Abstract
Current regulations emphasize that good husbandry practices allow animals to engage in species appropriate postural adjustments without touching the enclosure walls. This study evaluated the well-being of rats housed in a commercially available multilevel rat caging system, with or without access to the upper level of the caging. The evaluation methodologies included assessment of behavioral observations in the home cage, physiological assessment of metabolism and immune function, and determination of the affective state using a spatial cognitive bias assay. The study determined that rats that were provided access to the full multilevel cage during testing after initial restriction to the lower level of the cage demonstrated behavioral changes consistent with a positive affective state, while those with no changes to their housing situation had no significant differences in their affective states. Rats that were consistently housed with access restricted to the lower level of the cage exhibited a tendency to increased neutrophil:lymphocyte ratios as compared with those provided with access to all levels of the multilevel cage. There were no differences in body weight demonstrated between the experimental groups. Overall use of the cage space, as documented through analysis of behavioral observations in the home cage, demonstrated no significant differences in preferred location in the cage during the light or dark cycles, though rats with access to both levels of the cage were significantly more active during the light cycle. The results of this study suggest that the use of a multilevel caging system may improve the well-being of rats used in research.
It has been long recognized that protecting the health and well-being of animals used in research improves the reliability of studies that use animal models, ensuring that the results from these studies are as accurate as possible.1–3 Current regulations put increased emphasis on ensuring that husbandry practices are consistent with animal needs, including provision of cage complexity and manipulanda that allow expression of species appropriate behaviors, the ability to engage in species appropriate postural adjustments without touching the enclosure walls, and contact with conspecifics for social animals.4,5
The current standard laboratory rat caging system consists of a square or rectangular box that facilitates husbandry practices and bioprotection. Social housing is the preferred default environmental enhancement for providing social buffering and for minimizing potential distress,6–8 but social housing of rats is not always possible for scientific or clinical concerns. Whether individually or socially housed, modifications to the standard caging systems to create complex or super-enriched environments that allow enhanced social interactions or opportunities to engage in species appropriate behaviors have been associated with significant behavioral changes such as decreases in aggressive behaviors,9–12 improved performance on behavioral tasks13,14 and neural development,15–18 and significant improvements in overall health and well-being.19,20 These studies suggest that there are opportunities for raising current animal housing standards to improve the well-being of rats used in research.
Although European and American regulatory guidelines specify that animals should be able to engage in normal postures and make appropriate postural adjustments without touching the enclosure walls, these regulations recommend a cage height of 17.8 to 19.1 cm (7 to 7.5 inches) for rats. As rats frequently engage in bipedal stances, especially when exploring their environment (Figure 1) and adult rats measure an average of 15.2 cm (6 inches) from nose to tail, this height is insufficient for allowing rats to engage in normal postural positions.
21
Although studies of the effects of complex housing environments have included increases in vertical space,19,22 these studies also included increased social opportunities and provision of manipulanda, challenging the evaluation of the effect of cage size. Only one study has specifically looked at the utilization of vertical space by rats.
23
In this study, the author provided ACI, Lewis, and Sprague Dawley rats with vertical space ranging from 19 to 30 cm (7.5 to 11.8 inches) and tracked how they used it through the use of passive infrared detectors. They concluded that upright standing beyond the regulatory standard of 19 cm (7.5 inches) is an integral part of the rat behavioral repertoire.
The current regulatory standard of cage height for rats of 17.8 to 18 cm (7 to 7.5 inches) prevents adult rats from standing in a full bipedal stance.
Recently, one commercial caging system manufacturer has developed a multilevel caging system for rats (Double Decker™; Tecniplast USA, West Chester, PA, USA). This system has twice the vertical space than is currently recommended for rats with a cage height of 40.4 cm (15.89 inches). Additionally, the cage has a shelf in the center of the cage that represents approximately two-thirds of the floor space of the cage. This shelf increases the available floor space by over 50%, increasing the number of animals that can be housed in the caging system under current regulatory guidelines. It also provides rats with a variable level environment that would be expected to encourage activity, exploratory behaviors, and general improvements in health consistent with those seen in complex housing environments.
In this study, we evaluated measurements of the well-being of individually-housed rats in a commercially available multilevel rat caging system, with or without access to the upper level of the caging. The rats were individually housed to remove the potential for social buffering on the affective state of the animals in the study6–8 and to approximate the benefits that can be provided by this caging system in situations where the rat must be individually housed for scientific or clinical reasons. The evaluation methodologies included the assessment of behavioral observations in the home cage, physiological assessment of metabolism and immune function, and determination of emotional state using a spatial cognitive bias assay.
Materials and methods
Overview of the experimental design
All the projects were approved by the Institutional Animal Care and Use Committee prior to initiation of the study. To determine the effect of housing on the affective state of rats, 42 male Sprague Dawley outbred rats (Harlan, Indianapolis, IN, USA) were randomly and equally assigned to one of five treatment groups upon arrival: housed in a standard shoebox rat cage (control); full access to all levels of the multilevel caging system during the entire study (FF); access restricted to the lower portion of the multilevel caging system during the entire study (RR); rats that began the study with access restricted to the lower portion of the multilevel caging system during habituation and training, but were provided full access during the testing phase (RF); or rats that began the study with full access to all levels of the multilevel caging system during habituation and training, but had their access restricted to the lower portion of the multilevel caging system during the testing phase (FR) (Figure 2). For the rats that changed style of housing, their initial housing condition was maintained throughout acclimation (approximately one week), habituation (3–5 days) and training (approximately 15–23 days, depending on the pace of learning of the individual rat) of the spatial cognitive bias assay. They then experienced the new housing condition during the testing phase (seven days). They were tested three times, every other day, during the seven-day testing phase. Those rats that did not change housing type remained in their assigned housing type for the same period of time (approximately 5–6 weeks, depending on the pace of learning of the individual rat).
Overview of experimental design. F = full access to all levels of the multilevel caging. R = access is restricted to the lower level of the multilevel caging. Housing configuration changes after training phase and prior to initiation of testing phase with first letter indicating initial housing configuration and second letter indicating final housing configuration. For example, FF had full access to all levels of the multilevel caging throughout the entire study while FR had full access to all levels of the multilevel caging through the training phase of the study, but access was restricted to the lower level of the multilevel caging prior to initiation of the testing phase.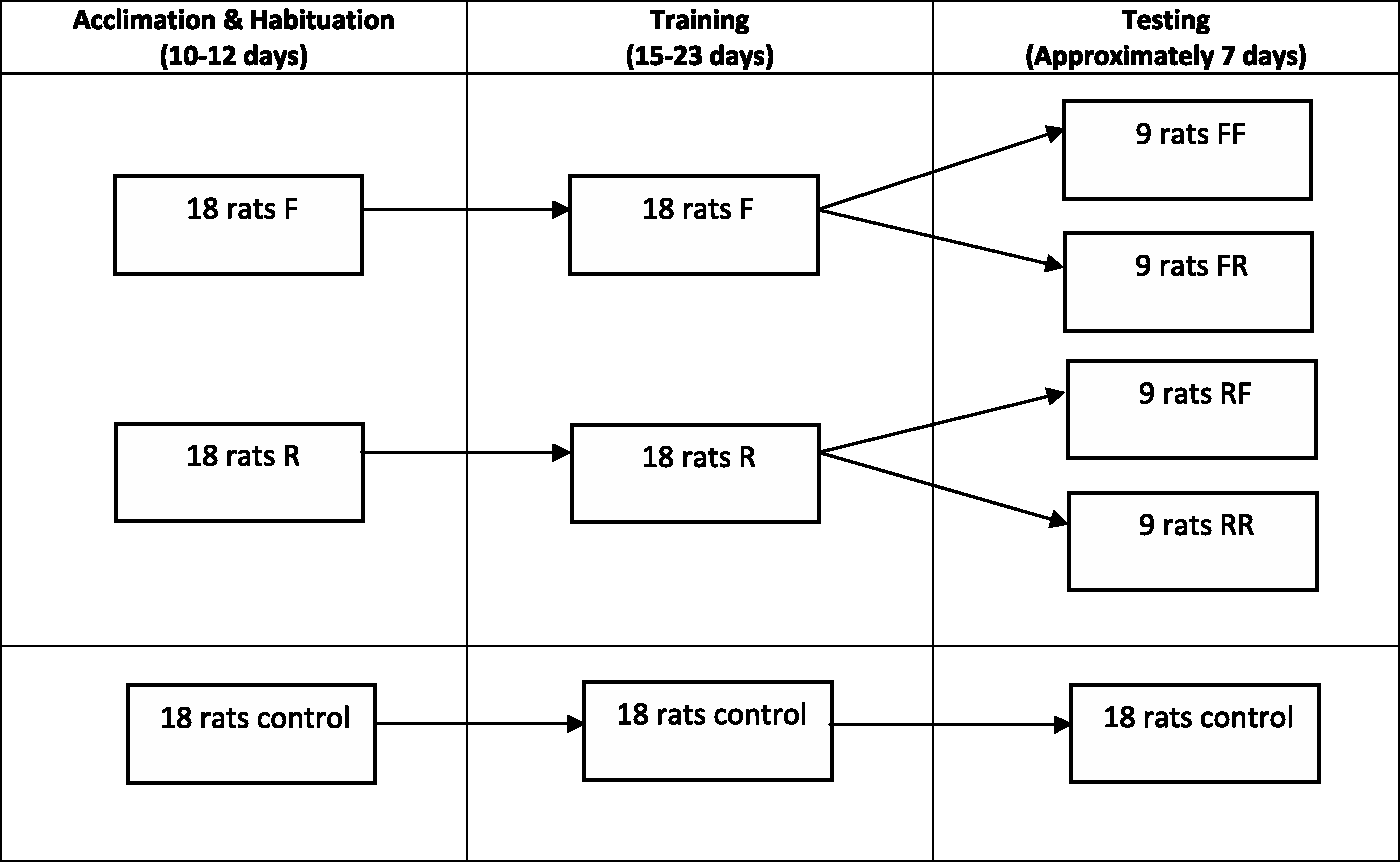
Animals and husbandry
Animals
Forty-two male Sprague Dawley outbred rats (Harlan) were used in this study. Each rat weighed between 180 and 225 g (approximately seven weeks of age) at the initiation of the study. The rat colonies in this facility were screened quarterly for the following pathogens: rat coronavirus, Sendai virus, pneumonia virus of mice, parvovirus,
Caging
The control cage unit (control) was a standard rat shoebox cage (Lab Products, Seaford, DE, USA). This cage measured 55.88 cm (L) × 31.75 cm (W) × 20.32 cm (D) (22 inches × 12.5 inches × 8 inches). The experimental cage unit was a multilevel rat caging system (Double Decker™, Tecniplast USA). This cage measured 46.2 cm (L) × 40.3 cm (W) × 40.4 cm (D) (18 inches × 16 inches × 16 inches). When rats were housed in the ‘full access’ housing (F), they had access to both the top and bottom levels of the cage (Figure 3a). When rats were housed in the ‘restricted access’ housing (R), a weighted plastic insert was placed over the interior shelf, creating a false ceiling and preventing the rat from accessing the top portion of the cage (Figure 3b). This created a cage which measured 46.2 cm (L) × 40.3 cm (W) × 20.2 cm (D) (18 inches × 16 inches × 8 inches), and which was consistent with regulatory standards.
(a) Commercially available multilevel caging system with all components demonstrated (Tecniplast USA, West Chester, PA, USA). (b) Commercially available multilevel caging system demonstrating placement of temporary divider (divider is highlighted with stripes for purposes of illustration only, no stripes were present on the divider during the study).
Husbandry
The rats were limit-fed to provide motivation for the performance of the spatial cognitive bias assay. Additionally, the limit feeding procedure allowed the laboratory to perform daily handling of each rat to ensure good clinical health as each rat was weighed daily and provided with its daily calculated ration assuming 8 g of feed per 100 g of body weight per day. If a rat failed to gain weight, a supplemental feed was provided. The rats were individually housed to facilitate monitoring of feed intake and behavioral observations in the home cage. The cages were lined with hardwood bedding (Sani-Chip; Harlan Teklad, Indianapolis, IN, USA). Municipal water was provided via a water bottle ad libitum. Cages were changed on a weekly basis. The macroenvironment was maintained at a temperature of 18 to 24℃ and relative humidity of within 30–70%, with a 12:12 h light:dark cycle, lights on at 07:00 h.
Cognitive bias assay
A previously described spatial cognitive bias assay was used to evaluate the positive or negative affective state of each rat. 24 The assay is divided into three phases: (1) acclimation and habituation, (2) training, and (3) testing.
Acclimation and habituation
Upon arrival, the rats were randomly assigned to a treatment group and allowed to acclimate to the facility, individually housed, for seven days. After the acclimation period, the rats were habituated to the training arena by placing them in the arena for a period of 10 min, with 10 treats (Fruit Loops; Kellogg’s, Battle Creek, MI, USA) randomly distributed on the floor of the arena. This procedure was repeated for 3–5 days, with the number of treats eaten and the number of fecal pellets deposited recorded daily for each rat.
Training
The training and testing procedures were conducted using previously published procedures. 24 Briefly, each rat was trained to discriminate between a rewarded (accessible treat) goal pot and an unrewarded (inaccessible treat) goal pot in an arena. Rats were trained every other day with 12 trials per day, with either a rewarded or non-rewarded goal pot provided in each trial and no more than two of the same goal pot type offered sequentially (e.g. ++−+−−+−++−−). The latency time to enter the goal pot was recorded for each trial. Discrimination was achieved when the latency times of entry between the rewarded and non-rewarded trials were statistically significant for two concurrent days of training, and this generally took approximately 15–23 days, depending on the pace of learning for each individual rat. Upon demonstration of discrimination, the rat was moved to its new housing configuration, if applicable, and testing began within 18 to 36 h of this change.
Testing
During the testing phase, sessions consisted of 13 trials, with rewarded and non-rewarded pots distributed randomly, with no more than two rewarded or non-rewarded trials offered consecutively. However, trials 5, 9, and 13 were randomly assigned ambiguous probes, with the pot being placed midway between the location of the rewarded or non-rewarded pot (neutral), between the neutral pot and the rewarded pot (rewarded plus [R+]), or between the neutral and the non-rewarded pot (non-rewarded plus [NR+]). The latency times to approach the reference (rewarded and non-rewarded) and the ambiguous probe (R+, neutral, and NR+) locations were recorded and analyzed to determine the affective states of the rats in each group. Each testing session was performed every other day for three sessions, resulting in three latency values for each ambiguous probe location for each rat.
Hematological analysis
Two blood samples were collected: one at the completion of the training phase and before initiation of the testing phase, and a second at the completion of the testing phase. Each blood sample was collected from the medial saphenous vein of the rat. A total of 50 uL was collected and placed in an EDTA treated blood collection tube. The blood sample was analyzed on a Hemavet® 950 (Drew Scientific, Oxford, CT, USA) which provided absolute and percentage values for all leukocytes. The total number of white blood cells, neutrophils, and lymphocytes were compared between groups. Additionally, the neutrophil/lymphocyte ratio was calculated. Increases in white blood cell numbers, especially as represented by increased neutrophil/lymphocyte ratios, are associated with chronic conditions of distress of at least seven days in length.25,26
Physiological assessment
Body weights at the time of the initial blood collection were compared with body weights at the final blood collection and the percent change was compared between groups (interval of approximately one week).
Behavioral observations in the home cage
Each cage was recorded between 16:00 and 00:00 h each day for the first three days that the rats were placed into their novel housing. This time was selected to be at the end of the day, when traffic in the room would be significantly decreased, and which would provide digital footage of both the light and dark periods. Digital footage was time- and date-stamped at the time of collection. For evaluation, digital footage between 16:00 and 18:00 h (light period) and between 22:00 and 00:00 h (dark period) was scored using a scanning method. Every 30 s, an observer to treatment recorded the rat as ‘active’ (e.g. moving around the cage, grooming, eating) versus ‘inactive’ (e.g. sleeping, sitting in one place). The observer also recorded the location of the rat in the cage as follows: top and front, bottom and front, top and rear, and bottom and rear. These data were coded and analyzed between rats that were housed with full access versus rats that were housed with restricted access, regardless of previous exposure. Although the observer could not be blinded to the condition that the animal that they were observing was currently experiencing (e.g. the presence of the plastic insert and any inability of the rat to obtain access to the upper level of the caging system would be obvious on the video recording), the observer was blinded to the experimental treatment group of the rats that they were scoring.
Statistical analyses
The choice between parametric and non-parametric alternatives was decided based upon evaluation of the data distribution for normality (inspection of residual plots), with transformations being performed if needed. This evaluation demonstrated that all data met the criteria for normality and homogeneity of variance. The main dependent variable for body weight, latency to approach reference and ambiguous probe locations, behavioral observations in the home cage, and neutrophil/lymphocyte ratios were analyzed with a full factorial analysis of variance (ANOVA) model in which the experimental housing condition served as the main factor (SAS version 10.0.0; SAS Institute Inc, Cary, NC, USA). Similarly, time was included as another factor in the analyses of habituation-related variables to evaluate possible differences in these variables over the 3 days of habituation using a repeated measures ANOVA. Whenever the ANOVA indicated a significant main effect of a factor or interactions between factors, least square means were compared using the Bonferroni test. Significance level for all tests and post-hoc comparisons were set at
Results
Cognitive bias assay
Testing
To analyze the latency times during the testing phase, the average time to enter each goal pot was calculated for each location. The initial analysis was to determine if there was a difference in the latency times to enter reference locations during testing or if this response differed between the five treatments. There was a significant difference between the latency times to enter the two reference locations, with the time to enter the non-rewarded location being significantly longer than the rewarded location (
To further investigate this significant interaction, we performed an ANOVA to compare treatments for each individual probe location, with the average latency to the reference location used as a covariate as described above. A significant effect of treatment was identified in the latency to approach time for the non-rewarded probe (NR+) location ( Latency times to approach the (a) non-reward plus [NR+], (b) neutral [N], and (c) reward plus [R+]. Differences between experimental groups and the control group are indicated by (*) (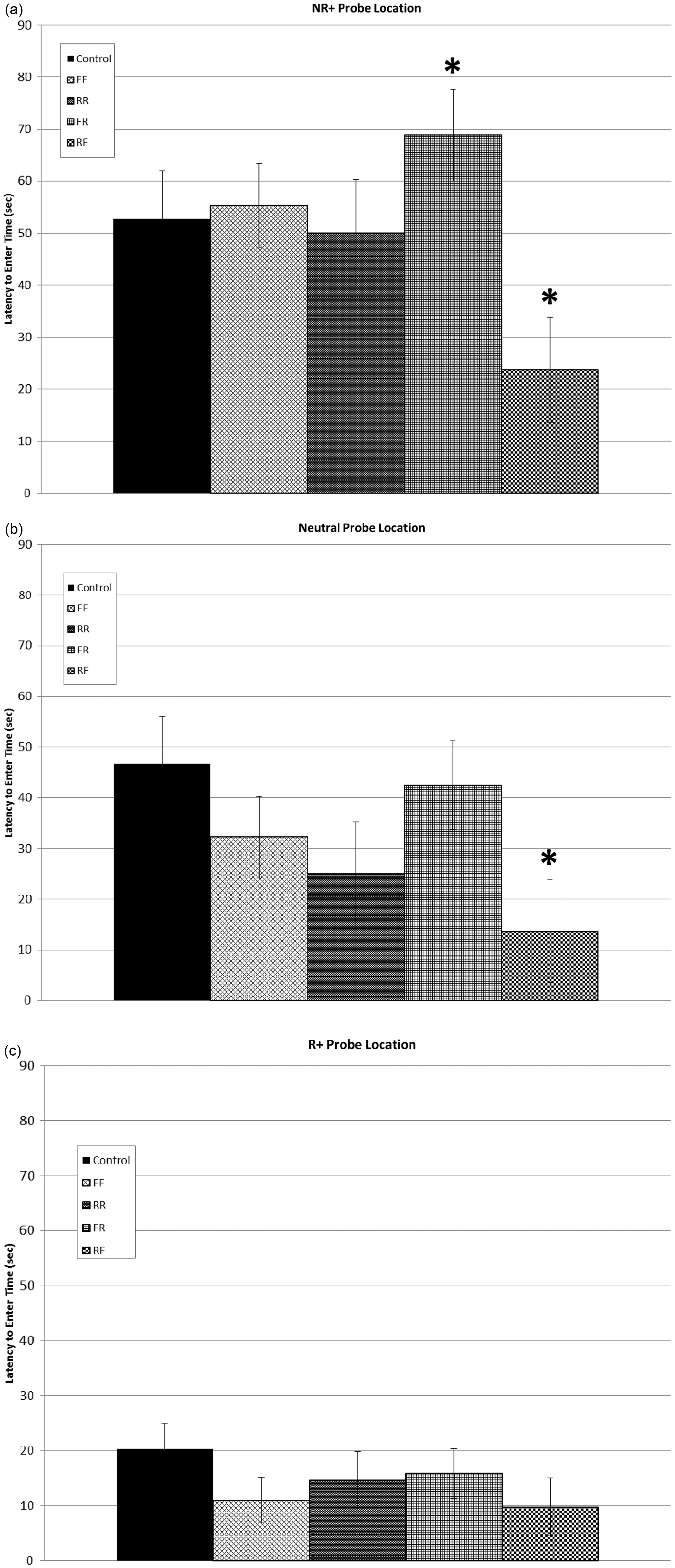
Hematological analysis
Mean and standard error of baseline leukocyte values for each treatment group as compared with the control group.

Significance was set at
Mean and standard error of final leukocyte values for each treatment group as compared with the control group.
WBC: white blood cells, NE: neutrophils, LY: lymphocytes.
Body weights
The differences in weight between the first and last blood collection were compared between the control group and the four treatment groups. There were no statistically significant differences between these groups (
Behavioral observations in the home cage
There were no significant differences between rats housed with restricted access (R) as compared with those housed with full access (F) when overall active behaviors were compared for the dark cycle (
The majority of the active behaviors were noted in the bottom level of the cage. During the dark cycle, the rats with access to the full cage were active in the top level of the cage approximately 9% of the observed time, and exhibited active behaviors in the bottom level of the cage approximately 27% of the time. The rats with access only to the bottom level of the cage exhibited active behaviors approximately 32% of the time. During the light cycle, the rats with access to the full cage exhibited active behaviors in the top level of the cage approximately 7% of the observed time and exhibited active behaviors in the bottom level of the cage approximately 19% of the observed time. There were no significant differences in active behaviors occurring in the bottom level of the caging system between the rats with access to the full cage and those limited to the bottom level of the cage during the dark cycle (
The rats demonstrated a preference for the rear of the cage, though there were no differences between rats that were provided with access to the entire caging system as compared with those with access limited to the bottom of the cage (
Discussion
The behavioral observations in the home cage analysis demonstrated that the rats with access to all levels of the multilevel cage (FF) used both levels to engage in active behaviors during both the light and the dark phases of observation, though it was not readily apparent why the rats that had access to all levels of the multilevel cage (FF) exhibited increased activity during the light cycle. The lower level was used primarily for sleeping and it was hypothesized that this was due to the shade from the room light that was provided by the position of the intra-cage shelf, especially as the back was preferred to the front by all the rats, regardless of treatment group. Because the multilevel caging system is made of transparent polysulfonate and has a light weight, open frame construction for the rack, ambient light is able to easily penetrate the cage with limited areas for the rats to take shelter and facilitate sleeping behaviors during the light cycle. Overall, the rats in this study were relatively inactive during both the light and dark periods, probably due to the individual housing and lack of conspecifics with which to interact. 11 If the rats were pair- or group-housed, more interactions would have been seen using all of the available space during our observation periods, as was reported by the manufacturer (Tecniplast which made the ‘Double Decker’ – a new and radical approach to individually ventilated cage (IVC) rat caging by the industry for the industry).
Cognitive bias assays are designed to objectively measure the affective state of animals, to determine if they are in a positive affective state (‘optimistic’) versus a negative affective state (‘pessimistic’).24,27–31 Because these affective states are generally not stable for long periods of time in a clinically and mentally healthy individual, the assay provides a reasonable reflection of how any factors that have changed in the immediate past have affected the animal’s affective state.27,29,32 The results of the cognitive bias assay in this study suggest that a change in environment has a significant effect on the affective state of rats which lasts through approximately the first week following the change. The rats in the FR group that were provided with full access to the multilevel caging system initially, but then had their access restricted to the lower level of the cage during testing, demonstrated significantly increased latency times to approach the rewarded reference and NR+ probe locations, and overall increased latency times to approach the other locations. The rats in the RF group that had their access restricted to the lower levels of the multilevel caging system during training, but were then allowed full access during testing demonstrated significantly decreased latency times to approach the NR+ location, and overall decreased latency times to approach the other locations. These findings suggest that the change in environment induced a negative affective state in those rats which found their environment restricted, while the provision of an enhanced environment induced a positive affective state. It is possible that those rats which were provided with full access to the full cage after access had been restricted to the lower portion of the cage (RF) had shorter latency times for the rewarded reference and the NR+ probe location because these rats were more active and therefore more hungry and motivated to seek the treat. Likewise, the rats that had been provided with full access to the cage initially but were then restricted to the lower level (FR) may have exhibited longer latency times because they were using less energy by being more sedentary. However, our interpretation of the cognitive bias assay as a measure of the affective state is consistent with the results of others who have demonstrated that the provision of environmental enhancements induces a positive affective state,14,28,33 regardless of activity levels.
The two groups of rats that had no change in their housing environment (FF and RR) showed no significant differences in their latency times to approach the reference locations or the ambiguous probe locations. This result is not unexpected, as the rats in these two groups had had over three weeks to habituate to their housing environment. Any changes in affective state would have been expected to occur in approximately the first week after being put in the new environment, a parameter that requires further exploration in future studies.
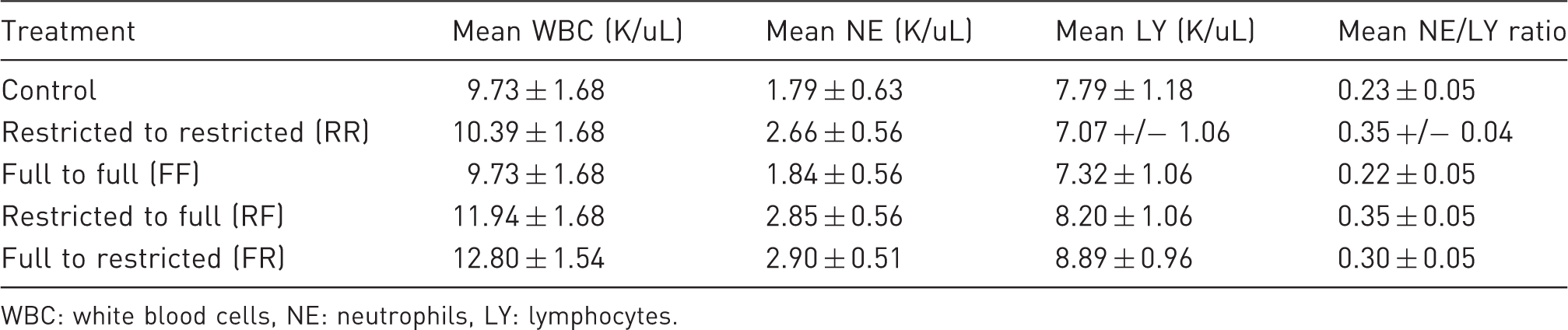
To determine if chronic housing with full versus restricted access to the multilevel caging system was beneficial, we examined the hematologic data that were collected during this study. After approximately three weeks of housing with either full or restricted access to the multilevel caging, the rats with full access had increased average white blood cell counts, increased average total number of neutrophils, and increased average total number of lymphocytes as compared with the control group. However, none of these increases were significant and all were within the normal ranges previously published for male Sprague Dawley rats, with the exception of the increase in the total number of neutrophils, calling into question the biological relevancy of these increases. 34
When the neutrophil/lymphocyte ratio of the control group was compared across all four experimental groups, the rats with continuous full access to the multilevel caging system (FF) demonstrated a decreased neutrophil/lymphocyte ratio, but the remaining groups had similar elevations, though none of these increases were statistically significant. As an increase in neutrophil/lymphocyte ratio has been described as consistent with distress, 25 these elevations were hypothesized to be due to a response to the chronic distress of the inability to access all levels of the multilevel caging (FR and RR) and the distress associated with a change in the environment for the other groups (FR and RF). Although it was not significant, when the neutrophil/lymphocyte ratio was compared between the two groups with no environmental change throughout the study (RR and FF), the neutrophil/lymphocyte ratio was decreased in the rats that had full access (FF), suggesting that the latter experienced less distress associated with this housing configuration. 25
There were no significant differences in weight across subjects, suggesting that neither full nor restricted access to the multilevel housing created different weights according to cage size. However, these rats were three months old on arrival and the times between their arrivals and baseline days of training varied, so restrictions of food intake while growing may have contributed to the differences in weight between cage groups. Additional further study is needed to assess the effect of long-term housing in the multilevel caging system on growth and overall body condition.
This study concluded that the provision of access to all levels of a multilevel caging system induced a positive affective state in rats. Rats that were chronically housed with full access to this multilevel caging system (FF) also demonstrated a reduction in their neutrophil/lymphocyte ratio, suggesting an improvement in their overall well-being. The results of this study suggest that the use of a multilevel caging system that provides rats with more space and complexity in structure may improve the well-being of rats that are used in research. However, additional studies that evaluate other physiological parameters of animal well-being (such as reproductive success and changes in body weight over months) should be considered to determine if there is a long-term benefit to the use of a multilevel caging system.
Footnotes
Funding
This study was supported through internal funding provided by the School of Medicine at Indiana University in Indianapolis. The rack and caging system was loaned to the institution by Tecniplast and subsequently purchased by the institution.
Declaration of conflicting interests
The authors declare that they have no competing interests.
Acknowledgements
The authors wish to thank Nate Ooms and Amy Treat for their assistance in the collection and entering of data for this study.
