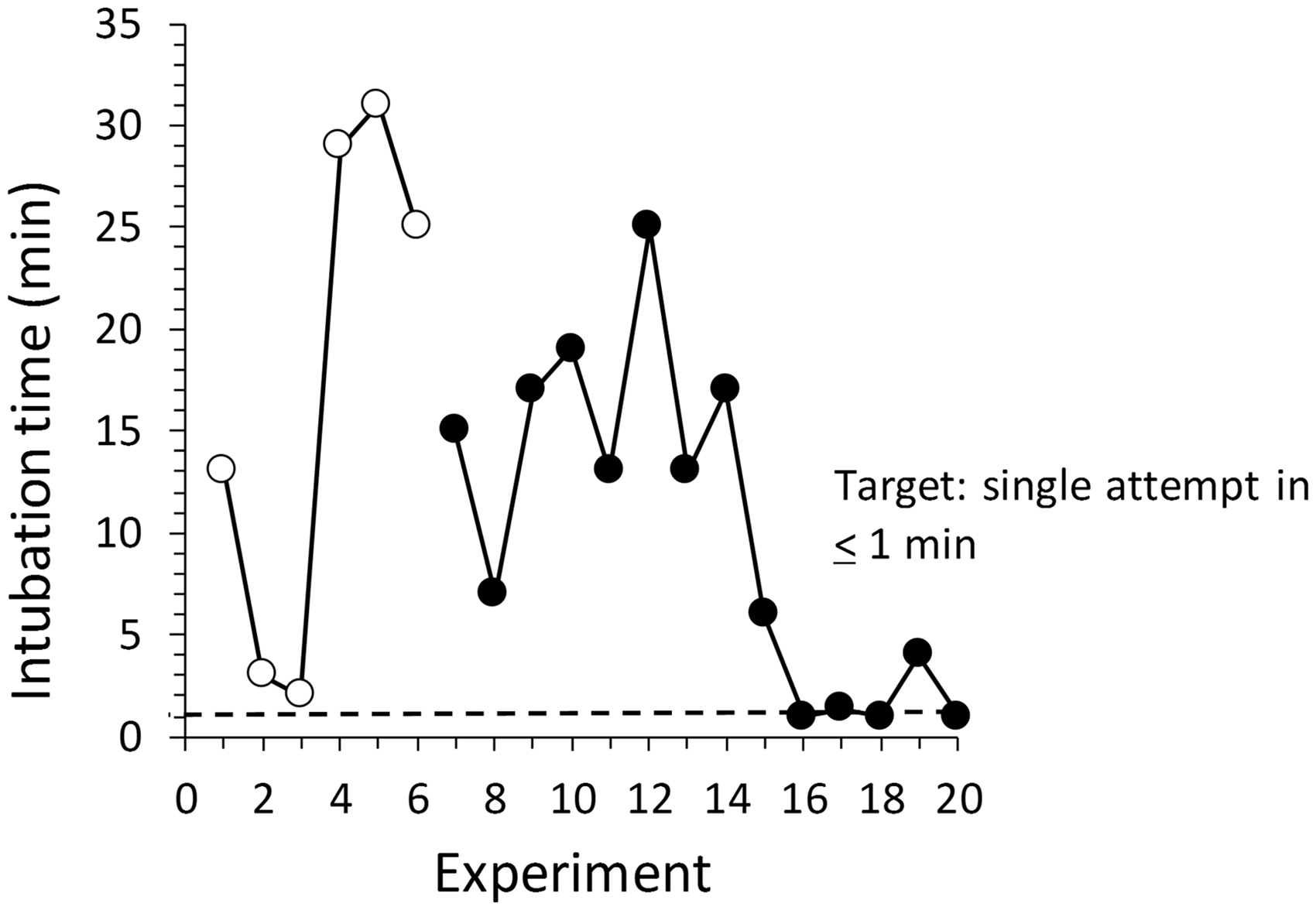
Discussion
Animal models of disease and injury are complex and logistically challenging. Variation in both protocol procedures and performance are frequently overlooked and can significantly affect research outcomes. Therefore, pre-interventional procedures must be comprehensively researched, documented and, if possible, tested prior to the primary trial. 1 Reactionary, scattershot approaches to problems are wasteful and may not achieve reliable solutions.16,17 In contrast, well-designed pilot studies can be extremely useful for early identification of logistic and welfare-related issues and for informing design of the main study.
In this study, we report how application of quality improvement techniques in a pilot trial enabled standardization of a complex protocol and identification of major procedural problems and changes to be made before beginning the primary trial. We focused on task identification, execution and control, items often overlooked in study planning. Because this pilot study was observational, we cannot ascribe a causal relationship between implementation of quality improvement techniques, improvements noted in performance and animal model stability. However, this approach resulted in a model system where animals achieved predefined physiological targets, variation was reduced and a clinically significant ‘reliable’ coagulopathy was produced.
Essential elements of a DMAIC quality improvement effort (Figure 1) include clear definition of tasks (work-stream maps are recommended for complex protocols); measurement of simple performance metrics; frequent assessment and evaluation of compliance; and ongoing control measures (including checklists and visual aids). Strengths of this approach are the use of simple routine measurements and tracking procedures to identify problems and guide decisions, and inclusion of all team members as contributors to the improvement process (especially the technical staff most involved with the day to day work). The main barriers to effective implementation are, first, the immense amount of planning and organization required before the study can begin and second, the sometimes radical cultural and behavioural changes required of all members of the team. Planning is often downplayed, especially if there is pressure to get the project started without further ‘waste of time’. However, investment in planning before the primary trial greatly increases the chances that the experiment will be completed successfully, with meaningful and informative data and without wasting animals.
Convincing team members to shift focus from task completion to task quality may be difficult. Because emphasis on quality may result in radical changes from the status quo, it is essential that investigators commit to providing adequate guidance and oversight, especially during the early stages of implementation. This does not imply micro-management: overly-specific goals encourage ‘cheating’, prevent learning from experience and result in overall declines in performance 36 because of errors caused by carelessness, haste, or inattention to the animal’s physiological condition. Instead, project leaders should provide clear and concise directives, clear communication of expectations and training and coaching as required. Checklists are invaluable in reducing error by placing less reliance on memory, and they empower staff by increasing investment in the experimental process, rather than relying on the laboratory chief for protocol implementation and monitoring. Checklists are used over a broad range of complex procedures and disciplines, from flying a plane to thoracic surgery. 29 By making procedures more effective and simplifying management, checklists could greatly benefit animal experimentation.
Although most of the technical team had the success of the trial as a priority, nearly all personnel tended to relapse into old habits as the study progressed. It is essential to communicate the value of quality assessment changes to usual practice, especially if the status quo is more comfortable or less threatening to personnel. Our data show that even for experienced motivated teams, different human performance measures stabilize or improve at very different rates and that others (such as airway management) presented serious and previously unidentified problems. Therefore, even with a standardized protocol in place, investigators must be diligent to ensure ‘control’ is ongoing to ensure major performance targets are met and protocol compliance is maintained.
An often-overlooked part of the experimental process, and a key refinement procedure, is habituation of experimental animals to handling, to reduce fear and distress prior to experimentation. 37 Habituation through positive reinforcement training has been shown to improve data quality and decrease variability, as well as having direct welfare benefits.19,20,38,39 Significant procoagulant changes occur with acute psychological stress, adrenergic upregulation and exercise. 40 In this study, habituation training was essential to enable rapid sedation without causing undue distress (as would occur if the animals had to be chased and/or restrained) and to reduce potential effects of stress on haemostatic function. Incorporation of habituation tasks into the work-stream map was essential to ensure this critical refinement step was not overlooked.
This paper focused only on quality improvement measures. Not discussed here are other major factors in experimental design more directly concerned with quality of evidence and bias minimization, such as randomization, blinding and formal statistical experimental design. Investigators are encouraged to consult specific guidelines such as ARRIVE 28 and DEPART, 41 and planning tools such as the Experimental Design Assistant (EDA; available at www.nc3rs.org.uk/experimental-design-assistant-eda).
Well-designed pilot studies with clearly specified objectives and measurable success metrics are useful tools for informing best research practice.5,6,42 They are also critical for the 3Rs principles of refinement and reduction. Integration by investigators of quality improvement processes and procedures in the early stages of a research project 11 would result in rapid improvement of research quality, better use of animals and improve translatability. However, integrating quality into animal-based research will require substantial cultural change among investigators. 11
Supplemental Material
Supplemental material for Informing efficient pilot development of animal trauma models through quality improvement strategies
Supplemental material for Informing efficient pilot development of animal trauma models through quality improvement strategies by Penny S Reynolds, Jacquelyn McCarter, Christopher Sweeney, Bassem M Mohammed, Donald F. Brophy, Bernard Fisher, Erika J Martin and Ramesh Natarajan in Laboratory Animals
Footnotes
Acknowledgements
We wish to thank: BD Spiess and J Halem (Anesthesiology), R Sabo and S Ketchum (Biostatistics), M Ellenberg (Chemistry), M Dance (Division of Animal Resources), C Blocher (Surgery) and P Lunn (deceased) and A Sharma (Transplant Surgery) for their generous assistance with the development of this animal model. We also thank an anonymous reviewer for constructive criticism and useful suggestions that greatly improved the manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship and/or publication of this article: This work was supported by the United States Department of Defense USAMRMC Contract Number W81XWH-15-2-0064 to R Natarajan.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
