Abstract
Intra-bone marrow transplantation (IBMT) may improve the seeding efficiency of transplanted hematopoietic stem cells compared to the routinely used intravenous injection. Current IBMT protocols are optimized for ease of use and to improve experimental results. However, there have been no investigations to assess the impact of IBMT on animal welfare. Here, we report the results of pain assessment after IBMT and the effects of refinements to the current standard procedure. IBMT was performed in either the tibia or the femur of a recipient mouse under general anesthesia. Impact was determined using clinical scoring of different parameters (lameness, grip capacity, body weight loss, footprint analysis), behavioural tests (burrowing, open-field), monitoring of stress hormones and post-mortem histology. The results revealed that IBMT definitely induces severe post-operative distress. Although IBMT in the tibia is technically easier, the degree of impairment and the distress observed were consistently higher than for transplantation in the femur. A refinement for IBMT in the tibia was achieved by using 30- instead of 26-gauge needles and by sparing the patellar tendon. Consequently, for IBMT, we recommend either using the femur, or if the tibia is required due to its better feasibility, using our refined protocol. Furthermore, IBMT should definitely be limited to one leg per animal.
The transplantation of human hematopoietic stem cells into immunodeficient mouse models is traditionally conducted by intravenous (IV) injection into the lateral tail vein. In order to properly proliferate and differentiate, the infused cells have to enter the bone marrow (BM) of the recipient. However, the seeding efficiency of IV transplanted cells in the recipient’s BM to subsequently initiate hematopoiesis is low.1–3 The marginal proportion of transplanted cells which establish in the marrow after entering from the blood (‘homing’) suggests that there is no preferential uptake of cells by the BM. Instead, the infused cells leave circulation shortly after injection and are predominantly found in other organs and tissues.4,5 This is particularly relevant when only a limited number of donor cells are available. To overcome this problem, intra-bone marrow transplantation (IBMT) has been adapted to mouse models as an alternative method of stem or progenitor cell transplantation. 6 Since the cells are deposited directly into the recipient’s BM, it has a higher rate of donor-cell engraftment compared to the IV transplantation route.7,8
During the last decade, murine IBMT has been applied in studies ranging from stem cell therapy to cancer research.9–16 Intraosseous transplantation is routinely conducted in the tibial shaft of the mouse after a small incision on the thigh. To reach the bone cavity, a hole for the microsyringe is drilled by inserting a 26- or 27-gauge needle into the joint surface of the tibia through the patellar tendon. The experimental procedure is performed under general anesthesia. Transplantations in the femur have also been described but are rare in comparison to tibia injection.17,18
To our knowledge, there has not yet been a comprehensive analysis of the impact of IBMT on animal welfare. However, this surgery could potentially result in considerable post-operative pain and distress. Therefore, the aim of this study was to evaluate the actual impact of standard IBMT in the tibia of mice. In addition, we determined the effects of using the femur as an alternative injection site and refined the standard procedure to reduce distress in mice from IBMT.
Materials and methods
Animals
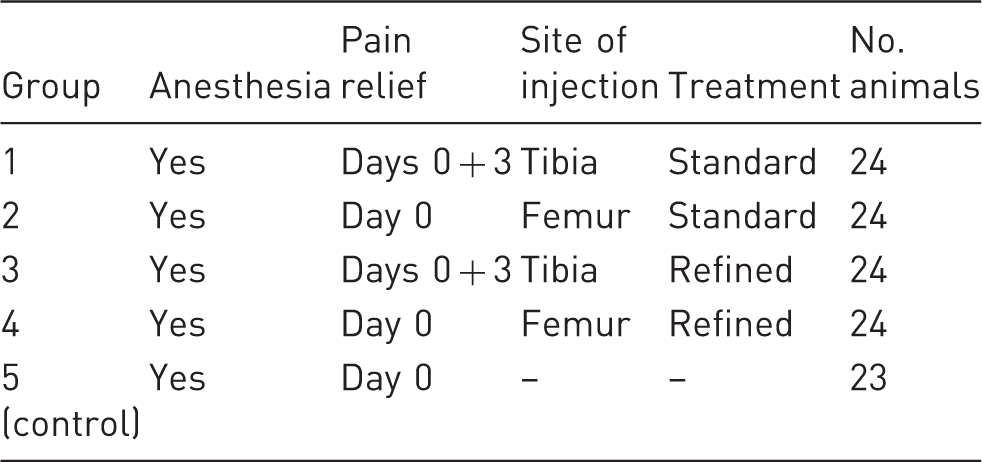
Number of animals in different treatment and control groups.
The distress induced by the IBMT was prospectively categorized as ‘medium’. The study was approved by the institutional ethics committee of the University of Veterinary Medicine Vienna, and an experiment license was granted under BMWF-68.205/0183-II/3b/2011 (Austrian Federal Ministry of Science and Research).
Experimental design
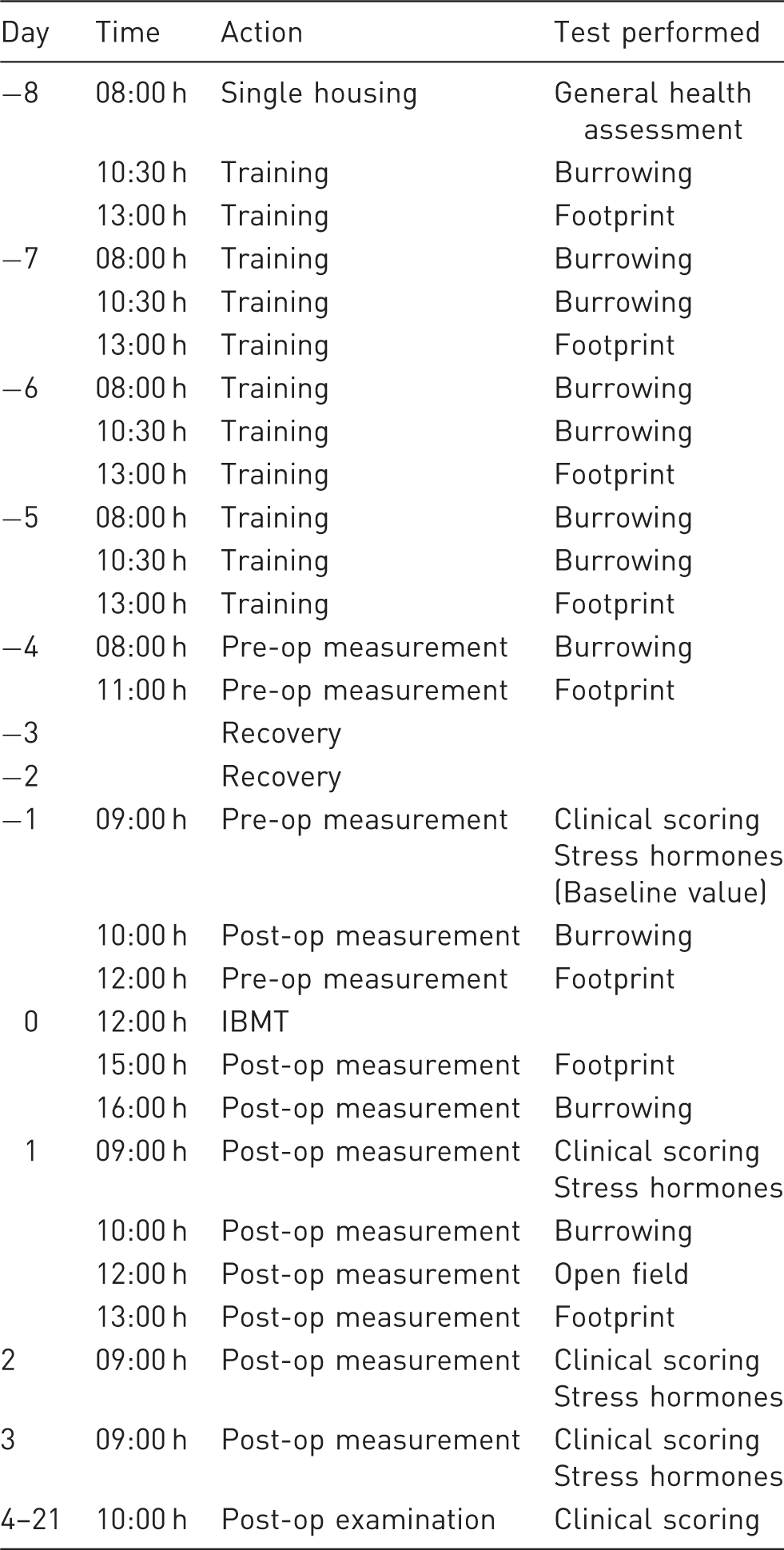
Schedule for the experimental procedures and tests before and after intra-bone marrow transplantation (IBMT).
Anesthesia and analgesia
For IBMT, the animals received a combined anesthesia of midazolam (0.7 mg/kg, Dormicum®; Roche, Vienna, Austria), medetomidine (7 mg/kg, Domitor®; Pfizer, Vienna, Austria), and fentanyl (0.07 mg/kg, Fentanyl-Janssen®; Janssen–Cilag Pharma GmbH, Vienna, Austria) (MMF) via intraperitoneal (IP) injection. Before the IBMT was performed, the surgical tolerance was tested by the toe-pinch withdrawal reflex. The anesthesia was antagonized after 40 min with a subcutaneous (SC) injection of a mixture of atipamezole (2.6 mg/kg, Antisedan®; Pfizer) and flumazenil (0.53 mg/kg, Anexate®; Roche). In order to keep the analgesic effect of fentanyl, its antagonist naloxone was replaced by 0.9% NaCl (Fresenius Kabi, Graz, Austria). In addition, buprenorphine (0.1 mg/kg, Temgesic®; AESCA Traiskirchen, Austria) was administered SC during IBMT. In order to assess the real impact of the treatment and to avoid side-effects from analgesic drugs, we originally intended to conduct the IBMT without further pain relief. However, during a pilot study it became obvious that the animals suffered significantly from post-operative pain for an extended time. Therefore, for ethical reasons, a second injection of buprenorphine was given 3 h post-IBMT to all animals. To avoid affecting the stress and pain indicators, additional analgesia was avoided until the footprint analysis, open-field test, stress hormone, and burrowing tests were complete (day 3). However, it appeared to be unethical to allow the animals to be in pain for the time necessary to complete the clinical scoring so those that still had a high clinical score after performing the post-operation measurements (both tibia transplantation groups), received an additional administration of buprenorphine on day 3.
Surgical procedure
Anesthetized mice were placed on a warming plate to prevent hypothermia. Eyes were protected with eye ointment (Oleovit; Fresenius Kabi). In contrast to the method described in the literature, we generally avoided making an incision on the thigh. For better visualization of the anatomical structures, the region around the knee was depilated with cream (Veet; Reckitt Benckiser AG, Wallisellen, Switzerland) before disinfection with 70% ethanol (EtOH). Only one leg (the right) was treated per animal.
For the standard IBMT in the tibia, the knee was flexed at about 90° and a 26 G × 12 mm needle was inserted into the joint surface through the patellar tendon and drilled into the BM cavity. The refined method for the IBMT was conducted in the same way except with a 30 G × 12 mm needle and avoided the patellar tendon (Figure 1a). In both procedures, the needle was removed and a microsyringe (10 µL; Bartelt GmbH, Graz, Austria, RN, 30 G × 12 mm) was introduced into the drilled tunnel to inject 5 µL human lymphocyte culture medium (RPMI 1640 medium, PAA, Cat. No. E15-842) into the BM cavity (Figure 1b). The transplantation into the medullar cavity of the distal femur was conducted in a similar manner as described for the tibia. However, due to anatomical reasons it was not possible to avoid a patellar tendon lesion. Therefore, the refined method for the femur transplantation differed from the standard procedure only in the reduction of the size of the needle used to drill the bone hole (Figure 1c).
(a) For intra-bone marrow transplantation (IBMT) the needle was inserted into the joint surface and drilled into the bone marrow cavity of the tibia (refined method). (b) Introduction of the microsyringe into the drilled tunnel for IBMT in the tibia. (c) Inserted needle in the bone marrow cavity of the femur after drilling through the joint surface (standard method).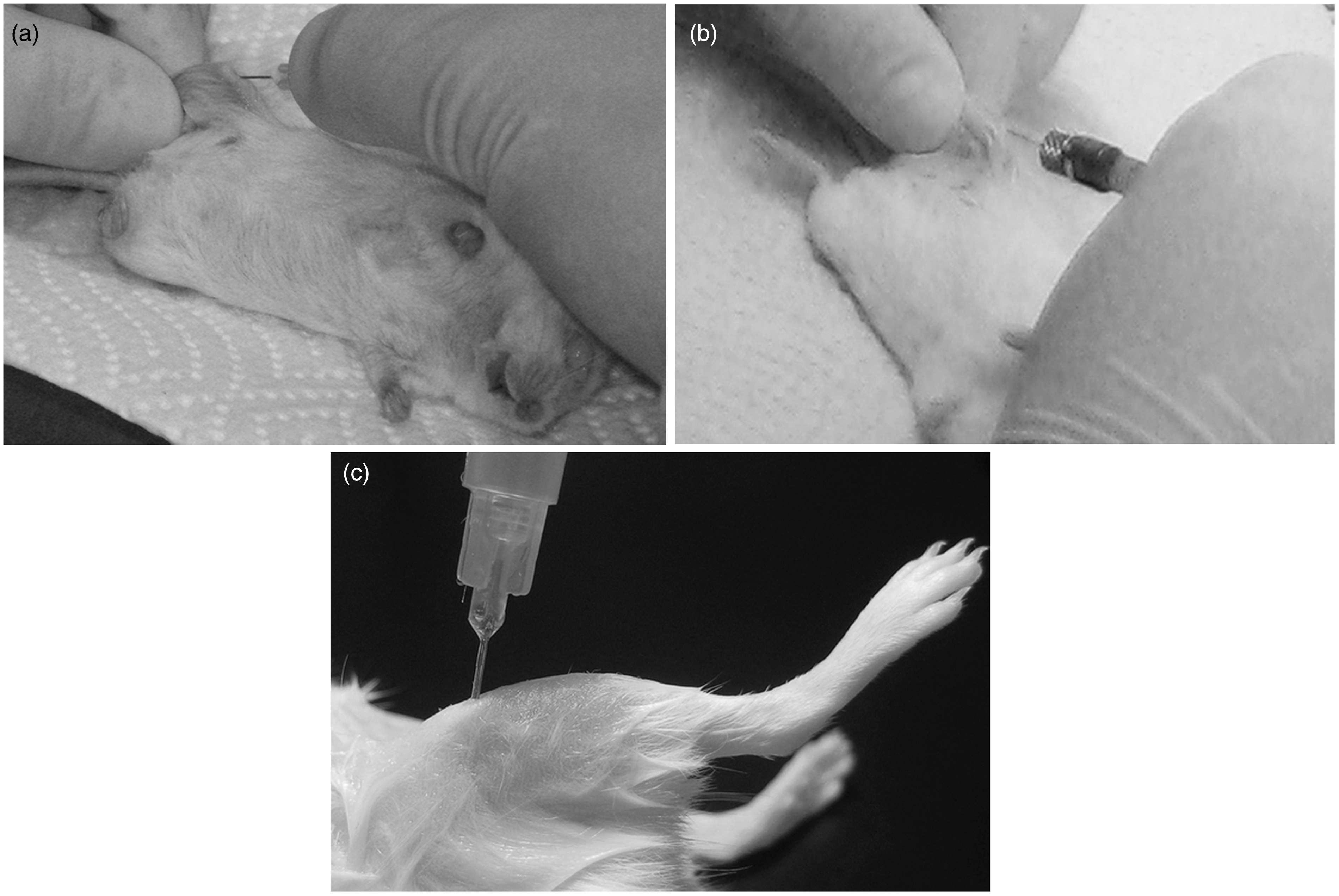
Clinical scoring
Clinical scoring scheme.

The grip capacity was assessed by placing the mouse with its hind legs on the edge of the home cage, and the mouse’s ability to grip the edge of the cage with the injected leg was noted. This was done three times in a row for each mouse and the results were averaged.
Body weight was measured for seven days post-operation using an electronic scale.
The set-up for footprint analysis consisted of a runway of 60 cm, guiding the mice from a light tunnel through a dark tunnel into a mouse house, similar to that from their home cage. Sunflower seeds in the house additionally motivated the animals to take the runway. To record the footprints of the mice, two paper sheets (DIN A4) were placed along the runway. The hind paws of the mice were painted with non-toxic blue ink immediately before their run. Gait analysis was carried out by measurement of the step length and step width of the footprints. 19 In addition, post-operative changes such as sharpening of footsteps, toe position, balance, compensation steps and rhythmic/linear movement were noted.
Behavioural scoring
The burrowing test was performed according to Deacon and Jirkof et al. with minor modifications.20–22 In brief, two standard water bottles (250 mL), one filled with food pellets and one left empty, were provided in combination with nesting material in the home cage. During the experiment, burrowing was visually observed for a maximum of 30 min after each morning refill. Burrowing behaviour was defined as the spontaneous removal of more than five pellets in 10 s from the burrowing tube without the need to feed, to hoard or to hide. The latency to burrow was not considered. In addition, overnight burrowing was recorded in the morning of days 0, 1, 2 and 3.
In order to measure general locomotor activity and willingness to explore, the open-field test was applied on day 1 post-operation. Individual mice were placed in the centre of a square arena (40 cm × 40 cm × 60 cm) and were allowed to explore the open field freely for 5 min. Standard parameters for locomotor activity (total distance, average speed, resting time, transits through the centre and time spent on the edges) were recorded with the automated video-computational TiBeSplit1.0 tracking system (Sony Color Video Camera CVX-V18NSP).
Monitoring of stress hormones
Concentrations of corticosterone metabolites were measured in the feces of the animals before (baseline value) and after IBMT. Fresh feces were collected while the animals were housed individually in an empty cage for one hour. The lag time for corticosterone metabolites in feces was estimated to be about 8–10 h. Therefore, fecal collection was scheduled in the morning to avoid any impact from the other tests performed the day before. Samples were placed immediately on ice and stored at –18℃. For the analysis, fecal samples were homogenized, and 0.05 g was mixed with 1 mL of 80% methanol. The samples were vortexed, centrifuged, and the supernatant was diluted 1:20 with assay buffer. Concentrations of corticosterone metabolites were analyzed by an in-house 5α-pregnane-3β,11β,21-triol-20-one enzyme immunoassay (EIA). For further details of the EIA and cross-reactivity of the antibody see Touma et al.23,24
Histological examination
After completion of the tests, the animals with the lowest and the highest clinical scores in each treatment group were sacrificed by cervical dislocation and the treated (right) hind legs were dissected. In order to examine the immediate post-operative impact of IBMT, age-matched BALB/c females were sacrificed and a standard IBMT was performed in either the femur or the tibia of one hind leg. Afterwards, treated and untreated legs were isolated for histological examination. The collected specimens were immediately fixed in 4% buffered formaldehyde solution, decalcified in 8% EDTA and subsequently embedded in paraffin. Serial sections were stained with hematoxylin & eosin (H&E).
Statistics
Results are expressed as means. Differences were assessed by repeated measurement and one-way analysis of variance (ANOVA) with Dunnet’s post-hoc procedure. If ANOVA was not appropriate, Kruskal–Wallis test, Mann–Whitney U-test and Wilcoxon W-test were used. Also, Spearman’s rank correlation coefficient, Pearson’s correlation coefficient and Pearson’s chi-square test were used to detect correlations among results. Data were analyzed using IBM SPSS v19.0 (SPSS Inc, Chicago, IL, USA), and a P value of <5% (P < 0.05) was considered to be significant.
Results
Clinical scoring
The clinical scoring clearly demonstrates the adverse effects of IBMT on the mice for all measured parameters, independent of the transplantation site and the refinement of the procedure (Figures 2a, 2b and 2c). See also Supplementary Figures S1a, S1b and S2 (all supplementary materials can be found online with this article at http://lan.sagepub.com).
Results of the clinical scoring. (a) Lameness scores of animals in different treatment groups from day 1 to day 21 after intra-bone marrow transplantation (IBMT). (b) Grip capacity scores of animals in different treatment groups from day 1 to day 21 after IBMT. (c) Body weight of animals in different groups from day 1 to day 7 after treatment.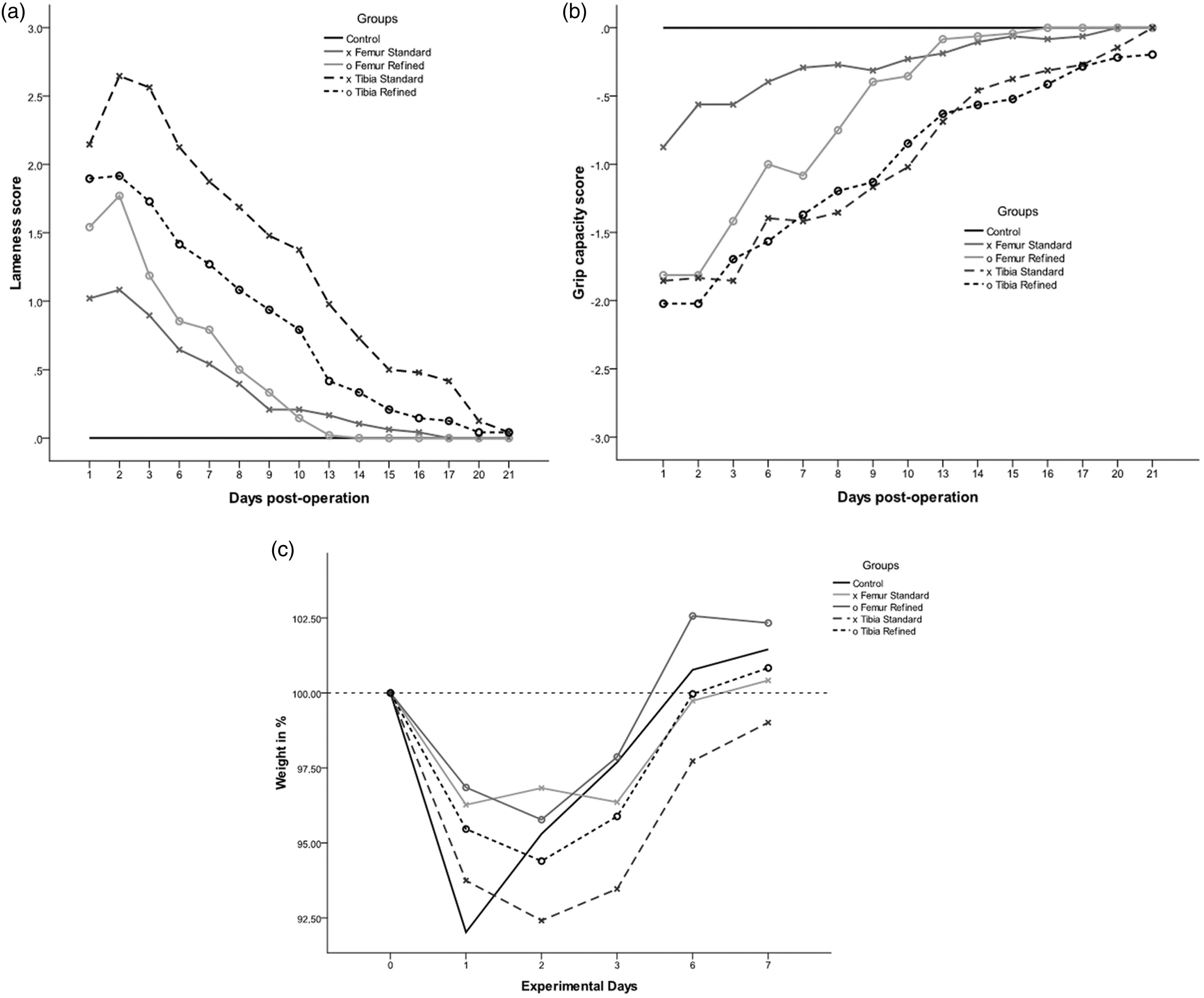
The degree of lameness and the reduction of grip capacity were more distinct for tibia groups compared to the femur groups, suggesting a higher and more prolonged burden after tibia injection (Figures 2a and 2b). There were significant differences in lameness scores between the femur groups and the controls until day 8 (standard method, P = 0.008) and day 9 (refined method, P = 0.015) post-operation whereas animals after standard tibia injection remained significantly different until day 20 post-operation (P = 0.009). The refinement of the injection technique was most effective for tibia injection, resulting in a generally lower degree of lameness and recovery on day 16 post-operation, four days earlier than with the standard method.
A similar impact of IBMT was observed regarding the grip capacity of the treated leg. The femur groups recovered on days 9 and 10 post-operation, respectively. Although both tibia groups also exhibited continuous improvement, the grip capacity remained significantly impaired until day 17 of the observation period for the standard method (P = 0.027) and until day 21 post-operation for the refined method (P = 0.021) (Figure 2b). Boxplots for lameness and grip capacity reveal a reciprocal development of both parameters over time (Supplementary Figures S1a and S1b). The lameness decreased and the grip capacity improved continuously over the post-operative observation period. This was confirmed by strong negative Spearman rank correlation coefficients from r = −0.66 to −0.78 between lameness and grip capacity for the post-operative observation period.
All experimental groups (including the control groups) reacted to the anesthesia with a body weight loss of 4–8% on day 1 post-operation (Figure 2c). With the exception of the standard tibia group, the other animals needed about six days to gain back their pre-operative body weight. Although the animals of the refined tibia group still exhibited a significant reduction in body weight on day 3 post-operation (P = 0.041), their body weights then rapidly increased to pre-operative levels. By contrast, the body weights of the standard tibia group were still significantly lower (P = 0.011) than the controls even seven days post-operation (Figure 2c).
Scoring results for lameness and grip capacity were supported by observed changes in the footprint profile for injected mice (Supplementary Figure S2). On day 1 post-operation we observed nonlinear movement, compensation steps of the untreated leg, and a lack of definition of the footprints. Measurement of step lengths and step widths revealed no significant differences between experimental groups and controls (data not shown).
Behavioural scoring
To assess any distress or discomfort due to IBMT, we also investigated spontaneous burrowing behaviour. All mice burrowed rigorously on day −1 (pre-operation). About 3 h post-operation, the burrowing behaviour was reduced in all groups, suggesting the antagonized anesthesia continued to have a strong effect on the animals at this time point. There were no significant differences between any experimental group and the controls on day 0 and day 1. However, 24 h post-operation, burrowing of both refined groups returned nearly to their pre-operative levels whereas post-operative burrowing was at the lowest level (not significant) in the tibia group injected using the standard method (Figure 3). All animals but one (from the standard tibia group) engaged in overnight burrowing even during the first night after surgery (data not shown).
Scoring results for burrowing behavior of different experimental groups and the controls before and after treatment.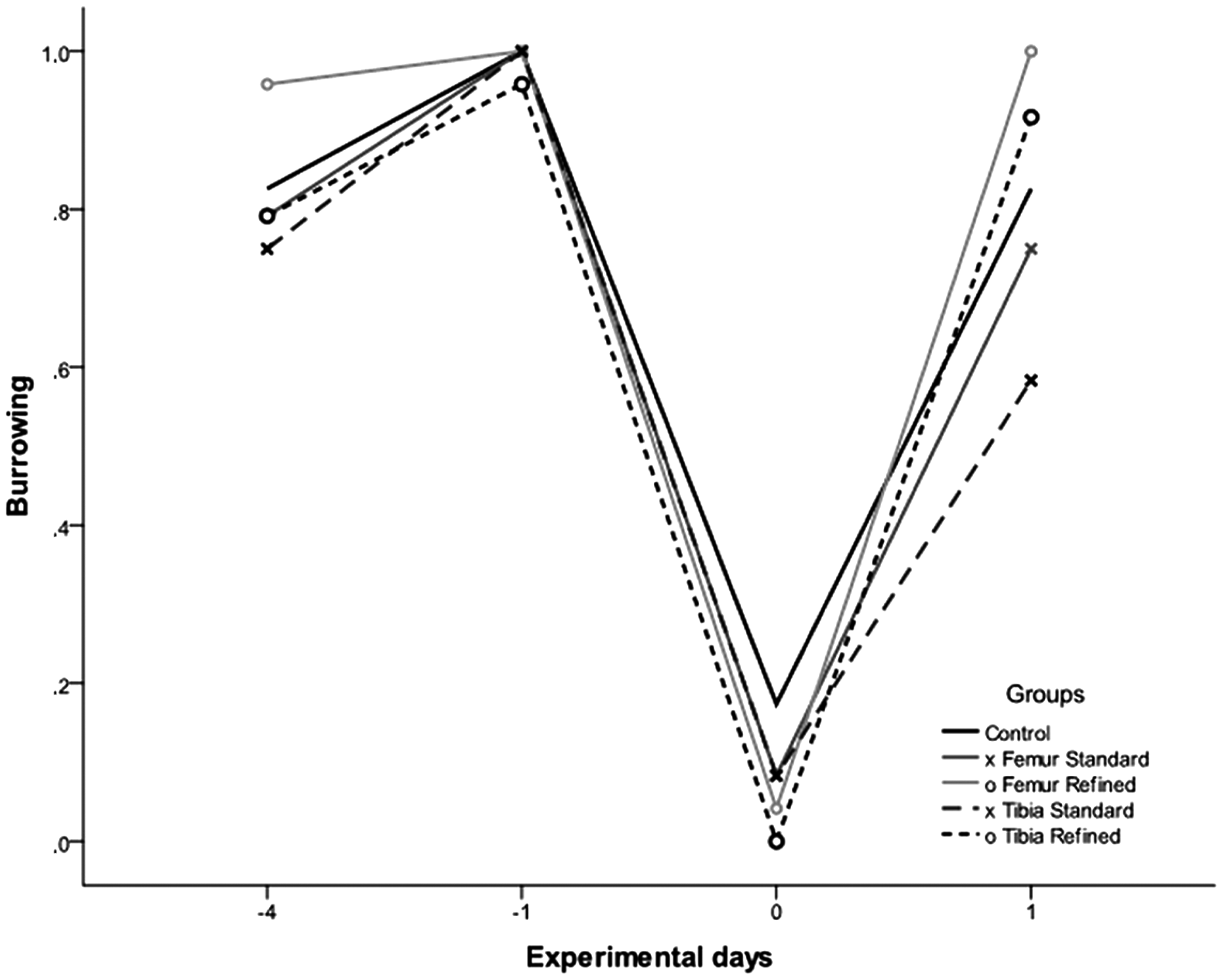
The open-field test was conducted on day 1 post-operation. No significant differences to the control group were observed in any measured parameter in both femur IBMT groups. The tibia groups did not differ in the transits through the center and time spent on the edges (data not shown). However, the distances travelled were significantly reduced in both the standard tibia group (P = 0.027) and the refined tibia group (P = 0.041) (Supplementary Figure S3). Furthermore, we observed a decreased speed in both tibia groups which was significant in animals from the refined tibia group (P = 0.036; Supplementary Figure S4).
Monitoring of stress hormones
To assess the pain and suffering of the animals after IBMT, we monitored stress hormone metabolites in the feces. There was a significant increase of stress hormones for the first three days post-operation in all groups including the controls. A significant increase of corticosterone metabolites compared to the controls was only detected for the standard tibia group on day 3 post-operation (P = 0.03; Figure 4).
Boxplots for corticosterone metabolites measured in the feces of different experimental groups and controls before and after treatment. Medians (lines in boxes), 25–75% interquartile range (iqr) (boxes), range from smallest to highest value excluding extreme values (whiskers), extreme values more than 1.5 times iqr above q3 or below q1 (•) or more than 3 times iqr above q3 (*). IBMT: intra-bone marrow transplantation.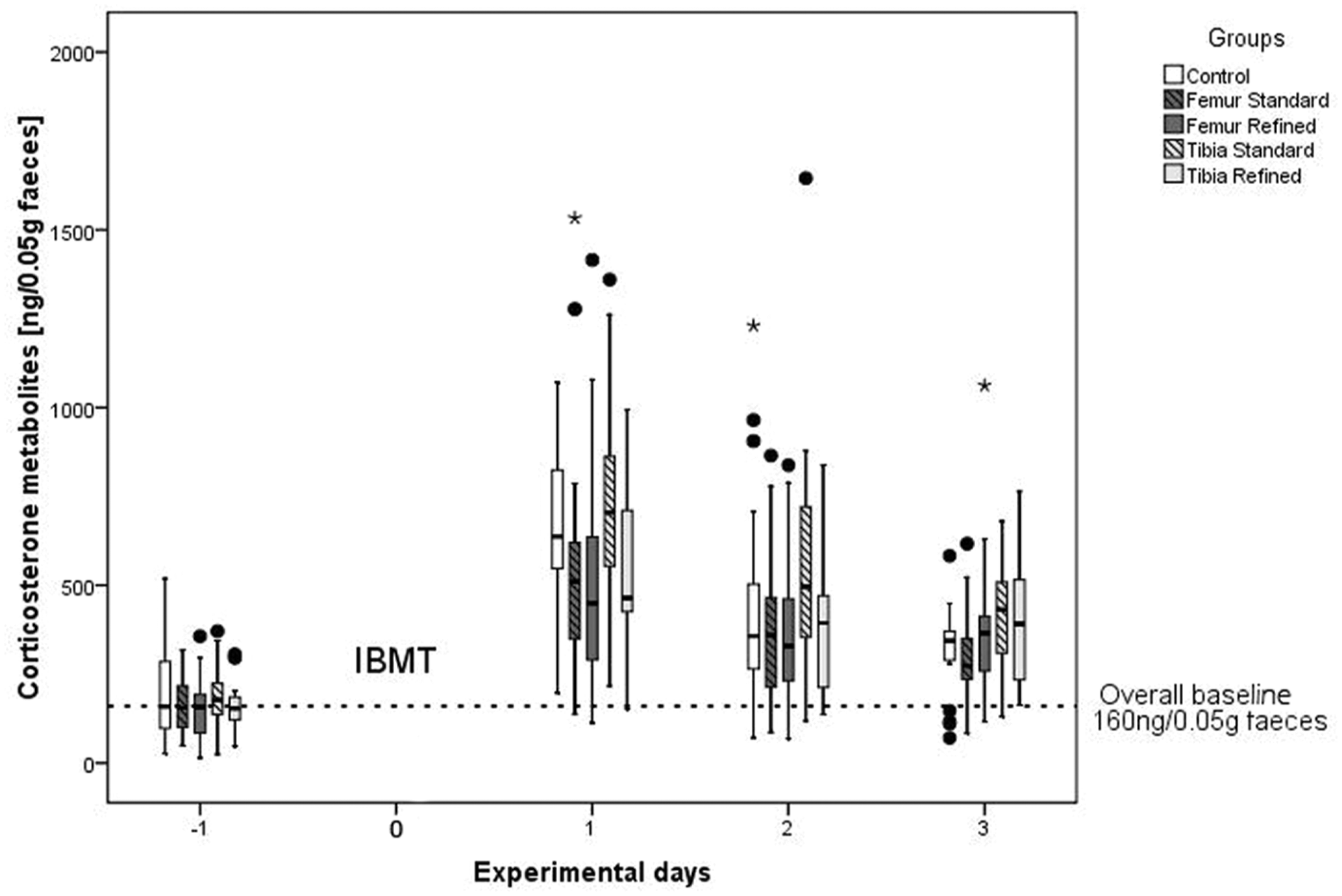
Histology
Figure 5a shows a histological section through the knee joint of a control animal depicting femur, tibia and the cruciate ligaments in the center of the joint cavity. The section through the knee joint of directly treated animals clearly shows the drilled channel penetrating the epiphysis and growth plate, reaching the BM cavity of the diaphysis (Figures 5b and 5c). Remnants of the previous penetration were visible as bony formations within the growth plate three weeks after treatment (Figures 5d and 5e). In addition, severe connective tissue proliferation and scar formation were repeatedly detected in animals treated by the standard tibia method (Figure 5d) but not by the femur route despite penetrating the patellar tendon. In the refined tibia group, connective tissue proliferation was found for one animal with a high clinical score; however it was much less intense than in the standard treatment (data not shown).
Histological section through the knee joint of a (a) control animal depicting femur (F), tibia (T) and the cruciate ligaments (CL) in the centre of the joint cavity. The knee joint of animals directly injected with the standard method in (b) the tibia and (c) the femur clearly shows a channel penetrating the epiphysis and growth plate and reaching the bone marrow cavity of the diaphysis (black arrow). Section through the knee joint of an animal three weeks after treatment with the standard method in (d) the tibia and with the refined method in (e) the femur demonstrating remnants of the previous penetration as bony formations within the growth plate (arrow). In addition, severe connective tissue proliferation and scar formation (asterisk) is seen at the site of the CL after standard tibia injection (d). Staining with hematoxylin & eosin (H&E), scale bar = 300 µm.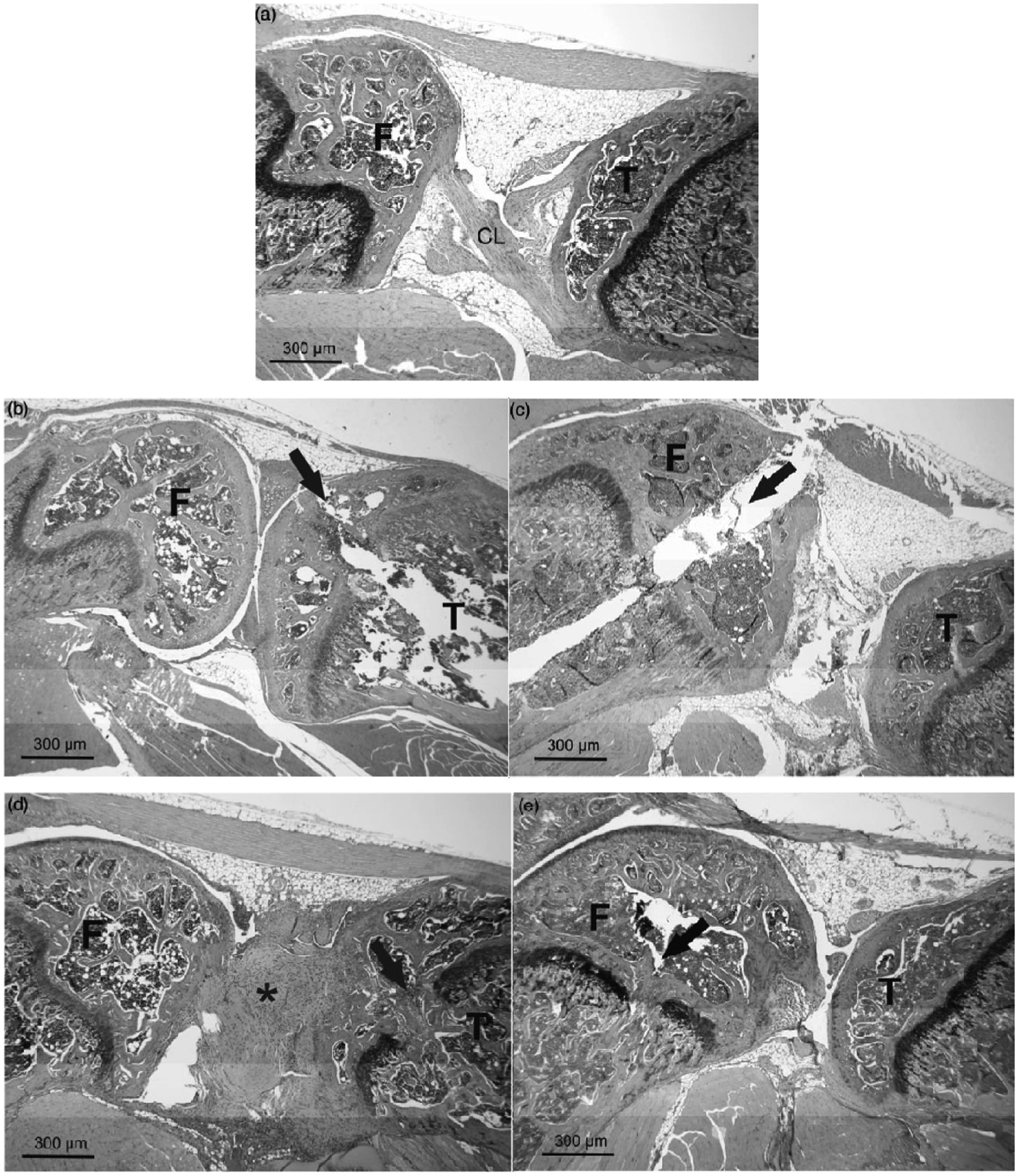
Discussion
IBMT in mouse models is a method for delivering (malignant) hematopoietic stem cells directly to their preferred engraftment site. It has been shown that the BM microenvironment promotes the survival and proliferation of the transplanted cells.7,25 The presented study was conducted to determine the distress induced by IBMT in the tibia and the femur of laboratory mice. Clinical scoring, behavioural tests, measurement of corticosterone metabolites and post-mortem histology revealed a significant impact on the well-being of treated animals. This finding was generally more pronounced for transplantation into the tibia compared to the femur. The adopted refinement of tibia injection by using smaller injection needles and avoiding the patellar tendon resulted in a measurable reduction of the impairment and accelerated recovery of treated animals compared to the standard method (except grip capacity, Figure 2b).
IBMT in mice is currently routinely conducted in the tibia. Due to the larger muscles in the thigh, the femur is poorly accessible for fixation, making IBMT more difficult for the experimenter. However, a possible advantage of the femur is that the condyles can serve as a guiding structure for the cannula into the BM cavity. In addition, in a flexed position, the anterior and posterior cruciate ligaments will not be encountered upon penetration. The higher burden seen after tibia injection coincides with severe connective tissue proliferation and scar formation around the cruciate ligaments (Figure 5d) since they cannot be avoided when injecting into the tibia. This malformation was mostly seen in animals after standard IBMT in the tibia but not found after femur injection. However, in the refined tibia group, connective tissue proliferation was observed at a lower degree and only in an animal with a bad clinical score (data not shown).
Pain signs such as increased lameness, decreased grip capacity and abnormal walking patterns occurred in all groups with IBMT. Furthermore, we found destroyed nests in the morning of day 1 even though all the mice were highly motivated pre-operation to build a nest. Nest destruction was observed in both experimental groups and the controls, suggesting a strong impact of the MMF anesthesia on the well-being and behavior of the animals during the first two days post-operation. The observations coincided with the results of the body weight and the burrowing test. Both seemed to be partially superimposed by the anesthesia since the control group exhibited similar changes to animals of the IBMT groups over the first days post-operation (Figures 2c and 3). Therefore, the results of body weight and behavioural scoring during the early post-operative period were obviously biased for the assessment of the level of discomfort and harm inflicted by IBMT.
In contrast to lameness and grip capacity, whose slow recovery reflected the impairment of the affected limbs, burrowing soon recovered to pre-operative levels. Good performance of spontaneous home cage behaviours like burrowing is thought to be indicative of well-being.21,22,26 Assuming that home cage behaviours are negatively affected in suffering mice, the fast resumption of burrowing after IBMT suggests that mice can cope with the impairment of one affected limb. Furthermore, the rapid return to pre-operative levels of this self-rewarding behaviour suggests no high and lasting levels of pain and distress after IBMT, despite the lingering of the disability.
Stress hormones were analyzed in the feces of treated animals and controls (Figure 4). Although the animals were undisturbed for two days before collecting feces for the pre-operative measurement, the overall baseline for corticosterone metabolites determined at day −1 was generally higher, as could have been expected from our previous measurement data. 27 As we can exclude external stressors, we assume that this is a residual impact from the prior five-day training period.
The levels of stress hormone metabolites increased significantly after IBMT in all treatment groups and in the control group, revealing that, post-operation, MMF anesthesia also has an effect on stress hormones that was obviously not compensated by the analgesic treatment. A significant increase in levels of stress hormone metabolites compared to those in the non-injected controls was only observable on day 3 for the standard tibia group.
In conclusion, the presented study demonstrates that IBMT in the tibia and the femur of mice has a significant impact on the well-being of the animals. Different tests have revealed that the technically simpler, and thus more common, tibia injection is more harmful to the animals, probably due to the lesions in the cruciate ligaments of the knee followed by connective tissue proliferation and scar formation. Thus, the femur should be the favored IBMT injection site. However, if this is not possible we strongly recommend a refinement by reducing the size of the injection needle and by sparing the patellar tendon. Because the treated limb cannot be used for up to three weeks, IBMT should definitely be limited to one leg per animal, irrespective of the bone chosen.
Footnotes
Acknowledgements
Special thanks go to Edith Klobetz-Rassam for corticosterone metabolite analysis, Michael Balka for support with the TiBe1.0-System and Karina Hrazdirova for support with the animal care. Special thanks also go to Christiana Winding and Auke Boersma for help in establishing the experimental work and for fruitful discussions of the data.
Declaration of conflicting interests
None declared.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
